Professional Documents
Culture Documents
0 ratings0% found this document useful (0 votes)
26 viewsMass-Moles Go
Mass-Moles Go
Uploaded by
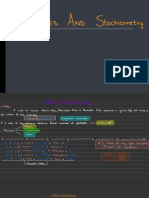
api-291560513This document provides instructions for converting between mass and moles of a substance by using Avogadro's constant of 6.022 x 1023 particles. To determine moles from mass, look up the substance on the periodic table to find its molar mass and divide the mass by that value. To determine mass from moles, multiply the number of moles by the molar mass of the substance found on the periodic table.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
You might also like
- Week12 Mole Student 2019Document35 pagesWeek12 Mole Student 2019api-4915646430% (1)
- 1st Sec Catalyst 4 Unit 2Document1 page1st Sec Catalyst 4 Unit 2yassinyaser95No ratings yet
- Chapter 1 PensyarahDocument56 pagesChapter 1 PensyarahAdi BaddNo ratings yet
- Chapter 2 Pensyarah - Stoichiometry of Formulas and EquationsDocument58 pagesChapter 2 Pensyarah - Stoichiometry of Formulas and Equationsnadiahchehussin24No ratings yet
- Stoichiometry of Formulas and EquationsDocument45 pagesStoichiometry of Formulas and EquationsNitha CwectiaNo ratings yet
- Moles and StoichiometryDocument46 pagesMoles and StoichiometryInspector Chulbul PandayNo ratings yet
- Chemistry Unit 4 MoleDocument1 pageChemistry Unit 4 MoleAdrián GarcíaNo ratings yet
- ACFrOgAsZJDlN9z fZja3YuD8P628as3XS-D8W2CT6nF02noP kYFimqGNFSpFwsZ0ECWDWxzBUO0JZs1f9RtWfcbdKro90aVpvzRB76QkBIect860ffb-HOv8djE6Xs0DH497mKXGfDU3ssMDOoDocument8 pagesACFrOgAsZJDlN9z fZja3YuD8P628as3XS-D8W2CT6nF02noP kYFimqGNFSpFwsZ0ECWDWxzBUO0JZs1f9RtWfcbdKro90aVpvzRB76QkBIect860ffb-HOv8djE6Xs0DH497mKXGfDU3ssMDOoAinalnaem GhidariNo ratings yet
- Chem 12 (2nd) PDFDocument36 pagesChem 12 (2nd) PDFRyan100% (1)
- Univariate Analysis of Variance: NotesDocument5 pagesUnivariate Analysis of Variance: NotesSiti AisyahNo ratings yet
- Worksheet Package 2.7-2.10Document13 pagesWorksheet Package 2.7-2.10Waynessboro HarleyNo ratings yet
- Table of IQS 2270 Part 11Document4 pagesTable of IQS 2270 Part 11Sudhanandh VSNo ratings yet
- Notes 5 7Document3 pagesNotes 5 7Dimas Hakri LenaldiNo ratings yet
- STR STR 613 613: Advanced: Advanced Numerical Analysis Numerical AnalysisDocument5 pagesSTR STR 613 613: Advanced: Advanced Numerical Analysis Numerical AnalysisHamza El-masryNo ratings yet
- Screening Analysis EXPERIMENTDocument10 pagesScreening Analysis EXPERIMENTJohnNo ratings yet
- Answers - H2 Topical Chemistry 2014Document99 pagesAnswers - H2 Topical Chemistry 2014Ruel Arila Jr.No ratings yet
- Detergency Performances of Liquid Detergent FormulationsDocument2 pagesDetergency Performances of Liquid Detergent FormulationsFaisal IqbalNo ratings yet
- Sample CalculationDocument1 pageSample CalculationElizaga ElizagaNo ratings yet
- Completed Roadmap To Moles OrganizerDocument1 pageCompleted Roadmap To Moles Organizerapi-481387154No ratings yet
- Mathongo Solution Some Basic Concepts of Chemistry Jee Main 2021 March Chapterwise obm9Ha1LeuPr5bOFa2JuDocument4 pagesMathongo Solution Some Basic Concepts of Chemistry Jee Main 2021 March Chapterwise obm9Ha1LeuPr5bOFa2JuSTUDY FOR JEENo ratings yet
- Calc BC 6.14 PacketDocument4 pagesCalc BC 6.14 PacketWilliam MaNo ratings yet
- Practical TipsDocument5 pagesPractical TipsPari KaleNo ratings yet
- On Equivariant Poincar e Duality, Gysin Morphisms and Euler ClassesDocument80 pagesOn Equivariant Poincar e Duality, Gysin Morphisms and Euler ClassesHuWalterNo ratings yet
- GronwallDocument197 pagesGronwallChương Quách VănNo ratings yet
- DataBase TransPlants BIL161Document6 pagesDataBase TransPlants BIL161Mariana TabaresNo ratings yet
- Academic Vocabulary-MolesDocument1 pageAcademic Vocabulary-Molesapi-291560513No ratings yet
- What Is A Mole SummativeDocument8 pagesWhat Is A Mole Summativeapi-291560513No ratings yet
