Professional Documents
Culture Documents
Graph of Energy Vs I.E. of Number of Magnesium
Graph of Energy Vs I.E. of Number of Magnesium
Uploaded by
Michelle Ivashkov0 ratings0% found this document useful (0 votes)
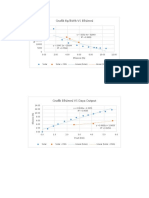
10 views1 pageThis graph shows the relationship between energy in kilojoules and the ionization energy number of magnesium. As the ionization energy number increases from 1 to 8, the energy required also increases steadily from around 5000 kJ to over 25,000 kJ, indicating that more energy is needed to remove each additional electron from the magnesium atom.
Original Description:
ionisation energy
Original Title
Magnesium IE
Copyright
© © All Rights Reserved
Available Formats
DOCX, PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentThis graph shows the relationship between energy in kilojoules and the ionization energy number of magnesium. As the ionization energy number increases from 1 to 8, the energy required also increases steadily from around 5000 kJ to over 25,000 kJ, indicating that more energy is needed to remove each additional electron from the magnesium atom.
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
Download as docx, pdf, or txt
0 ratings0% found this document useful (0 votes)
10 views1 pageGraph of Energy Vs I.E. of Number of Magnesium
Graph of Energy Vs I.E. of Number of Magnesium
Uploaded by
Michelle IvashkovThis graph shows the relationship between energy in kilojoules and the ionization energy number of magnesium. As the ionization energy number increases from 1 to 8, the energy required also increases steadily from around 5000 kJ to over 25,000 kJ, indicating that more energy is needed to remove each additional electron from the magnesium atom.
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
Download as docx, pdf, or txt
You are on page 1of 1
Graph of Energy vs I.E.
of Number of Magnesium
30000
25000
20000
Energy (kJ)
15000
10000
5000
0
4
I.E. Number
Energy (kJ)
You might also like
- The Woman Speaks To The Man Who Has Employed Her SonDocument3 pagesThe Woman Speaks To The Man Who Has Employed Her SonKerry-Ann Williams40% (5)
- Chemguide - Questions: Further Ionisation EnergiesDocument2 pagesChemguide - Questions: Further Ionisation EnergiesRoshae SinclairNo ratings yet
- Figure 7: Relation Between UHV & GCV (Jharia Coalfields)Document2 pagesFigure 7: Relation Between UHV & GCV (Jharia Coalfields)Manish KumarNo ratings yet
- Exercises Day 1Document6 pagesExercises Day 1LLEHCIMNo ratings yet
- Conversion Factors For Units of EnergyDocument1 pageConversion Factors For Units of EnergyAfonso CuambaNo ratings yet
- Experiment 3Document6 pagesExperiment 3rastgonikoNo ratings yet
- Grafik RP/KWH Vs EfisiensiDocument2 pagesGrafik RP/KWH Vs EfisiensiM Afta SyaifudinNo ratings yet
- Electron Structure, Ionisation Energy WorksheetDocument1 pageElectron Structure, Ionisation Energy Worksheetyathinp822No ratings yet
- Photovoltaic Simulation Tools ComparisonDocument11 pagesPhotovoltaic Simulation Tools ComparisonIvan BevandaNo ratings yet
- OLS - Forecasting Daily Monthly Demand PowerDocument20 pagesOLS - Forecasting Daily Monthly Demand PowerRajat AggarwalNo ratings yet
- Electric Vehicle Fire Testing: Dean D. MacneilDocument19 pagesElectric Vehicle Fire Testing: Dean D. Macneilluke hainesNo ratings yet
- UDL On Simply Supported Beam123Document6 pagesUDL On Simply Supported Beam123bhagvat daveNo ratings yet
