Professional Documents
Culture Documents
#Design Equation D (X) /D (W) (-Raprime) / Fao X (0) 0 X (F) 0.70 W (0) 0
#Design Equation D (X) /D (W) (-Raprime) / Fao X (0) 0 X (F) 0.70 W (0) 0
Uploaded by
AzmiHafifi0 ratings0% found this document useful (0 votes)
13 views2 pagesThis document contains the design equations, rate law, stoichiometry, parameters, and differential equations for modeling a chemical reaction. The key differential equation is d(x)/d(w) = (-raprime) / Fao, where raprime is the rate of the reaction and depends on rate constant k, reactants pa and pb, which in turn depend on x. The parameters such as rate constant k, temperature To, gas constant R, and Fao are provided, and the initial and final values of variables are given.
Original Description:
aa
Original Title
Design Equation
Copyright
© © All Rights Reserved
Available Formats
DOCX, PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentThis document contains the design equations, rate law, stoichiometry, parameters, and differential equations for modeling a chemical reaction. The key differential equation is d(x)/d(w) = (-raprime) / Fao, where raprime is the rate of the reaction and depends on rate constant k, reactants pa and pb, which in turn depend on x. The parameters such as rate constant k, temperature To, gas constant R, and Fao are provided, and the initial and final values of variables are given.
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
Download as docx, pdf, or txt
0 ratings0% found this document useful (0 votes)
13 views2 pages#Design Equation D (X) /D (W) (-Raprime) / Fao X (0) 0 X (F) 0.70 W (0) 0
#Design Equation D (X) /D (W) (-Raprime) / Fao X (0) 0 X (F) 0.70 W (0) 0
Uploaded by
AzmiHafifiThis document contains the design equations, rate law, stoichiometry, parameters, and differential equations for modeling a chemical reaction. The key differential equation is d(x)/d(w) = (-raprime) / Fao, where raprime is the rate of the reaction and depends on rate constant k, reactants pa and pb, which in turn depend on x. The parameters such as rate constant k, temperature To, gas constant R, and Fao are provided, and the initial and final values of variables are given.
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
Download as docx, pdf, or txt
You are on page 1of 2
#design equation
d(x)/d(w)= (-raprime)/ Fao
x(0)=0
x(f)=0.70
w(0)=0
#rate law
raprime=-(k*pa*pb)
#stochiometry
pb=pao*(3-3*x)
pa=pao*(1-x)
pao=1 #atm
k=1.67*10^-3 #kmol/hr.kgcat.atm2
To=245+273 #K
R=1.987 #cal/Kmol
Fao=68.89 #kmol/hr
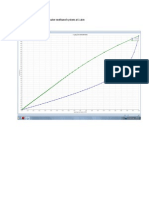
POLYMATH Report No Title
Ordinary Differential Equations 26-Mei-2014
Calculated values of DEQ variables
Variable Initial value Minimal value Maximal value Final value
1 Fao 68.89 68.89 68.89 68.89
2 k 0.00167 0.00167 0.00167 0.00167
3 pa 1. 0.3 1. 0.3
4 pao 1. 1. 1. 1.
