Professional Documents
Culture Documents
CHM 103s
CHM 103s
Uploaded by
snualpeCopyright:
Available Formats
You might also like
- หลักวิศวกรรมอาหาร 1Document7 pagesหลักวิศวกรรมอาหาร 1Sura C. JirNo ratings yet
- คู่มือครู เคมี เพิ่มเติม ม4 เล่ม 1 PDFDocument268 pagesคู่มือครู เคมี เพิ่มเติม ม4 เล่ม 1 PDFsnualpe89% (9)
- M460B1 PDFDocument172 pagesM460B1 PDFMamarine Wannasiri ThiphayamongkolNo ratings yet
- โครงการประเมินทักษะภาษาอังกฤษระดับประเทศ ครั้งที่ 1 Test Paper M1-3Document22 pagesโครงการประเมินทักษะภาษาอังกฤษระดับประเทศ ครั้งที่ 1 Test Paper M1-3snualpe100% (3)
- Scincem1 2Document324 pagesScincem1 2Montira SakaetongNo ratings yet
- เอกสารประกอบการสอน general physics I-rev1-5 PDFDocument199 pagesเอกสารประกอบการสอน general physics I-rev1-5 PDFNattawut NoreeNo ratings yet
- !!02207311 Eng-Hydrology Final-All 2557 PDFDocument403 pages!!02207311 Eng-Hydrology Final-All 2557 PDFสาธิต ปริ นทร์ทอง67% (3)
- แผนฯ หน้าที่พลเมืองฯ ม.4-6 เล่ม 1Document139 pagesแผนฯ หน้าที่พลเมืองฯ ม.4-6 เล่ม 1snualpe88% (8)
- รายละเอียดคอร์สsummer 63 ม5Document4 pagesรายละเอียดคอร์สsummer 63 ม5ครูพี่กาย โรงเรียนกวดวิชาวิคเตอร์No ratings yet
- Microsoft TeamDocument2 pagesMicrosoft Teamporamath33No ratings yet
- ตารางสอบวัดระดับด้านภาษาอังกฤษฯและดิจิทัลฉบับจริงDocument6 pagesตารางสอบวัดระดับด้านภาษาอังกฤษฯและดิจิทัลฉบับจริงP Danuphong ThitithananonNo ratings yet
- รายละเอียดคอร์สsummer 63 ม6Document4 pagesรายละเอียดคอร์สsummer 63 ม6ครูพี่กาย โรงเรียนกวดวิชาวิคเตอร์No ratings yet
- รายงานประชุมห้องเรียนพิเศษวิทยาศาสตร์ฯ ครั้งที่ - 2 - 2566Document4 pagesรายงานประชุมห้องเรียนพิเศษวิทยาศาสตร์ฯ ครั้งที่ - 2 - 2566Tutah RecordsNo ratings yet
- รายละเอียดคอร์สsummer 63 ม4Document4 pagesรายละเอียดคอร์สsummer 63 ม4ครูพี่กาย โรงเรียนกวดวิชาวิคเตอร์No ratings yet
- Course Outline - À À À À CHM 129 - 66 - LMSDocument5 pagesCourse Outline - À À À À CHM 129 - 66 - LMSaathitityNo ratings yet
- AMEDocument4 pagesAMEMek PunyawichNo ratings yet
- ปฏิทินสอบDocument7 pagesปฏิทินสอบจิรัฐิวัฒน์ เลาหวรรณธนะNo ratings yet
- 3. มฐ. และ ตชว. กลุ่มสาระการเรียนรู้วิทยาศาสตร์ (ฉบับปรับปรุง พ.ศ. 2560)Document273 pages3. มฐ. และ ตชว. กลุ่มสาระการเรียนรู้วิทยาศาสตร์ (ฉบับปรับปรุง พ.ศ. 2560)Scandoo DeeNo ratings yet
- A-Level 64 Phy วิชาฟิสิกส์ - TCAS66 BlueprintDocument1 pageA-Level 64 Phy วิชาฟิสิกส์ - TCAS66 Blueprint52mydjt2r8No ratings yet
- ตารางเรียน สาขาวิชาการสอนภาษาอังกฤษDocument4 pagesตารางเรียน สาขาวิชาการสอนภาษาอังกฤษUltimateNo ratings yet
- D 1Document277 pagesD 1ปรารถนา ปลื้มNo ratings yet
- à¸à¸³à¸à¸§à¸à¸£à¸²à¸¢à¸à¸·à¹à¸à¸à¸¹à¹à¸ªà¸³à¹à¸£à¹à¸à¸à¸²à¸£à¸¨à¸¶à¸à¸©à¸²-à¸à¸µà¸à¸²à¸£à¸¨à¸¶à¸à¸©à¸²-2564-à¸à¸£à¸¸à¸¨à¸²à¸ªà¸à¸£à¹Document55 pagesà¸à¸³à¸à¸§à¸à¸£à¸²à¸¢à¸à¸·à¹à¸à¸à¸¹à¹à¸ªà¸³à¹à¸£à¹à¸à¸à¸²à¸£à¸¨à¸¶à¸à¸©à¸²-à¸à¸µà¸à¸²à¸£à¸¨à¸¶à¸à¸©à¸²-2564-à¸à¸£à¸¸à¸¨à¸²à¸ªà¸à¸£à¹ploymaneerat.tNo ratings yet
