Professional Documents
Culture Documents
0 ratings0% found this document useful (0 votes)
44 viewsAldaba Josue
Aldaba Josue
Uploaded by
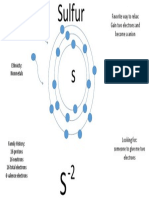
api-263909505Sulfur is a non-metal with 16 protons, 16 neutrons, and 16 electrons, with 6 valence electrons. It has the ability to gain two electrons to form an anion, as sulfur is an element that others can donate electrons to. Sulfur's atomic number is 16 and it has 6 valence electrons available for bonding.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
You might also like
- Topic 3: Chemical BondsDocument49 pagesTopic 3: Chemical BondsFlores DavidNo ratings yet
- Chemical BondingDocument30 pagesChemical BondingRhian Geena VelasquezNo ratings yet
- 03-Lesson 3 Forming IonsDocument24 pages03-Lesson 3 Forming IonsAnnabel PatarataNo ratings yet
- Chem 2Document3 pagesChem 2Mega MegaNo ratings yet
- Properties of Particles Include:: Key IdeasDocument8 pagesProperties of Particles Include:: Key IdeasEmily McCullochNo ratings yet
- Structure and Bonding - Lesson 3 - Ionic BondingDocument19 pagesStructure and Bonding - Lesson 3 - Ionic Bondingcharlie markouNo ratings yet
- Chemical Bonding NotesDocument23 pagesChemical Bonding Noteskaylee chiaNo ratings yet
- Chapter - 02 NewDocument36 pagesChapter - 02 NewmaheshNo ratings yet
- Electrochemistry Daniell CellDocument16 pagesElectrochemistry Daniell CellSiew Kiong Wong100% (1)
- Electrons WS 5 Electron Configuration IonsDocument3 pagesElectrons WS 5 Electron Configuration IonsAna Marie Corales TabunarNo ratings yet
- Reaction Mechanism NotesDocument17 pagesReaction Mechanism NotesMahalaksshmi .DNo ratings yet
- Ikatan KimiaDocument46 pagesIkatan KimiaFla Syafa SabitnaNo ratings yet
- Chemistry Notes (Electrolysis)Document2 pagesChemistry Notes (Electrolysis)Teo Jia Ming Nickolas100% (3)
- F4 C5 Chemical BondingDocument23 pagesF4 C5 Chemical BondingkaiqianNo ratings yet
- Chapter 2: Atomic Structure & Interatomic Bonding: Issues To Address..Document29 pagesChapter 2: Atomic Structure & Interatomic Bonding: Issues To Address..MPRajNo ratings yet
- Class 10 - Chapter - 2Document41 pagesClass 10 - Chapter - 2Nitin SNo ratings yet
- Atomic Structure Bonding Periodic TableDocument59 pagesAtomic Structure Bonding Periodic TableCaribelatinaNo ratings yet
- Ni Laterite Short Course 050512Document214 pagesNi Laterite Short Course 050512Slamet SetyowibowoNo ratings yet
- AQA Chemistry SampleDocument2 pagesAQA Chemistry SampleHugh MunguzNo ratings yet
- Numbers of Electrons in The Outermost ShellDocument46 pagesNumbers of Electrons in The Outermost ShellGianna SaludoNo ratings yet
- Bonding in CarbonDocument33 pagesBonding in CarbonSatyam PrakashNo ratings yet
- Ion Activity SheetDocument1 pageIon Activity SheetXhialvea KeziahNo ratings yet
- Atomic Structures: © 2011 Project Lead The Way, Inc. Magic of ElectronsDocument25 pagesAtomic Structures: © 2011 Project Lead The Way, Inc. Magic of ElectronsFaiad KamalNo ratings yet
- Assignment 3:: Smallest - A & Largest - DDocument2 pagesAssignment 3:: Smallest - A & Largest - DBarbara BananaNo ratings yet
- Electrolysis Cheat Sheet: by ViaDocument2 pagesElectrolysis Cheat Sheet: by Viaaziz ahmadNo ratings yet
