Professional Documents
Culture Documents
Year 11 Chemistry Worksheet: Air and Water
Year 11 Chemistry Worksheet: Air and Water
Uploaded by
Mohamad El MawlaCopyright:
Available Formats
You might also like
- Comprehensive Interview Material - Onlinestudy4uDocument61 pagesComprehensive Interview Material - Onlinestudy4uvikram mehta100% (2)
- POT Trinidad Fact SheetDocument8 pagesPOT Trinidad Fact SheetLok Chen ChuangNo ratings yet
- Ion Exchange Resins and Adsorbents in Chemical Processing: Second EditionFrom EverandIon Exchange Resins and Adsorbents in Chemical Processing: Second EditionRating: 5 out of 5 stars5/5 (1)
- Survey of Industrial Chemestry - Philip J. ChenierDocument13 pagesSurvey of Industrial Chemestry - Philip J. ChenierBiain A SecasNo ratings yet
- Lecture 3 Industrial Gases+ Ammonia 2019Document49 pagesLecture 3 Industrial Gases+ Ammonia 2019Mohamed AbdelaalNo ratings yet
- Flashcards - Topic 11 Air and Water - CAIE Chemistry IGCSEDocument77 pagesFlashcards - Topic 11 Air and Water - CAIE Chemistry IGCSEKendrickNo ratings yet
- Annealing of Wire TechDocument4 pagesAnnealing of Wire TechJayabharath KrishnanNo ratings yet
- BTEX Removal From Natural GasDocument46 pagesBTEX Removal From Natural GasSurendar Babu S100% (1)
- Petrochemical Industry - Production ProcessDocument40 pagesPetrochemical Industry - Production ProcessAyie Arie AyitNo ratings yet
- Air and Water ChemistryDocument24 pagesAir and Water ChemistryShaman Samuel GodfreyNo ratings yet
- 1صناعاتDocument15 pages1صناعاتroaanaseem267No ratings yet
- Chemical Monitoring and Management: Chemist XDocument11 pagesChemical Monitoring and Management: Chemist XLNo ratings yet
- C1b HomeworkDocument7 pagesC1b HomeworkWendywitch123No ratings yet
- Fertiliser Manufacturing Processes and Its Environmental ProblemsDocument69 pagesFertiliser Manufacturing Processes and Its Environmental ProblemsAulizar MarioNo ratings yet
- Haber AmmoniaDocument22 pagesHaber AmmoniaKabilanNo ratings yet
- Haber Bosch ProcessDocument44 pagesHaber Bosch ProcessyudiapnNo ratings yet
- Chem 30Document3 pagesChem 30micahanedwardsNo ratings yet
- Ethylene OxideDocument6 pagesEthylene OxideRabiya SaeedNo ratings yet
- Episode 3: Production of Synthesis Gas by Steam Methane ReformingDocument31 pagesEpisode 3: Production of Synthesis Gas by Steam Methane ReformingSAJJAD KHUDHUR ABBASNo ratings yet
- UreaDocument18 pagesUreaDian Anggraini PurbaNo ratings yet
- Production of Ammonia by Haber Bosch ProcessDocument5 pagesProduction of Ammonia by Haber Bosch ProcesszahidNo ratings yet
- Haber ProcessDocument10 pagesHaber ProcessTon5698No ratings yet
- Motors in Chemical Eng.Document6 pagesMotors in Chemical Eng.Guillermo CúmezNo ratings yet
- 1 AirDocument51 pages1 AirAli HarbNo ratings yet
- Ethylene 2520oxide Methods 2520of 2520productionDocument25 pagesEthylene 2520oxide Methods 2520of 2520productionmartin_2104No ratings yet
- Athmosphere and Environment Research For O LevelsDocument12 pagesAthmosphere and Environment Research For O LevelsAsim HussainNo ratings yet
- Synthesis Gas Preparation First Methane Is Cleaned To Remove Impurities That Would Poison The CatalystsDocument44 pagesSynthesis Gas Preparation First Methane Is Cleaned To Remove Impurities That Would Poison The CatalystsabdoNo ratings yet
