Professional Documents
Culture Documents
Group Transfer Polymerization: Concept
Group Transfer Polymerization: Concept
Uploaded by
cuongtran_siegenOriginal Description:
Original Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Group Transfer Polymerization: Concept
Group Transfer Polymerization: Concept
Uploaded by
cuongtran_siegenCopyright:
Available Formats
Group Transfer Polymerization
Concept
Getting to know your Silicon
Group Transfer Polymerization
Concept
Getting to know your Silicon
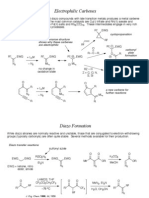
R FR R Si W FR R
F Si W R
-1 FFR R
F F Si W R
-2
Group Transfer Polymerization
Concept
R FR R Si W W FR R Si R F -1
Si
F-
W-
Si
Group Transfer Polymerization
Concept
R FR R Si W FR R
F Si W R
-1 F
-
F R R Si W F R
-2
F-
Five coordinate Silicon has greater Lewis acidity a. Because of charge or b. Because of greater resonance stabilization of six coordinate silicon
Group Transfer Polymerization
Initiator
Si
Si + H
O OR
O OR
Silyl ketene acetal
Group Transfer Polymerization
Mechanism: Propagation
OSi(CH3)3
OCH3
X- (X = F, RCOO, etc) OCH3 -
X Si(CH 3)3
Group Transfer Polymerization
Mechanism: Propagation
OCH3 X Si(CH 3)3 O -
MMA OCH3 OCH3 O O X Si(CH 3)3
Group Transfer Polymerization
Mechanism: Propagation
OCH3 OCH3 O O X Si(CH3)3
OCH3 OCH3 O O
X Si(CH3)3
Group Transfer Polymerization
Mechanism: Propagation
OCH3 OCH3 O O X Si(CH3)3
OCH3 OCH3 O O
X Si(CH3)3
Group Transfer Polymerization
Mechanism: Propagation
OCH3 OCH3 O O
OCH3 X Si(CH3)3 OCH3 O OCH 3 O O X Si(CH3)3
Group Transfer Polymerization
Mechanism: Propagation
OCH3 OCH3 O OCH3 O O X Si(CH3)3
OCH3 OCH3 O OCH3 O O X Si(CH3)3
Group Transfer Polymerization
Mechanism: Propagation
OCH3 OCH3 O OCH3 O O X Si(CH3)3
OCH3 OCH3 O OCH3 O O X Si(CH3)3
Group Transfer Polymerization
Mechanism: Termination
OCH3 OCH3 O OCH3 O O X H Si(CH3)3 Cl OCH3 OCH3 O OCH3 O OH X + Cl Si(CH3)3
OCH3 OCH3 O OCH3 O O
Advantages of GTP
Group Transfer Polymerization
1. Living (PDI 1) 2. Can do this reaction at room temperature (ionic requires low temp) 3. Can make telechelic polymers 4. Obtain random copolymers (reactivity ratios less of a consideration) 5. Air stable (but not water stable) 6. Can use DMF as solvent 7. Tolerant of other functional groups, especially vinyl side chains that would polymerize during radical polymerizations. Cant use acids (MAA)
O
Group Transfer Polymerization
Disadvantages of GTP
1. Only use for acrylate or methacrylate monomers (or aldehyde as discussed next) 2. More expensive than radical, 3. Requires water free conditions (pure reagents)
Group Transfer Polymerization
Aldol GTP: Things to know
1. Should use less than 5% anionic catalyst (F- , or CH3COO-) to avoid total catalyst displacement 2. Rates slower than radical polym. (by approx an order of magnitude) 3. Can get competing radical polymerization.
