Professional Documents
Culture Documents
1 P PV RT: Sifat Gas Ideal: Sifat Gas Nyata
1 P PV RT: Sifat Gas Ideal: Sifat Gas Nyata
Uploaded by
Teo Wijayarto0 ratings0% found this document useful (0 votes)
11 views1 pageThis document discusses the properties of ideal gases and real gases. Ideal gases are characterized by molecules with negligible intermolecular attractions and volume, following Boyle's, Charles', Avogadro's, Dalton's, Graham's, and Amagat's laws. Real gases exhibit intermolecular attractions and molecular volume, deviating from ideal behavior. Real gas properties are expressed using van der Waals equation with constants a and b accounting for attraction and molecular size/compressibility. The document also discusses relationships between molar mass, composition, amount of substance, pressure, volume, and temperature for ideal gases.
Original Description:
Rangkuman Gas Kuliah
Original Title
Rangkuman Gas
Copyright
© © All Rights Reserved
Available Formats
PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentThis document discusses the properties of ideal gases and real gases. Ideal gases are characterized by molecules with negligible intermolecular attractions and volume, following Boyle's, Charles', Avogadro's, Dalton's, Graham's, and Amagat's laws. Real gases exhibit intermolecular attractions and molecular volume, deviating from ideal behavior. Real gas properties are expressed using van der Waals equation with constants a and b accounting for attraction and molecular size/compressibility. The document also discusses relationships between molar mass, composition, amount of substance, pressure, volume, and temperature for ideal gases.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
Download as pdf or txt
0 ratings0% found this document useful (0 votes)
11 views1 page1 P PV RT: Sifat Gas Ideal: Sifat Gas Nyata
1 P PV RT: Sifat Gas Ideal: Sifat Gas Nyata
Uploaded by
Teo WijayartoThis document discusses the properties of ideal gases and real gases. Ideal gases are characterized by molecules with negligible intermolecular attractions and volume, following Boyle's, Charles', Avogadro's, Dalton's, Graham's, and Amagat's laws. Real gases exhibit intermolecular attractions and molecular volume, deviating from ideal behavior. Real gas properties are expressed using van der Waals equation with constants a and b accounting for attraction and molecular size/compressibility. The document also discusses relationships between molar mass, composition, amount of substance, pressure, volume, and temperature for ideal gases.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
Download as pdf or txt
You are on page 1of 1
barnas holil
0812 21 6996 1 / 0818 20 4321
e-mail: barnas@chem.itb.ac.id / barnasholil@yahoo.com
Gas Ideal
Gas Nyata
Sifat gas ideal:
antaraksi antar molekul gas diabaikan
volume molekul gas diabaikan
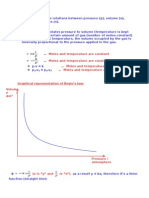
1. Boyle
V~
1
P
(n & T tetap)
2. Charles & Gay Lussac
V~T
(n & P tetap)
Sifat gas nyata:
antaraksi antar molekul gas tidak dapat diabaikan
volume molekul gas tidak dapat diabaikan
pada tekanan rendah (P0), semua gas nyata bersifat
ideal
penyimpangan dari keadaan ideal dinayatakan oleh faktor
PV
daya mampat, Z, dengan Z =
RT
Z=1
gas ideal
Z1
gas nyata
3. Avogadro
V~n
(T & P tetap)
Persamaan van der Waals
P = Pt = P i
(V & T tetap)
Hukum-hukum gas ideal
4. Dalton
the measured
pressure
the measured
volume
Pi = xi.P
5. Amagat
V = Vt = P i
(P & T tetap)
Vi = xi.V
6. Graham
v=r=~
v2
=
v1
1
=
2
n 2a
Pmeas V 2 Vmeas nb nRT
Mr
M r,1
M r,2
(T & P tetap)
correction to bring
measured P up to the
pressure an idal gas
would exert
correction to reduce
measured V to the volume
an ideal gas would have
Hubungan massa molar campuran gas dan komposisi gas
Mr = xi.Mr,i
The term a is a measure of attractive force between the
molecules.
Persamaan keadaan gas ideal
PV = nRT
The term b is due to the finite volume of the molecules and to
their general incompressibility.
Elaborasi persamaan gas ideal
is the effective volume of molecules in one mole of gas.