- Chemical MonitoringDocument28 pagesChemical MonitoringdasdaNo ratings yet
- LECTURE - 6: Ethylene Derivatives: Ethylene Oxide and Ethanol Amines 6.1 Ethylene OxideDocument7 pagesLECTURE - 6: Ethylene Derivatives: Ethylene Oxide and Ethanol Amines 6.1 Ethylene Oxideمحمود محمدNo ratings yet
- Component Percentage% Nitrogen Oxygen Argon Carbon Dioxide Neon Krypton Nitrogen Dioxide Methane Ozone Helium Hydrogen XenonDocument4 pagesComponent Percentage% Nitrogen Oxygen Argon Carbon Dioxide Neon Krypton Nitrogen Dioxide Methane Ozone Helium Hydrogen XenonVitor GarciaNo ratings yet
- Patterns and Properties of Non-MetalsDocument14 pagesPatterns and Properties of Non-MetalsiluvreadingbooksNo ratings yet
- CH 19Document8 pagesCH 19JayelleNo ratings yet
- Chemistry Year 11 Depth Study: Frank DingDocument7 pagesChemistry Year 11 Depth Study: Frank DingFrankNo ratings yet
- Wet Air Oxidation of Alkanolamines: Chapter No. 3Document6 pagesWet Air Oxidation of Alkanolamines: Chapter No. 3Azam KhanNo ratings yet
- Science Notes Year 10Document34 pagesScience Notes Year 10Rishi Máran100% (2)
- TechDocument8 pagesTechNagendra KumarNo ratings yet
- Joshua Campe Sunny Chawla Brandon HardwickeDocument19 pagesJoshua Campe Sunny Chawla Brandon HardwickeWalida AdauwiyahNo ratings yet
- New Microsoft Word DocumentDocument19 pagesNew Microsoft Word DocumentVishal RajNo ratings yet
- Chapter 13 - Nitrogen and SulfurDocument10 pagesChapter 13 - Nitrogen and SulfurNabindra RuwaliNo ratings yet
- Ammonia PlantDocument16 pagesAmmonia Plantganeshan67% (6)
- Production of AmmoniaDocument29 pagesProduction of AmmoniaBhavna Bajpai83% (6)
- CH 11 Air and WaterDocument6 pagesCH 11 Air and Waterlayan alharbiNo ratings yet
- Introduction To AmmoniaDocument15 pagesIntroduction To AmmoniaHameed Akhtar100% (1)
- Ammonia Process InformationDocument4 pagesAmmonia Process InformationRishikesh AwaleNo ratings yet
- AmmoniaDocument33 pagesAmmoniaaregawi zemchealNo ratings yet
- Sem1Fin SummaryDocument6 pagesSem1Fin SummarySharonNo ratings yet
- Ammonia and Urea ProductionDocument10 pagesAmmonia and Urea Productionwaheed_bhattiNo ratings yet
- 9.5 Industrial ChemistryDocument33 pages9.5 Industrial ChemistrykevNo ratings yet
- As Notes Final 2023docxDocument53 pagesAs Notes Final 2023docxMuhammad HashimNo ratings yet
- Natural Gas Applications: FertilzersDocument20 pagesNatural Gas Applications: FertilzersTamanna Kumari100% (1)
- Topic.10 Chemistry of Our EnviromentDocument12 pagesTopic.10 Chemistry of Our EnviromentJoyce AmirNo ratings yet
- Chapter 2Document7 pagesChapter 2pragati agrawalNo ratings yet
- Nitrogen and Sulfur AS LevelDocument5 pagesNitrogen and Sulfur AS LevelThapelo SebolaiNo ratings yet
- Core Topic 3 - Chemical Monitoring and ManagementDocument20 pagesCore Topic 3 - Chemical Monitoring and ManagementD RayNo ratings yet
- Air Quality ManagementFrom EverandAir Quality ManagementSuresh T. NesaratnamNo ratings yet
- Thermodynamic analysis of geothermal heat pumps for civil air-conditioningFrom EverandThermodynamic analysis of geothermal heat pumps for civil air-conditioningRating: 5 out of 5 stars5/5 (2)
- Natural Gas: Consumers and Consuming Industry: A Handbook for Students of the Natural Gas IndustryFrom EverandNatural Gas: Consumers and Consuming Industry: A Handbook for Students of the Natural Gas IndustryNo ratings yet
