Professional Documents
Culture Documents
0 ratings0% found this document useful (0 votes)
4 viewsGrafik PH Meter
Grafik PH Meter
Uploaded by
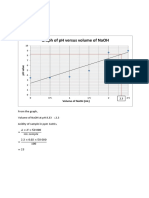
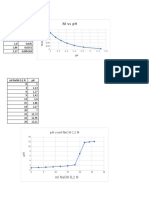
Ratu Nabila AzzahraThis document shows a graph of the relationship between the volume of 0.1 M NaOH solution and pH. The graph lines show that as the volume of NaOH increases, the pH also increases in a nonlinear fashion, with the greatest rise in pH occurring between volumes of 0-20 ml NaOH. Above 20 ml, additional NaOH has a diminishing effect on pH increases.
Copyright:
Attribution Non-Commercial (BY-NC)
Available Formats
Download as DOC, PDF, TXT or read online from Scribd
You might also like
- Phys 1Document5 pagesPhys 1Sandra Phan50% (2)
- Disassociation Constant Estimation Using Acetic Acid and Sodium Hydroxide TitrationDocument19 pagesDisassociation Constant Estimation Using Acetic Acid and Sodium Hydroxide Titrationwani280475% (4)
- Report 2Document10 pagesReport 2Maria Anh Thư0% (1)
- Sample Titration LabDocument11 pagesSample Titration LabJENGABOYNo ratings yet
- Potentiometric Titration of An Acid MixtureDocument5 pagesPotentiometric Titration of An Acid Mixtureabhishek sharmaNo ratings yet
- Titration 1Document19 pagesTitration 1Yvette Malubay AlpuertoNo ratings yet
- Experiment 3 Conductometric Titration: University of Bahrain College of Science Department of ChemistryDocument7 pagesExperiment 3 Conductometric Titration: University of Bahrain College of Science Department of ChemistryFaisal MumtazNo ratings yet
- 1 0 Konsentrasi: HCL HCL 0Document6 pages1 0 Konsentrasi: HCL HCL 0Fina HandayaniNo ratings yet
- Y-Values: A. Making Titration CurveDocument3 pagesY-Values: A. Making Titration CurveFatin AtikahNo ratings yet
- Quantitative Analysis of Phosphoric Acid Found in Commercial Soft Drink "Pool Cola" Via Potentiometric TitrationDocument4 pagesQuantitative Analysis of Phosphoric Acid Found in Commercial Soft Drink "Pool Cola" Via Potentiometric TitrationSofía EscobarNo ratings yet
- Grafik PH Sampel 1 Vs Volume NaohDocument1 pageGrafik PH Sampel 1 Vs Volume Naohnisa methildaNo ratings yet
- 7.2 Acid-Base Titrations 1718Document93 pages7.2 Acid-Base Titrations 1718P YNo ratings yet
- Titration Curves Lab ReportDocument2 pagesTitration Curves Lab Reportapi-584392101No ratings yet
- Chemistry LaboratoryDocument31 pagesChemistry LaboratoryBunty KhiljiNo ratings yet
- Determination of Phosphoric Acid Content in Pepsi Light Soft Drink PDFDocument4 pagesDetermination of Phosphoric Acid Content in Pepsi Light Soft Drink PDFXiomara Galeano100% (1)
- Lab InstrumentalDocument3 pagesLab InstrumentalVictor VerbeeckNo ratings yet
- Lab 1 QADocument3 pagesLab 1 QADionna HooperNo ratings yet
- Module 3 Titrimetric AnalysisDocument3 pagesModule 3 Titrimetric AnalysisNikoh Anthony EwayanNo ratings yet
