Professional Documents
Culture Documents
HW 1 Key
HW 1 Key
Uploaded by
winadoo87789697Original Description:
Original Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
HW 1 Key
HW 1 Key
Uploaded by
winadoo87789697Copyright:
Available Formats
CHM 380
Fall 2013
Homework #1 Due September 12th
Please answer all questions on additional paper
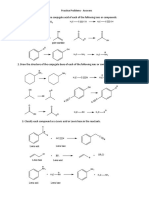
1. For the following acids & bases with the given pKas i) ii) iii) draw the acid and base (protonated and deprotonated) forms and label them use Henderson Hasselbalch to calculate the ratio of the two forms at pH 7.86 calculate the concentration of the base form if the acid form is 0.1 M at pH 7.86. NOTE: The concentration may be extremely large or small. ), pKa = 4.86
A) Propanoic Acid ( a. Acid: Base: a. pH = pKa + log
[CH 3CH 2COO ] [CH 3CH 2COOH ] [CH 3CH 2COO ] [CH 3CH 2COOH ]
7.86 = 4.86 + log
3.00 = log
[CH 3CH 2COO ] [CH 3CH 2COOH ] [CH 3CH 2COO ] [CH 3CH 2COOH ]
[CH 3CH 2COO ] (0.1M )
1.0 x 103 =
b. 1.0 x 103 =
CH3CH2COO- =1.0 x 103 x 0.1 M = 1.0 x 102 M or 100 M
CHM 380
Fall 2013
B) Isobutylamine (
), pKa = 10.56
a. Acid:
+
3
Base: [C4 H 9 NH 2] [C4 H 9 NH 3+ ] [C4 H 9 NH 2] [C4 H 9 NH 3+ ]
b. pH = pKa + log
7.86 = 10.56 + log
-2.70 = log
[C4 H 9 NH 2] [C4 H 9 NH 3+ ] [C4 H 9 NH 2] [C4 H 9 NH 3+ ]
2.0 x 10-3 =
c. 2.0 x 10-3 =
[C4 H 9 NH 2] (0.1M )
C4H9NH2 =2.0 x 10-3 x 0.1 M = 2.0 x 10-4 M 2. Draw hydrogen bonding between the following pairs of molecules. Draw a dashed line from the hydrogen being donated and the hydrogen bond acceptor a. Two water molecules
HOH HOH
CHM 380
Fall 2013
b. Water & 3-Pentanone (
HOH
c. Phenylacetic acid (
) and 1-propylamine (
)(Draw two
possible answers to this question; remember, a carboxylic acid can be a hydrogen bond donor OR acceptor)
H
O CH2
H CH2 CH2 CH3 CH2 O H N CH2 CH2 CH3
OH
OH
3. For the following pairs of compounds, draw the product formed by their acid catalyzed condensation. Identify the category of organic compound the product falls into:
A.
Ester
B. +
Amide (primary)
CHM 380
Fall 2013
C.
Amide (secondary)
D.
+ 1
Hemiacetal
CHM 380
Fall 2013
4.
Identify the following structures as:
A.
Fatty acid Triacylglycerol Steroid B.
