Professional Documents
Culture Documents
Quality Assurance Procedures Index: Qap Rev Title
Quality Assurance Procedures Index: Qap Rev Title
Uploaded by
shifa0 ratings0% found this document useful (0 votes)
9 views1 pageThis document outlines quality assurance procedures for an organization, including procedures for controlling documents and records, management commitment and review, competence and training, purchasing, identification and traceability, inspection of equipment and materials, audits, handling non-conforming materials, and continual improvement.
Original Description:
QAP
Original Title
QAP
Copyright
© © All Rights Reserved
Available Formats
DOCX, PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentThis document outlines quality assurance procedures for an organization, including procedures for controlling documents and records, management commitment and review, competence and training, purchasing, identification and traceability, inspection of equipment and materials, audits, handling non-conforming materials, and continual improvement.
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
Download as docx, pdf, or txt
0 ratings0% found this document useful (0 votes)
9 views1 pageQuality Assurance Procedures Index: Qap Rev Title
Quality Assurance Procedures Index: Qap Rev Title
Uploaded by
shifaThis document outlines quality assurance procedures for an organization, including procedures for controlling documents and records, management commitment and review, competence and training, purchasing, identification and traceability, inspection of equipment and materials, audits, handling non-conforming materials, and continual improvement.
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
Download as docx, pdf, or txt
You are on page 1of 1
QUALITY ASSURANCE PROCEDURES INDEX
QAP REV TITLE
4.2.3 A Control of Documents
4.2.4 A Control of Records
5.1 A Management Commitment
5.2 A Customer Focus
5.4 A Planning
5.6 A Management Review
6.2.2 A Competence, Awareness & Training
6.4 N/C F.O.D. Detection and Prevention
7.2.1 A Contract Review
7.4 B Purchasing
7.5.3 A Identification and Traceability
7.5.4 A Preservation of Customer Supplied Material
7.6 A Control of Inspection, Measuring & Test Equipment
8.2 A Test & Inspection Methods
8.2.1 A Customer Satisfaction
8.2.2 A Internal Quality Audits
8.2.4 A Receiving Inspection
8.2.4A A First Article Inspection
8.2.4B A In-Process Inspection
8.2.4C A Final Inspection
8.3 A Control of Non-Conforming Material
8.3A A Control and Customer Notification of Suspect Material
8.5.1 A Continual Improvement
8.5.2 A Corrective Action
8.5.3 A Preventive Action
You might also like
- Documents To Be Elaborated in Order To Meet ISO 22716 PDFDocument3 pagesDocuments To Be Elaborated in Order To Meet ISO 22716 PDFamayorga86% (22)
- Content Audits and Inventories: A Handbook for Content AnalysisFrom EverandContent Audits and Inventories: A Handbook for Content AnalysisNo ratings yet
- ISO 9001 Quality Manual TemplateDocument43 pagesISO 9001 Quality Manual TemplateNC Rigor Luis93% (14)
- Internal Audit MatrixDocument1 pageInternal Audit MatrixTravisNo ratings yet
- IATF 16949 Gap Assessment ChecklistDocument7 pagesIATF 16949 Gap Assessment ChecklistBharath100% (1)
- Some Common Standards IncludeDocument46 pagesSome Common Standards IncludekardglNo ratings yet
- Clause Matrix Iatf 16949Document10 pagesClause Matrix Iatf 16949ikesh mNo ratings yet
- API Specification Q1 9th Edition Errata 3 20191017Document1 pageAPI Specification Q1 9th Edition Errata 3 20191017shifaNo ratings yet
- API Q1 9th Edition Mandatory Records: No. Clause No. DescriptionDocument1 pageAPI Q1 9th Edition Mandatory Records: No. Clause No. Descriptionzae nuddin100% (2)