- Graph of PH Versus Volume of Naoh: Acidity and Alkalinity 1.0 Data 1.1 AcidityDocument3 pagesGraph of PH Versus Volume of Naoh: Acidity and Alkalinity 1.0 Data 1.1 AcidityNasrulNo ratings yet
- PH Meter It's Applications: Practical Advance Pharmaceutical AnalysisDocument19 pagesPH Meter It's Applications: Practical Advance Pharmaceutical AnalysisarjunkumbharchemNo ratings yet
- GraficasDocument3 pagesGraficasfabio bermudezNo ratings yet
- PDF PH TITRATIONDocument16 pagesPDF PH TITRATIONعبدالله هنيةNo ratings yet
- Darimbang - PPT - BSFT - Quantitative Chemistry 10 - Nadzma DarimbangDocument13 pagesDarimbang - PPT - BSFT - Quantitative Chemistry 10 - Nadzma DarimbangLoraine Ruth LumenNo ratings yet
- Phenolphthalein-Naoh KineticsDocument8 pagesPhenolphthalein-Naoh KineticsKamran AliNo ratings yet
- Standardization of Naoh: Tables and ObservationsDocument1 pageStandardization of Naoh: Tables and ObservationsDon CasimeroNo ratings yet
- Haldia Institute of Technology Engineering Chemistry Laboratory (CH 191 &CH 291)Document29 pagesHaldia Institute of Technology Engineering Chemistry Laboratory (CH 191 &CH 291)Shresth Sanskar100% (1)
- Kimi-An - Asidi AlkalimetriDocument69 pagesKimi-An - Asidi Alkalimetrisri rahayuNo ratings yet
- Acids, Bases, and SaltsDocument41 pagesAcids, Bases, and SaltsdelugedcityNo ratings yet
- Experiment 2Document8 pagesExperiment 2Alok VermaNo ratings yet
- ACID - BASE TITRATIONS Determination of Purity of Potassium AcidDocument4 pagesACID - BASE TITRATIONS Determination of Purity of Potassium Acidhilary0622No ratings yet
- Jce 2007 P 0124 WDocument25 pagesJce 2007 P 0124 WAlexaNo ratings yet
- Chem Lab ManualDocument63 pagesChem Lab ManualBala NandaNo ratings yet
- Ch312 Lecture05 Ch9 10 11 w09Document38 pagesCh312 Lecture05 Ch9 10 11 w09Diyu MNo ratings yet
- WjceDocument5 pagesWjceshmohiuddinNo ratings yet
- 022data SheetDocument6 pages022data SheetKIPNGENO EMMANUELNo ratings yet
- Experi 3 Acid Base TitrationDocument4 pagesExperi 3 Acid Base TitrationOromay EliasNo ratings yet
- Naoh Vol. Buret Max. Value Ph1 ### Ph2 Ph3 Ph4 - 1.65 HCL Va 5.9 ML Ma 0.06 M Naoh VB 10 ML MB 0 M KWDocument4 pagesNaoh Vol. Buret Max. Value Ph1 ### Ph2 Ph3 Ph4 - 1.65 HCL Va 5.9 ML Ma 0.06 M Naoh VB 10 ML MB 0 M KWafra afifahNo ratings yet
- Report Form PE1Document10 pagesReport Form PE1st19000941No ratings yet
- Soft DrinksDocument3 pagesSoft DrinkskiiieeeshieeeNo ratings yet
- 20L Weak Acid Analysis GraphsDocument4 pages20L Weak Acid Analysis GraphsEdison KendrickNo ratings yet
- Lab 4 - Soda Titrations 1Document9 pagesLab 4 - Soda Titrations 1api-385516219No ratings yet
- Acids, Bases, and PH: Review ExercisesDocument10 pagesAcids, Bases, and PH: Review ExercisesmihtautNo ratings yet
- SPECTROPHOTOMETRIC DETERMINATION OF AN ACID DISSOCIATION CONSTANT - EditedDocument5 pagesSPECTROPHOTOMETRIC DETERMINATION OF AN ACID DISSOCIATION CONSTANT - Editedsindiswamngadi076No ratings yet