- Tomic Structure and Interatomic Bonding: Issues To Address..Document22 pagesTomic Structure and Interatomic Bonding: Issues To Address..Yahya AbdulsalamNo ratings yet
- Tomic Structure and Interatomic Bonding: Issues To Address..Document22 pagesTomic Structure and Interatomic Bonding: Issues To Address..mohamd jehadNo ratings yet
- Naming of Compounds - Part2Document2 pagesNaming of Compounds - Part2Linh NguyễnNo ratings yet
- Learning Targets:: Writing Chemical Formula, D. Naming Ionic CompoundsDocument16 pagesLearning Targets:: Writing Chemical Formula, D. Naming Ionic CompoundsJecky LorenzoNo ratings yet
- (Trans) Organic Lecture Chapter 2Document4 pages(Trans) Organic Lecture Chapter 2jilldlrs06No ratings yet
- Ionic Bonding: © 2013 Marshall Cavendish International (Singapore) Private LimitedDocument51 pagesIonic Bonding: © 2013 Marshall Cavendish International (Singapore) Private LimitedSuhaan HussainNo ratings yet
- Ionic BondingDocument51 pagesIonic BondingAaditya MK100% (1)
- BS Chemistry MCQsDocument6 pagesBS Chemistry MCQsRamsha saeedNo ratings yet
- ch02 0310Document26 pagesch02 0310pasternack2379No ratings yet
- When Atoms Meet: Chemical BondingDocument88 pagesWhen Atoms Meet: Chemical BondingWilsonNo ratings yet
- 2nd Quarter Reviewer - CHEMISTRYDocument2 pages2nd Quarter Reviewer - CHEMISTRYKate Ashley CailingNo ratings yet
- Atomic StructureDocument13 pagesAtomic Structurerenan.masangya-18No ratings yet
- Chemical Bonding - Ionic BondDocument56 pagesChemical Bonding - Ionic BondFianna GalleroNo ratings yet
- Structure and Bonding: A Guide For A Level StudentsDocument86 pagesStructure and Bonding: A Guide For A Level StudentshaidarNo ratings yet
- Atomic StructureDocument25 pagesAtomic Structuremichaeldevid7890No ratings yet
- Bonding Forces and Energies Al Francis Asuncion EE 3bDocument8 pagesBonding Forces and Energies Al Francis Asuncion EE 3bAce John OliverNo ratings yet
- Materials Science - Lecture #2 - Atomic StructureDocument62 pagesMaterials Science - Lecture #2 - Atomic Structureisura678hasankaNo ratings yet
- Semi Conductor2Document60 pagesSemi Conductor2mythonyNo ratings yet
- Chapter 7 Ionic and Metallic BondingDocument56 pagesChapter 7 Ionic and Metallic BondingCharles GibbsNo ratings yet
- Dwnload Full Descriptive Inorganic Chemistry 6th Edition Rayner Canham Solutions Manual PDFDocument35 pagesDwnload Full Descriptive Inorganic Chemistry 6th Edition Rayner Canham Solutions Manual PDFmarieramirezsaqdmkpfgb100% (16)
- Lec 4Document10 pagesLec 4Maina PhysicistNo ratings yet
- Chemical BondingDocument19 pagesChemical Bondinghassanyaraanka28No ratings yet
- Chemistry Reviewer: Capacity of Atoms To From Bonds. Elements in The Periodic TableDocument4 pagesChemistry Reviewer: Capacity of Atoms To From Bonds. Elements in The Periodic TableKatrina BernardinoNo ratings yet
- Chemical BondDocument64 pagesChemical BondangelieshaynnmtalagtagNo ratings yet
- G9.MODULE 2 Lesson 6.2 Formation of IonsDocument10 pagesG9.MODULE 2 Lesson 6.2 Formation of IonsAndrina Binogwal TocgongnaNo ratings yet
- UntitledDocument6 pagesUntitledSuzuki YatoNo ratings yet
- The Atom Activities: Discovery of Electron Democritus Proposes The Word "Atom" Current ModelDocument8 pagesThe Atom Activities: Discovery of Electron Democritus Proposes The Word "Atom" Current ModelHunter PpollNo ratings yet
