Professional Documents
Culture Documents
Estandarización: DPH / DV Vs Volumen Naoh
Estandarización: DPH / DV Vs Volumen Naoh
Uploaded by
Eduardo Camarena Sullca0 ratings0% found this document useful (0 votes)
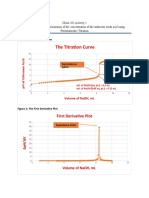
11 views1 pageThis graph shows the change in pH (dpH/dV) of a solution as increasing volumes of sodium hydroxide (NaOH) are added. The dpH/dV decreases rapidly with small additions of NaOH at first, then levels off and approaches zero as more NaOH is added, indicating the solution has reached its equivalence point and maximum pH change.
Original Description:
1V-dpH (std)
1V-dpH (std)
1V-dpH (std)
Original Title
1V-dpH (std)
Copyright
© © All Rights Reserved
Available Formats
DOCX, PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentThis graph shows the change in pH (dpH/dV) of a solution as increasing volumes of sodium hydroxide (NaOH) are added. The dpH/dV decreases rapidly with small additions of NaOH at first, then levels off and approaches zero as more NaOH is added, indicating the solution has reached its equivalence point and maximum pH change.
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
Download as docx, pdf, or txt
0 ratings0% found this document useful (0 votes)
11 views1 pageEstandarización: DPH / DV Vs Volumen Naoh
Estandarización: DPH / DV Vs Volumen Naoh
Uploaded by
Eduardo Camarena SullcaThis graph shows the change in pH (dpH/dV) of a solution as increasing volumes of sodium hydroxide (NaOH) are added. The dpH/dV decreases rapidly with small additions of NaOH at first, then levels off and approaches zero as more NaOH is added, indicating the solution has reached its equivalence point and maximum pH change.
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
Download as docx, pdf, or txt
You are on page 1of 1
Estandarizacin: dpH / dV vs Volumen NaOH
5.000
4.500
4.000
3.500
dpH / dV
3.000
2.500
2.000
1.500
1.000
0.500
0.000
0
10
15
Volumen (mL)
20
25
You might also like
- Student Exploration: TitrationDocument7 pagesStudent Exploration: TitrationMF - 11AK 827776 Central Peel SSNo ratings yet
- Act 3 Lab ReportDocument6 pagesAct 3 Lab ReportHassan ZakariaNo ratings yet
- Sample Problem #22Document8 pagesSample Problem #22Dozdi0% (1)
- Determination of Sodium Hypophosphite PDFDocument3 pagesDetermination of Sodium Hypophosphite PDFHans TorresNo ratings yet
- Experiment 3 Conductometric Titration: University of Bahrain College of Science Department of ChemistryDocument7 pagesExperiment 3 Conductometric Titration: University of Bahrain College of Science Department of ChemistryFaisal MumtazNo ratings yet
- Colligative Properties 1Document19 pagesColligative Properties 1Jie Ann Faith AusmoloNo ratings yet
- SolutionDocument3 pagesSolutionbriefcinemablitzNo ratings yet
- Colligative Properties 1Document21 pagesColligative Properties 1Jie Ann Faith AusmoloNo ratings yet
- Determination of The Acid Concentration in JuiceDocument4 pagesDetermination of The Acid Concentration in Juiceapi-642164099No ratings yet
- Kurva Titrasi PH Vs Volume Penambahan Naoh Kurva Titrasi PH Vs Volume Penambahan NaohDocument1 pageKurva Titrasi PH Vs Volume Penambahan Naoh Kurva Titrasi PH Vs Volume Penambahan NaohMaulinda Imansari ArmaytabinoyaNo ratings yet
- Lab 4 - Soda Titrations 1Document9 pagesLab 4 - Soda Titrations 1api-385516219No ratings yet