- Exp8-Phenolphthalein NaOH KineticsDocument7 pagesExp8-Phenolphthalein NaOH KineticsKamran AliNo ratings yet
- Titration Curves ReportDocument26 pagesTitration Curves ReportMa-anJaneDiamosNo ratings yet
- Titration Curves: Chem 18.1 Experiment 8Document26 pagesTitration Curves: Chem 18.1 Experiment 8Ghadeer M HassanNo ratings yet
- PH and Buffer LabDocument13 pagesPH and Buffer LabAdellaine Lois GreyNo ratings yet
- General Chemistry Laboratory Report: Experiment 2 PH and BuffersDocument11 pagesGeneral Chemistry Laboratory Report: Experiment 2 PH and BuffersBùi Nhật MaiNo ratings yet
- SUBJECT1Document7 pagesSUBJECT1Alexander DemongNo ratings yet
- SUBJECT1Document7 pagesSUBJECT1Alexander DemongNo ratings yet
- Abstract #5Document3 pagesAbstract #5Christian Deleg100% (1)
- FST556 Preparation and Standardisation of Base and Acid SolutionDocument6 pagesFST556 Preparation and Standardisation of Base and Acid SolutionNurizzatiainiNo ratings yet
- Acid Base Titration Lab ReportDocument10 pagesAcid Base Titration Lab ReportSiti Rania Norazli ShamNo ratings yet
- CC NGSS Phosphoric Acid LQDocument4 pagesCC NGSS Phosphoric Acid LQleonmcgrgrNo ratings yet
- TItrasi Potensiometri PH MetriDocument19 pagesTItrasi Potensiometri PH MetriNabila Putri SabilaNo ratings yet
- PH of Buffers Hydrolysed SaltsDocument27 pagesPH of Buffers Hydrolysed SaltsFmipa Khaeru NisaaNo ratings yet
- Chapter 14 Principle of Neutralization TDocument29 pagesChapter 14 Principle of Neutralization TS. MartinezNo ratings yet
- ChemistryDocument14 pagesChemistryKishore KishoreNo ratings yet
- Report 2Document13 pagesReport 2Linh Giao Nguyễn TrầnNo ratings yet
- Grafik Distilasi VakumDocument5 pagesGrafik Distilasi VakumRatu Nabila AzzahraNo ratings yet
- Cover PQ Ruang Filling Syrup IIDocument1 pageCover PQ Ruang Filling Syrup IIRatu Nabila AzzahraNo ratings yet
- Grafik Dtilasi FraksionasiDocument4 pagesGrafik Dtilasi FraksionasiRatu Nabila AzzahraNo ratings yet
- Sampul AskefDocument2 pagesSampul AskefRatu Nabila AzzahraNo ratings yet
- Grafik Ekstraksi Cair - Cair - ExcelDocument2 pagesGrafik Ekstraksi Cair - Cair - ExcelRatu Nabila AzzahraNo ratings yet
- Tabel HE PlateDocument3 pagesTabel HE PlateRatu Nabila AzzahraNo ratings yet
- PolariDocument1 pagePolariRatu Nabila AzzahraNo ratings yet
- Konsentr Asi (PPM) Serapan 0.2 0.195 0.4 0.346 0.6 0.561 0.8 0.826Document2 pagesKonsentr Asi (PPM) Serapan 0.2 0.195 0.4 0.346 0.6 0.561 0.8 0.826Ratu Nabila AzzahraNo ratings yet
Grafik PH Meter
Grafik PH Meter
Uploaded by
Ratu Nabila Azzahra0 ratings0% found this document useful (0 votes)
4 views3 pagesThis document shows a graph of the relationship between the volume of 0.1 M NaOH solution and pH. The graph lines show that as the volume of NaOH increases, the pH also increases in a nonlinear fashion, with the greatest rise in pH occurring between volumes of 0-20 ml NaOH. Above 20 ml, additional NaOH has a diminishing effect on pH increases.