- Conversion GoDocument1 pageConversion Goapi-291560513No ratings yet
- Lab Report Rubric: Score Points 100% 85% 80% 70% 0%Document4 pagesLab Report Rubric: Score Points 100% 85% 80% 70% 0%api-291560513No ratings yet
- 1 Making ConversionsDocument23 pages1 Making Conversionsapi-291560513No ratings yet
- University of BahrainDocument35 pagesUniversity of Bahrainapi-291560513No ratings yet
- Photovoltaic Cell Close ReadDocument3 pagesPhotovoltaic Cell Close Readapi-291560513No ratings yet
- Inquiry Worksheet: Name - Period: - Date: - What Do You Know? 1Document5 pagesInquiry Worksheet: Name - Period: - Date: - What Do You Know? 1api-291560513No ratings yet
- DSSC Lesson PlanDocument3 pagesDSSC Lesson Planapi-291560513No ratings yet
- ArgumentationDocument2 pagesArgumentationapi-291560513No ratings yet
- Scientific ArgumentationDocument11 pagesScientific Argumentationapi-291560513No ratings yet
- Summative PadletDocument3 pagesSummative Padletapi-291560513No ratings yet
- M M Lab Lesson PlanDocument4 pagesM M Lab Lesson Planapi-291560513No ratings yet
- Informal Argumentation FormatDocument2 pagesInformal Argumentation Formatapi-291560513No ratings yet
- Academic Vocabulary-MolesDocument1 pageAcademic Vocabulary-Molesapi-291560513No ratings yet
Mass-Moles Go
Mass-Moles Go
Uploaded by
api-2915605130 ratings0% found this document useful (0 votes)
26 views1 pageThis document provides instructions for converting between mass and moles of a substance by using Avogadro's constant of 6.022 x 1023 particles. To determine moles from mass, look up the substance on the periodic table to find its molar mass and divide the mass by that value. To determine mass from moles, multiply the number of moles by the molar mass of the substance found on the periodic table.
Original Description:
Original Title
mass-moles go
Copyright
© © All Rights Reserved
Available Formats
PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentThis document provides instructions for converting between mass and moles of a substance by using Avogadro's constant of 6.022 x 1023 particles. To determine moles from mass, look up the substance on the periodic table to find its molar mass and divide the mass by that value. To determine mass from moles, multiply the number of moles by the molar mass of the substance found on the periodic table.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
Download as pdf or txt
0 ratings0% found this document useful (0 votes)
26 views1 pageMass-Moles Go
Mass-Moles Go
Uploaded by
api-291560513This document provides instructions for converting between mass and moles of a substance by using Avogadro's constant of 6.022 x 1023 particles. To determine moles from mass, look up the substance on the periodic table to find its molar mass and divide the mass by that value. To determine mass from moles, multiply the number of moles by the molar mass of the substance found on the periodic table.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
Download as pdf or txt
You are on page 1of 1
Mass in Grams
Moles of ______
Use Periodic
Table
Use Periodic Table
6.022 x 1023
(Avogadro's Constant)
Particles (Things)
You might also like
- Week12 Mole Student 2019Document35 pagesWeek12 Mole Student 2019api-4915646430% (1)
- 1st Sec Catalyst 4 Unit 2Document1 page1st Sec Catalyst 4 Unit 2yassinyaser95No ratings yet
- Chapter 1 PensyarahDocument56 pagesChapter 1 PensyarahAdi BaddNo ratings yet
- Chapter 2 Pensyarah - Stoichiometry of Formulas and EquationsDocument58 pagesChapter 2 Pensyarah - Stoichiometry of Formulas and Equationsnadiahchehussin24No ratings yet
- Stoichiometry of Formulas and EquationsDocument45 pagesStoichiometry of Formulas and EquationsNitha CwectiaNo ratings yet
- Moles and StoichiometryDocument46 pagesMoles and StoichiometryInspector Chulbul PandayNo ratings yet