- รายละเอียดของวิชา 315102-SC501001-General Physics I - 1-2564Document3 pagesรายละเอียดของวิชา 315102-SC501001-General Physics I - 1-2564มัชฌิมา ลีซ้ายNo ratings yet
- มคอ.2 - วท.ม.สัตวศาสตร์ ฉ.2563เสนอ อว. update 24 June 2020Document147 pagesมคอ.2 - วท.ม.สัตวศาสตร์ ฉ.2563เสนอ อว. update 24 June 2020Chai YawatNo ratings yet
- การประชุมคณะกรรมการสถานศึกษา 23 ก.พ. 64Document17 pagesการประชุมคณะกรรมการสถานศึกษา 23 ก.พ. 64Puthiput ChotsutaworakulNo ratings yet
- ตารางห้องเรียนพิเศษ 1 - 2566Document9 pagesตารางห้องเรียนพิเศษ 1 - 2566bravepun28No ratings yet
- ระเบียบวาระการประชุมครู๕๕Document11 pagesระเบียบวาระการประชุมครู๕๕nuumbpNo ratings yet
- ตารางเรียน 2-65Document18 pagesตารางเรียน 2-65Adaris SupaNo ratings yet
- ปฏิทินการศึกษาโรงเรียนอาชีวะดอนบอสโก บ้านโป่งปีการศึกษา 2553Document8 pagesปฏิทินการศึกษาโรงเรียนอาชีวะดอนบอสโก บ้านโป่งปีการศึกษา 2553bsctech08No ratings yet
- ประมวลรายวิชาโลก ดาราศาสตร์ฯ ม.4 ภาค 2 2556 ว30104 PDFDocument4 pagesประมวลรายวิชาโลก ดาราศาสตร์ฯ ม.4 ภาค 2 2556 ว30104 PDFDezolate AlcNo ratings yet
- Scincem2 2Document350 pagesScincem2 2Montira SakaetongNo ratings yet
- ตารางสอบกลางภาค2 66Document6 pagesตารางสอบกลางภาค2 66Kanokwan KoedkrungNo ratings yet
- Mid2 2566new1Document5 pagesMid2 2566new1boonnapat.narinNo ratings yet
- แบบฝึกปริมาณสารสัมพันธ์2Document64 pagesแบบฝึกปริมาณสารสัมพันธ์2NoonTeachathanyakul67% (6)
- À À À °à ¡À À À À À À À À À 336-215 À À À À À À À À À À À À À À À À À À À À À © 2-2Document5 pagesÀ À À °à ¡À À À À À À À À À 336-215 À À À À À À À À À À À À À À À À À À À À À © 2-2vywave2No ratings yet
- Chapter 1Document98 pagesChapter 1BanyaphonNo ratings yet
- Bio60 170925043402 PDFDocument20 pagesBio60 170925043402 PDFวิสาร ศศิวิบูลย์วงศ์No ratings yet
- แต่งตั้งรับสมัครDocument10 pagesแต่งตั้งรับสมัครทิพยรัตน์ ทองจันทร์No ratings yet
- Course Syllabus 2758501 65-2 Jan 66Document9 pagesCourse Syllabus 2758501 65-2 Jan 66ploypilin pokinnantakulNo ratings yet
- Part II Manufacturing PDFDocument157 pagesPart II Manufacturing PDFChaiyuth ArmyforceNo ratings yet
- 02. ตารางสอบเค้าโครงวิจัย 3 บท ภาค 2 - 66Document3 pages02. ตารางสอบเค้าโครงวิจัย 3 บท ภาค 2 - 66koisamran17pelNo ratings yet
- Mee Mne EseDocument10 pagesMee Mne EseKornkanok PMNo ratings yet
- KMUNB Thesis 2558Document133 pagesKMUNB Thesis 2558Arkom ThaicharoenNo ratings yet
- TQF 3 QC1 SummaryDocument13 pagesTQF 3 QC1 SummaryT. SaritaNo ratings yet
- ตารางสอบปลายภาคเรียนที่ 2Document1 pageตารางสอบปลายภาคเรียนที่ 2อนัญญา สรวงกุดเรือNo ratings yet
- Prepare Book Reading TestDocument3 pagesPrepare Book Reading TestPerter DerminNo ratings yet
- Scincem2 1Document250 pagesScincem2 1Montira SakaetongNo ratings yet