- Clean Ironmaking and Steelmaking Processes: Efficient Technologies for Greenhouse Emissions AbatementFrom EverandClean Ironmaking and Steelmaking Processes: Efficient Technologies for Greenhouse Emissions AbatementNo ratings yet
- Indicator SpeciesDocument6 pagesIndicator SpeciesMohamad El MawlaNo ratings yet
- Waves 7 - Refractive Index HWDocument3 pagesWaves 7 - Refractive Index HWMohamad El MawlaNo ratings yet
- Desert Ecosystems: Case Study by Kim KerrDocument13 pagesDesert Ecosystems: Case Study by Kim KerrMohamad El MawlaNo ratings yet
- The Kidney: Filtration & Re-Absorption: Pickering ReferenceDocument8 pagesThe Kidney: Filtration & Re-Absorption: Pickering ReferenceMohamad El MawlaNo ratings yet
- Refugee Blues - Analytical Essay: W. H. AudenDocument1 pageRefugee Blues - Analytical Essay: W. H. AudenMohamad El MawlaNo ratings yet
- Cold War TimelineDocument2 pagesCold War TimelineMohamad El MawlaNo ratings yet
- Ethylene Glycol HeatDocument6 pagesEthylene Glycol Heatdalton2004No ratings yet
- Answers For Frequently Asked Java QuestionsDocument29 pagesAnswers For Frequently Asked Java QuestionsS.S. AmmarNo ratings yet
- Loss of PCDocument50 pagesLoss of PCAlMahmudNo ratings yet
- JNTUA-B.Tech.4-1 CSE-R15-SYLLABUSDocument16 pagesJNTUA-B.Tech.4-1 CSE-R15-SYLLABUSYallala Srihari ReddyNo ratings yet
- Credit Card Fraud Detection Using Enhanced Random Forest Classifier For Imbalanced DataDocument11 pagesCredit Card Fraud Detection Using Enhanced Random Forest Classifier For Imbalanced Datanarendramatta18No ratings yet
- Coagulation Profile in Diabetes MellitusDocument5 pagesCoagulation Profile in Diabetes MellitusAsfandyar RoghaniNo ratings yet
- Activate TreadmillDocument2 pagesActivate Treadmillajmal_2kNo ratings yet
- CHAP 1 Interference of Light New Syllabus-1Document14 pagesCHAP 1 Interference of Light New Syllabus-1Cradon FernandesNo ratings yet
- RC Beam Analysis & Design (En1992-1) in Accordance With UK National AnnexDocument7 pagesRC Beam Analysis & Design (En1992-1) in Accordance With UK National Annexmishu_cojoNo ratings yet
- MP3 DFPlayer Mini Module ENGDocument36 pagesMP3 DFPlayer Mini Module ENGmariguanixNo ratings yet
- Statistical Models in Simulation: Course LeaderDocument23 pagesStatistical Models in Simulation: Course LeadershyNo ratings yet
- Rothoblaas - Alumidi Bracket With Holes - Item Specification - enDocument4 pagesRothoblaas - Alumidi Bracket With Holes - Item Specification - enAndrei GheorghicaNo ratings yet
- Crank System - Piston and CylinderDocument16 pagesCrank System - Piston and Cylinder270329No ratings yet
- Aggregate Training ManualDocument266 pagesAggregate Training ManualFanuel MsangiNo ratings yet
- AA SL 24 Revision Geometry and Trigonometry Part 2Document18 pagesAA SL 24 Revision Geometry and Trigonometry Part 2MeeraNo ratings yet
- N13072012 2Document85 pagesN13072012 2Ng Hock HengNo ratings yet
- Module Week 5Document13 pagesModule Week 5DM BartolayNo ratings yet
- Machine-Learning Analysis For The Iris Dataset: ArticleDocument16 pagesMachine-Learning Analysis For The Iris Dataset: ArticleBarrey WaneNo ratings yet
- Backup and Restore Logical Volume Using LVM SnapshotDocument25 pagesBackup and Restore Logical Volume Using LVM SnapshotpalkybdNo ratings yet