Original Description:
DUH65U56U76
Original Title
Grafik pH Meter
Copyright
© Attribution Non-Commercial (BY-NC)
Available Formats
DOC, PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentThis document shows a graph of the relationship between the volume of 0.1 M NaOH solution and pH. The graph lines show that as the volume of NaOH increases, the pH also increases in a nonlinear fashion, with the greatest rise in pH occurring between volumes of 0-20 ml NaOH. Above 20 ml, additional NaOH has a diminishing effect on pH increases.
Copyright:
Attribution Non-Commercial (BY-NC)
Available Formats
Download as DOC, PDF, TXT or read online from Scribd
Download as doc, pdf, or txt
0 ratings0% found this document useful (0 votes)
4 views3 pagesGrafik PH Meter
Grafik PH Meter
Uploaded by
Ratu Nabila AzzahraThis document shows a graph of the relationship between the volume of 0.1 M NaOH solution and pH. The graph lines show that as the volume of NaOH increases, the pH also increases in a nonlinear fashion, with the greatest rise in pH occurring between volumes of 0-20 ml NaOH. Above 20 ml, additional NaOH has a diminishing effect on pH increases.
Copyright:
Attribution Non-Commercial (BY-NC)
Available Formats
Download as DOC, PDF, TXT or read online from Scribd
Download as doc, pdf, or txt
You are on page 1of 3
Kurva Hubungan Volume NaOH Vs pH
13
12
11
10
9
pH
8
7
6
5
4
3
2
1
0
0
10
20
30
Volume NaOH 0,1 M
40
50
pH
Kurva Hubungan volume NaOH Vs pH
13
12
11
10
9
8
7
6
5
4
3
2
1
0
0
10
20
30
40
Volume NaOH 0,1 M (ml)
50
60
You might also like
- Phys 1Document5 pagesPhys 1Sandra Phan50% (2)
- Disassociation Constant Estimation Using Acetic Acid and Sodium Hydroxide TitrationDocument19 pagesDisassociation Constant Estimation Using Acetic Acid and Sodium Hydroxide Titrationwani280475% (4)
- Report 2Document10 pagesReport 2Maria Anh Thư0% (1)
- Sample Titration LabDocument11 pagesSample Titration LabJENGABOYNo ratings yet
- Potentiometric Titration of An Acid MixtureDocument5 pagesPotentiometric Titration of An Acid Mixtureabhishek sharmaNo ratings yet
- Titration 1Document19 pagesTitration 1Yvette Malubay AlpuertoNo ratings yet
- Experiment 3 Conductometric Titration: University of Bahrain College of Science Department of ChemistryDocument7 pagesExperiment 3 Conductometric Titration: University of Bahrain College of Science Department of ChemistryFaisal MumtazNo ratings yet
- 1 0 Konsentrasi: HCL HCL 0Document6 pages1 0 Konsentrasi: HCL HCL 0Fina HandayaniNo ratings yet
- Y-Values: A. Making Titration CurveDocument3 pagesY-Values: A. Making Titration CurveFatin AtikahNo ratings yet
- Quantitative Analysis of Phosphoric Acid Found in Commercial Soft Drink "Pool Cola" Via Potentiometric TitrationDocument4 pagesQuantitative Analysis of Phosphoric Acid Found in Commercial Soft Drink "Pool Cola" Via Potentiometric TitrationSofía EscobarNo ratings yet
- Grafik PH Sampel 1 Vs Volume NaohDocument1 pageGrafik PH Sampel 1 Vs Volume Naohnisa methildaNo ratings yet