- คู่มือครูรายวิชาพื้นฐานวิทยาศาสตร์และเทคโนโลยี ม.3 เล่ม 2 (Update 081164)Document383 pagesคู่มือครูรายวิชาพื้นฐานวิทยาศาสตร์และเทคโนโลยี ม.3 เล่ม 2 (Update 081164)39889 wichienmatuNo ratings yet
- รายละเอียดคอร์สเทอม1 67 ม1ถึงม3Document2 pagesรายละเอียดคอร์สเทอม1 67 ม1ถึงม3ครูพี่กาย โรงเรียนกวดวิชาวิคเตอร์No ratings yet
- Pdftimetable - 164405100087 8Document1 pagePdftimetable - 164405100087 8supatsara2420No ratings yet
- Dokumen - Tips Aaaaaaaaaaaaaaaaaaaaaaaaoe-Webmastermssqldatachemt2040108Document49 pagesDokumen - Tips Aaaaaaaaaaaaaaaaaaaaaaaaoe-Webmastermssqldatachemt2040108Nipaporn SimsomNo ratings yet
- ปรับแก้ 17 สค 63 กอ211ภาค1 2563 7Document4 pagesปรับแก้ 17 สค 63 กอ211ภาค1 2563 7กุลทรัพย์ ชัยอมรทรัพย์No ratings yet
- Anc 670103Document3 pagesAnc 670103zoo28444No ratings yet
- ภาพถ่ายหน้าจอ 2565-09-24 เวลา 00.01.57Document6 pagesภาพถ่ายหน้าจอ 2565-09-24 เวลา 00.01.57Kanokwan KoedkrungNo ratings yet
- คำสั่ง แต่งตั้งคณะกรรมการคุมสอบกลางภาคภาคเรียนที่๒Document14 pagesคำสั่ง แต่งตั้งคณะกรรมการคุมสอบกลางภาคภาคเรียนที่๒มนตรี จิตจักรNo ratings yet
- XF28 2555 2Document6 pagesXF28 2555 2Kun SlipperNo ratings yet
- 00 กำหนดการ-รุ่น24Document1 page00 กำหนดการ-รุ่น24เสกสรรค์ จันทร์สุขปลูกNo ratings yet
- Lecture 1 Physics-Of-Measurement-And-Vector (3383)Document37 pagesLecture 1 Physics-Of-Measurement-And-Vector (3383)Worachate AriyasubkamolNo ratings yet
- รายงานการวิเคราะห์ปริมาณรังสีสะสมของซากDocument163 pagesรายงานการวิเคราะห์ปริมาณรังสีสะสมของซากคุณครู ลั้นลาNo ratings yet
- หลักสูตรปรัชญาดุษฎีบัณฑิต สาขาวิชาเทคโนโลยีชีวภาพDocument67 pagesหลักสูตรปรัชญาดุษฎีบัณฑิต สาขาวิชาเทคโนโลยีชีวภาพมานพ อุดมศรีNo ratings yet
- 1 ตรวจสอบความเข้าใจ 1 - 7Document2 pages1 ตรวจสอบความเข้าใจ 1 - 7snualpeNo ratings yet
- Alevel64 - Yt - Eng NirinDocument29 pagesAlevel64 - Yt - Eng NirinsnualpeNo ratings yet
- No1 Grammar TOEIC GAT O-NETDocument27 pagesNo1 Grammar TOEIC GAT O-NETsnualpeNo ratings yet
- แผนที่ 4 การมองเห็นและความสว่างต่อสิ่งมีชีวิตDocument34 pagesแผนที่ 4 การมองเห็นและความสว่างต่อสิ่งมีชีวิตsnualpeNo ratings yet
- แผนที่ 2 การหักเหของแสงDocument43 pagesแผนที่ 2 การหักเหของแสงsnualpeNo ratings yet
- 1 130928043832 Phpapp02Document25 pages1 130928043832 Phpapp02snualpe100% (1)
- แผนที่ 3 การสะท้อนกลับหมดของแสง และประโยชน์การสะท้อน การหักเหDocument34 pagesแผนที่ 3 การสะท้อนกลับหมดของแสง และประโยชน์การสะท้อน การหักเหsnualpeNo ratings yet
- Conic 28-9-2555Document31 pagesConic 28-9-2555snualpeNo ratings yet
- แผนที่ 1 การสะท้อนของแสงDocument45 pagesแผนที่ 1 การสะท้อนของแสงsnualpeNo ratings yet
- - ม.ปลาย - ฟิสิกส์ - ไฟฟ้ากระแสตรง 1 19Document13 pages- ม.ปลาย - ฟิสิกส์ - ไฟฟ้ากระแสตรง 1 19snualpeNo ratings yet