- Offshore Structures and Ship Impact Analysis MRT 608Document17 pagesOffshore Structures and Ship Impact Analysis MRT 608Emmanuel Olisa Umenwa100% (2)
- Topic 4 - StoichiometryDocument34 pagesTopic 4 - StoichiometryRhyn Ruz DohilagNo ratings yet
- Convergence IndicatorDocument21 pagesConvergence Indicatorsikandar100% (1)
- Experiment No. 3 Measurement of Specific Weight and Pressure Intensity of LiquidsDocument4 pagesExperiment No. 3 Measurement of Specific Weight and Pressure Intensity of LiquidsChristine KimNo ratings yet
- LIFTDocument37 pagesLIFTNek ManNo ratings yet
- DSI Brochure 2016Document36 pagesDSI Brochure 2016Petro NimchukNo ratings yet
- Consumer Choice: ECON1005 Principles of Economics I (Microeconomics)Document33 pagesConsumer Choice: ECON1005 Principles of Economics I (Microeconomics)Daniel SarchaevNo ratings yet
- Sadtem - Catalogue - Transformateurs de Courant Intérieurs - 2009Document24 pagesSadtem - Catalogue - Transformateurs de Courant Intérieurs - 2009ZorbanfrNo ratings yet
- Brosur Pipa JackingDocument1 pageBrosur Pipa JackingDavid SiburianNo ratings yet
- Classes of CompoundsDocument12 pagesClasses of CompoundsReynaldo VirtucioNo ratings yet
Year 11 Chemistry Worksheet: Air and Water
Year 11 Chemistry Worksheet: Air and Water
Uploaded by
Mohamad El MawlaOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Year 11 Chemistry Worksheet: Air and Water
Year 11 Chemistry Worksheet: Air and Water
Uploaded by
Mohamad El MawlaCopyright:
Available Formats
Year 11 chemistry worksheet: Air and water
Read pages 182-189 in the textbook completely before
attempting questions, and then use these pages to
research proper answers to the following questions
Complete the following table showing the composition of dry,
unpolluted air. List the most abundant gases first.
Gas
Oxygen
Carbon dioxide
Nitrogen
Argon
Formula
O2
CO2
N2
Ar
Abundance %
21
0.04
78
0.9
The natural humidity of air varies across the globe. What is the
general range of % water in the atmosphere? Between 1% and
4%.
Name and briefly describe the process used to obtain pure
nitrogen and oxygen gases from air.
Air is cooled and compressed to form a liquid, and then the liquid is fractionally distilled
to obtain each of the different components in the air.
Name three uses for pure oxygen gas (do not include normal
breathing).
-
Oxygen gas is poisonous to bacteria that cause gangrene. Therefore, it is used to kill
them.
Oxygen is used as a step in the production of antifreeze and polyester polymers. The
polymers are then used to make plastic and fabrics.
Pure oxygen is used to ensure the complete combustion of different chemicals.
Name two uses for Nitrogen gas, and explain why it is so useful.
-
Nitrogen is used in high temperature thermometers where mercury cannot be used. This
is because mercury boils at 356 C and cannot be used in such thermometers.
Nitrogen mixed with argon is used in electric bulbs to provide an inert atmosphere. It
helps in prevention of oxidation and evaporation of the filament of the bulb, giving it a
longer life.
Name an industrial process that uses nitrogen gas as a reactant
(Hint: you should have revised this process by now!) and state the
main products.