- ISO 9001 - 2008 RequirementDocument9 pagesISO 9001 - 2008 RequirementNavnath TamhaneNo ratings yet
- Breakout C (LAC 9 New WS#9)Document3 pagesBreakout C (LAC 9 New WS#9)Suleman JahangirNo ratings yet
- Template - ScheduleDocument3 pagesTemplate - Schedulerodolfo1649100% (1)
- List of QMS Process PDFDocument2 pagesList of QMS Process PDFkarthikkandaNo ratings yet
- Quality Management System - ISO 9001 MIND MAP April 3, 2012: Compiled by Ravi Kiran Muddha Ref: ISO 9001:2008Document1 pageQuality Management System - ISO 9001 MIND MAP April 3, 2012: Compiled by Ravi Kiran Muddha Ref: ISO 9001:2008KarthiNo ratings yet
- IATF IA CHecklistDocument2 pagesIATF IA CHecklistDamodaran RajanayagamNo ratings yet
- Iso 9001:2000 Sample Audit Plan Schedule (Registrar)Document0 pagesIso 9001:2000 Sample Audit Plan Schedule (Registrar)Muhammad ShafiNo ratings yet
- API Spec Q1 9th Edition - Mandatory Documented Procedures: No. Clause No. DescriptionDocument1 pageAPI Spec Q1 9th Edition - Mandatory Documented Procedures: No. Clause No. Descriptionzae nuddinNo ratings yet
- Qms Guidelines Appendix A 0Document3 pagesQms Guidelines Appendix A 0BranErikNo ratings yet
- Company Name Here. Quality Manual: Section 0Document2 pagesCompany Name Here. Quality Manual: Section 0arunradNo ratings yet
- Checklist of Mandatory Documentation Required by Iso 13485 2016Document3 pagesChecklist of Mandatory Documentation Required by Iso 13485 2016Ami Jariwala100% (1)
- AS9100 Mandatory RecordsDocument3 pagesAS9100 Mandatory Recordsocardenas9160No ratings yet
- Iso 9001 Clause ChartDocument2 pagesIso 9001 Clause ChartSUBODHHNo ratings yet
- Compare ISO 9001 AS9100c PDFDocument1 pageCompare ISO 9001 AS9100c PDFRamses SalazarNo ratings yet
- 9001 2015 9001 2008 Comparison Table 22 July 2016Document2 pages9001 2015 9001 2008 Comparison Table 22 July 2016Islam MarzokyNo ratings yet
- QC Plan TOC ExampleDocument4 pagesQC Plan TOC ExampleAbdul hafizNo ratings yet
- Process ListDocument3 pagesProcess ListManoj ManeNo ratings yet
- Quality Management System: Understanding Our QMS For The Achievement of Organization and Business SuccessDocument56 pagesQuality Management System: Understanding Our QMS For The Achievement of Organization and Business SuccessNoor AhmedNo ratings yet
- ISO PrinciplesDocument4 pagesISO PrincipleshasibNo ratings yet
- Iso 9001 Document RequirementsDocument47 pagesIso 9001 Document Requirementsksbbs100% (2)
- Quality Management SystemDocument56 pagesQuality Management SystemTatiana Rilo100% (6)
- DFC 4.1Document1 pageDFC 4.1Muhammad WaqasNo ratings yet
- IMS Systems DirectoryDocument3 pagesIMS Systems DirectoryCandiceNo ratings yet
- ISO 9001:2015 To ISO 9001:2008 Correlation MatrixDocument6 pagesISO 9001:2015 To ISO 9001:2008 Correlation MatrixAstrid Dwitha ManikNo ratings yet
- REQ. ClausulDocument35 pagesREQ. ClausulAdeAAANo ratings yet
- P018 Internal Audit Procedure: ISO 9001:2008 Clause 8.2.2Document9 pagesP018 Internal Audit Procedure: ISO 9001:2008 Clause 8.2.2Álvaro Martínez Fernández100% (1)
- ISO 9001-Correlation MatricesDocument6 pagesISO 9001-Correlation MatricesLailla MarlinaNo ratings yet
- ISO 9001:2015 Clause: Page 1 of 4Document4 pagesISO 9001:2015 Clause: Page 1 of 4aamirNo ratings yet
- 2.1.3 QANPD Guidance 01 APR 2014 PDFDocument65 pages2.1.3 QANPD Guidance 01 APR 2014 PDFSebastian CotinghiuNo ratings yet
- List of Mandatory Records For ISO 9001Document2 pagesList of Mandatory Records For ISO 9001NC Rigor Luis100% (2)
- ISO 9001 - Documentaion Requirement SummaryDocument7 pagesISO 9001 - Documentaion Requirement SummaryminhajurrehmanNo ratings yet
- Version 2008 Version 2015 What We Do .Document6 pagesVersion 2008 Version 2015 What We Do .GVS RaoNo ratings yet
