Professional Documents
Culture Documents
Add NH Solution Cation Solution: Coloured White
Add NH Solution Cation Solution: Coloured White
Uploaded by
chongpeisi0 ratings0% found this document useful (0 votes)
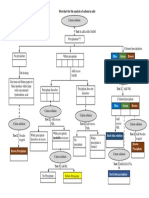
9 views1 pageA cation solution was added to NH3 solution which caused precipitates to form that were white, green, or brown depending on the cation. Excess NH3 turned the solution blue, and some precipitates dissolved while others remained undissolved.
Original Description:
Original Title
Cation Test2
Copyright
© © All Rights Reserved
Available Formats
DOC, PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentA cation solution was added to NH3 solution which caused precipitates to form that were white, green, or brown depending on the cation. Excess NH3 turned the solution blue, and some precipitates dissolved while others remained undissolved.
Copyright:
© All Rights Reserved
Available Formats
Download as DOC, PDF, TXT or read online from Scribd
Download as doc, pdf, or txt
0 ratings0% found this document useful (0 votes)
9 views1 pageAdd NH Solution Cation Solution: Coloured White
Add NH Solution Cation Solution: Coloured White
Uploaded by
chongpeisiA cation solution was added to NH3 solution which caused precipitates to form that were white, green, or brown depending on the cation. Excess NH3 turned the solution blue, and some precipitates dissolved while others remained undissolved.
Copyright:
© All Rights Reserved
Available Formats
Download as DOC, PDF, TXT or read online from Scribd
Download as doc, pdf, or txt
You are on page 1of 1
Add NH3 solution
Cation solution
Percipitates
Unchanges
Coloured
White
Green
MC
Brown
Excess NH3
Blue
Dissolved
Undissolved
You might also like
- Flowchart For The Analysis of Cations in Salts: Blue Green BrownDocument1 pageFlowchart For The Analysis of Cations in Salts: Blue Green BrownKuan LoongNo ratings yet
- Kate Coleen D. Galera BS in Chemistry II May 4, 2017 Experiment 12 Amines, Amino Acids and ProteinsDocument9 pagesKate Coleen D. Galera BS in Chemistry II May 4, 2017 Experiment 12 Amines, Amino Acids and ProteinsKateNo ratings yet
- Lab 5 Properties of Acids and BasesDocument5 pagesLab 5 Properties of Acids and BasesmicahalanhuismanNo ratings yet
- Pink Color + Precipitate Pink Color + PrecipitateDocument3 pagesPink Color + Precipitate Pink Color + PrecipitateAnggraini Nugroho PNo ratings yet
- Chemistry 130.1 NOR-AINADocument19 pagesChemistry 130.1 NOR-AINACriszia Mae FloresNo ratings yet
- Ready Reference ChartDocument6 pagesReady Reference Chartjayprakashverma2006No ratings yet
- Food Tests Bio Excercise NotesDocument8 pagesFood Tests Bio Excercise NotesmelyndasiimweNo ratings yet
- Carbonyl Compounds and Carbohydrates I.Data and Results Compound Homogeneity ObservationsDocument11 pagesCarbonyl Compounds and Carbohydrates I.Data and Results Compound Homogeneity ObservationsKateNo ratings yet
- Qualitative AnalysisDocument4 pagesQualitative AnalysisVictor OkosunNo ratings yet
- CHM 361 3 FullDocument7 pagesCHM 361 3 FullIsmi FadliNo ratings yet
- Chemistry Practical RevisionDocument23 pagesChemistry Practical RevisionRachel RachNo ratings yet
- 49 Question Trial SPM ChemistryDocument1 page49 Question Trial SPM ChemistrychongpeisiNo ratings yet
- PPD Pasir Gudang Johor Modul Hots Kbat Kimia Tingkatan 5Document1 pagePPD Pasir Gudang Johor Modul Hots Kbat Kimia Tingkatan 5chongpeisiNo ratings yet
- 2016 Pcpaper 1Document28 pages2016 Pcpaper 1chongpeisiNo ratings yet
- Module 5 Mole Concept in Chemical EquationDocument7 pagesModule 5 Mole Concept in Chemical EquationchongpeisiNo ratings yet
- QuizDocument2 pagesQuizchongpeisiNo ratings yet
- Ex 6 - Empirical FormulaDocument2 pagesEx 6 - Empirical FormulachongpeisiNo ratings yet
- Anion Test: 1 Reageant Anion X Observation Inferences 1 A B C D 2 1 A 2 B C D 3 1 A 2 B C D 4 1 A 2 B 3 C DDocument1 pageAnion Test: 1 Reageant Anion X Observation Inferences 1 A B C D 2 1 A 2 B C D 3 1 A 2 B C D 4 1 A 2 B 3 C DchongpeisiNo ratings yet
- Module 1 Chemical FormulaeDocument5 pagesModule 1 Chemical FormulaechongpeisiNo ratings yet
- Acid N BaseDocument2 pagesAcid N BasechongpeisiNo ratings yet
- ENGLISH Scheme of Work FORM 5 2016 - KamalaDocument12 pagesENGLISH Scheme of Work FORM 5 2016 - Kamalachongpeisi100% (1)
- Technique of Answering SPM Chemistry: Disediakan Oleh Chong Pei SiDocument8 pagesTechnique of Answering SPM Chemistry: Disediakan Oleh Chong Pei SichongpeisiNo ratings yet
- CH 7 ObjDocument4 pagesCH 7 ObjchongpeisiNo ratings yet
- Ex 8 - Numerical Problems Using Chemical EquationsDocument1 pageEx 8 - Numerical Problems Using Chemical EquationschongpeisiNo ratings yet
- Revision Champion2Document62 pagesRevision Champion2chongpeisiNo ratings yet
- Terengganu Chemistry Paper 1,2,3 (Included Marking Scheme)Document71 pagesTerengganu Chemistry Paper 1,2,3 (Included Marking Scheme)chongpeisiNo ratings yet
- Exercise ElectrochemistryDocument1 pageExercise ElectrochemistrychongpeisiNo ratings yet
- Analytical Exercise ElectrochemistryDocument1 pageAnalytical Exercise ElectrochemistrychongpeisiNo ratings yet
- Chemical Formula AND Chemical Equations: No. Ionic Compound Formula No. Ionic Compound FormulaDocument2 pagesChemical Formula AND Chemical Equations: No. Ionic Compound Formula No. Ionic Compound FormulachongpeisiNo ratings yet