- ChembondDocument51 pagesChembondNovelynLozano-EdrosoNo ratings yet
- ch02 RevDocument22 pagesch02 RevFasih Ul HaqNo ratings yet
- Forming IonsDocument22 pagesForming IonsSamantha Shane P. ConumNo ratings yet
- Covalent BondsDocument6 pagesCovalent BondsimunasingheNo ratings yet
- ENS167 Lecture 2Document22 pagesENS167 Lecture 2JANNA MARIE NInA BATENo ratings yet
- Chem 1 MY18 Lec Session 6, 7 PDFDocument13 pagesChem 1 MY18 Lec Session 6, 7 PDFBytehubNetcafeNo ratings yet
- Lecture 1Document23 pagesLecture 1sanjunaNo ratings yet
- Tema 3. Chemical BondingDocument68 pagesTema 3. Chemical Bondingkarthik tvkNo ratings yet
- Track Your LearningDocument1 pageTrack Your Learningapi-263909505No ratings yet
- Group ContractDocument1 pageGroup Contractapi-263909505No ratings yet
- Group ContractDocument3 pagesGroup Contractapi-263909505No ratings yet
- Angel EsquedaDocument1 pageAngel Esquedaapi-263909505No ratings yet
- Track Your LearningDocument1 pageTrack Your Learningapi-263909505No ratings yet
- Delilah GonzalezDocument1 pageDelilah Gonzalezapi-263909505No ratings yet
- Acids and BasesDocument57 pagesAcids and Basesapi-263909505No ratings yet
- Stoich Quick RefresherDocument1 pageStoich Quick Refresherapi-263909505No ratings yet
- Bertha ChavezDocument1 pageBertha Chavezapi-263909505No ratings yet
- Density Floating ActivityDocument1 pageDensity Floating Activityapi-263909505No ratings yet
- Ayala AilynDocument1 pageAyala Ailynapi-263909505No ratings yet
- Chavez VictorDocument1 pageChavez Victorapi-263909505No ratings yet
- Sandoval DemetriDocument1 pageSandoval Demetriapi-263909505No ratings yet
- Intermolecular ForcesDocument18 pagesIntermolecular Forcesapi-263909505No ratings yet
- Banda JenniferDocument1 pageBanda Jenniferapi-263909505No ratings yet
- Naming Simple MoleculesDocument1 pageNaming Simple Moleculesapi-263909505No ratings yet
- Ink ChromatographyDocument9 pagesInk Chromatographyapi-2639095050% (1)
- Leyva AriamDocument1 pageLeyva Ariamapi-263909505No ratings yet
- Ramirez JoseDocument1 pageRamirez Joseapi-263909505No ratings yet
- Franco NikkoDocument1 pageFranco Nikkoapi-263909505No ratings yet
- Brite JimmyDocument1 pageBrite Jimmyapi-263909505No ratings yet
- Alavarez ArmandoDocument1 pageAlavarez Armandoapi-263909505No ratings yet
- Llanas AlexisDocument1 pageLlanas Alexisapi-263909505No ratings yet
- Flores SammyDocument1 pageFlores Sammyapi-263909505No ratings yet
- Marzan MiguelDocument1 pageMarzan Miguelapi-263909505No ratings yet
- Ortega DavidDocument1 pageOrtega Davidapi-263909505No ratings yet
- Martinez KevinDocument1 pageMartinez Kevinapi-263909505No ratings yet
- Torres CassieDocument1 pageTorres Cassieapi-263909505No ratings yet
- Hernandez CrystalDocument1 pageHernandez Crystalapi-263909505No ratings yet
- Esqueda DanielleDocument1 pageEsqueda Danielleapi-263909505No ratings yet
Aldaba Josue
Aldaba Josue
Uploaded by
api-2639095050 ratings0% found this document useful (0 votes)
44 views1 pageSulfur is a non-metal with 16 protons, 16 neutrons, and 16 electrons, with 6 valence electrons. It has the ability to gain two electrons to form an anion, as sulfur is an element that others can donate electrons to. Sulfur's atomic number is 16 and it has 6 valence electrons available for bonding.