Group Transfer Polymerization
Aldol GTP-Initiation
OSiMe2t-Bu O ZnBr2
ZnBr2 SiMe2t-Bu
O
Group Transfer Polymerization
Aldol GTP: Initiation and Propagation
ZnBr2 SiMe2t-Bu
O O
ZnBr2 SiMe2t-Bu
O
O O
SiMe2t-Bu ZnBr2
ZnBr2 SiMe2t-Bu
O
Group Transfer Polymerization
Aldol GTP
O O
SiMe2t-Bu ZnBr2 SiMe2t-Bu
O
O O
SiMe2t-Bu SiMe2t-Bu ZnBr2
Group Transfer Polymerization
Aldol GTP
109 Si
X-
90 Si
Group Transfer Polymerization
Aldol GTP
New catalyst-free initiator
Si
OCH3
You might also like
- Casting Acrylic ProcessDocument32 pagesCasting Acrylic ProcessFirman Muttaqin100% (3)
- Schaum's Easy Outline of Organic Chemistry, Second EditionFrom EverandSchaum's Easy Outline of Organic Chemistry, Second EditionRating: 3.5 out of 5 stars3.5/5 (2)
- Biochemistry of Carbohydrates PDFDocument7 pagesBiochemistry of Carbohydrates PDFAshley Beatriz Pascual100% (1)
- Coursesaver (Chad) College Physics OutlinesDocument40 pagesCoursesaver (Chad) College Physics Outlinescalong558No ratings yet
- Chapter 10-Structure and Synthesis of AlcoholsDocument22 pagesChapter 10-Structure and Synthesis of Alcohols張湧浩No ratings yet
- MCAT Biology Complete OutlinesDocument34 pagesMCAT Biology Complete OutlinesJacob Mikhail90% (10)
- High School Chemistry Grade 10-12Document486 pagesHigh School Chemistry Grade 10-12Todd95% (39)
- This Difference Between Living and NonDocument1 pageThis Difference Between Living and Noncuongtran_siegenNo ratings yet
- Particle Technology and Applications PDFDocument335 pagesParticle Technology and Applications PDFUzair Javaid50% (2)
- Module 3 Class 4 J BatchDocument20 pagesModule 3 Class 4 J BatchVedurupaka Venkata SaiNo ratings yet
- Amino Acids and Protein (Saifur)Document45 pagesAmino Acids and Protein (Saifur)Anggry SolihinNo ratings yet
- Biochemistry of CarbohydratesDocument7 pagesBiochemistry of CarbohydratesRobin TolentinoNo ratings yet
- Aturan PaulingDocument39 pagesAturan PaulingMeyga Evi Ferama SariNo ratings yet
- RadicalDocument13 pagesRadicalSaharNo ratings yet
- Saturn GC/MS: Quick Reference GuideDocument10 pagesSaturn GC/MS: Quick Reference Guidechemistry_se2636No ratings yet
- Csir Ugc JRF Net Chemistry Paper 1 (Part B) Series - 1Document22 pagesCsir Ugc JRF Net Chemistry Paper 1 (Part B) Series - 1polamrajuNo ratings yet
- Autoxidation of GasolineDocument32 pagesAutoxidation of GasolineNguyễn Ngọc ThụyNo ratings yet
- Carbenoids 2Document15 pagesCarbenoids 2costea0028No ratings yet
- Carbenes and Carbene ComplexesDocument11 pagesCarbenes and Carbene ComplexesAlex CervantesNo ratings yet
- Chapter 5 Alkyl HalidesDocument32 pagesChapter 5 Alkyl HalidesMohd HanafiahNo ratings yet
- Wilkinson CatalystDocument19 pagesWilkinson Catalystjagabandhu_patraNo ratings yet
- Modern Organic Synthesis An IntroductionDocument37 pagesModern Organic Synthesis An IntroductionHanh DuongNo ratings yet
- Properties of SiliconDocument16 pagesProperties of SiliconGabriela ResendeNo ratings yet
- 1 Phase Transfer ReactionsDocument51 pages1 Phase Transfer ReactionsHiren BhendwalNo ratings yet
- CH 10 ReviewDocument5 pagesCH 10 ReviewxryceuNo ratings yet
- mf11 Revision 2Document37 pagesmf11 Revision 2api-250366166No ratings yet
- Pyrolytic Syn EliminationsDocument20 pagesPyrolytic Syn EliminationsSulagna DasNo ratings yet