Haber Process
This reaction is reversible.
The forward reaction (from left to right) is exothermic.
Nitrogen + hydrogen
N2(g)
3H2(g)
ammonia (+ heat).
2NH3(g) (+ heat)
The industrial conditions are
1) Temperature between 450 C and 500 C.
2) Pressure of 200 atm (200 atmospheres).
3) Use an iron catalyst.
Only about 15% of the reactants are converted into products
under these conditions.
Ammonia is cooled and liquefied at the reaction pressure,
and then removed as liquid ammonia.
The remaining mix of nitrogen and hydrogen gases (85%)
are recycled and fed in at the reactant stage.
The process operates continuously.
List the most common pollutants in the table, with their sources
and hazards.
Pollutant
Source
Hazard
Why do nitrogen oxides form in exhaust gases? Write a balanced
equation.
What is fitted to car exhausts to remove nitrogen oxides? Write
and equation to show how two toxic gases are reacted together to
produce less harmful gases.
One of these less harmful gases is still a major environmental
concern. Which gas is it, and what is the concern?
There are four main greenhouse gases. List them in the table
below.
Greenhouse gas
Carbon dioxide
Methane
Source
Burning fossil fuels in power stations
and cars.
Cows, pigs and sheep release a lot of
methane by bacterial action in their
digestive systems.
Produced by bacteria in the soil.
Nitrous oxide
CFCs
Depletion of the ozone layer which
surrounds earth.
Read pages 180-181 in the textbook completely before
attempting questions, and then use these pages to
research proper answers to the following questions
There are two chemical tests for water. Name them both, and
write equations to explain and demonstrate the colour changes.
1: Copper (II) sulfate
2:
Label the diagram showing the stages of water treatment.
Settlement tank
Aluminum
sulfate added
Chlorine
Sand filter
Why is chlorine added to the water supply?
Chlorine is a highly efficient disinfectant, and is added to public water supplies to kill
disease-causing pathogens, such as bacteria and viruses that commonly grow in water
supply reservoirs, on the walls of water mains and in storage tanks.
You might also like
- Comprehensive Interview Material - Onlinestudy4uDocument61 pagesComprehensive Interview Material - Onlinestudy4uvikram mehta100% (2)
- POT Trinidad Fact SheetDocument8 pagesPOT Trinidad Fact SheetLok Chen ChuangNo ratings yet
- Ion Exchange Resins and Adsorbents in Chemical Processing: Second EditionFrom EverandIon Exchange Resins and Adsorbents in Chemical Processing: Second EditionRating: 5 out of 5 stars5/5 (1)
- Survey of Industrial Chemestry - Philip J. ChenierDocument13 pagesSurvey of Industrial Chemestry - Philip J. ChenierBiain A SecasNo ratings yet
- Lecture 3 Industrial Gases+ Ammonia 2019Document49 pagesLecture 3 Industrial Gases+ Ammonia 2019Mohamed AbdelaalNo ratings yet
- Flashcards - Topic 11 Air and Water - CAIE Chemistry IGCSEDocument77 pagesFlashcards - Topic 11 Air and Water - CAIE Chemistry IGCSEKendrickNo ratings yet
- Annealing of Wire TechDocument4 pagesAnnealing of Wire TechJayabharath KrishnanNo ratings yet
- BTEX Removal From Natural GasDocument46 pagesBTEX Removal From Natural GasSurendar Babu S100% (1)
- Petrochemical Industry - Production ProcessDocument40 pagesPetrochemical Industry - Production ProcessAyie Arie AyitNo ratings yet