- P 43027622133Document49 pagesP 43027622133snualpeNo ratings yet
- Complex NumberDocument42 pagesComplex NumbersnualpeNo ratings yet
- คู่มือครูเคมี เล่ม 2 PDFDocument223 pagesคู่มือครูเคมี เล่ม 2 PDFSatul Qalbai100% (1)
- 14ไฟฟ้าสถิตDocument33 pages14ไฟฟ้าสถิตsnualpeNo ratings yet
- math ม ต้น 2560 PDFDocument1 pagemath ม ต้น 2560 PDFsnualpeNo ratings yet
- คู่มือการใช้งานระบบ TCAS62 สำหรับผู้สมัคร V01 - 25611127 1: เปิด Browser แล้วระบุ URL: เลือกเมนู TCAS62 หรือระบุ URLDocument24 pagesคู่มือการใช้งานระบบ TCAS62 สำหรับผู้สมัคร V01 - 25611127 1: เปิด Browser แล้วระบุ URL: เลือกเมนู TCAS62 หรือระบุ URLsnualpeNo ratings yet
- ElectrostaticsDocument287 pagesElectrostaticssnualpeNo ratings yet
- การเคลื่อนที่ เรื่อง แนวตรง.pdf33 PDFDocument128 pagesการเคลื่อนที่ เรื่อง แนวตรง.pdf33 PDFsnualpe67% (3)
- 1วิธีเรียงสับเปลี่ยนและจัดหมู่p PDFDocument25 pages1วิธีเรียงสับเปลี่ยนและจัดหมู่p PDFsnualpeNo ratings yet
- การเคลื่อนที่แนวตรง PDFDocument5 pagesการเคลื่อนที่แนวตรง PDFsnualpeNo ratings yet
- แผนฯ หน้าที่พลเมืองฯ ม.4-6 เล่ม 2Document143 pagesแผนฯ หน้าที่พลเมืองฯ ม.4-6 เล่ม 2snualpe67% (6)
- การเคลื่อนมี่ 119 PDFDocument42 pagesการเคลื่อนมี่ 119 PDFsnualpeNo ratings yet
- การเคลื่อนมี่ 120 PDFDocument16 pagesการเคลื่อนมี่ 120 PDFsnualpeNo ratings yet
- Linear MotionDocument28 pagesLinear MotionsnualpeNo ratings yet
CHM 103s
CHM 103s
Uploaded by
snualpeCopyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
CHM 103s
CHM 103s
Uploaded by
snualpeCopyright:
Available Formats
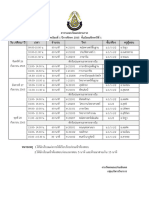
1
Course Title
Semester/ Year
Prerequisite
Lecturer
Examination
Time of Lecture
4
Weekday
Time of
Lecture
Room No.
Teaching Program
Department of Chemistry, Faculty of Science, KMUTT
CHM 103 Fundamental Chemistry ( 4)
2 / 2556
. .
28 2557 9.00 12.00 .
12 2557 9.00 12.00 .
MTE 1 A, MTE 1 B, PPT 1 A, PPT 1 B, PTE 1 A
Monday
Tuesday
10.30-12.20 .
Sc 2109
Wednesday
Thursday
Friday
9.30 10.20 .
Sc 2109
Course Description
Objectives
1.
2. /
Teaching Method :
1.
2. -
3.
Materials :
power points
Text Books :
1. Chang R., 2005, Chemistry (International Edition), 8th edition, McGraw Hill, Singapore.
2. McMurry J. and Fay R.C., 2004, Chemistry (International Edition), 4 th edition, Pearson
Prentice Hall, New Jersey.
References :
1. , 2541, 1 2,
Measurement :
(Quiz)
40
40
10
10
%
%
%
%
2
3
4
5
6
7
8
CHM103 (2/2556)
4 MTE 1 A, MTE 1 B, PPT 1 A, PPT 1 B, PTE 1 A
()
7 .. 57
Introduction & Basic of the Atomic Theory and Electronic .
Structure of Atoms (2)
10 .. 57 Basic of the Atomic Theory and Electronic Structure of
Atoms (1)
14 .. 57 Periodic Properties (2)
.
17 .. 57 Representative Elements (1)
.
21 .. 57 Representative Elements (1),
.
21 .. 57 Transition Metals (1)
.
24 .. 57 Transition Metals (1)
.