- Final Exam KeyDocument12 pagesFinal Exam KeykitthiNo ratings yet
- Recent Advances in Olefin Metathesis and Its Application in Organic SynthesisDocument38 pagesRecent Advances in Olefin Metathesis and Its Application in Organic SynthesisTiago Breve da SilvaNo ratings yet
- Polym ReactDocument8 pagesPolym ReactPradeep KumarNo ratings yet
- Book 0chapter 15Document20 pagesBook 0chapter 15Bich Hue NguyenNo ratings yet
- Synthesis of PolymersDocument14 pagesSynthesis of PolymersMohamad AzaniNo ratings yet
- Questions - Answers Bank Class - Xii Subject - Chemistry UNIT-5 (Surface Chemistry)Document6 pagesQuestions - Answers Bank Class - Xii Subject - Chemistry UNIT-5 (Surface Chemistry)Abhay BharadwajNo ratings yet
- Topic 12 - Spectroscopy Revision Notes 1) Infrared SpectrosDocument7 pagesTopic 12 - Spectroscopy Revision Notes 1) Infrared SpectrosChiwe Thando MatutaNo ratings yet
- Synthesis of Drug - 1Document42 pagesSynthesis of Drug - 1'Nurirjawati ElRuri KawangNo ratings yet
- Condensation PolymerizationDocument14 pagesCondensation PolymerizationLester John VeraNo ratings yet
- Preparation of Alkyl Halides, R-X Reaction of Alkanes With CL & BR (F Is Too Reactive, I Is Unreactive)Document20 pagesPreparation of Alkyl Halides, R-X Reaction of Alkanes With CL & BR (F Is Too Reactive, I Is Unreactive)davidtomyNo ratings yet
- 1 IER FundamentalsDocument54 pages1 IER FundamentalsAdam FendrychNo ratings yet
- Reaction MechanismDocument68 pagesReaction MechanismSiddarth Singh73% (11)
- Organometallic TurotialDocument2 pagesOrganometallic TurotialkhemrajmahadewNo ratings yet
- General Information OF Organic Chemistry: Jan 10, 2016 Usamvb Timisoara Facultatea de Medicină Veterinară 1Document23 pagesGeneral Information OF Organic Chemistry: Jan 10, 2016 Usamvb Timisoara Facultatea de Medicină Veterinară 1Flaviu TomuțaNo ratings yet
- Lecture 22Document16 pagesLecture 22imania shaumiNo ratings yet
- The Most Well-Known Rearrangements in Organic Chemistry at HandDocument32 pagesThe Most Well-Known Rearrangements in Organic Chemistry at HandAnkit JagetiaNo ratings yet
- Introduction To Polymer StabilizationDocument59 pagesIntroduction To Polymer Stabilizationvsi100% (1)
- HW 9Document5 pagesHW 9Suryakant Pandey0% (1)
- Cox ReducedDocument149 pagesCox ReducedmcgilicuttyNo ratings yet
- Free RadicalsDocument24 pagesFree RadicalsHALFGHOSTNo ratings yet
- +1 Chemistry - Some Important Questions & Answers PART 2Document12 pages+1 Chemistry - Some Important Questions & Answers PART 2ahammedkayangalkayangalNo ratings yet
- Adv RetrosynthesisDocument29 pagesAdv RetrosynthesisSurya Dewi Wahyuningrum100% (1)
- This PDF Is The Sample PDF Taken From Our Comprehensive Study Material For IIT-JEE Main & AdvancedDocument13 pagesThis PDF Is The Sample PDF Taken From Our Comprehensive Study Material For IIT-JEE Main & AdvancedGod is every whereNo ratings yet
- Oksidasi BiologiDocument62 pagesOksidasi BiologisenadaNo ratings yet
- 11 Chemistry Revision Book 2017 2018 Chapter 12Document20 pages11 Chemistry Revision Book 2017 2018 Chapter 12Thingnam Jotin SinghNo ratings yet
- 11 Chemistry Revision Book 2017 2018 Chapter 12 PDFDocument20 pages11 Chemistry Revision Book 2017 2018 Chapter 12 PDFManoj kumarNo ratings yet
- Heterogeneous CatalysisDocument38 pagesHeterogeneous CatalysisGnanaprakasam ANo ratings yet
