Professional Documents
Culture Documents
Ayala Ailyn
Ayala Ailyn
Uploaded by
api-2639095050 ratings0% found this document useful (0 votes)
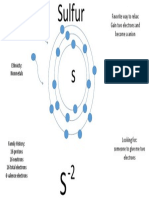
18 views1 pageBromine is a nonmetal halogen with 35 protons, 35 electrons, and 45 neutrons. It has 7 valence electrons and forms an anion called Br-1 by gaining 1 electron. Bromine's favorite way to relax is by gaining an electron to become a negatively charged ion.
Original Description:
Original Title
ayala ailyn
Copyright
© © All Rights Reserved
Available Formats
PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentBromine is a nonmetal halogen with 35 protons, 35 electrons, and 45 neutrons. It has 7 valence electrons and forms an anion called Br-1 by gaining 1 electron. Bromine's favorite way to relax is by gaining an electron to become a negatively charged ion.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
Download as pdf or txt
0 ratings0% found this document useful (0 votes)
18 views1 pageAyala Ailyn
Ayala Ailyn
Uploaded by
api-263909505Bromine is a nonmetal halogen with 35 protons, 35 electrons, and 45 neutrons. It has 7 valence electrons and forms an anion called Br-1 by gaining 1 electron. Bromine's favorite way to relax is by gaining an electron to become a negatively charged ion.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
Download as pdf or txt
You are on page 1of 1
Group
Bromine
Nonmetal
Halogens
35 protons
35 electrons
45 neutrons
7 valence
electrons
Br-1
Favorite way to
relax: Gain 1
electron and
become
anion
Looking for
someone to give
me 1 electron
You might also like
- Radical Substitution 1Document11 pagesRadical Substitution 1will hayNo ratings yet
- Chapter 6 CompoundsDocument49 pagesChapter 6 CompoundsUrooj GulNo ratings yet
- Class01 Science G10 NotesDocument17 pagesClass01 Science G10 Notesemmazou18No ratings yet
- Grade 11 ReviewDocument14 pagesGrade 11 ReviewAiza Casinillo CabatinganNo ratings yet
- Atomic Structure Revision QuestionsDocument1 pageAtomic Structure Revision Questionsrachael.knightNo ratings yet
- Class02 Chemistry G11 Notes Jul 31Document16 pagesClass02 Chemistry G11 Notes Jul 31luengsaraNo ratings yet
- 4.1 - 4.2 & 4.3 The Ionic Bond-1Document24 pages4.1 - 4.2 & 4.3 The Ionic Bond-1서태현No ratings yet
- Year 10 Chemistry: (Made Using Pages Taken From Old Bangor Revision Guides)Document32 pagesYear 10 Chemistry: (Made Using Pages Taken From Old Bangor Revision Guides)jaeNo ratings yet
- Organic Chemistry: IsomerismDocument21 pagesOrganic Chemistry: IsomerismIsabellaNo ratings yet
- Periodic TableDocument59 pagesPeriodic TableDelano PeteNo ratings yet
- Chapter 8 Models of Matter Atoms and MoleculesDocument38 pagesChapter 8 Models of Matter Atoms and MoleculesLin Xiao FenNo ratings yet
- Atomic Structure: Mrs Griffiths Wellington SchoolDocument65 pagesAtomic Structure: Mrs Griffiths Wellington SchoolSaiyed Qamar RizwanNo ratings yet
- Atomic Numbers.: Done By: Kimmi Lee (7) Group KryptonDocument7 pagesAtomic Numbers.: Done By: Kimmi Lee (7) Group KryptonKimmileexqNo ratings yet
- Welcome To SCIE 150: The Science of EverythingDocument46 pagesWelcome To SCIE 150: The Science of Everythingandroidc660No ratings yet
- Ch13 ElectrolysisDocument35 pagesCh13 ElectrolysishahaNo ratings yet
- Chemical Bond: A Quick Review . - A Bond Results From The Attraction of Nuclei For ElectronsDocument35 pagesChemical Bond: A Quick Review . - A Bond Results From The Attraction of Nuclei For ElectronsAlessandra CastroNo ratings yet