PV = nRT
m
RT
=
Mr
m
PMr =
RT
V
= RT
Perhitungan stoikiometri gas ideal dapat menggunakan satuan
jumlah zat dalam:
mol
volume
(pada T & P tetap)
tekanan
(pada T & V tetap)
gas_rangkuman/19-Oct-14 9:20:40 PM
Notice that the constant a involves a correction to the pressure
term of the ideal gas law, so the size of a would indicate
something about attractions between molecules. Larger values
of a mean stronger attractive forces between molecules. Thus,
the most easily liquified substances, like water and ethyl
alcohol, have the largest values of the van der Waals constant
a, suggesting relatively strong attractive forces between their
molecules.
The constant b helps to correct for the volume occupied by the
molecules themselves, so the size of b indicates something
about the sizes of particles in the gas. Larger values of b
mean larger molecule sizes.
You might also like
- (Lec5) Properties of GasesDocument52 pages(Lec5) Properties of GasesdinurjNo ratings yet
- Unit 1Document26 pagesUnit 1firehywotNo ratings yet
- Chemical Thermodynamicse All-1Document245 pagesChemical Thermodynamicse All-1danimiresa961No ratings yet
- Gas Behaviour EOSDocument59 pagesGas Behaviour EOSMurugavel ChandranNo ratings yet
- Behaviour of GasesDocument29 pagesBehaviour of GasesAli RazaNo ratings yet
- G19RA Chap5 - GasesDocument37 pagesG19RA Chap5 - GasesaidaNo ratings yet
- SC RE Chap5-GasesDocument49 pagesSC RE Chap5-Gasesweldsv0% (1)
- Wa0001.Document31 pagesWa0001.Stefani KavangoNo ratings yet
- States of Matter (Gas)Document27 pagesStates of Matter (Gas)Siti NuraqidahNo ratings yet
- Chemistry QuizDocument3 pagesChemistry QuizCielo PulmaNo ratings yet
- 4.1 Ideal GasesDocument22 pages4.1 Ideal GasesAnonymous o97HYLpe0No ratings yet
- Eservoir OdelingDocument21 pagesEservoir Odelingعلي سعيد سعودNo ratings yet
- Class-11 Chemistry Chapter-5 States of Matter Part-IIDocument7 pagesClass-11 Chemistry Chapter-5 States of Matter Part-IINevin ShajiNo ratings yet
- PGE 362 - Fluid Prop ch2-2Document17 pagesPGE 362 - Fluid Prop ch2-2Turki AlshammariNo ratings yet
- EOS For Real Gas FinalDocument17 pagesEOS For Real Gas FinalrutvikNo ratings yet
- Unit8 1 TNSDocument11 pagesUnit8 1 TNSSylvesterMcLaneNo ratings yet
- SCH 103 NotesDocument50 pagesSCH 103 NotesJacqueseNo ratings yet
- Gas Laws:: P V K VDocument18 pagesGas Laws:: P V K VFarah Zu'biNo ratings yet
- Ideal GasDocument17 pagesIdeal GasPoonamNo ratings yet
- The Gas Laws: Equations of StateDocument32 pagesThe Gas Laws: Equations of Stateyiye rubyNo ratings yet
- 5.0 States of MatterDocument106 pages5.0 States of MatterTasya KassimNo ratings yet
- Week 3 PPT AD CHEMDocument8 pagesWeek 3 PPT AD CHEMSophia Ysabelle EstradaNo ratings yet
- Chap 03 - Gaseous State MindNote Class 11th by Arnav SirDocument5 pagesChap 03 - Gaseous State MindNote Class 11th by Arnav SirSmeet MehtaNo ratings yet
- Sample Problem PPDocument12 pagesSample Problem PPcallielizzaNo ratings yet
- Lecture 4 Gas Laws and RelationsDocument28 pagesLecture 4 Gas Laws and RelationsArsal SohrabNo ratings yet
- CH 10 Gases StudentDocument48 pagesCH 10 Gases StudentTrọng NguyễnNo ratings yet
- Dalton's Law of Partial Pressure: MathematicallyDocument6 pagesDalton's Law of Partial Pressure: MathematicallyNabila Azzahra SalsabilaNo ratings yet
- Class 11 Chemistry Chapter 5 Study MaterialDocument33 pagesClass 11 Chemistry Chapter 5 Study MaterialmeghaNo ratings yet
- Chapter 10Document20 pagesChapter 10Kyrie IrvingNo ratings yet