Original Description:
Original Title
aldaba josue
Copyright
© © All Rights Reserved
Available Formats
PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentSulfur is a non-metal with 16 protons, 16 neutrons, and 16 electrons, with 6 valence electrons. It has the ability to gain two electrons to form an anion, as sulfur is an element that others can donate electrons to. Sulfur's atomic number is 16 and it has 6 valence electrons available for bonding.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
Download as pdf or txt
0 ratings0% found this document useful (0 votes)
44 views1 pageAldaba Josue
Aldaba Josue
Uploaded by
api-263909505Sulfur is a non-metal with 16 protons, 16 neutrons, and 16 electrons, with 6 valence electrons. It has the ability to gain two electrons to form an anion, as sulfur is an element that others can donate electrons to. Sulfur's atomic number is 16 and it has 6 valence electrons available for bonding.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
Download as pdf or txt
You are on page 1of 1
sulfur
hometown:
6A
-2
S
Gain 2 which would
Be anion
Ethenicity:
non_metal
Someone to gain
Electrons from.
Family history:
16 protons
16 nutrons
16 electrons
6 valence
electrons
You might also like
- Topic 3: Chemical BondsDocument49 pagesTopic 3: Chemical BondsFlores DavidNo ratings yet
- Chemical BondingDocument30 pagesChemical BondingRhian Geena VelasquezNo ratings yet
- 03-Lesson 3 Forming IonsDocument24 pages03-Lesson 3 Forming IonsAnnabel PatarataNo ratings yet
- Chem 2Document3 pagesChem 2Mega MegaNo ratings yet
- Properties of Particles Include:: Key IdeasDocument8 pagesProperties of Particles Include:: Key IdeasEmily McCullochNo ratings yet
- Structure and Bonding - Lesson 3 - Ionic BondingDocument19 pagesStructure and Bonding - Lesson 3 - Ionic Bondingcharlie markouNo ratings yet
- Chemical Bonding NotesDocument23 pagesChemical Bonding Noteskaylee chiaNo ratings yet
- Chapter - 02 NewDocument36 pagesChapter - 02 NewmaheshNo ratings yet
- Electrochemistry Daniell CellDocument16 pagesElectrochemistry Daniell CellSiew Kiong Wong100% (1)
- Electrons WS 5 Electron Configuration IonsDocument3 pagesElectrons WS 5 Electron Configuration IonsAna Marie Corales TabunarNo ratings yet
- Reaction Mechanism NotesDocument17 pagesReaction Mechanism NotesMahalaksshmi .DNo ratings yet
- Ikatan KimiaDocument46 pagesIkatan KimiaFla Syafa SabitnaNo ratings yet
- Chemistry Notes (Electrolysis)Document2 pagesChemistry Notes (Electrolysis)Teo Jia Ming Nickolas100% (3)
- F4 C5 Chemical BondingDocument23 pagesF4 C5 Chemical BondingkaiqianNo ratings yet
- Chapter 2: Atomic Structure & Interatomic Bonding: Issues To Address..Document29 pagesChapter 2: Atomic Structure & Interatomic Bonding: Issues To Address..MPRajNo ratings yet
- Class 10 - Chapter - 2Document41 pagesClass 10 - Chapter - 2Nitin SNo ratings yet
- Atomic Structure Bonding Periodic TableDocument59 pagesAtomic Structure Bonding Periodic TableCaribelatinaNo ratings yet
- Ni Laterite Short Course 050512Document214 pagesNi Laterite Short Course 050512Slamet SetyowibowoNo ratings yet
- AQA Chemistry SampleDocument2 pagesAQA Chemistry SampleHugh MunguzNo ratings yet
- Numbers of Electrons in The Outermost ShellDocument46 pagesNumbers of Electrons in The Outermost ShellGianna SaludoNo ratings yet
- Bonding in CarbonDocument33 pagesBonding in CarbonSatyam PrakashNo ratings yet