- Grade 8 Elements and CompoundsDocument68 pagesGrade 8 Elements and CompoundsreddypoulomiNo ratings yet
- Form 4: The Structure of The AtomDocument9 pagesForm 4: The Structure of The AtomannatsubakiNo ratings yet
- Chem1A Study Guide 1Document2 pagesChem1A Study Guide 1Nairl BoltonNo ratings yet
- GCSE Science ChemistryDocument48 pagesGCSE Science ChemistryRobsonNo ratings yet
- Aromatic Compounds EditedDocument42 pagesAromatic Compounds EditedHabtamu GetaNo ratings yet
- Molecular PolarityDocument31 pagesMolecular PolarityRCMI GMANo ratings yet
- Chemistry GcseDocument68 pagesChemistry GcseSir BirdyNo ratings yet
- Atom_&_Atomic_StructureDocument7 pagesAtom_&_Atomic_StructuretonyNo ratings yet
- Electronic Take Home Exam (e-THE) - Chemistry - Smart EDGE Fiber To The HOME APR 2024 (F2F)Document22 pagesElectronic Take Home Exam (e-THE) - Chemistry - Smart EDGE Fiber To The HOME APR 2024 (F2F)Ellie SalvadorNo ratings yet
- Atoms Molecules Ions PDFDocument41 pagesAtoms Molecules Ions PDFJoriel GeroquiaNo ratings yet
- ATOMIC STRUCTUReeDocument20 pagesATOMIC STRUCTUReeCahayaniFanihuNo ratings yet
- Track Your LearningDocument1 pageTrack Your Learningapi-263909505No ratings yet
- Group ContractDocument1 pageGroup Contractapi-263909505No ratings yet
- Group ContractDocument3 pagesGroup Contractapi-263909505No ratings yet
- Angel EsquedaDocument1 pageAngel Esquedaapi-263909505No ratings yet
- Track Your LearningDocument1 pageTrack Your Learningapi-263909505No ratings yet
- Delilah GonzalezDocument1 pageDelilah Gonzalezapi-263909505No ratings yet
- Acids and BasesDocument57 pagesAcids and Basesapi-263909505No ratings yet
- Stoich Quick RefresherDocument1 pageStoich Quick Refresherapi-263909505No ratings yet
- Bertha ChavezDocument1 pageBertha Chavezapi-263909505No ratings yet
- Density Floating ActivityDocument1 pageDensity Floating Activityapi-263909505No ratings yet
- Franco NikkoDocument1 pageFranco Nikkoapi-263909505No ratings yet
- Brite JimmyDocument1 pageBrite Jimmyapi-263909505No ratings yet
- Ramirez JoseDocument1 pageRamirez Joseapi-263909505No ratings yet
- Intermolecular ForcesDocument18 pagesIntermolecular Forcesapi-263909505No ratings yet
- Flores SammyDocument1 pageFlores Sammyapi-263909505No ratings yet
- Naming Simple MoleculesDocument1 pageNaming Simple Moleculesapi-263909505No ratings yet
- Ink ChromatographyDocument9 pagesInk Chromatographyapi-2639095050% (1)
- Leyva AriamDocument1 pageLeyva Ariamapi-263909505No ratings yet
- Banda JenniferDocument1 pageBanda Jenniferapi-263909505No ratings yet
- Alavarez ArmandoDocument1 pageAlavarez Armandoapi-263909505No ratings yet
- Aldaba JosueDocument1 pageAldaba Josueapi-263909505No ratings yet
- Sandoval DemetriDocument1 pageSandoval Demetriapi-263909505No ratings yet
- Llanas AlexisDocument1 pageLlanas Alexisapi-263909505No ratings yet
- Chavez VictorDocument1 pageChavez Victorapi-263909505No ratings yet
- Marzan MiguelDocument1 pageMarzan Miguelapi-263909505No ratings yet
- Ortega DavidDocument1 pageOrtega Davidapi-263909505No ratings yet
- Martinez KevinDocument1 pageMartinez Kevinapi-263909505No ratings yet
- Torres CassieDocument1 pageTorres Cassieapi-263909505No ratings yet
- Hernandez CrystalDocument1 pageHernandez Crystalapi-263909505No ratings yet
- Esqueda DanielleDocument1 pageEsqueda Danielleapi-263909505No ratings yet