- Air and Water ChemistryDocument24 pagesAir and Water ChemistryShaman Samuel GodfreyNo ratings yet
- 1صناعاتDocument15 pages1صناعاتroaanaseem267No ratings yet
- Chemical Monitoring and Management: Chemist XDocument11 pagesChemical Monitoring and Management: Chemist XLNo ratings yet
- C1b HomeworkDocument7 pagesC1b HomeworkWendywitch123No ratings yet
- Fertiliser Manufacturing Processes and Its Environmental ProblemsDocument69 pagesFertiliser Manufacturing Processes and Its Environmental ProblemsAulizar MarioNo ratings yet
- Haber AmmoniaDocument22 pagesHaber AmmoniaKabilanNo ratings yet
- Haber Bosch ProcessDocument44 pagesHaber Bosch ProcessyudiapnNo ratings yet
- Chem 30Document3 pagesChem 30micahanedwardsNo ratings yet
- Ethylene OxideDocument6 pagesEthylene OxideRabiya SaeedNo ratings yet
- Episode 3: Production of Synthesis Gas by Steam Methane ReformingDocument31 pagesEpisode 3: Production of Synthesis Gas by Steam Methane ReformingSAJJAD KHUDHUR ABBASNo ratings yet
- UreaDocument18 pagesUreaDian Anggraini PurbaNo ratings yet
- Production of Ammonia by Haber Bosch ProcessDocument5 pagesProduction of Ammonia by Haber Bosch ProcesszahidNo ratings yet
- Haber ProcessDocument10 pagesHaber ProcessTon5698No ratings yet
- Motors in Chemical Eng.Document6 pagesMotors in Chemical Eng.Guillermo CúmezNo ratings yet
- 1 AirDocument51 pages1 AirAli HarbNo ratings yet
- Ethylene 2520oxide Methods 2520of 2520productionDocument25 pagesEthylene 2520oxide Methods 2520of 2520productionmartin_2104No ratings yet
- Athmosphere and Environment Research For O LevelsDocument12 pagesAthmosphere and Environment Research For O LevelsAsim HussainNo ratings yet
- Synthesis Gas Preparation First Methane Is Cleaned To Remove Impurities That Would Poison The CatalystsDocument44 pagesSynthesis Gas Preparation First Methane Is Cleaned To Remove Impurities That Would Poison The CatalystsabdoNo ratings yet
- Chemical MonitoringDocument28 pagesChemical MonitoringdasdaNo ratings yet
- LECTURE - 6: Ethylene Derivatives: Ethylene Oxide and Ethanol Amines 6.1 Ethylene OxideDocument7 pagesLECTURE - 6: Ethylene Derivatives: Ethylene Oxide and Ethanol Amines 6.1 Ethylene Oxideمحمود محمدNo ratings yet
- Component Percentage% Nitrogen Oxygen Argon Carbon Dioxide Neon Krypton Nitrogen Dioxide Methane Ozone Helium Hydrogen XenonDocument4 pagesComponent Percentage% Nitrogen Oxygen Argon Carbon Dioxide Neon Krypton Nitrogen Dioxide Methane Ozone Helium Hydrogen XenonVitor GarciaNo ratings yet
- Patterns and Properties of Non-MetalsDocument14 pagesPatterns and Properties of Non-MetalsiluvreadingbooksNo ratings yet
- CH 19Document8 pagesCH 19JayelleNo ratings yet
- Chemistry Year 11 Depth Study: Frank DingDocument7 pagesChemistry Year 11 Depth Study: Frank DingFrankNo ratings yet
- Wet Air Oxidation of Alkanolamines: Chapter No. 3Document6 pagesWet Air Oxidation of Alkanolamines: Chapter No. 3Azam KhanNo ratings yet
- Science Notes Year 10Document34 pagesScience Notes Year 10Rishi Máran100% (2)
- TechDocument8 pagesTechNagendra KumarNo ratings yet
- Joshua Campe Sunny Chawla Brandon HardwickeDocument19 pagesJoshua Campe Sunny Chawla Brandon HardwickeWalida AdauwiyahNo ratings yet