Sphingolipid Phospholipid
Steroid
C. D.
Phospholipid
Sphingolipid
E
Triglyceride
Fatty Acid
CHM 380
Fall 2013
5. Draw the structure of the a phosphatidylethanolamine containing oleic acid and lauric acid
oleic acid
lauric acid
H
ethanolamine
CH2 O
-
OH
P O-
O-
CH
OH
CH2
OH
Phosphate
Glycerol
CH2
O O
O O H2N OP O
CH CH2
You might also like
- Acid Base Outside Solution PDFDocument19 pagesAcid Base Outside Solution PDFcsh891129100% (3)
- 7.1 (149 Marks) : MarkschemeDocument51 pages7.1 (149 Marks) : MarkschemeSemwezi Enock100% (1)
- 1aa3 2012 Test1 SolutionsDocument23 pages1aa3 2012 Test1 SolutionsDavidNo ratings yet
- Chapter 24 ProblemsDocument13 pagesChapter 24 Problemslynette-wuNo ratings yet
- Chapter 17 Acids and Bases HWDocument9 pagesChapter 17 Acids and Bases HWAlejo CardoNo ratings yet
- A CIDITYDocument17 pagesA CIDITYApex InstituteNo ratings yet
- Bansal Classes Organic Chemistry Study Material For IIT JEEDocument477 pagesBansal Classes Organic Chemistry Study Material For IIT JEEAditya Kavuluri40% (5)
- Alkyne AllDocument28 pagesAlkyne Allsushantkadam75100% (3)
- The Arrhenius Theory (Section 9.1) 9.1: Solutions To Selected Problems in Chap. 9 & 10Document20 pagesThe Arrhenius Theory (Section 9.1) 9.1: Solutions To Selected Problems in Chap. 9 & 10Riaz LourencoNo ratings yet
- Tutorial 7 PCDocument4 pagesTutorial 7 PCAmylia NatashaNo ratings yet
- 17 Petrucci10e CSMDocument104 pages17 Petrucci10e CSMElah PalaganasNo ratings yet
- Chapter 2 ExrecicesDocument24 pagesChapter 2 Exrecicespaulinhagraebin100% (4)
- CHM 1321 Assignment 5 Answers: 1) Name The Following CompoundsDocument15 pagesCHM 1321 Assignment 5 Answers: 1) Name The Following CompoundsSara Yuen100% (1)
- Chapter 6Document26 pagesChapter 6janNo ratings yet
- ChemDocument6 pagesChembighneshrath1No ratings yet
- Tutorial 6 AlcoholDocument5 pagesTutorial 6 Alcoholwan arifahNo ratings yet
- Chapter 7 Exercises.Document5 pagesChapter 7 Exercises.jimmysroujiNo ratings yet
- WR1.3 Acid-Base Chemistry (Solns)Document3 pagesWR1.3 Acid-Base Chemistry (Solns)Person GainableNo ratings yet
- Tutorial 6 (Alcohols, Aldehydes, Haloalkanes, Carboxylic Acids)Document5 pagesTutorial 6 (Alcohols, Aldehydes, Haloalkanes, Carboxylic Acids)dasani93No ratings yet
- Homework # 2Document1 pageHomework # 2Cong Chinh Bui ThanhNo ratings yet
- XI Mid Term QPDocument3 pagesXI Mid Term QPtechnical SiteNo ratings yet
- Chemistry s5 Theory and Pract.Document29 pagesChemistry s5 Theory and Pract.ngabonzizayusuf9No ratings yet
- Acidicity Basicity & H - Bonding Tautomerism (Q.B.) 12thDocument16 pagesAcidicity Basicity & H - Bonding Tautomerism (Q.B.) 12thAritra Lahiri100% (1)
- Oganic Cem PDFDocument2 pagesOganic Cem PDFaulaNo ratings yet
- AL-CHEM Chemistry of Carbon Compounds (03-06)Document24 pagesAL-CHEM Chemistry of Carbon Compounds (03-06)AmyLinNo ratings yet
- S6 Chemistry: Exam 2Document12 pagesS6 Chemistry: Exam 2Aine VisionNo ratings yet
- Carboxylic Acids and Acid Derivatives Carbonyl PDFDocument124 pagesCarboxylic Acids and Acid Derivatives Carbonyl PDFrvignesh2809No ratings yet
- CHM271 - Tutorial 3 - Ionic EquilibriumDocument3 pagesCHM271 - Tutorial 3 - Ionic Equilibriumfiefy zmrNo ratings yet
- IB-checklist-for-students-HL-Acids Ib Topic 8Document6 pagesIB-checklist-for-students-HL-Acids Ib Topic 8Hana BessalahNo ratings yet
- PS11 S07 SolnDocument5 pagesPS11 S07 SolnJerika ArceoNo ratings yet