- 7.2 Acid-Base Titrations 1718Document93 pages7.2 Acid-Base Titrations 1718P YNo ratings yet
- Titration Curves Lab ReportDocument2 pagesTitration Curves Lab Reportapi-584392101No ratings yet
- Chemistry LaboratoryDocument31 pagesChemistry LaboratoryBunty KhiljiNo ratings yet
- Determination of Phosphoric Acid Content in Pepsi Light Soft Drink PDFDocument4 pagesDetermination of Phosphoric Acid Content in Pepsi Light Soft Drink PDFXiomara Galeano100% (1)
- Lab InstrumentalDocument3 pagesLab InstrumentalVictor VerbeeckNo ratings yet
- Lab 1 QADocument3 pagesLab 1 QADionna HooperNo ratings yet
- Module 3 Titrimetric AnalysisDocument3 pagesModule 3 Titrimetric AnalysisNikoh Anthony EwayanNo ratings yet
- Graph of PH Versus Volume of Naoh: Acidity and Alkalinity 1.0 Data 1.1 AcidityDocument3 pagesGraph of PH Versus Volume of Naoh: Acidity and Alkalinity 1.0 Data 1.1 AcidityNasrulNo ratings yet
- PH Meter It's Applications: Practical Advance Pharmaceutical AnalysisDocument19 pagesPH Meter It's Applications: Practical Advance Pharmaceutical AnalysisarjunkumbharchemNo ratings yet
- GraficasDocument3 pagesGraficasfabio bermudezNo ratings yet
- PDF PH TITRATIONDocument16 pagesPDF PH TITRATIONعبدالله هنيةNo ratings yet
- Darimbang - PPT - BSFT - Quantitative Chemistry 10 - Nadzma DarimbangDocument13 pagesDarimbang - PPT - BSFT - Quantitative Chemistry 10 - Nadzma DarimbangLoraine Ruth LumenNo ratings yet
- Phenolphthalein-Naoh KineticsDocument8 pagesPhenolphthalein-Naoh KineticsKamran AliNo ratings yet
- Standardization of Naoh: Tables and ObservationsDocument1 pageStandardization of Naoh: Tables and ObservationsDon CasimeroNo ratings yet
- Haldia Institute of Technology Engineering Chemistry Laboratory (CH 191 &CH 291)Document29 pagesHaldia Institute of Technology Engineering Chemistry Laboratory (CH 191 &CH 291)Shresth Sanskar100% (1)
- Kimi-An - Asidi AlkalimetriDocument69 pagesKimi-An - Asidi Alkalimetrisri rahayuNo ratings yet
- Acids, Bases, and SaltsDocument41 pagesAcids, Bases, and SaltsdelugedcityNo ratings yet
- Experiment 2Document8 pagesExperiment 2Alok VermaNo ratings yet
- ACID - BASE TITRATIONS Determination of Purity of Potassium AcidDocument4 pagesACID - BASE TITRATIONS Determination of Purity of Potassium Acidhilary0622No ratings yet
- Jce 2007 P 0124 WDocument25 pagesJce 2007 P 0124 WAlexaNo ratings yet
- Chem Lab ManualDocument63 pagesChem Lab ManualBala NandaNo ratings yet
- Ch312 Lecture05 Ch9 10 11 w09Document38 pagesCh312 Lecture05 Ch9 10 11 w09Diyu MNo ratings yet
- WjceDocument5 pagesWjceshmohiuddinNo ratings yet
- 022data SheetDocument6 pages022data SheetKIPNGENO EMMANUELNo ratings yet
- Experi 3 Acid Base TitrationDocument4 pagesExperi 3 Acid Base TitrationOromay EliasNo ratings yet
- Naoh Vol. Buret Max. Value Ph1 ### Ph2 Ph3 Ph4 - 1.65 HCL Va 5.9 ML Ma 0.06 M Naoh VB 10 ML MB 0 M KWDocument4 pagesNaoh Vol. Buret Max. Value Ph1 ### Ph2 Ph3 Ph4 - 1.65 HCL Va 5.9 ML Ma 0.06 M Naoh VB 10 ML MB 0 M KWafra afifahNo ratings yet
- Report Form PE1Document10 pagesReport Form PE1st19000941No ratings yet
