Professional Documents
Culture Documents
Páginas DesdeProperty Description of Thermodynamic Package
Páginas DesdeProperty Description of Thermodynamic Package
Uploaded by
nestorsoft0 ratings0% found this document useful (0 votes)
24 views1 pageEquation of State models have proven to be very reliable in predicting the properties of most hydrocarbon based fluids over a wide range of operating conditions. The GCEOS model allows you to define and implement your own generalized cubic Equation of State including mixing rules and volume translation. The Kabadi Danner model is a modification of the original SRK equation of state, enhanced to improve the vapour-liquid-liquid equilibria calculations for waterhydrocarbon systems.
Original Description:
Copyright
© © All Rights Reserved
Available Formats
PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentEquation of State models have proven to be very reliable in predicting the properties of most hydrocarbon based fluids over a wide range of operating conditions. The GCEOS model allows you to define and implement your own generalized cubic Equation of State including mixing rules and volume translation. The Kabadi Danner model is a modification of the original SRK equation of state, enhanced to improve the vapour-liquid-liquid equilibria calculations for waterhydrocarbon systems.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
Download as pdf or txt
0 ratings0% found this document useful (0 votes)
24 views1 pagePáginas DesdeProperty Description of Thermodynamic Package
Páginas DesdeProperty Description of Thermodynamic Package
Uploaded by
nestorsoftEquation of State models have proven to be very reliable in predicting the properties of most hydrocarbon based fluids over a wide range of operating conditions. The GCEOS model allows you to define and implement your own generalized cubic Equation of State including mixing rules and volume translation. The Kabadi Danner model is a modification of the original SRK equation of state, enhanced to improve the vapour-liquid-liquid equilibria calculations for waterhydrocarbon systems.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
Download as pdf or txt
You are on page 1of 1
1
Property Package Descriptions
Equations of State (EOS)
Equation of State models have proven to be very reliable in predicting the properties of
most hydrocarbon based fluids over a wide range of operating conditions. Their
application focuses on primarily non-polar or slightly polar components.
GCEOS
The GCEOS model allows you to define and implement your own generalized cubic
equation of state including mixing rules and volume translation.
Kabadi Danner
The Kabadi Danner model is a modification of the original SRK equation of state,
enhanced to improve the vapour-liquid-liquid equilibria calculations for water-
hydrocarbon systems, particularly in dilute regions.
The model is an improvement over previous attempts which were limited in the region of
validity. The modification is based on an asymmetric mixing rule, whereby the
interaction in the water phase (with its strong H
2
bonding) is calculated based on both the
interaction between the hydrocarbons and the H
2
O, and on the perturbation by
hydrocarbon on the H
2
O-H
2
O interaction (due to its structure).
The Kabadi Danner model uses the Kabadi Danner method to calculate VLE and uses
SRK to calculate Enthalphy and Entropy.
Lee-Kessler Plocker
The Lee-Kesler Plocker model is the most accurate general method for non-polar
substances and mixtures and is recommended for Ethylene Towers. LKP uses the Lee-
Kesler-Plocker method to calculate VLE and uses the Lee Kesler method to calculate
Enthalpy and Entropy.
Plcker applied the Lee Kesler equation to mixtures, which itself was modified from the
BWR equation.
Peng-Robinson
The Peng-Robinson (PR) model is ideal for VLE calculations as well as calculating liquid
densities for hydrocarbon systems. Several enhancements to the original PR model were
made to extend its range of applicability and to improve its predictions for some non-
ideal systems. However, in situations where highly non-ideal systems are encountered,
the use of Activity Models is recommended.