- Dokumen.tips Bansal Classes Organic Chemistry Study Material for Iit Jee 1Document267 pagesDokumen.tips Bansal Classes Organic Chemistry Study Material for Iit Jee 1Soumya Ranjan SahooNo ratings yet
- IUPAC Nomenclature 11thDocument4 pagesIUPAC Nomenclature 11thNitya AroraNo ratings yet
- Acids, Bases and Buffers Exercise HA 2021 KeyDocument2 pagesAcids, Bases and Buffers Exercise HA 2021 KeyIsabelle ManaloNo ratings yet
- Test For AcidsDocument2 pagesTest For AcidsBeeNo ratings yet
- F AlkanesAlkenesStereochemTutorial 3Document4 pagesF AlkanesAlkenesStereochemTutorial 3Leong Yue YanNo ratings yet
- Organic Ps Chapter 7Document33 pagesOrganic Ps Chapter 7Mond DamascoNo ratings yet
- CHM 1321 Assignment 1 Answers: CN H H H H HDocument10 pagesCHM 1321 Assignment 1 Answers: CN H H H H HSara YuenNo ratings yet
- HW8 Soln PDFDocument9 pagesHW8 Soln PDFPatricia de Leon100% (1)
- Chemistry 101 Solutions To In-Class Assignment 10 (Due by Nov. 12th) Acids and BasesDocument3 pagesChemistry 101 Solutions To In-Class Assignment 10 (Due by Nov. 12th) Acids and BasesmikecostantiniNo ratings yet
- HW11 F06 KeyDocument6 pagesHW11 F06 KeySOFIA MELENDEZ RODRIGUEZNo ratings yet
- Organic Chemistry 8th Edition Bruice Test Bank download pdf full chapterDocument35 pagesOrganic Chemistry 8th Edition Bruice Test Bank download pdf full chaptermashiiratis100% (8)
- Exercícios Resolvidos - Cap. 10 (Ímpares) - Ácidos e Bases - Princípios de Química - AtkinsDocument40 pagesExercícios Resolvidos - Cap. 10 (Ímpares) - Ácidos e Bases - Princípios de Química - AtkinsJaoJaoNo ratings yet
- Olympiad Questions Acid-Base ChemDocument2 pagesOlympiad Questions Acid-Base ChemGadde Gopala Krishna50% (2)
- Alde & Ket-4Document14 pagesAlde & Ket-4Anusha SinghalNo ratings yet
- Tutorial 9Document3 pagesTutorial 9Vasant KelvinNo ratings yet
- First Exam: PH of 7.3Document11 pagesFirst Exam: PH of 7.3wastequestNo ratings yet
- Problems For Chapter 1 & 2 ANSWERS: 2xH 2 2xN 10 O 6Document6 pagesProblems For Chapter 1 & 2 ANSWERS: 2xH 2 2xN 10 O 6JibrilAttawarahNo ratings yet
- Assignment 1 - Aldehyde and Ketone Mac-Jul 2013Document2 pagesAssignment 1 - Aldehyde and Ketone Mac-Jul 2013anessismanisNo ratings yet
- Chem 2206 Unit 2Document69 pagesChem 2206 Unit 2Danica Rose ZapanzaNo ratings yet
- Answers To ROH Tutorial PDFDocument12 pagesAnswers To ROH Tutorial PDFCorvo Haosen Al-Han0% (1)
- Acids Bases QP - 2Document48 pagesAcids Bases QP - 2wnh8wyq97gNo ratings yet
- Chapter 16 Acid Base Equilibria and Solubility Equilibria PDFDocument120 pagesChapter 16 Acid Base Equilibria and Solubility Equilibria PDFlorrainebarandonNo ratings yet
- Chemistry 1CDocument6 pagesChemistry 1CKaiJie HanNo ratings yet
- Organic Chemistry 7th Edition Bruice Test BankDocument10 pagesOrganic Chemistry 7th Edition Bruice Test Bankmelissa100% (29)
- 2423 e 2Document24 pages2423 e 2Agustin KurniatiNo ratings yet
- Assignment 2 CHEM 215Document6 pagesAssignment 2 CHEM 215Abdullah AlteneijiNo ratings yet
- Gas Hydrates 1: Fundamentals, Characterization and ModelingFrom EverandGas Hydrates 1: Fundamentals, Characterization and ModelingDaniel BrosetaNo ratings yet
- Hyrdogen Storage TechnologiesFrom EverandHyrdogen Storage TechnologiesMehmet SankirNo ratings yet
- Gas Hydrates 2: Geoscience Issues and Potential Industrial ApplicationsFrom EverandGas Hydrates 2: Geoscience Issues and Potential Industrial ApplicationsLivio RuffineNo ratings yet
