Professional Documents
Culture Documents
Physics
Physics
Uploaded by
Ainin Akasyah Aziz0 ratings0% found this document useful (0 votes)
9 views4 pagesThermal equilibrium occurs when two objects in contact transfer heat such that they reach the same temperature and maintain it without further transfer to other objects. Thermal equilibrium is the subject of the Zeroth Law of Thermodynamics, which states that if two systems are each in thermal equilibrium with a third system, they are in thermal equilibrium with each other. Heat transfer can occur via conduction within a material, convection through mass motion of a fluid, or radiation through electromagnetic wave emission.
Original Description:
huhuhuhuhu
Copyright
© © All Rights Reserved
Available Formats
DOCX, PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentThermal equilibrium occurs when two objects in contact transfer heat such that they reach the same temperature and maintain it without further transfer to other objects. Thermal equilibrium is the subject of the Zeroth Law of Thermodynamics, which states that if two systems are each in thermal equilibrium with a third system, they are in thermal equilibrium with each other. Heat transfer can occur via conduction within a material, convection through mass motion of a fluid, or radiation through electromagnetic wave emission.
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
Download as docx, pdf, or txt
0 ratings0% found this document useful (0 votes)
9 views4 pagesPhysics
Physics
Uploaded by
Ainin Akasyah AzizThermal equilibrium occurs when two objects in contact transfer heat such that they reach the same temperature and maintain it without further transfer to other objects. Thermal equilibrium is the subject of the Zeroth Law of Thermodynamics, which states that if two systems are each in thermal equilibrium with a third system, they are in thermal equilibrium with each other. Heat transfer can occur via conduction within a material, convection through mass motion of a fluid, or radiation through electromagnetic wave emission.
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
Download as docx, pdf, or txt
You are on page 1of 4
THERMAL EQUILIBRIUM
A higher temperature object which is in
contact with a lower temperature object
will transfer heat to the lower
temperature object
The objects will approach the same temperature,
and in the absence of loss to other objects, they
will then maintain a constant temperature. They
are then said to be in thermal equilibrium.
Thermal equilibrium is the subject of the Zeroth
Law of Thermodynamics(two systems are at the
same time in thermal equilibrium with a third
system, they are in thermal equilibrium)
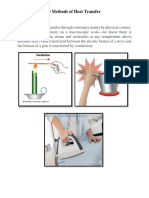
CONDUCTION is heat transfer by means of
molecular agitation within a material without any
motion of the material as a whole.
CONVECTION is heat transfer by mass motion
of a fluid such as air or water when the heated fluid is
caused to move away from the source of heat, carrying
energy with it.
RADIATION is heat transfer by the emission of
electromagnetic waves which carry energy away from
the emmiting object.
You might also like
- Reflection "Heat Transfer"Document2 pagesReflection "Heat Transfer"Lorie Joy De VillaNo ratings yet
- Thermal Energy TransferDocument13 pagesThermal Energy Transfernadamohey99No ratings yet
- HEAT WPLDocument9 pagesHEAT WPLKaira Shin MayngoNo ratings yet
- Heat - Grade 7Document32 pagesHeat - Grade 7ChelleNo ratings yet
- Mod 2 Part 2 ThermoDocument16 pagesMod 2 Part 2 ThermoJohn Rey DecanoNo ratings yet
- .... Yousef's Physics Project....Document4 pages.... Yousef's Physics Project....Yousef Abdel HamidNo ratings yet
- Thermal Engineering Thermal Energy Heat Thermal Conduction Thermal Convection Thermal Radiation Phase ChangesDocument1 pageThermal Engineering Thermal Energy Heat Thermal Conduction Thermal Convection Thermal Radiation Phase ChangesMat ComiaNo ratings yet
- Thermodynamics HandoutsDocument5 pagesThermodynamics HandoutsEnigma YTNo ratings yet
- Name:: Balen Karim MR - Ayad HussenDocument6 pagesName:: Balen Karim MR - Ayad HussenBalen KarimNo ratings yet
- Heat Is The Transfer of Energy From One Part of A Substance ToDocument2 pagesHeat Is The Transfer of Energy From One Part of A Substance ToJhune Dominique GalangNo ratings yet
- Project in Science: Charrie R. Nuñez Jercel D. Arcilla Ehra Lianne FernandezDocument4 pagesProject in Science: Charrie R. Nuñez Jercel D. Arcilla Ehra Lianne FernandezBrigs EckaNo ratings yet
- HeatDocument2 pagesHeatsharihNo ratings yet
- Mee407 1 20232024Document21 pagesMee407 1 20232024Adeboye BusayoNo ratings yet
- Written Report LabDocument3 pagesWritten Report LabDaniel CaquilalaNo ratings yet
- Menoufia UniversityDocument4 pagesMenoufia Universityelking.ahmed1562002No ratings yet