28 .. 57 Chemical Bonding (2)
.
31 .. 57 Chemical Bonding (1)
Quiz 1
4 .. 57
Chemical Bonding (2)
.
7 .. 57
Stoichiometry (1)
11 .. 57 Stoichiometry (2)
.
14 .. 57 Stoichiometry (1) ()
18 .. 57 Gases (2)
.
21 .. 57 Gases (1)
Quiz 2
28 .. 57
9.00 12.00 . Atomic Structure, Periodic Properties, Representative and
Transitions Elements, Chemical Bonding, Stoichiometry,
Gases
24 .. 57 4 .. 57
*** *** Quiz 2
Quiz 1 (1) Atomic Structure (2) Periodic Properties (3) Representative Elements,
Nonmetal and Transition Metals 30
Quiz 2 (1) Chemical Bonding (2) Stoichiometry 20
7 .. 57
()
Solid (1)
Solid (2)
10
11 .. 57 Solid (1) Liuid (1)
.
14 .. 57 Liuid (1)
11
18 .. 57 Solution (2)
.
21 .. 57 Chemical Kinetics (1)
12
25 .. 57 Chemical Kinetics (2)
.
28 .. 57 Chemical Kinetics (1)
Quiz 3
13
1 .. 57 Chemical Equilibrium (2)
.
4 .. 57 Chemical Equilibrium (1)
14
8 .. 57 Ionic Equilibrium (2)
.
11 .. 57 Ionic Equilibrium (1)
18 .. 57 ****** (1)
15
22 .. 57 Ionic Equilibrium (2)
.
25 .. 57 Electrochemistry (1)
16
29 .. 57 Electrochemistry (2)
.
2 .. 57
Electrochemistry (1)
Quiz 4
17-18
12 .. 2557
9.00 12.00 . Liquid-Solution, Solid, Chemical Kinetics,
Chemical Equilibrium, Ionic Equilibrium,
Electrochemistry
6 16 .. 57
*** *** Quiz 2
Quiz 3 (1) Liquid and Solution (2) Solid (3) Chemical Kinetics 30
Quiz 4 (1) Chemical Equilibrium (2) Ionic Equilibrium 20
You might also like
- หลักวิศวกรรมอาหาร 1Document7 pagesหลักวิศวกรรมอาหาร 1Sura C. JirNo ratings yet
- คู่มือครู เคมี เพิ่มเติม ม4 เล่ม 1 PDFDocument268 pagesคู่มือครู เคมี เพิ่มเติม ม4 เล่ม 1 PDFsnualpe89% (9)
- M460B1 PDFDocument172 pagesM460B1 PDFMamarine Wannasiri ThiphayamongkolNo ratings yet
- โครงการประเมินทักษะภาษาอังกฤษระดับประเทศ ครั้งที่ 1 Test Paper M1-3Document22 pagesโครงการประเมินทักษะภาษาอังกฤษระดับประเทศ ครั้งที่ 1 Test Paper M1-3snualpe100% (3)
- Scincem1 2Document324 pagesScincem1 2Montira SakaetongNo ratings yet
- เอกสารประกอบการสอน general physics I-rev1-5 PDFDocument199 pagesเอกสารประกอบการสอน general physics I-rev1-5 PDFNattawut NoreeNo ratings yet
- !!02207311 Eng-Hydrology Final-All 2557 PDFDocument403 pages!!02207311 Eng-Hydrology Final-All 2557 PDFสาธิต ปริ นทร์ทอง67% (3)
- แผนฯ หน้าที่พลเมืองฯ ม.4-6 เล่ม 1Document139 pagesแผนฯ หน้าที่พลเมืองฯ ม.4-6 เล่ม 1snualpe88% (8)
- รายละเอียดคอร์สsummer 63 ม5Document4 pagesรายละเอียดคอร์สsummer 63 ม5ครูพี่กาย โรงเรียนกวดวิชาวิคเตอร์No ratings yet
- Microsoft TeamDocument2 pagesMicrosoft Teamporamath33No ratings yet
- ตารางสอบวัดระดับด้านภาษาอังกฤษฯและดิจิทัลฉบับจริงDocument6 pagesตารางสอบวัดระดับด้านภาษาอังกฤษฯและดิจิทัลฉบับจริงP Danuphong ThitithananonNo ratings yet
- รายละเอียดคอร์สsummer 63 ม6Document4 pagesรายละเอียดคอร์สsummer 63 ม6ครูพี่กาย โรงเรียนกวดวิชาวิคเตอร์No ratings yet