- Ionic Equilibrium PyqDocument20 pagesIonic Equilibrium Pyqrakeshrenu24127No ratings yet
- ASSIGNMENT3Document9 pagesASSIGNMENT3Eternal MiracleNo ratings yet
- Critical Review AdsorptionDocument111 pagesCritical Review AdsorptionMohamed ElmessoudiNo ratings yet
- Kopecki-Fjetland 2019 Vignette #1: Introducing Active Learning To Improve Student Performance On Threshold Concepts in BiochemistryDocument14 pagesKopecki-Fjetland 2019 Vignette #1: Introducing Active Learning To Improve Student Performance On Threshold Concepts in BiochemistryJavier PNo ratings yet
- The Chemistry of Water: 2.1. Water As A SolventDocument6 pagesThe Chemistry of Water: 2.1. Water As A SolventEva MoonNo ratings yet
- Amino Acid: Navigation SearchDocument11 pagesAmino Acid: Navigation SearchSamiuddin YezdaniNo ratings yet
- Biochem Lab Prelim NotesDocument72 pagesBiochem Lab Prelim NotesMary Mae DequiñaNo ratings yet
- AAMC Sample Test SolutionsDocument53 pagesAAMC Sample Test SolutionsKarina Sanders100% (1)
- Acid BassDocument41 pagesAcid BassRobert Edwards100% (1)
- AminoAcids QPDocument24 pagesAminoAcids QPAnirudh RaoNo ratings yet
- Buffering AgentDocument3 pagesBuffering Agents.sabapathyNo ratings yet
- Master of Science (Chemistry-Physical) Sem-3-4Document15 pagesMaster of Science (Chemistry-Physical) Sem-3-4SudharsananNo ratings yet
- Vinita Katlamudi 20Cs01024 Section 4 Group 1: Ka (H+) (MR) / (HMR) Pka Ph-Log (MR) / (HMR)Document10 pagesVinita Katlamudi 20Cs01024 Section 4 Group 1: Ka (H+) (MR) / (HMR) Pka Ph-Log (MR) / (HMR)VINITA KATLAMUDINo ratings yet
- Revisions MaterialDocument187 pagesRevisions MaterialCarol SoiNo ratings yet
- New Microsoft Word DocumentDocument42 pagesNew Microsoft Word DocumentGnana Deepak JuvvaNo ratings yet
- McKee Chapter5 Sample PDFDocument60 pagesMcKee Chapter5 Sample PDFadane aynalemNo ratings yet
- Test Bank Organic Chemistry 8Th Edition Bruice 013404228X 978013404228 Full Chapter PDFDocument33 pagesTest Bank Organic Chemistry 8Th Edition Bruice 013404228X 978013404228 Full Chapter PDFaaron.dixon139100% (10)
- Chitosan and Aloe Vera: Two Gifts of NatureDocument15 pagesChitosan and Aloe Vera: Two Gifts of NatureEllen Mynelle MabulacNo ratings yet
- Chapter 2 Water: Multiple Choice QuestionsDocument10 pagesChapter 2 Water: Multiple Choice QuestionsFrank WuNo ratings yet
- OrganicChemistry10e PowerPoint Ch02Document69 pagesOrganicChemistry10e PowerPoint Ch02RanilouNo ratings yet
- Chemistry PDFDocument30 pagesChemistry PDFAnanta KhanalNo ratings yet
- Chem 40.1 - Midterms Reviewer Discussion: Buffers: - (Aq) 2 4 - (Aq) 4 2 - (Aq) 2Document5 pagesChem 40.1 - Midterms Reviewer Discussion: Buffers: - (Aq) 2 4 - (Aq) 4 2 - (Aq) 2Steffi GatdulaNo ratings yet
- ASOE Chemistry 2021 Reduced-FS-1Document43 pagesASOE Chemistry 2021 Reduced-FS-1kastonoNo ratings yet
- Waste ProcessDocument86 pagesWaste ProcessacidoanimalNo ratings yet
- Organic Chemistry Week 2 HomeworkDocument6 pagesOrganic Chemistry Week 2 Homeworkplus oneNo ratings yet
- Treatment For Multiple Chem EquilDocument157 pagesTreatment For Multiple Chem EquilEndah SuarsihNo ratings yet
- Tutorial SessionDocument5 pagesTutorial SessionKhánh LinhNo ratings yet
- Acid-Base Balance: George A. Tanner, PH.DDocument22 pagesAcid-Base Balance: George A. Tanner, PH.DDawlat SlamaNo ratings yet
- MOD 5 DDS Manuf PhyspharmDocument33 pagesMOD 5 DDS Manuf PhyspharmTsukishima KeiNo ratings yet