5 pb 3. 0.9 3. 0.9
6 R 1.987 1.987 1.987 1.987
7 raprime -0.00501 -0.00501 -0.0004509 -0.0004509
8 To 518. 518. 518. 518.
9 w 0 0 0.7 0.7
10 x 0 0 5.09E-05 5.09E-05
Differential equations
1 d(x)/d(w) =(-raprime) / Fao
Explicit equations
1 k = 1.67*10^-3
kmol/hr.kgcat.atm2
2 pao = 1
atm
3 pb = pao*(3-3*x)
4 pa = pao*(1-x)
5 raprime = -(k*pa*pb)
6 To = 245+273
K
7 R = 1.987
cal/Kmol
8 Fao = 68.89
kmol/hr
General
Total number of equations 9
Number of differential equations 1
Number of explicit equations 8
Elapsed time 2.315 sec
Solution method RKF_56
Step size guess. h 0.000001
Truncation error tolerance. eps 0.000001
You might also like
- Computer Organization and Design Mips Edition 5th Edition Patterson Solutions ManualDocument9 pagesComputer Organization and Design Mips Edition 5th Edition Patterson Solutions Manualmatthewmorrisonsmwqkifnjx94% (17)
- Me 402Document3 pagesMe 402jared100% (4)
- Essentials of Chemical Reaction Engineering 1st Edition Fogler Solutions ManualDocument38 pagesEssentials of Chemical Reaction Engineering 1st Edition Fogler Solutions Manualspikyflagworm84dyts100% (20)
- Introduction To Wireless Systems - P M Shankar - Solutions Manual PDFDocument246 pagesIntroduction To Wireless Systems - P M Shankar - Solutions Manual PDFmienimeez94% (17)
- Exercise 5: Frequency Analysis:) AgainstDocument9 pagesExercise 5: Frequency Analysis:) AgainstBikas C. BhattaraiNo ratings yet
- Chapter 1 - Modelling, Computers and Error AnalysisDocument35 pagesChapter 1 - Modelling, Computers and Error AnalysisAzmiHafifiNo ratings yet
- Mat Lab 2Document38 pagesMat Lab 2packiaraja lNo ratings yet
- Fluids Mechanics IiDocument16 pagesFluids Mechanics IiJesús MorenoNo ratings yet
- L12prob Plan2 04Document50 pagesL12prob Plan2 04eviroyerNo ratings yet
- Chempute1994 PDFDocument17 pagesChempute1994 PDFvlcmstne04No ratings yet
- Annex A To Appendix 9: Atpc Example CalculationsDocument9 pagesAnnex A To Appendix 9: Atpc Example Calculationsعبد الغفور الشرقاويNo ratings yet
- Analog Book AnswersDocument5 pagesAnalog Book AnswersnmaravindNo ratings yet
- Weighted Cycle Prefix OFDMDocument4 pagesWeighted Cycle Prefix OFDMRama Subrahmanyam BNo ratings yet
- Essentials of Chemical Reaction Engineering 1st Edition Fogler Solutions Manual Full Chapter PDFDocument42 pagesEssentials of Chemical Reaction Engineering 1st Edition Fogler Solutions Manual Full Chapter PDFJulieMorrisfazg100% (13)
- LRFD Steel BeamDocument30 pagesLRFD Steel Beam정영철100% (1)
- Dist IllationDocument31 pagesDist IllationMuntazer QasimNo ratings yet
- CHE654 2012 Homework2 Solutions PDFDocument21 pagesCHE654 2012 Homework2 Solutions PDFmbolantenainaNo ratings yet
- Variable Initial Value Minimal Value Maximal Value Final ValueDocument4 pagesVariable Initial Value Minimal Value Maximal Value Final Valuej0haNN3sNo ratings yet
- Decline CurvesDocument13 pagesDecline Curvesalok_789100% (1)
- Hrast BasicDocument48 pagesHrast Basicgord12No ratings yet
- Engine Propeller Matching: Kelompok 1 Rega Ardian Syah Angga Wahyu P Pandika DarmawanDocument15 pagesEngine Propeller Matching: Kelompok 1 Rega Ardian Syah Angga Wahyu P Pandika DarmawanyuniardimzNo ratings yet
- Cantilever Sheet Pile Wall Design 19.03.2024Document7 pagesCantilever Sheet Pile Wall Design 19.03.2024Aamir SuhailNo ratings yet
- (Mobile Communication) Take Home Exam Solution Ryid Al-Kahtani ID#423121512Document16 pages(Mobile Communication) Take Home Exam Solution Ryid Al-Kahtani ID#423121512Muqeet Khaja UddinNo ratings yet