- รายงานประชุมห้องเรียนพิเศษวิทยาศาสตร์ฯ ครั้งที่ - 2 - 2566Document4 pagesรายงานประชุมห้องเรียนพิเศษวิทยาศาสตร์ฯ ครั้งที่ - 2 - 2566Tutah RecordsNo ratings yet
- รายละเอียดคอร์สsummer 63 ม4Document4 pagesรายละเอียดคอร์สsummer 63 ม4ครูพี่กาย โรงเรียนกวดวิชาวิคเตอร์No ratings yet
- Course Outline - À À À À CHM 129 - 66 - LMSDocument5 pagesCourse Outline - À À À À CHM 129 - 66 - LMSaathitityNo ratings yet
- AMEDocument4 pagesAMEMek PunyawichNo ratings yet
- ปฏิทินสอบDocument7 pagesปฏิทินสอบจิรัฐิวัฒน์ เลาหวรรณธนะNo ratings yet
- 3. มฐ. และ ตชว. กลุ่มสาระการเรียนรู้วิทยาศาสตร์ (ฉบับปรับปรุง พ.ศ. 2560)Document273 pages3. มฐ. และ ตชว. กลุ่มสาระการเรียนรู้วิทยาศาสตร์ (ฉบับปรับปรุง พ.ศ. 2560)Scandoo DeeNo ratings yet
- A-Level 64 Phy วิชาฟิสิกส์ - TCAS66 BlueprintDocument1 pageA-Level 64 Phy วิชาฟิสิกส์ - TCAS66 Blueprint52mydjt2r8No ratings yet
- ตารางเรียน สาขาวิชาการสอนภาษาอังกฤษDocument4 pagesตารางเรียน สาขาวิชาการสอนภาษาอังกฤษUltimateNo ratings yet
- D 1Document277 pagesD 1ปรารถนา ปลื้มNo ratings yet
- à¸à¸³à¸à¸§à¸à¸£à¸²à¸¢à¸à¸·à¹à¸à¸à¸¹à¹à¸ªà¸³à¹à¸£à¹à¸à¸à¸²à¸£à¸¨à¸¶à¸à¸©à¸²-à¸à¸µà¸à¸²à¸£à¸¨à¸¶à¸à¸©à¸²-2564-à¸à¸£à¸¸à¸¨à¸²à¸ªà¸à¸£à¹Document55 pagesà¸à¸³à¸à¸§à¸à¸£à¸²à¸¢à¸à¸·à¹à¸à¸à¸¹à¹à¸ªà¸³à¹à¸£à¹à¸à¸à¸²à¸£à¸¨à¸¶à¸à¸©à¸²-à¸à¸µà¸à¸²à¸£à¸¨à¸¶à¸à¸©à¸²-2564-à¸à¸£à¸¸à¸¨à¸²à¸ªà¸à¸£à¹ploymaneerat.tNo ratings yet
- รายละเอียดของวิชา 315102-SC501001-General Physics I - 1-2564Document3 pagesรายละเอียดของวิชา 315102-SC501001-General Physics I - 1-2564มัชฌิมา ลีซ้ายNo ratings yet
- มคอ.2 - วท.ม.สัตวศาสตร์ ฉ.2563เสนอ อว. update 24 June 2020Document147 pagesมคอ.2 - วท.ม.สัตวศาสตร์ ฉ.2563เสนอ อว. update 24 June 2020Chai YawatNo ratings yet
- การประชุมคณะกรรมการสถานศึกษา 23 ก.พ. 64Document17 pagesการประชุมคณะกรรมการสถานศึกษา 23 ก.พ. 64Puthiput ChotsutaworakulNo ratings yet
- ตารางห้องเรียนพิเศษ 1 - 2566Document9 pagesตารางห้องเรียนพิเศษ 1 - 2566bravepun28No ratings yet
- ระเบียบวาระการประชุมครู๕๕Document11 pagesระเบียบวาระการประชุมครู๕๕nuumbpNo ratings yet
- ตารางเรียน 2-65Document18 pagesตารางเรียน 2-65Adaris SupaNo ratings yet
- ปฏิทินการศึกษาโรงเรียนอาชีวะดอนบอสโก บ้านโป่งปีการศึกษา 2553Document8 pagesปฏิทินการศึกษาโรงเรียนอาชีวะดอนบอสโก บ้านโป่งปีการศึกษา 2553bsctech08No ratings yet
- ประมวลรายวิชาโลก ดาราศาสตร์ฯ ม.4 ภาค 2 2556 ว30104 PDFDocument4 pagesประมวลรายวิชาโลก ดาราศาสตร์ฯ ม.4 ภาค 2 2556 ว30104 PDFDezolate AlcNo ratings yet
- Scincem2 2Document350 pagesScincem2 2Montira SakaetongNo ratings yet
- ตารางสอบกลางภาค2 66Document6 pagesตารางสอบกลางภาค2 66Kanokwan KoedkrungNo ratings yet
- Mid2 2566new1Document5 pagesMid2 2566new1boonnapat.narinNo ratings yet