- New Microsoft Word DocumentDocument19 pagesNew Microsoft Word DocumentVishal RajNo ratings yet
- Chapter 13 - Nitrogen and SulfurDocument10 pagesChapter 13 - Nitrogen and SulfurNabindra RuwaliNo ratings yet
- Ammonia PlantDocument16 pagesAmmonia Plantganeshan67% (6)
- Production of AmmoniaDocument29 pagesProduction of AmmoniaBhavna Bajpai83% (6)
- CH 11 Air and WaterDocument6 pagesCH 11 Air and Waterlayan alharbiNo ratings yet
- Introduction To AmmoniaDocument15 pagesIntroduction To AmmoniaHameed Akhtar100% (1)
- Ammonia Process InformationDocument4 pagesAmmonia Process InformationRishikesh AwaleNo ratings yet
- AmmoniaDocument33 pagesAmmoniaaregawi zemchealNo ratings yet
- Sem1Fin SummaryDocument6 pagesSem1Fin SummarySharonNo ratings yet
- Ammonia and Urea ProductionDocument10 pagesAmmonia and Urea Productionwaheed_bhattiNo ratings yet
- 9.5 Industrial ChemistryDocument33 pages9.5 Industrial ChemistrykevNo ratings yet
- As Notes Final 2023docxDocument53 pagesAs Notes Final 2023docxMuhammad HashimNo ratings yet
- Natural Gas Applications: FertilzersDocument20 pagesNatural Gas Applications: FertilzersTamanna Kumari100% (1)
- Topic.10 Chemistry of Our EnviromentDocument12 pagesTopic.10 Chemistry of Our EnviromentJoyce AmirNo ratings yet
- Chapter 2Document7 pagesChapter 2pragati agrawalNo ratings yet
- Nitrogen and Sulfur AS LevelDocument5 pagesNitrogen and Sulfur AS LevelThapelo SebolaiNo ratings yet
- Core Topic 3 - Chemical Monitoring and ManagementDocument20 pagesCore Topic 3 - Chemical Monitoring and ManagementD RayNo ratings yet
- Air Quality ManagementFrom EverandAir Quality ManagementSuresh T. NesaratnamNo ratings yet
- Thermodynamic analysis of geothermal heat pumps for civil air-conditioningFrom EverandThermodynamic analysis of geothermal heat pumps for civil air-conditioningRating: 5 out of 5 stars5/5 (2)
- Natural Gas: Consumers and Consuming Industry: A Handbook for Students of the Natural Gas IndustryFrom EverandNatural Gas: Consumers and Consuming Industry: A Handbook for Students of the Natural Gas IndustryNo ratings yet
- Clean Ironmaking and Steelmaking Processes: Efficient Technologies for Greenhouse Emissions AbatementFrom EverandClean Ironmaking and Steelmaking Processes: Efficient Technologies for Greenhouse Emissions AbatementNo ratings yet
- Indicator SpeciesDocument6 pagesIndicator SpeciesMohamad El MawlaNo ratings yet
- Waves 7 - Refractive Index HWDocument3 pagesWaves 7 - Refractive Index HWMohamad El MawlaNo ratings yet
- Desert Ecosystems: Case Study by Kim KerrDocument13 pagesDesert Ecosystems: Case Study by Kim KerrMohamad El MawlaNo ratings yet
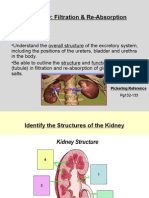
- The Kidney: Filtration & Re-Absorption: Pickering ReferenceDocument8 pagesThe Kidney: Filtration & Re-Absorption: Pickering ReferenceMohamad El MawlaNo ratings yet
- Refugee Blues - Analytical Essay: W. H. AudenDocument1 pageRefugee Blues - Analytical Essay: W. H. AudenMohamad El MawlaNo ratings yet
- Cold War TimelineDocument2 pagesCold War TimelineMohamad El MawlaNo ratings yet
- Ethylene Glycol HeatDocument6 pagesEthylene Glycol Heatdalton2004No ratings yet
- Answers For Frequently Asked Java QuestionsDocument29 pagesAnswers For Frequently Asked Java QuestionsS.S. AmmarNo ratings yet