- C Ci (P) : Ex - No: Date: Economic Dispatch Problem AimDocument7 pagesC Ci (P) : Ex - No: Date: Economic Dispatch Problem AimsasitsnNo ratings yet
- Bode Plot Design PDFDocument15 pagesBode Plot Design PDFsmileuplease8498No ratings yet
- Problems 13 1 To 13 17Document39 pagesProblems 13 1 To 13 17Carlos Huete FelixNo ratings yet
- Reaktor-01Document16 pagesReaktor-01Agung Prasetyo NugrohoNo ratings yet
- Sludge ThickenerDocument15 pagesSludge ThickenerDavid Lambert100% (1)
- Reactor Design Python CodeDocument23 pagesReactor Design Python Codealiggulam16No ratings yet
- Lecture 2Document22 pagesLecture 2pongpumNo ratings yet
- Economic Dispatch Thermal Units and Methods of SolutionDocument15 pagesEconomic Dispatch Thermal Units and Methods of SolutionFulki Kautsar SNo ratings yet
- Virtual Lecture - 3-Tutorials-ThermodynamicsDocument20 pagesVirtual Lecture - 3-Tutorials-ThermodynamicsRukmani DeviNo ratings yet
- Chemical Reactors: FoglerDocument34 pagesChemical Reactors: FoglerMike PoulinNo ratings yet
- Lecture 2Document25 pagesLecture 2asmrsamir5No ratings yet
- Exam 1 Heat and MassDocument2 pagesExam 1 Heat and MassTodd MixNo ratings yet
- New - OOI CHEL GEE - 005093 (RED2&3)Document17 pagesNew - OOI CHEL GEE - 005093 (RED2&3)Choo Shyan LeeNo ratings yet
- ProblemsDocument12 pagesProblemsThothef AliNo ratings yet
- Density Functional TheoryDocument10 pagesDensity Functional Theorymommy2hNo ratings yet
- FounDocument18 pagesFounSisay GashaNo ratings yet
- Flight Mechanics Numericals With SolutionDocument27 pagesFlight Mechanics Numericals With Solutionmanikandan_murugaiahNo ratings yet
- Systemverilog - Lecture 1Document62 pagesSystemverilog - Lecture 1Shilpa ReddyNo ratings yet
- Solution Manual Elements of Chemical Reaction Engineering 4th Edition WWW - Elsolucionario.org 573 680Document108 pagesSolution Manual Elements of Chemical Reaction Engineering 4th Edition WWW - Elsolucionario.org 573 680Jose Maria Quintas GironNo ratings yet
- 2 EC Cryptography: 2.1 Elliptic Curve ArithmeticDocument8 pages2 EC Cryptography: 2.1 Elliptic Curve ArithmeticHadis MehariNo ratings yet
- Efficient Solution of Multiple-Model, Multiple-Algorithm Problems in Undergraduate and Graduate EducationDocument24 pagesEfficient Solution of Multiple-Model, Multiple-Algorithm Problems in Undergraduate and Graduate EducationAzar DeenNo ratings yet
- Ti 89 Titanium Exercise 3Document10 pagesTi 89 Titanium Exercise 3kalimbaloNo ratings yet
- EE633 HOWM WORK 3 With SolutionDocument15 pagesEE633 HOWM WORK 3 With Solutionbelal maNo ratings yet
- Diffusion of Liquids Through Stagnant Non Diffusing Air GRP ReportDocument23 pagesDiffusion of Liquids Through Stagnant Non Diffusing Air GRP ReportCharlyn Joy RamirezNo ratings yet
- Tuning PI Controllers For Integrator/Dead Time ProcessesDocument4 pagesTuning PI Controllers For Integrator/Dead Time ProcessesRaadBassamNo ratings yet
- Audio Algorithm Notes PDFDocument9 pagesAudio Algorithm Notes PDFJimmy KodyNo ratings yet
- OLS Estimation and Monte Carlo Simulation: Introductory Notes and CommentsDocument38 pagesOLS Estimation and Monte Carlo Simulation: Introductory Notes and CommentsTHINH LYNo ratings yet
- 10-27-Noted-Practice Midterm With SolutionDocument5 pages10-27-Noted-Practice Midterm With SolutionK163726 Syed Ashar AliNo ratings yet