- Chap15 16 Ana Chem. Report22222222222222222Document15 pagesChap15 16 Ana Chem. Report22222222222222222Ian Rey AbellaNo ratings yet
- SolutionsDocument38 pagesSolutionsakshaysinghofficial007No ratings yet
- Experiment No. 4: Adamson University College of Engineering Chemical Engineering DepartmentDocument10 pagesExperiment No. 4: Adamson University College of Engineering Chemical Engineering DepartmentRon PascualNo ratings yet
- Titration SeDocument9 pagesTitration Ses2100016No ratings yet
- Monographie Sucrose - British PharmacopoeiaDocument5 pagesMonographie Sucrose - British Pharmacopoeiaasmae.labindusNo ratings yet
- Quantitative Analysis of Phosphoric Acid Found in Commercial Soft Drink "Pool Cola" Via Potentiometric TitrationDocument4 pagesQuantitative Analysis of Phosphoric Acid Found in Commercial Soft Drink "Pool Cola" Via Potentiometric TitrationSofía EscobarNo ratings yet
- Potentiometric Determination of Phosphor PDFDocument7 pagesPotentiometric Determination of Phosphor PDFFlex GodNo ratings yet
- Potentiometric Determination of Phosphoric Acid in Unknown SampleDocument7 pagesPotentiometric Determination of Phosphoric Acid in Unknown SamplekahullanyNo ratings yet
- Determination of The Molecular Weight of A Non-Volatile Solid by The Cryoscopic MethodDocument13 pagesDetermination of The Molecular Weight of A Non-Volatile Solid by The Cryoscopic Methodariana religiosoNo ratings yet
- Determining The Concentration of NaOH Solution.Document6 pagesDetermining The Concentration of NaOH Solution.Ck WongNo ratings yet
- Experiment 12 Lab ReportDocument3 pagesExperiment 12 Lab Reporttehani541No ratings yet
- Grafik Hubungan Antara PH CH Cooh Dengan Volume Total NaohDocument4 pagesGrafik Hubungan Antara PH CH Cooh Dengan Volume Total NaohAji SaputraNo ratings yet
- Atmospheric Thermodynamics ProblemsDocument5 pagesAtmospheric Thermodynamics Problemssathish22No ratings yet
- Densidad IR IRo European Pharmacopoeia 8 0Document3 pagesDensidad IR IRo European Pharmacopoeia 8 0Marco AronesNo ratings yet
- CH 11Document38 pagesCH 11Chala1989No ratings yet
- Ebook Chemistry An Atoms First Approach 2Nd Edition Zumdahl Solutions Manual Full Chapter PDFDocument62 pagesEbook Chemistry An Atoms First Approach 2Nd Edition Zumdahl Solutions Manual Full Chapter PDFthuygladys5x0100% (14)
- Solutions Manual Chapter11Document37 pagesSolutions Manual Chapter11zemr6No ratings yet
- Vnaohvsph: Analisis Data 1. Tabel Volume Naoh Vs PHDocument3 pagesVnaohvsph: Analisis Data 1. Tabel Volume Naoh Vs PHulul dwi yuliandaNo ratings yet
- Instant Download PDF Chemistry An Atoms First Approach 2nd Edition Zumdahl Solutions Manual Full ChapterDocument64 pagesInstant Download PDF Chemistry An Atoms First Approach 2nd Edition Zumdahl Solutions Manual Full Chaptermvondogxagxa4100% (6)
- BuffersDocument3 pagesBuffersIshak Ika Kovac100% (1)
- Experiment No 1Document3 pagesExperiment No 1NEHA BANSALNo ratings yet
- Bits PilaniDocument30 pagesBits PilaniVenkata Bhaskar DameraNo ratings yet
- Potentiometric Titration of A Weak Acid: Chemistry 135 Clark CollegeDocument14 pagesPotentiometric Titration of A Weak Acid: Chemistry 135 Clark CollegeMay LeeNo ratings yet
- Acid Base Titration Lab ReportDocument10 pagesAcid Base Titration Lab ReportSiti Rania Norazli ShamNo ratings yet