- Iso9001-As9100 Check ListDocument19 pagesIso9001-As9100 Check ListJohn Rajesh100% (1)
- ISO 9001 Documentation Toolkit ListDocument3 pagesISO 9001 Documentation Toolkit Listdumisani ManentsaNo ratings yet
- QMSDocument1 pageQMSVirginia GrandoNo ratings yet
- 9 Iatf 16949 Gap Assessment Checklist 170601102322Document8 pages9 Iatf 16949 Gap Assessment Checklist 170601102322Homero JanuncioNo ratings yet
- Documents and Records Required For An ISO/IEC 17025:2017Document3 pagesDocuments and Records Required For An ISO/IEC 17025:2017Aris Rahman100% (2)
- All WimDocument8 pagesAll WimMaruan MuhammadNo ratings yet
- Comparisson Matrix ISO 9001Document6 pagesComparisson Matrix ISO 9001Suhendra SangkalaNo ratings yet
- As 9100 D AssenceDocument5 pagesAs 9100 D Assencemhk665No ratings yet
- Iso 9001:2008 Iso/ts 16949:2009Document3 pagesIso 9001:2008 Iso/ts 16949:2009timkoidNo ratings yet
- TS 16949 Standard in One PageDocument1 pageTS 16949 Standard in One Pagepkd1963100% (3)
- Compariosn Between IATF and TS RequirementsDocument32 pagesCompariosn Between IATF and TS RequirementsDamodaran RajanayagamNo ratings yet
- Company Name: Quality System ManualDocument21 pagesCompany Name: Quality System ManualBrett BensleyNo ratings yet
- Internal AuditDocument2 pagesInternal AuditAkhilesh KumarNo ratings yet
- Iso 9001:2008 Iso 9001:2015Document5 pagesIso 9001:2008 Iso 9001:2015vpreityNo ratings yet
- ISO 9001 Required Documentation and StepsDocument3 pagesISO 9001 Required Documentation and StepsganandmailNo ratings yet
- Quality System Manual ISO 9001:2008 Internal Audit Check ListDocument14 pagesQuality System Manual ISO 9001:2008 Internal Audit Check ListDivakar Panigrahi100% (1)
- The Structure Is The Old OneDocument8 pagesThe Structure Is The Old One601026No ratings yet
- Establishing A CGMP Laboratory Audit System: A Practical GuideFrom EverandEstablishing A CGMP Laboratory Audit System: A Practical GuideNo ratings yet
- Cracking the Case of ISO 9001:2015 for Service: A Simple Guide to Implementing Quality Management in Service OrganizationsFrom EverandCracking the Case of ISO 9001:2015 for Service: A Simple Guide to Implementing Quality Management in Service OrganizationsNo ratings yet
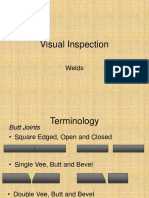
- Visual Inspection WeldDocument53 pagesVisual Inspection Weldshifa100% (1)
- Torque For B7 L7 Stud BoltsDocument2 pagesTorque For B7 L7 Stud BoltsshifaNo ratings yet
- Visual Inspection Checklist PipingDocument2 pagesVisual Inspection Checklist PipingshifaNo ratings yet
- API Specification 6A 21st Edition Errata 2 20200625Document4 pagesAPI Specification 6A 21st Edition Errata 2 20200625shifaNo ratings yet
- QA Manual Rev6Document44 pagesQA Manual Rev6shifa100% (1)
- Quality Manual Revision L Edition 2Document32 pagesQuality Manual Revision L Edition 2shifa100% (1)
- API Internal Audit ProgrammeDocument72 pagesAPI Internal Audit Programmeshifa100% (1)
- Non-Destructive TestingDocument78 pagesNon-Destructive TestingshifaNo ratings yet
- TS 0070Document1 pageTS 0070shifaNo ratings yet
- API MonoDocument1 pageAPI MonoshifaNo ratings yet
- Personal Protective EquipmentsDocument1 pagePersonal Protective EquipmentsshifaNo ratings yet
- About API Monogram ProgramDocument1 pageAbout API Monogram ProgramshifaNo ratings yet
- Radha Enterprises: Centrifuge Tube & StandDocument1 pageRadha Enterprises: Centrifuge Tube & StandshifaNo ratings yet
- Introduction of The CompanyDocument1 pageIntroduction of The CompanyshifaNo ratings yet
- Va 7.3-4eDocument4 pagesVa 7.3-4eshifaNo ratings yet
- ValveDocument1 pageValveshifaNo ratings yet
- Shah Promoters & Developers: Sr. No. Details of Changes SignatureDocument1 pageShah Promoters & Developers: Sr. No. Details of Changes Signatureshifa0% (1)