- Soft DrinksDocument3 pagesSoft DrinkskiiieeeshieeeNo ratings yet
- 20L Weak Acid Analysis GraphsDocument4 pages20L Weak Acid Analysis GraphsEdison KendrickNo ratings yet
- Lab 4 - Soda Titrations 1Document9 pagesLab 4 - Soda Titrations 1api-385516219No ratings yet
- Acids, Bases, and PH: Review ExercisesDocument10 pagesAcids, Bases, and PH: Review ExercisesmihtautNo ratings yet
- SPECTROPHOTOMETRIC DETERMINATION OF AN ACID DISSOCIATION CONSTANT - EditedDocument5 pagesSPECTROPHOTOMETRIC DETERMINATION OF AN ACID DISSOCIATION CONSTANT - Editedsindiswamngadi076No ratings yet
- Exp8-Phenolphthalein NaOH KineticsDocument7 pagesExp8-Phenolphthalein NaOH KineticsKamran AliNo ratings yet
- Titration Curves ReportDocument26 pagesTitration Curves ReportMa-anJaneDiamosNo ratings yet
- Titration Curves: Chem 18.1 Experiment 8Document26 pagesTitration Curves: Chem 18.1 Experiment 8Ghadeer M HassanNo ratings yet
- PH and Buffer LabDocument13 pagesPH and Buffer LabAdellaine Lois GreyNo ratings yet
- General Chemistry Laboratory Report: Experiment 2 PH and BuffersDocument11 pagesGeneral Chemistry Laboratory Report: Experiment 2 PH and BuffersBùi Nhật MaiNo ratings yet
- SUBJECT1Document7 pagesSUBJECT1Alexander DemongNo ratings yet
- SUBJECT1Document7 pagesSUBJECT1Alexander DemongNo ratings yet
- Abstract #5Document3 pagesAbstract #5Christian Deleg100% (1)
- FST556 Preparation and Standardisation of Base and Acid SolutionDocument6 pagesFST556 Preparation and Standardisation of Base and Acid SolutionNurizzatiainiNo ratings yet
- Acid Base Titration Lab ReportDocument10 pagesAcid Base Titration Lab ReportSiti Rania Norazli ShamNo ratings yet
- CC NGSS Phosphoric Acid LQDocument4 pagesCC NGSS Phosphoric Acid LQleonmcgrgrNo ratings yet
- TItrasi Potensiometri PH MetriDocument19 pagesTItrasi Potensiometri PH MetriNabila Putri SabilaNo ratings yet
- PH of Buffers Hydrolysed SaltsDocument27 pagesPH of Buffers Hydrolysed SaltsFmipa Khaeru NisaaNo ratings yet
- Chapter 14 Principle of Neutralization TDocument29 pagesChapter 14 Principle of Neutralization TS. MartinezNo ratings yet
- ChemistryDocument14 pagesChemistryKishore KishoreNo ratings yet
- Report 2Document13 pagesReport 2Linh Giao Nguyễn TrầnNo ratings yet
- Grafik Distilasi VakumDocument5 pagesGrafik Distilasi VakumRatu Nabila AzzahraNo ratings yet
- Cover PQ Ruang Filling Syrup IIDocument1 pageCover PQ Ruang Filling Syrup IIRatu Nabila AzzahraNo ratings yet
- Grafik Dtilasi FraksionasiDocument4 pagesGrafik Dtilasi FraksionasiRatu Nabila AzzahraNo ratings yet
- Sampul AskefDocument2 pagesSampul AskefRatu Nabila AzzahraNo ratings yet
- Grafik Ekstraksi Cair - Cair - ExcelDocument2 pagesGrafik Ekstraksi Cair - Cair - ExcelRatu Nabila AzzahraNo ratings yet
- Tabel HE PlateDocument3 pagesTabel HE PlateRatu Nabila AzzahraNo ratings yet
- PolariDocument1 pagePolariRatu Nabila AzzahraNo ratings yet
- Konsentr Asi (PPM) Serapan 0.2 0.195 0.4 0.346 0.6 0.561 0.8 0.826Document2 pagesKonsentr Asi (PPM) Serapan 0.2 0.195 0.4 0.346 0.6 0.561 0.8 0.826Ratu Nabila AzzahraNo ratings yet