- Chem 1101 L2Document28 pagesChem 1101 L2katieamills59No ratings yet
- 5.1 Pressure: Chapter 5: GasesDocument4 pages5.1 Pressure: Chapter 5: GasesSam ChungNo ratings yet
- States - of - Matter Gas Law Cape Module 1Document40 pagesStates - of - Matter Gas Law Cape Module 1devoydouglasNo ratings yet
- Properties of Gases (Report)Document19 pagesProperties of Gases (Report)Rex LapisNo ratings yet
- Gases and The Atmosphere: Robert Boyle 1627-1691. Jacques Charles 1746-1823. J. Charles 1783Document34 pagesGases and The Atmosphere: Robert Boyle 1627-1691. Jacques Charles 1746-1823. J. Charles 1783ade dosmariaNo ratings yet
- Gases and The Atmosphere: Robert Boyle 1627-1691. Jacques Charles 1746-1823. J. Charles 1783Document34 pagesGases and The Atmosphere: Robert Boyle 1627-1691. Jacques Charles 1746-1823. J. Charles 1783ade dosmariaNo ratings yet
- Gas LawsDocument2 pagesGas LawsNeil BrazaNo ratings yet
- Equations of State For Real GasesDocument19 pagesEquations of State For Real GasesShamim khanNo ratings yet
- Gas Properties I 2018Document55 pagesGas Properties I 2018Johny ImitazNo ratings yet
- Gas Laws: M. L. WatsonDocument25 pagesGas Laws: M. L. WatsonAbhishek ChakrabartiNo ratings yet
- 05 Behavior of Gas and OilsDocument12 pages05 Behavior of Gas and OilsfarajNo ratings yet
- Gases: Theodore L. Brown H. Eugene Lemay, Jr. and Bruce E. BurstenDocument28 pagesGases: Theodore L. Brown H. Eugene Lemay, Jr. and Bruce E. BurstenAmalia AnggreiniNo ratings yet
- CHAPTER 5.1 GasDocument18 pagesCHAPTER 5.1 GasZARITH SOFHIA BINTI MD KHARODIN KM-PelajarNo ratings yet
- Chapter 4 States of Matter 2021Document24 pagesChapter 4 States of Matter 2021suh mey chongNo ratings yet
- Gas Properties I 2018Document55 pagesGas Properties I 2018Akib ImtihanNo ratings yet
- Any Two Molecules of A Solid Is Very Large. LiquidsDocument1 pageAny Two Molecules of A Solid Is Very Large. LiquidsBarunMondalNo ratings yet
- Mechnotes: Unit - 1 ObjectiveDocument25 pagesMechnotes: Unit - 1 ObjectiveKaran SelvaNo ratings yet
- 9.pertemuan 6. GasesDocument28 pages9.pertemuan 6. GasesMUHAMMAD HEFFIQRI RIADYNo ratings yet
- Chemistry Notes Ideal Gas LawsDocument19 pagesChemistry Notes Ideal Gas LawsAbhishek MasneNo ratings yet
- Fundamentals of Gas LawsDocument9 pagesFundamentals of Gas LawsMangojuice MbeleNo ratings yet
- Introduction To Physical Chemistry: 2. Real Gases and Van Der Waals EquationsDocument13 pagesIntroduction To Physical Chemistry: 2. Real Gases and Van Der Waals EquationsDery RachmandaniNo ratings yet
- Chapter3 IdealgaslawDocument45 pagesChapter3 Idealgaslaw翁绍棠No ratings yet
- Lec 21 22 - CH 13Document21 pagesLec 21 22 - CH 13samhameed2No ratings yet
- REN5415 Y19 Lec11&12Document69 pagesREN5415 Y19 Lec11&12Abdulla MohammadNo ratings yet
- 5 - Behaviour of GasesDocument37 pages5 - Behaviour of Gasessiaskel100% (1)
- The Van Der Waals' Gas (VDW.G.) : NRT RT P V VDocument5 pagesThe Van Der Waals' Gas (VDW.G.) : NRT RT P V VSoji AdimulaNo ratings yet
- Gaseous State: Ideal GasesDocument13 pagesGaseous State: Ideal GasesM.S.A. Mobashwer HossenNo ratings yet
- Atkins' Physical Chemistry: Peter Atkins - Julio de PaulaDocument37 pagesAtkins' Physical Chemistry: Peter Atkins - Julio de PaulaAmalia AnggreiniNo ratings yet
- Chapter 12Document27 pagesChapter 12احمد الدلالNo ratings yet
- States of Matter Subjective TypeDocument8 pagesStates of Matter Subjective TypePrajesh BiswasNo ratings yet