- แบบฝึกปริมาณสารสัมพันธ์2Document64 pagesแบบฝึกปริมาณสารสัมพันธ์2NoonTeachathanyakul67% (6)
- À À À °à ¡À À À À À À À À À 336-215 À À À À À À À À À À À À À À À À À À À À À © 2-2Document5 pagesÀ À À °à ¡À À À À À À À À À 336-215 À À À À À À À À À À À À À À À À À À À À À © 2-2vywave2No ratings yet
- Chapter 1Document98 pagesChapter 1BanyaphonNo ratings yet
- Bio60 170925043402 PDFDocument20 pagesBio60 170925043402 PDFวิสาร ศศิวิบูลย์วงศ์No ratings yet
- แต่งตั้งรับสมัครDocument10 pagesแต่งตั้งรับสมัครทิพยรัตน์ ทองจันทร์No ratings yet
- Course Syllabus 2758501 65-2 Jan 66Document9 pagesCourse Syllabus 2758501 65-2 Jan 66ploypilin pokinnantakulNo ratings yet
- Part II Manufacturing PDFDocument157 pagesPart II Manufacturing PDFChaiyuth ArmyforceNo ratings yet
- 02. ตารางสอบเค้าโครงวิจัย 3 บท ภาค 2 - 66Document3 pages02. ตารางสอบเค้าโครงวิจัย 3 บท ภาค 2 - 66koisamran17pelNo ratings yet
- Mee Mne EseDocument10 pagesMee Mne EseKornkanok PMNo ratings yet
- KMUNB Thesis 2558Document133 pagesKMUNB Thesis 2558Arkom ThaicharoenNo ratings yet
- TQF 3 QC1 SummaryDocument13 pagesTQF 3 QC1 SummaryT. SaritaNo ratings yet
- ตารางสอบปลายภาคเรียนที่ 2Document1 pageตารางสอบปลายภาคเรียนที่ 2อนัญญา สรวงกุดเรือNo ratings yet
- Prepare Book Reading TestDocument3 pagesPrepare Book Reading TestPerter DerminNo ratings yet
- Scincem2 1Document250 pagesScincem2 1Montira SakaetongNo ratings yet
- คู่มือครูรายวิชาพื้นฐานวิทยาศาสตร์และเทคโนโลยี ม.3 เล่ม 2 (Update 081164)Document383 pagesคู่มือครูรายวิชาพื้นฐานวิทยาศาสตร์และเทคโนโลยี ม.3 เล่ม 2 (Update 081164)39889 wichienmatuNo ratings yet
- รายละเอียดคอร์สเทอม1 67 ม1ถึงม3Document2 pagesรายละเอียดคอร์สเทอม1 67 ม1ถึงม3ครูพี่กาย โรงเรียนกวดวิชาวิคเตอร์No ratings yet
- Pdftimetable - 164405100087 8Document1 pagePdftimetable - 164405100087 8supatsara2420No ratings yet
- Dokumen - Tips Aaaaaaaaaaaaaaaaaaaaaaaaoe-Webmastermssqldatachemt2040108Document49 pagesDokumen - Tips Aaaaaaaaaaaaaaaaaaaaaaaaoe-Webmastermssqldatachemt2040108Nipaporn SimsomNo ratings yet
- ปรับแก้ 17 สค 63 กอ211ภาค1 2563 7Document4 pagesปรับแก้ 17 สค 63 กอ211ภาค1 2563 7กุลทรัพย์ ชัยอมรทรัพย์No ratings yet
- Anc 670103Document3 pagesAnc 670103zoo28444No ratings yet
- ภาพถ่ายหน้าจอ 2565-09-24 เวลา 00.01.57Document6 pagesภาพถ่ายหน้าจอ 2565-09-24 เวลา 00.01.57Kanokwan KoedkrungNo ratings yet
- คำสั่ง แต่งตั้งคณะกรรมการคุมสอบกลางภาคภาคเรียนที่๒Document14 pagesคำสั่ง แต่งตั้งคณะกรรมการคุมสอบกลางภาคภาคเรียนที่๒มนตรี จิตจักรNo ratings yet
- XF28 2555 2Document6 pagesXF28 2555 2Kun SlipperNo ratings yet
- 00 กำหนดการ-รุ่น24Document1 page00 กำหนดการ-รุ่น24เสกสรรค์ จันทร์สุขปลูกNo ratings yet
- Lecture 1 Physics-Of-Measurement-And-Vector (3383)Document37 pagesLecture 1 Physics-Of-Measurement-And-Vector (3383)Worachate AriyasubkamolNo ratings yet