- Ion Activity SheetDocument1 pageIon Activity SheetXhialvea KeziahNo ratings yet
- Atomic Structures: © 2011 Project Lead The Way, Inc. Magic of ElectronsDocument25 pagesAtomic Structures: © 2011 Project Lead The Way, Inc. Magic of ElectronsFaiad KamalNo ratings yet
- Assignment 3:: Smallest - A & Largest - DDocument2 pagesAssignment 3:: Smallest - A & Largest - DBarbara BananaNo ratings yet
- Electrolysis Cheat Sheet: by ViaDocument2 pagesElectrolysis Cheat Sheet: by Viaaziz ahmadNo ratings yet
- Tomic Structure and Interatomic Bonding: Issues To Address..Document22 pagesTomic Structure and Interatomic Bonding: Issues To Address..Yahya AbdulsalamNo ratings yet
- Tomic Structure and Interatomic Bonding: Issues To Address..Document22 pagesTomic Structure and Interatomic Bonding: Issues To Address..mohamd jehadNo ratings yet
- Naming of Compounds - Part2Document2 pagesNaming of Compounds - Part2Linh NguyễnNo ratings yet
- Learning Targets:: Writing Chemical Formula, D. Naming Ionic CompoundsDocument16 pagesLearning Targets:: Writing Chemical Formula, D. Naming Ionic CompoundsJecky LorenzoNo ratings yet
- (Trans) Organic Lecture Chapter 2Document4 pages(Trans) Organic Lecture Chapter 2jilldlrs06No ratings yet
- Ionic Bonding: © 2013 Marshall Cavendish International (Singapore) Private LimitedDocument51 pagesIonic Bonding: © 2013 Marshall Cavendish International (Singapore) Private LimitedSuhaan HussainNo ratings yet
- Ionic BondingDocument51 pagesIonic BondingAaditya MK100% (1)
- BS Chemistry MCQsDocument6 pagesBS Chemistry MCQsRamsha saeedNo ratings yet
- ch02 0310Document26 pagesch02 0310pasternack2379No ratings yet
- When Atoms Meet: Chemical BondingDocument88 pagesWhen Atoms Meet: Chemical BondingWilsonNo ratings yet
- 2nd Quarter Reviewer - CHEMISTRYDocument2 pages2nd Quarter Reviewer - CHEMISTRYKate Ashley CailingNo ratings yet
- Atomic StructureDocument13 pagesAtomic Structurerenan.masangya-18No ratings yet
- Chemical Bonding - Ionic BondDocument56 pagesChemical Bonding - Ionic BondFianna GalleroNo ratings yet
- Structure and Bonding: A Guide For A Level StudentsDocument86 pagesStructure and Bonding: A Guide For A Level StudentshaidarNo ratings yet
- Atomic StructureDocument25 pagesAtomic Structuremichaeldevid7890No ratings yet
- Bonding Forces and Energies Al Francis Asuncion EE 3bDocument8 pagesBonding Forces and Energies Al Francis Asuncion EE 3bAce John OliverNo ratings yet
- Materials Science - Lecture #2 - Atomic StructureDocument62 pagesMaterials Science - Lecture #2 - Atomic Structureisura678hasankaNo ratings yet
- Semi Conductor2Document60 pagesSemi Conductor2mythonyNo ratings yet
- Chapter 7 Ionic and Metallic BondingDocument56 pagesChapter 7 Ionic and Metallic BondingCharles GibbsNo ratings yet
- Dwnload Full Descriptive Inorganic Chemistry 6th Edition Rayner Canham Solutions Manual PDFDocument35 pagesDwnload Full Descriptive Inorganic Chemistry 6th Edition Rayner Canham Solutions Manual PDFmarieramirezsaqdmkpfgb100% (16)
- Lec 4Document10 pagesLec 4Maina PhysicistNo ratings yet
- Chemical BondingDocument19 pagesChemical Bondinghassanyaraanka28No ratings yet
- Chemistry Reviewer: Capacity of Atoms To From Bonds. Elements in The Periodic TableDocument4 pagesChemistry Reviewer: Capacity of Atoms To From Bonds. Elements in The Periodic TableKatrina BernardinoNo ratings yet