- Loss of PCDocument50 pagesLoss of PCAlMahmudNo ratings yet
- JNTUA-B.Tech.4-1 CSE-R15-SYLLABUSDocument16 pagesJNTUA-B.Tech.4-1 CSE-R15-SYLLABUSYallala Srihari ReddyNo ratings yet
- Credit Card Fraud Detection Using Enhanced Random Forest Classifier For Imbalanced DataDocument11 pagesCredit Card Fraud Detection Using Enhanced Random Forest Classifier For Imbalanced Datanarendramatta18No ratings yet
- Coagulation Profile in Diabetes MellitusDocument5 pagesCoagulation Profile in Diabetes MellitusAsfandyar RoghaniNo ratings yet
- Activate TreadmillDocument2 pagesActivate Treadmillajmal_2kNo ratings yet
- CHAP 1 Interference of Light New Syllabus-1Document14 pagesCHAP 1 Interference of Light New Syllabus-1Cradon FernandesNo ratings yet
- RC Beam Analysis & Design (En1992-1) in Accordance With UK National AnnexDocument7 pagesRC Beam Analysis & Design (En1992-1) in Accordance With UK National Annexmishu_cojoNo ratings yet
- MP3 DFPlayer Mini Module ENGDocument36 pagesMP3 DFPlayer Mini Module ENGmariguanixNo ratings yet
- Statistical Models in Simulation: Course LeaderDocument23 pagesStatistical Models in Simulation: Course LeadershyNo ratings yet
- Rothoblaas - Alumidi Bracket With Holes - Item Specification - enDocument4 pagesRothoblaas - Alumidi Bracket With Holes - Item Specification - enAndrei GheorghicaNo ratings yet
- Crank System - Piston and CylinderDocument16 pagesCrank System - Piston and Cylinder270329No ratings yet
- Aggregate Training ManualDocument266 pagesAggregate Training ManualFanuel MsangiNo ratings yet
- AA SL 24 Revision Geometry and Trigonometry Part 2Document18 pagesAA SL 24 Revision Geometry and Trigonometry Part 2MeeraNo ratings yet
- N13072012 2Document85 pagesN13072012 2Ng Hock HengNo ratings yet
- Module Week 5Document13 pagesModule Week 5DM BartolayNo ratings yet
- Machine-Learning Analysis For The Iris Dataset: ArticleDocument16 pagesMachine-Learning Analysis For The Iris Dataset: ArticleBarrey WaneNo ratings yet
- Backup and Restore Logical Volume Using LVM SnapshotDocument25 pagesBackup and Restore Logical Volume Using LVM SnapshotpalkybdNo ratings yet
- Offshore Structures and Ship Impact Analysis MRT 608Document17 pagesOffshore Structures and Ship Impact Analysis MRT 608Emmanuel Olisa Umenwa100% (2)
- Topic 4 - StoichiometryDocument34 pagesTopic 4 - StoichiometryRhyn Ruz DohilagNo ratings yet
- Convergence IndicatorDocument21 pagesConvergence Indicatorsikandar100% (1)
- Experiment No. 3 Measurement of Specific Weight and Pressure Intensity of LiquidsDocument4 pagesExperiment No. 3 Measurement of Specific Weight and Pressure Intensity of LiquidsChristine KimNo ratings yet
- LIFTDocument37 pagesLIFTNek ManNo ratings yet
- DSI Brochure 2016Document36 pagesDSI Brochure 2016Petro NimchukNo ratings yet
- Consumer Choice: ECON1005 Principles of Economics I (Microeconomics)Document33 pagesConsumer Choice: ECON1005 Principles of Economics I (Microeconomics)Daniel SarchaevNo ratings yet
- Sadtem - Catalogue - Transformateurs de Courant Intérieurs - 2009Document24 pagesSadtem - Catalogue - Transformateurs de Courant Intérieurs - 2009ZorbanfrNo ratings yet
- Brosur Pipa JackingDocument1 pageBrosur Pipa JackingDavid SiburianNo ratings yet
- Classes of CompoundsDocument12 pagesClasses of CompoundsReynaldo VirtucioNo ratings yet