- รายงานการวิเคราะห์ปริมาณรังสีสะสมของซากDocument163 pagesรายงานการวิเคราะห์ปริมาณรังสีสะสมของซากคุณครู ลั้นลาNo ratings yet
- หลักสูตรปรัชญาดุษฎีบัณฑิต สาขาวิชาเทคโนโลยีชีวภาพDocument67 pagesหลักสูตรปรัชญาดุษฎีบัณฑิต สาขาวิชาเทคโนโลยีชีวภาพมานพ อุดมศรีNo ratings yet
- 1 ตรวจสอบความเข้าใจ 1 - 7Document2 pages1 ตรวจสอบความเข้าใจ 1 - 7snualpeNo ratings yet
- Alevel64 - Yt - Eng NirinDocument29 pagesAlevel64 - Yt - Eng NirinsnualpeNo ratings yet
- No1 Grammar TOEIC GAT O-NETDocument27 pagesNo1 Grammar TOEIC GAT O-NETsnualpeNo ratings yet
- แผนที่ 4 การมองเห็นและความสว่างต่อสิ่งมีชีวิตDocument34 pagesแผนที่ 4 การมองเห็นและความสว่างต่อสิ่งมีชีวิตsnualpeNo ratings yet
- แผนที่ 2 การหักเหของแสงDocument43 pagesแผนที่ 2 การหักเหของแสงsnualpeNo ratings yet
- 1 130928043832 Phpapp02Document25 pages1 130928043832 Phpapp02snualpe100% (1)
- แผนที่ 3 การสะท้อนกลับหมดของแสง และประโยชน์การสะท้อน การหักเหDocument34 pagesแผนที่ 3 การสะท้อนกลับหมดของแสง และประโยชน์การสะท้อน การหักเหsnualpeNo ratings yet
- Conic 28-9-2555Document31 pagesConic 28-9-2555snualpeNo ratings yet
- แผนที่ 1 การสะท้อนของแสงDocument45 pagesแผนที่ 1 การสะท้อนของแสงsnualpeNo ratings yet
- - ม.ปลาย - ฟิสิกส์ - ไฟฟ้ากระแสตรง 1 19Document13 pages- ม.ปลาย - ฟิสิกส์ - ไฟฟ้ากระแสตรง 1 19snualpeNo ratings yet
- P 43027622133Document49 pagesP 43027622133snualpeNo ratings yet
- Complex NumberDocument42 pagesComplex NumbersnualpeNo ratings yet
- คู่มือครูเคมี เล่ม 2 PDFDocument223 pagesคู่มือครูเคมี เล่ม 2 PDFSatul Qalbai100% (1)
- 14ไฟฟ้าสถิตDocument33 pages14ไฟฟ้าสถิตsnualpeNo ratings yet
- math ม ต้น 2560 PDFDocument1 pagemath ม ต้น 2560 PDFsnualpeNo ratings yet
- คู่มือการใช้งานระบบ TCAS62 สำหรับผู้สมัคร V01 - 25611127 1: เปิด Browser แล้วระบุ URL: เลือกเมนู TCAS62 หรือระบุ URLDocument24 pagesคู่มือการใช้งานระบบ TCAS62 สำหรับผู้สมัคร V01 - 25611127 1: เปิด Browser แล้วระบุ URL: เลือกเมนู TCAS62 หรือระบุ URLsnualpeNo ratings yet
- ElectrostaticsDocument287 pagesElectrostaticssnualpeNo ratings yet
- การเคลื่อนที่ เรื่อง แนวตรง.pdf33 PDFDocument128 pagesการเคลื่อนที่ เรื่อง แนวตรง.pdf33 PDFsnualpe67% (3)
- 1วิธีเรียงสับเปลี่ยนและจัดหมู่p PDFDocument25 pages1วิธีเรียงสับเปลี่ยนและจัดหมู่p PDFsnualpeNo ratings yet
- การเคลื่อนที่แนวตรง PDFDocument5 pagesการเคลื่อนที่แนวตรง PDFsnualpeNo ratings yet
- แผนฯ หน้าที่พลเมืองฯ ม.4-6 เล่ม 2Document143 pagesแผนฯ หน้าที่พลเมืองฯ ม.4-6 เล่ม 2snualpe67% (6)
- การเคลื่อนมี่ 119 PDFDocument42 pagesการเคลื่อนมี่ 119 PDFsnualpeNo ratings yet
- การเคลื่อนมี่ 120 PDFDocument16 pagesการเคลื่อนมี่ 120 PDFsnualpeNo ratings yet
- Linear MotionDocument28 pagesLinear MotionsnualpeNo ratings yet