- Uniformly Distributed Load On Simply Supported Beam 1 Purpose of CalculationDocument6 pagesUniformly Distributed Load On Simply Supported Beam 1 Purpose of CalculationRam LangheNo ratings yet
- Ch3 WS1Document1 pageCh3 WS1FerminNo ratings yet
- ResultsnklbkvjDocument2 pagesResultsnklbkvjMarielle PerejonNo ratings yet
- Electron Arrangement Successive Ionisation Energies For PotassiumDocument1 pageElectron Arrangement Successive Ionisation Energies For PotassiumMahfuz A. AzimNo ratings yet
- Ffects of Design Parameters On Operating: E Characteristics of An Electric Assisted Bicycle Using Fuel CellDocument12 pagesFfects of Design Parameters On Operating: E Characteristics of An Electric Assisted Bicycle Using Fuel CellSaurabh ChougaleNo ratings yet
- Case Study: HVAC System Optimization: SampleDocument1 pageCase Study: HVAC System Optimization: SampleGabrielMattosdeOliveiraNo ratings yet
- Experiment 4. Electrochemical Cells. Ocampo. 3CHEM1 PDFDocument6 pagesExperiment 4. Electrochemical Cells. Ocampo. 3CHEM1 PDFEsmeralda A OcampoNo ratings yet
- Gen Performance - Solar RooftopDocument6 pagesGen Performance - Solar RooftopShilpi GuptaNo ratings yet
- Model A Model B Mix Profits Total Profit Constraints Engine Assembly Metal Stamping Model A Assembly Model B AssemblyDocument2 pagesModel A Model B Mix Profits Total Profit Constraints Engine Assembly Metal Stamping Model A Assembly Model B AssemblyPNo ratings yet
- Escuela Académica Profesional de Ingeniería IndustrialDocument4 pagesEscuela Académica Profesional de Ingeniería IndustrialAlisson Sofia ASCOY IPARRAGUIRRENo ratings yet
- Ion Detection Efficiency of Microchannel Plates: 20 KV 20 KVDocument1 pageIon Detection Efficiency of Microchannel Plates: 20 KV 20 KVMohammadNo ratings yet
- Fourth Form Energy Notes - Fill in Andeev Notes 123Document1 pageFourth Form Energy Notes - Fill in Andeev Notes 123eviegracehaiandersonNo ratings yet
- System I (1 X Cent 1000 T.R) System II (1 X 1000 T.R) ABSORPTIONDocument1 pageSystem I (1 X Cent 1000 T.R) System II (1 X 1000 T.R) ABSORPTIONpicuNo ratings yet
- DQ of ElectrochemistryDocument25 pagesDQ of Electrochemistryabhinavsharmah101No ratings yet
- Magnetic Field Vs CurrentDocument2 pagesMagnetic Field Vs CurrentShubham ChandraNo ratings yet
- System I (1 X Cent 1000 T.R) System II (1 X 1000 T.R) ABSORPTIONDocument1 pageSystem I (1 X Cent 1000 T.R) System II (1 X 1000 T.R) ABSORPTIONpicuNo ratings yet
- Physics Criterion CDocument4 pagesPhysics Criterion CMs MsNo ratings yet
- EEGUCDocument135 pagesEEGUCR SKNo ratings yet
- An Anfis-Based Prediction For Monthly Clearness Index and Daily Solar Radiation: Application For Sizing of A Stand-Alone Photovoltaic SystemDocument21 pagesAn Anfis-Based Prediction For Monthly Clearness Index and Daily Solar Radiation: Application For Sizing of A Stand-Alone Photovoltaic SystemJhon Peñalva SanchezNo ratings yet
- Problem37 44Document1 pageProblem37 44IENCSNo ratings yet
- Construction and Analysis of Generator Actual Capability Curves Using The New MethodDocument11 pagesConstruction and Analysis of Generator Actual Capability Curves Using The New MethodMuthukumar SivaramanNo ratings yet
- Lab 3Document10 pagesLab 3AliNo ratings yet
- Unit Ii Balancing Part ADocument15 pagesUnit Ii Balancing Part AsureshkumarNo ratings yet
- 11TH Jee Level 1Document4 pages11TH Jee Level 1sayalis1604No ratings yet
- Ionisation EnergyDocument4 pagesIonisation EnergyKhadija PrescottNo ratings yet
- Sem1 Unit4 Periodic TableDocument10 pagesSem1 Unit4 Periodic Tableshehdilanun0% (1)