- Dynamic Modeling of A Batch Bioreactor For Trans-Esterification of Waste Vegetable OilDocument40 pagesDynamic Modeling of A Batch Bioreactor For Trans-Esterification of Waste Vegetable Oilaremunabeel5895No ratings yet
- CH01 Solution PDFDocument8 pagesCH01 Solution PDFanand_duraiswamyNo ratings yet
- Batch c7Document15 pagesBatch c7Karthik BalajiNo ratings yet
- 10.3 One-Dimensional Search With First DerivativesDocument4 pages10.3 One-Dimensional Search With First DerivativesRajivSadewaNo ratings yet
- Savitsky Short Prog-1Document10 pagesSavitsky Short Prog-1fellipe_veríssimoNo ratings yet
- PVT CorrelationsDocument6 pagesPVT CorrelationsFredy Andres Narvaez BohorquezNo ratings yet
- Analytical Modeling of Solute Transport in Groundwater: Using Models to Understand the Effect of Natural Processes on Contaminant Fate and TransportFrom EverandAnalytical Modeling of Solute Transport in Groundwater: Using Models to Understand the Effect of Natural Processes on Contaminant Fate and TransportNo ratings yet
- Numerical Methods for Simulation and Optimization of Piecewise Deterministic Markov Processes: Application to ReliabilityFrom EverandNumerical Methods for Simulation and Optimization of Piecewise Deterministic Markov Processes: Application to ReliabilityNo ratings yet
- Exercise 4 PDC Sem II 201415 STDDocument1 pageExercise 4 PDC Sem II 201415 STDAzmiHafifiNo ratings yet
- (A) Generate A Txy Diagram For The Water-Methanol System at 1 AtmDocument4 pages(A) Generate A Txy Diagram For The Water-Methanol System at 1 AtmAzmiHafifiNo ratings yet
- A) T-Xy Diagram For The Water-Methanol System at 1 AtmDocument2 pagesA) T-Xy Diagram For The Water-Methanol System at 1 AtmAzmiHafifiNo ratings yet
- (Selasa) : BIL Nama No. Matrik Alamat KolejDocument2 pages(Selasa) : BIL Nama No. Matrik Alamat KolejAzmiHafifiNo ratings yet
- Faculty of Chemical and Natural Resources Engineering: Research Proposal (20%)Document2 pagesFaculty of Chemical and Natural Resources Engineering: Research Proposal (20%)AzmiHafifiNo ratings yet
- Minor Report From Proposal PresentationDocument2 pagesMinor Report From Proposal PresentationAzmiHafifiNo ratings yet
- Lectures 8 9 10Document185 pagesLectures 8 9 10AzmiHafifiNo ratings yet
- Buku Log Briged Siswa (UQB1011) Semester 1 Sesi 2011/2012Document1 pageBuku Log Briged Siswa (UQB1011) Semester 1 Sesi 2011/2012AzmiHafifiNo ratings yet
- Roles of Engineer in Herbal Industy AND Warisan Kundur Resources SDN BHDDocument2 pagesRoles of Engineer in Herbal Industy AND Warisan Kundur Resources SDN BHDAzmiHafifiNo ratings yet
- Section 4: Chemical Reaction Engineering IiDocument1 pageSection 4: Chemical Reaction Engineering IiAzmiHafifiNo ratings yet
- Polymerization MethodDocument5 pagesPolymerization MethodAzmiHafifiNo ratings yet
- Final Report Format 2014AAAAAADocument18 pagesFinal Report Format 2014AAAAAAAzmiHafifiNo ratings yet
- Basic Calculation Mola, MolaliDocument22 pagesBasic Calculation Mola, MolaliAzmiHafifiNo ratings yet
- Properties Method Selection FlowchartDocument2 pagesProperties Method Selection FlowchartAzmiHafifiNo ratings yet
- Evaporation: DKK3433 Unit Operation Dr. Syed Mohd Saufi 2012/2013-IDocument43 pagesEvaporation: DKK3433 Unit Operation Dr. Syed Mohd Saufi 2012/2013-IAzmiHafifiNo ratings yet




























































![Catalan's Constant [Ramanujan's Formula]](https://imgv2-2-f.scribdassets.com/img/word_document/399691191/149x198/f7e96de7ef/1668683798?v=1)