The PR property package rigorously solves any single-, two-, or three-phase system with
a high degree of efficiency and reliability and is applicable over a wide range of
conditions:
Temperature Range >-271C or -456F
Pressure Range <100,000 kPa or 15,000 psia
You might also like
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeFrom EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeRating: 4 out of 5 stars4/5 (5822)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreFrom EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreRating: 4 out of 5 stars4/5 (1093)
- Never Split the Difference: Negotiating As If Your Life Depended On ItFrom EverandNever Split the Difference: Negotiating As If Your Life Depended On ItRating: 4.5 out of 5 stars4.5/5 (852)
- Grit: The Power of Passion and PerseveranceFrom EverandGrit: The Power of Passion and PerseveranceRating: 4 out of 5 stars4/5 (590)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceFrom EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceRating: 4 out of 5 stars4/5 (898)
- Shoe Dog: A Memoir by the Creator of NikeFrom EverandShoe Dog: A Memoir by the Creator of NikeRating: 4.5 out of 5 stars4.5/5 (540)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersFrom EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersRating: 4.5 out of 5 stars4.5/5 (349)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureFrom EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureRating: 4.5 out of 5 stars4.5/5 (474)
- Her Body and Other Parties: StoriesFrom EverandHer Body and Other Parties: StoriesRating: 4 out of 5 stars4/5 (823)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)From EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Rating: 4.5 out of 5 stars4.5/5 (122)
- The Emperor of All Maladies: A Biography of CancerFrom EverandThe Emperor of All Maladies: A Biography of CancerRating: 4.5 out of 5 stars4.5/5 (271)
- The Little Book of Hygge: Danish Secrets to Happy LivingFrom EverandThe Little Book of Hygge: Danish Secrets to Happy LivingRating: 3.5 out of 5 stars3.5/5 (403)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyFrom EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyRating: 3.5 out of 5 stars3.5/5 (2259)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaFrom EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaRating: 4.5 out of 5 stars4.5/5 (266)
- The Yellow House: A Memoir (2019 National Book Award Winner)From EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Rating: 4 out of 5 stars4/5 (98)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryFrom EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryRating: 3.5 out of 5 stars3.5/5 (231)
- Team of Rivals: The Political Genius of Abraham LincolnFrom EverandTeam of Rivals: The Political Genius of Abraham LincolnRating: 4.5 out of 5 stars4.5/5 (234)
- On Fire: The (Burning) Case for a Green New DealFrom EverandOn Fire: The (Burning) Case for a Green New DealRating: 4 out of 5 stars4/5 (74)
- The Unwinding: An Inner History of the New AmericaFrom EverandThe Unwinding: An Inner History of the New AmericaRating: 4 out of 5 stars4/5 (45)
- AspenTech AspenONE 8.0 InstallDocument6 pagesAspenTech AspenONE 8.0 InstallimoomexNo ratings yet
- Petroleomics and Structure-Function Relations of Crude Oils and AsphaltenesDocument1 pagePetroleomics and Structure-Function Relations of Crude Oils and AsphaltenesnestorsoftNo ratings yet
- Basic System Configuration: Wellhead and ManifoldDocument1 pageBasic System Configuration: Wellhead and ManifoldnestorsoftNo ratings yet
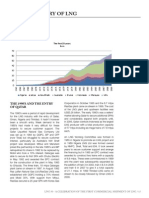
- A Brief History of LNG: The 1990'S and The Entry of QatarDocument1 pageA Brief History of LNG: The 1990'S and The Entry of QatarnestorsoftNo ratings yet
- Fire Water System and Fire Fighting Equipment PDFDocument3 pagesFire Water System and Fire Fighting Equipment PDFnestorsoftNo ratings yet
- 2012 06 Beacon SDocument1 page2012 06 Beacon SnestorsoftNo ratings yet
- ISA 750101 SPBD PDFDocument8 pagesISA 750101 SPBD PDFnestorsoft0% (1)
- Membrane Technology and Applications PDFDocument4 pagesMembrane Technology and Applications PDFnestorsoft100% (1)
- Nitrogen Asphyxiation Bulletin Training PresentationDocument24 pagesNitrogen Asphyxiation Bulletin Training PresentationnestorsoftNo ratings yet
- Oil Refinery Feasibility Study PDFDocument55 pagesOil Refinery Feasibility Study PDFPatrice Adjei96% (23)
- Feed DefinitionsDocument3 pagesFeed DefinitionsnestorsoftNo ratings yet