- Gaylord 1956Document8 pagesGaylord 1956Robert RicardoNo ratings yet
- Chemistry Sample Paper Class 12: General Instructions: Read The Following Instructions CarefullyDocument31 pagesChemistry Sample Paper Class 12: General Instructions: Read The Following Instructions CarefullyThala ThalapathyNo ratings yet
- ToxicokineticsDocument31 pagesToxicokineticsabdiukon100% (1)
- L. 9-Gas ChromatographyDocument24 pagesL. 9-Gas ChromatographyA. RebelNo ratings yet
- New Frontiers in Sciences, Engineering and the Arts: Vol. Ii the Chemistry of Initiation of Non-Ringed Monomers/CompoundsFrom EverandNew Frontiers in Sciences, Engineering and the Arts: Vol. Ii the Chemistry of Initiation of Non-Ringed Monomers/CompoundsNo ratings yet
- Graphene Oxide: Fundamentals and ApplicationsFrom EverandGraphene Oxide: Fundamentals and ApplicationsAyrat M. DimievNo ratings yet
- New Frontiers in Sciences, Engineering and the Arts: Volume Iii-B: the Chemistry of Initiation of Ringed, Ringed-Forming and Polymeric Monomers/CompoundsFrom EverandNew Frontiers in Sciences, Engineering and the Arts: Volume Iii-B: the Chemistry of Initiation of Ringed, Ringed-Forming and Polymeric Monomers/CompoundsNo ratings yet
- DLS Terms Defined MalvernDocument6 pagesDLS Terms Defined Malverncuongtran_siegenNo ratings yet
- PMMA - SynthesisDocument8 pagesPMMA - Synthesiscuongtran_siegenNo ratings yet
- Heat Conduction CalorimeterDocument7 pagesHeat Conduction Calorimetercuongtran_siegenNo ratings yet
- Lecture 3 Conventional-MicroscopeDocument45 pagesLecture 3 Conventional-Microscopearul1987No ratings yet
- Pmma - 2Document20 pagesPmma - 2cuongtran_siegenNo ratings yet
- PMMADocument7 pagesPMMAcuongtran_siegenNo ratings yet
- MDW QCM Web StuffDocument19 pagesMDW QCM Web Stuffcuongtran_siegenNo ratings yet
- Instructions For Quartz Crystal Microbalance (QCM) : Picture From: HTTP://WWW - Yb.Gmm - Gu.Se/UploadimagesDocument9 pagesInstructions For Quartz Crystal Microbalance (QCM) : Picture From: HTTP://WWW - Yb.Gmm - Gu.Se/Uploadimagescuongtran_siegenNo ratings yet
- Photon Scanning Tunneling Microscope: Detection of Evanescent WavesDocument5 pagesPhoton Scanning Tunneling Microscope: Detection of Evanescent Wavescuongtran_siegenNo ratings yet
- Group Transfer PolymerizationDocument22 pagesGroup Transfer Polymerizationcuongtran_siegenNo ratings yet
- Crosslinking - PDMSDocument139 pagesCrosslinking - PDMScuongtran_siegenNo ratings yet
- Stereochemistry and Synthesis - Exercises 4: H MemgbrDocument2 pagesStereochemistry and Synthesis - Exercises 4: H Memgbrcuongtran_siegenNo ratings yet
- Nitrile Containing Natural ProductsDocument10 pagesNitrile Containing Natural Productscuongtran_siegenNo ratings yet
- Molecular Structure and Adsorption of Dimethyl Sulfoxide at The Surface of Aqueous SolutionsDocument7 pagesMolecular Structure and Adsorption of Dimethyl Sulfoxide at The Surface of Aqueous Solutionsalex.pascu8580No ratings yet
- Polymers SynthesisDocument25 pagesPolymers Synthesisjose juan urbina sanchezNo ratings yet
- Styrene Butadiene RubberDocument9 pagesStyrene Butadiene Rubberdashali1100% (1)
- Molecules: ROMP Synthesis of Iron-Containing Organometallic PolymersDocument16 pagesMolecules: ROMP Synthesis of Iron-Containing Organometallic PolymersBetty WangNo ratings yet