- PH in Blood Experimental Model of A Blood Buffer and Its BufDocument5 pagesPH in Blood Experimental Model of A Blood Buffer and Its Bufbashar.adamat101No ratings yet
- Lab 1. ChemistryDocument3 pagesLab 1. ChemistryFame MoosNo ratings yet
- TSSM Topic 2Document13 pagesTSSM Topic 2sudotesterNo ratings yet
- Lab Experiment - Titration of Sulfuric Acid (H2SO4)Document2 pagesLab Experiment - Titration of Sulfuric Acid (H2SO4)gdfeiu dionwdnNo ratings yet
- Data SheetDocument5 pagesData Sheetkyle javierNo ratings yet
- Labsheet No.1: Kashif Husaain Sp-18-Bschem-004Document6 pagesLabsheet No.1: Kashif Husaain Sp-18-Bschem-004Muyammad Ahsan abbasNo ratings yet
- Quantitative Determination of Potassium Acid Phthalate KHPDocument17 pagesQuantitative Determination of Potassium Acid Phthalate KHPMichelle Cruz AbrilNo ratings yet
- Acid Rain IIDocument3 pagesAcid Rain IIMaxWittNo ratings yet
- Prac-8 - Colligative PropertiesDocument15 pagesPrac-8 - Colligative PropertiesCristiano PassarelliNo ratings yet
- 1 Theory: Buffers and Buffer CapacityDocument5 pages1 Theory: Buffers and Buffer Capacitygrim_ripperNo ratings yet
- Differential Index of Refraction, DN/DCDocument6 pagesDifferential Index of Refraction, DN/DCblablubgaloreNo ratings yet
- Changes in Vapour Pressure. (Vapour Pressure Lowering) : V - P Depends Only On The SolventDocument5 pagesChanges in Vapour Pressure. (Vapour Pressure Lowering) : V - P Depends Only On The SolventMarthy DayagNo ratings yet
- Larutan: Isana SYL Kimia - UNYDocument21 pagesLarutan: Isana SYL Kimia - UNYroza maiyarniNo ratings yet
- Diffusion in LiquidsDocument3 pagesDiffusion in LiquidsAdy FathNo ratings yet
- Intro Titration Mini-Lab: Objective BackgroundDocument3 pagesIntro Titration Mini-Lab: Objective BackgroundKhuslenNo ratings yet
- Expressions of ConcentrationsDocument36 pagesExpressions of Concentrationsgycht672qdNo ratings yet
- Jce 2007 P 0124 WDocument25 pagesJce 2007 P 0124 WAlexaNo ratings yet
- Buffer Index and Buffer Capacity For A Simple Buffer SolutionDocument3 pagesBuffer Index and Buffer Capacity For A Simple Buffer SolutionEduardo CastroNo ratings yet
- Solutions Class 12 Important Questions CBSE PDFDocument36 pagesSolutions Class 12 Important Questions CBSE PDFprathibha seenirajNo ratings yet
- Potentiometric Titration of An Acid MixtureDocument5 pagesPotentiometric Titration of An Acid Mixtureabhishek sharmaNo ratings yet
- Practica 2: Valoraciones: Estandarización y Titulación de Soluciones Acido-Base AutoresDocument5 pagesPractica 2: Valoraciones: Estandarización y Titulación de Soluciones Acido-Base AutoresDaniela Sanchez VargasNo ratings yet
- Sample Titration LabDocument11 pagesSample Titration LabJENGABOYNo ratings yet
- ML Naoh PH PH/ V PH/ VDocument3 pagesML Naoh PH PH/ V PH/ VLusiNo ratings yet
- Tit RationDocument7 pagesTit RationgautamahujaNo ratings yet
- CHY 47.1 Procedure Factors Affecting Buffers Capacity 1st Sem 2021-2022Document5 pagesCHY 47.1 Procedure Factors Affecting Buffers Capacity 1st Sem 2021-2022Kathryne May JinonNo ratings yet
- APG RiskBasedThinking2015Document3 pagesAPG RiskBasedThinking2015Luis LuceroNo ratings yet
- Uncertainty PDFDocument6 pagesUncertainty PDFNguyen Duc ToanNo ratings yet
- Expansion, Contraction, Pressure Drop PDFDocument3 pagesExpansion, Contraction, Pressure Drop PDFBubai111No ratings yet
- Tutorial ChemsepDocument4 pagesTutorial ChemsepEduardo Camarena SullcaNo ratings yet