- Solutions Acceleration of A Geared SystemDocument13 pagesSolutions Acceleration of A Geared SystemSam StideNo ratings yet
- Modeling and Analysis of PV Fed DC-DC Converters: P. Maithili, J. Kanakaraj, T.ViswanathanDocument4 pagesModeling and Analysis of PV Fed DC-DC Converters: P. Maithili, J. Kanakaraj, T.ViswanathanVishal KevatNo ratings yet
- Chapter (2) Atomic Structure and BondingDocument56 pagesChapter (2) Atomic Structure and BondingJessica De GuzmanNo ratings yet
- Newtons Second LawDocument10 pagesNewtons Second LawMike DesaiNo ratings yet
- Icmcer-2024Document25 pagesIcmcer-2024himanshumishra.nitrNo ratings yet
- 604 2014 1389 Moesm1 EsmDocument6 pages604 2014 1389 Moesm1 EsmDavid AriasNo ratings yet
- Design, Modeling and Control of A Reaction Wheel Balanced Inverted PendulumDocument9 pagesDesign, Modeling and Control of A Reaction Wheel Balanced Inverted PendulumCarlos AndresNo ratings yet
- Part 1 MD Simulation SaccharineDocument4 pagesPart 1 MD Simulation SaccharineNino AmigoNo ratings yet
- Article Text 511953 1 10 20210325 PDFDocument19 pagesArticle Text 511953 1 10 20210325 PDFderbalt, noraNo ratings yet
- Dr. Nikhil PPT On Start UP Program - NISEDocument68 pagesDr. Nikhil PPT On Start UP Program - NISEShadifNo ratings yet
- Chapter 3 - Periodic Table - QDocument4 pagesChapter 3 - Periodic Table - QhernaniabdullahNo ratings yet
- Determination of Trace Elements: ReagentsDocument4 pagesDetermination of Trace Elements: ReagentsGustavo Tovar GaitanNo ratings yet
- Hbojrn3oncp1m Eci1eocnepclm12e C - 2e - U2encpk - 2klecDocument1 pageHbojrn3oncp1m Eci1eocnepclm12e C - 2e - U2encpk - 2klecErik MendozaNo ratings yet
- AC TI: Department of EducationDocument7 pagesAC TI: Department of EducationAne Abon CandidoNo ratings yet
- Lab 5 AnalogDocument13 pagesLab 5 Analogabangcik0099No ratings yet
- Determination of The Effective Energy in X-Rays Standard Beams, Mammography LevelDocument6 pagesDetermination of The Effective Energy in X-Rays Standard Beams, Mammography LevelnyeinhsuthantharNo ratings yet
- Lab 08 Calculations and DiscussionDocument14 pagesLab 08 Calculations and DiscussionahmadNo ratings yet
- Exercise No. 3 Table 2Document1 pageExercise No. 3 Table 2rkdias005No ratings yet
- Inc Junior Model-B Physics NpyqDocument41 pagesInc Junior Model-B Physics NpyqDikshan M HollaNo ratings yet
- Centrifugal ForceDocument6 pagesCentrifugal ForceJayDee ReynosoNo ratings yet
- Hayat PumpDocument4 pagesHayat PumpshaheenikhtiyaralikhanNo ratings yet
- Introduction To Static MechanicsDocument17 pagesIntroduction To Static MechanicsMustafa JasimNo ratings yet
- Experiment 07Document12 pagesExperiment 07waleedNo ratings yet
- Trinidad Perspective After COP21Document5 pagesTrinidad Perspective After COP21Michelle IvashkovNo ratings yet
- T&T Among Most Polluted in World: PublishedDocument13 pagesT&T Among Most Polluted in World: PublishedMichelle IvashkovNo ratings yet
- Subsistence Hunting in BrazilDocument17 pagesSubsistence Hunting in BrazilMichelle IvashkovNo ratings yet
- Environment Reporter, BBC News 24 December 2015: by Mark KinverDocument9 pagesEnvironment Reporter, BBC News 24 December 2015: by Mark KinverMichelle IvashkovNo ratings yet
- Global InteractionsDocument20 pagesGlobal InteractionsMichelle IvashkovNo ratings yet
- MetaphorsDocument26 pagesMetaphorsMichelle IvashkovNo ratings yet
- "Once Upon A Time" by Gabriel OkaraDocument11 pages"Once Upon A Time" by Gabriel OkaraMichelle IvashkovNo ratings yet