- Styrene-Butadiene Rubber: Kirk-Othmer Encyclopedia of Chemical Technology. John Wiley & Sons, Inc. All Rights ReservedDocument17 pagesStyrene-Butadiene Rubber: Kirk-Othmer Encyclopedia of Chemical Technology. John Wiley & Sons, Inc. All Rights ReservedTHỊ HÀ NGUYỄNNo ratings yet
- Project On PolymerDocument18 pagesProject On Polymermfayaq1100% (1)
- Kiskan 2017Document61 pagesKiskan 2017Siraj AL sharifNo ratings yet
- PLA Synthesis and PolymerizationDocument36 pagesPLA Synthesis and PolymerizationddexterrNo ratings yet
- Synthesis and Characterization of Silicone Modified Acrylic Resin and Its Uses in The Emulsion PaintsDocument13 pagesSynthesis and Characterization of Silicone Modified Acrylic Resin and Its Uses in The Emulsion PaintsfredyNo ratings yet
- PaperDocument48 pagesPaperAnonymous MHKrd6RNo ratings yet
- Molecular Weight: CH CH - CH CH CH - CH CH - CH - CH - CH CH - (CH) CH CH - (CH) CH CH - (CH) CHDocument12 pagesMolecular Weight: CH CH - CH CH CH - CH CH - CH - CH - CH CH - (CH) CH CH - (CH) CH CH - (CH) CHbilal sheikhNo ratings yet
- Suspension PolymerizationDocument29 pagesSuspension PolymerizationHuynh Hoang HuyNo ratings yet
- Solid State PolymerizationDocument28 pagesSolid State PolymerizationDarkLugiaNo ratings yet
- PolymersDocument42 pagesPolymersMuhammad Waqas AkbarNo ratings yet
- Acrylic Binders For Low Voc PaintsDocument31 pagesAcrylic Binders For Low Voc PaintsMaina1206550% (2)
- Sempermed Informs Nr3 Virus Permeability EN 01Document4 pagesSempermed Informs Nr3 Virus Permeability EN 01Asanka SandakelumNo ratings yet
- 3 Rekayasa Reaksi PolimerisasiDocument86 pages3 Rekayasa Reaksi Polimerisasirudy_423522658No ratings yet
- POLYMER TECHNOLOGY SYLLABUS Polytechnic KeralaDocument3 pagesPOLYMER TECHNOLOGY SYLLABUS Polytechnic KeralaprasobhaNo ratings yet
- Role of Oxygen Inhibited Layer On Shear Bond Strength of CompositesDocument4 pagesRole of Oxygen Inhibited Layer On Shear Bond Strength of CompositesAli QawasNo ratings yet
- Acrylamide PolymerizationDocument16 pagesAcrylamide PolymerizationUmakanth Reddy Vudumula100% (1)
- PolymersDocument25 pagesPolymersSai Sasivardhan GampaNo ratings yet
- Macromolecules: Question PaperDocument17 pagesMacromolecules: Question PaperSumitNo ratings yet
- Picadora de RamasDocument4 pagesPicadora de RamasAriel AntonioNo ratings yet
- Resume Murthy Maddipatla-Nov2020 PDFDocument3 pagesResume Murthy Maddipatla-Nov2020 PDFKathy LaneNo ratings yet
- 1.build A Molecule of Ethane, Ethene and Ethyne. 2.build A Molecule of Butane and Then Build An Isomer of Butane (Isobutane)Document28 pages1.build A Molecule of Ethane, Ethene and Ethyne. 2.build A Molecule of Butane and Then Build An Isomer of Butane (Isobutane)Jamilur RahmanNo ratings yet
- Characteristics of RadicalsDocument54 pagesCharacteristics of RadicalsdesyNo ratings yet
- Catalytic Polymerisation of Light Gases Majorly Olefins To Produce Polymer GasolineDocument12 pagesCatalytic Polymerisation of Light Gases Majorly Olefins To Produce Polymer GasolineHadi H. HussenNo ratings yet
- Grade 11 Chemistry Lesson 2Document4 pagesGrade 11 Chemistry Lesson 2Rokeish RoweNo ratings yet
- 3 Polimerisasi HomogenDocument38 pages3 Polimerisasi Homogenutami10No ratings yet