- Chemical BondDocument64 pagesChemical BondangelieshaynnmtalagtagNo ratings yet
- G9.MODULE 2 Lesson 6.2 Formation of IonsDocument10 pagesG9.MODULE 2 Lesson 6.2 Formation of IonsAndrina Binogwal TocgongnaNo ratings yet
- UntitledDocument6 pagesUntitledSuzuki YatoNo ratings yet
- The Atom Activities: Discovery of Electron Democritus Proposes The Word "Atom" Current ModelDocument8 pagesThe Atom Activities: Discovery of Electron Democritus Proposes The Word "Atom" Current ModelHunter PpollNo ratings yet
- ChembondDocument51 pagesChembondNovelynLozano-EdrosoNo ratings yet
- ch02 RevDocument22 pagesch02 RevFasih Ul HaqNo ratings yet
- Forming IonsDocument22 pagesForming IonsSamantha Shane P. ConumNo ratings yet
- Covalent BondsDocument6 pagesCovalent BondsimunasingheNo ratings yet
- ENS167 Lecture 2Document22 pagesENS167 Lecture 2JANNA MARIE NInA BATENo ratings yet
- Chem 1 MY18 Lec Session 6, 7 PDFDocument13 pagesChem 1 MY18 Lec Session 6, 7 PDFBytehubNetcafeNo ratings yet
- Lecture 1Document23 pagesLecture 1sanjunaNo ratings yet
- Tema 3. Chemical BondingDocument68 pagesTema 3. Chemical Bondingkarthik tvkNo ratings yet
- Track Your LearningDocument1 pageTrack Your Learningapi-263909505No ratings yet
- Group ContractDocument1 pageGroup Contractapi-263909505No ratings yet
- Group ContractDocument3 pagesGroup Contractapi-263909505No ratings yet
- Angel EsquedaDocument1 pageAngel Esquedaapi-263909505No ratings yet
- Track Your LearningDocument1 pageTrack Your Learningapi-263909505No ratings yet
- Delilah GonzalezDocument1 pageDelilah Gonzalezapi-263909505No ratings yet
- Acids and BasesDocument57 pagesAcids and Basesapi-263909505No ratings yet
- Stoich Quick RefresherDocument1 pageStoich Quick Refresherapi-263909505No ratings yet
- Bertha ChavezDocument1 pageBertha Chavezapi-263909505No ratings yet
- Density Floating ActivityDocument1 pageDensity Floating Activityapi-263909505No ratings yet
- Ayala AilynDocument1 pageAyala Ailynapi-263909505No ratings yet
- Chavez VictorDocument1 pageChavez Victorapi-263909505No ratings yet
- Sandoval DemetriDocument1 pageSandoval Demetriapi-263909505No ratings yet
- Intermolecular ForcesDocument18 pagesIntermolecular Forcesapi-263909505No ratings yet
- Banda JenniferDocument1 pageBanda Jenniferapi-263909505No ratings yet
- Naming Simple MoleculesDocument1 pageNaming Simple Moleculesapi-263909505No ratings yet
- Ink ChromatographyDocument9 pagesInk Chromatographyapi-2639095050% (1)
- Leyva AriamDocument1 pageLeyva Ariamapi-263909505No ratings yet
- Ramirez JoseDocument1 pageRamirez Joseapi-263909505No ratings yet
- Franco NikkoDocument1 pageFranco Nikkoapi-263909505No ratings yet
- Brite JimmyDocument1 pageBrite Jimmyapi-263909505No ratings yet
- Alavarez ArmandoDocument1 pageAlavarez Armandoapi-263909505No ratings yet
- Llanas AlexisDocument1 pageLlanas Alexisapi-263909505No ratings yet
- Flores SammyDocument1 pageFlores Sammyapi-263909505No ratings yet
- Marzan MiguelDocument1 pageMarzan Miguelapi-263909505No ratings yet
- Ortega DavidDocument1 pageOrtega Davidapi-263909505No ratings yet
- Martinez KevinDocument1 pageMartinez Kevinapi-263909505No ratings yet
- Torres CassieDocument1 pageTorres Cassieapi-263909505No ratings yet
- Hernandez CrystalDocument1 pageHernandez Crystalapi-263909505No ratings yet
- Esqueda DanielleDocument1 pageEsqueda Danielleapi-263909505No ratings yet