- Heat TransfeerDocument26 pagesHeat TransfeerRonald AlisingNo ratings yet
- Heat and Mass Transfer Module 1 Lesson 1Document10 pagesHeat and Mass Transfer Module 1 Lesson 1cool kidNo ratings yet
- Science Subject For High School Heat TransferDocument21 pagesScience Subject For High School Heat Transferlj8075702No ratings yet
- Heat Transfer - G6 Lesson NotesDocument3 pagesHeat Transfer - G6 Lesson NotesJung Ahn HongNo ratings yet
- What Is HeatDocument2 pagesWhat Is HeatNagen TamilNo ratings yet
- HEATDocument13 pagesHEATJemabel RosarioNo ratings yet
- Zeroth Law of ThermodynamicsDocument4 pagesZeroth Law of Thermodynamicsmideepak007100% (1)
- Heat and Temperatu-ReDocument11 pagesHeat and Temperatu-ReFedora MefiaNo ratings yet
- S ThermochemistryDocument15 pagesS ThermochemistryJoselito UbaldoNo ratings yet
- AP PresentationDocument16 pagesAP PresentationRohit RajNo ratings yet
- Assignment Solution Saifullah UW-17-EE-BSC-059 Sol Q#1 (A) : ConductionDocument3 pagesAssignment Solution Saifullah UW-17-EE-BSC-059 Sol Q#1 (A) : ConductionSaif UllahNo ratings yet
- Difference Between Conduction, Convection and RadiationDocument10 pagesDifference Between Conduction, Convection and RadiationMaria Alejandra Diaz RomeroNo ratings yet
- Heat TransferDocument2 pagesHeat Transfervijay kumar honnaliNo ratings yet
- Lesson 7: Thermal EnergyDocument19 pagesLesson 7: Thermal EnergyAthena ArevaloNo ratings yet
- Lesson-1: TemperatureDocument59 pagesLesson-1: Temperaturemehaksinghgill098No ratings yet
- 1 IntroduccionConducciónDocument70 pages1 IntroduccionConducciónJhonfrey Bautista EscobarNo ratings yet
- Heat ActivityDocument3 pagesHeat ActivitySittie Jivaiza Malanji MalantocNo ratings yet
- Physics Grade 12 Year of 2015 Unit One Short NoteDocument13 pagesPhysics Grade 12 Year of 2015 Unit One Short NoteBahiruNo ratings yet
- Diccionario en InglésDocument9 pagesDiccionario en InglésLaura Giselle RomeroNo ratings yet
- ThermodynamicsDocument11 pagesThermodynamicsJan Dave DeocampoNo ratings yet
- Introduction and Basic Concepts: Heat and Mass TransferDocument40 pagesIntroduction and Basic Concepts: Heat and Mass TransferHugo MartinsNo ratings yet
- HeatDocument3 pagesHeatSheryl BorromeoNo ratings yet
- Heat Capacity of The Matters. It Is The Distinguishing Property ofDocument3 pagesHeat Capacity of The Matters. It Is The Distinguishing Property ofjaihogoluNo ratings yet
- Heat Transfer OperationsDocument10 pagesHeat Transfer OperationsShafique AhmedNo ratings yet
- WK2BSDocument3 pagesWK2BSojo ayodeji johnsonNo ratings yet
- Introduction and Basic Concepts: Heat and Mass Transfer: Fundamentals & ApplicationsDocument30 pagesIntroduction and Basic Concepts: Heat and Mass Transfer: Fundamentals & Applicationsdang kyNo ratings yet
- Black BodyDocument4 pagesBlack BodyRafael SantosNo ratings yet
- C Chemistry For EngineeringDocument9 pagesC Chemistry For EngineeringCathy LevreNo ratings yet
- Physics Thermal Equilibrium LessonDocument24 pagesPhysics Thermal Equilibrium LessonRufat IsmayilovNo ratings yet
- Introduction: Modes and Mechanisms of Heat Transfer: Lesson Plan No.1Document2 pagesIntroduction: Modes and Mechanisms of Heat Transfer: Lesson Plan No.1Ashish jauhariNo ratings yet
- Explain in Your Own Words What Heat Is and Explain The Difference Between Heat and EnergyDocument1 pageExplain in Your Own Words What Heat Is and Explain The Difference Between Heat and EnergypashupatishahNo ratings yet
- Modes of Heat TransferDocument6 pagesModes of Heat TransferfaisalNo ratings yet
- ACTIVIDAD DE CIENCIA DE LOS MATERIALES - Ingles y Español - Jose Daniel BallestasDocument15 pagesACTIVIDAD DE CIENCIA DE LOS MATERIALES - Ingles y Español - Jose Daniel BallestasJose Daniel BallestasNo ratings yet
- Quarter 3 Week 8: Thermal EnergyDocument3 pagesQuarter 3 Week 8: Thermal EnergyarinNo ratings yet
- Heat Transfer: Dr. Abdulbari AlboraniDocument36 pagesHeat Transfer: Dr. Abdulbari Alboraniنزار الدهاميNo ratings yet
- Physics - WRITTEN REPORT - WPS OfficeDocument5 pagesPhysics - WRITTEN REPORT - WPS OfficeLU NANo ratings yet
- Experiment #1Document8 pagesExperiment #1Talha AhmadNo ratings yet
- Heat TransferDocument2 pagesHeat TransferSakureNo ratings yet
- Heat TransferDocument12 pagesHeat Transferعبدالمحسن علي ENo ratings yet
- Heat TransferDocument3 pagesHeat TransferRoscell Ducusin ReyesNo ratings yet
- Unit - 1: (B) Difference Between Thermodynamcis and Heat TransferDocument66 pagesUnit - 1: (B) Difference Between Thermodynamcis and Heat TransferSayyadh Rahamath Baba100% (1)
- Fundamentals of Heat TransferDocument1 pageFundamentals of Heat TransferVarun KavvampalliNo ratings yet
- Heat Transfer Conduction Convection RadiationDocument3 pagesHeat Transfer Conduction Convection RadiationAMJAD100% (1)