- Chemistry Unit 4 MoleDocument1 pageChemistry Unit 4 MoleAdrián GarcíaNo ratings yet
- ACFrOgAsZJDlN9z fZja3YuD8P628as3XS-D8W2CT6nF02noP kYFimqGNFSpFwsZ0ECWDWxzBUO0JZs1f9RtWfcbdKro90aVpvzRB76QkBIect860ffb-HOv8djE6Xs0DH497mKXGfDU3ssMDOoDocument8 pagesACFrOgAsZJDlN9z fZja3YuD8P628as3XS-D8W2CT6nF02noP kYFimqGNFSpFwsZ0ECWDWxzBUO0JZs1f9RtWfcbdKro90aVpvzRB76QkBIect860ffb-HOv8djE6Xs0DH497mKXGfDU3ssMDOoAinalnaem GhidariNo ratings yet
- Chem 12 (2nd) PDFDocument36 pagesChem 12 (2nd) PDFRyan100% (1)
- Univariate Analysis of Variance: NotesDocument5 pagesUnivariate Analysis of Variance: NotesSiti AisyahNo ratings yet
- Worksheet Package 2.7-2.10Document13 pagesWorksheet Package 2.7-2.10Waynessboro HarleyNo ratings yet
- Table of IQS 2270 Part 11Document4 pagesTable of IQS 2270 Part 11Sudhanandh VSNo ratings yet
- Notes 5 7Document3 pagesNotes 5 7Dimas Hakri LenaldiNo ratings yet
- STR STR 613 613: Advanced: Advanced Numerical Analysis Numerical AnalysisDocument5 pagesSTR STR 613 613: Advanced: Advanced Numerical Analysis Numerical AnalysisHamza El-masryNo ratings yet
- Screening Analysis EXPERIMENTDocument10 pagesScreening Analysis EXPERIMENTJohnNo ratings yet
- Answers - H2 Topical Chemistry 2014Document99 pagesAnswers - H2 Topical Chemistry 2014Ruel Arila Jr.No ratings yet
- Detergency Performances of Liquid Detergent FormulationsDocument2 pagesDetergency Performances of Liquid Detergent FormulationsFaisal IqbalNo ratings yet
- Sample CalculationDocument1 pageSample CalculationElizaga ElizagaNo ratings yet
- Completed Roadmap To Moles OrganizerDocument1 pageCompleted Roadmap To Moles Organizerapi-481387154No ratings yet
- Mathongo Solution Some Basic Concepts of Chemistry Jee Main 2021 March Chapterwise obm9Ha1LeuPr5bOFa2JuDocument4 pagesMathongo Solution Some Basic Concepts of Chemistry Jee Main 2021 March Chapterwise obm9Ha1LeuPr5bOFa2JuSTUDY FOR JEENo ratings yet
- Calc BC 6.14 PacketDocument4 pagesCalc BC 6.14 PacketWilliam MaNo ratings yet
- Practical TipsDocument5 pagesPractical TipsPari KaleNo ratings yet
- On Equivariant Poincar e Duality, Gysin Morphisms and Euler ClassesDocument80 pagesOn Equivariant Poincar e Duality, Gysin Morphisms and Euler ClassesHuWalterNo ratings yet
- GronwallDocument197 pagesGronwallChương Quách VănNo ratings yet
- DataBase TransPlants BIL161Document6 pagesDataBase TransPlants BIL161Mariana TabaresNo ratings yet
- Academic Vocabulary-MolesDocument1 pageAcademic Vocabulary-Molesapi-291560513No ratings yet
- What Is A Mole SummativeDocument8 pagesWhat Is A Mole Summativeapi-291560513No ratings yet
- Conversion GoDocument1 pageConversion Goapi-291560513No ratings yet
- Lab Report Rubric: Score Points 100% 85% 80% 70% 0%Document4 pagesLab Report Rubric: Score Points 100% 85% 80% 70% 0%api-291560513No ratings yet
- 1 Making ConversionsDocument23 pages1 Making Conversionsapi-291560513No ratings yet
- University of BahrainDocument35 pagesUniversity of Bahrainapi-291560513No ratings yet
- Photovoltaic Cell Close ReadDocument3 pagesPhotovoltaic Cell Close Readapi-291560513No ratings yet
- Inquiry Worksheet: Name - Period: - Date: - What Do You Know? 1Document5 pagesInquiry Worksheet: Name - Period: - Date: - What Do You Know? 1api-291560513No ratings yet
- DSSC Lesson PlanDocument3 pagesDSSC Lesson Planapi-291560513No ratings yet
- ArgumentationDocument2 pagesArgumentationapi-291560513No ratings yet
- Scientific ArgumentationDocument11 pagesScientific Argumentationapi-291560513No ratings yet
- Summative PadletDocument3 pagesSummative Padletapi-291560513No ratings yet
- M M Lab Lesson PlanDocument4 pagesM M Lab Lesson Planapi-291560513No ratings yet
- Informal Argumentation FormatDocument2 pagesInformal Argumentation Formatapi-291560513No ratings yet
- Academic Vocabulary-MolesDocument1 pageAcademic Vocabulary-Molesapi-291560513No ratings yet