Professional Documents
Culture Documents
Microbateri in Food and Water
Microbateri in Food and Water
Uploaded by
Arief Happy RachmadhiCopyright:
Available Formats
You might also like
- Lab Report. Serial DilutionDocument5 pagesLab Report. Serial Dilutionanon_747148947100% (2)
- Report BacteriaDocument11 pagesReport BacteriaSuzeanni JalilNo ratings yet
- SPREad PlateDocument7 pagesSPREad PlateJermeLou BaoNo ratings yet
- 9-Food and WaterTHE MICROBIOLOGICAL EXAMINATION OF FOODS & WATERDocument16 pages9-Food and WaterTHE MICROBIOLOGICAL EXAMINATION OF FOODS & WATERArri KurniawanNo ratings yet
- EnumerationDocument6 pagesEnumerationJITHIN VNo ratings yet
- Microbiology Practical 3Document7 pagesMicrobiology Practical 3Kah Jun100% (1)
- Practical 3 Lab Report MicrobiologyDocument14 pagesPractical 3 Lab Report MicrobiologynurulakhmalNo ratings yet
- Chapter 5 - Basic Techniques in Food MicrobiologyDocument54 pagesChapter 5 - Basic Techniques in Food MicrobiologyNurhayati Hasanah100% (1)
- Direct MeasurementDocument33 pagesDirect MeasurementAira VillarinNo ratings yet
- Methods For Microbiological Examination of of FoodDocument5 pagesMethods For Microbiological Examination of of Food9qdd95pw9kNo ratings yet
- Counting BacteriaDocument30 pagesCounting Bacteriayuppie_raj2175100% (1)
- Experiment 3 - Air, Water and Soil PollutionDocument9 pagesExperiment 3 - Air, Water and Soil PollutionTama KalongaNo ratings yet
- Enumeration of Microorganisms: Microbiology BIOL 275Document10 pagesEnumeration of Microorganisms: Microbiology BIOL 275Diya GhosalNo ratings yet
- C5 Mic 254Document64 pagesC5 Mic 254Aqilah NajwaNo ratings yet
- Exp4 Total Count BacteriaDocument6 pagesExp4 Total Count Bacterialzero93100% (2)
- Full Report Bacterial CountDocument10 pagesFull Report Bacterial CountSuzeanni Jalil100% (1)
- Bacterial TitreDocument3 pagesBacterial TitreManjunatha Bukkambudi KrishnaswamyNo ratings yet
- Enumeration of Microorganisms: Microbiology BIOL 275Document10 pagesEnumeration of Microorganisms: Microbiology BIOL 275yudhieNo ratings yet
- EnumerationDocument7 pagesEnumerationSikin Sikin100% (1)
- Introduction To Lab Ex. 19: Enumeration of Bacteria - Pour Plate TechniqueDocument7 pagesIntroduction To Lab Ex. 19: Enumeration of Bacteria - Pour Plate TechniqueRenan RodriguesNo ratings yet
- Practical 12Document7 pagesPractical 125408 ManshaNo ratings yet
- Experiment 5Document5 pagesExperiment 5Nipuni Saubhagya RathnayakeNo ratings yet
- Exercise 17 Microbial Analysis of WaterDocument6 pagesExercise 17 Microbial Analysis of WaterNecka AmoloNo ratings yet
- Microbial Load: Pasteurized vs. Raw Milk Kathy Ziuzianska Biology 240, Section 301 October 19 2011 Robert Bellas, PHDDocument7 pagesMicrobial Load: Pasteurized vs. Raw Milk Kathy Ziuzianska Biology 240, Section 301 October 19 2011 Robert Bellas, PHDkasiapitrasiaNo ratings yet
- Measurement of GrowthDocument6 pagesMeasurement of GrowthPrerna NegiNo ratings yet
- Food Microbiology FullDocument6 pagesFood Microbiology FullhasifNo ratings yet
- Direct Measurements of Microbial Growth: Viable Counts: Experiment 4Document3 pagesDirect Measurements of Microbial Growth: Viable Counts: Experiment 4gsharkzNo ratings yet
- Food Micro Lab 1Document6 pagesFood Micro Lab 1Elcanah Joy BernardoNo ratings yet
- Laboratory Exercise No. 10 Viable Plate Counts Results and DiscussionDocument3 pagesLaboratory Exercise No. 10 Viable Plate Counts Results and Discussionvanessa olga100% (1)
- Bacterial CountsDocument5 pagesBacterial CountsAndrew LeeNo ratings yet
- Bacteriological Water AnalysisDocument6 pagesBacteriological Water Analysisramkrishna01No ratings yet
- UntitledDocument30 pagesUntitledLEE JIA XINNo ratings yet
- Microbial Analysis of FoodDocument77 pagesMicrobial Analysis of FoodAbdurrazaq YakubuNo ratings yet
- Lab 17 Serial DilutionDocument15 pagesLab 17 Serial DilutionCedricDiehlNo ratings yet
- Virology - Isolation & Cultivation of BacgteriophageDocument12 pagesVirology - Isolation & Cultivation of BacgteriophageProbioticsAnywhereNo ratings yet
- Total Bacterial CounrDocument10 pagesTotal Bacterial CounrIrish OrleansNo ratings yet
- Serial Dilution ProtocolsDocument5 pagesSerial Dilution ProtocolsYugendra Babu K100% (1)
- Food Microbiology LR 3 & 4Document17 pagesFood Microbiology LR 3 & 4rishabh bhardwajNo ratings yet
- Standard Plate Count Method: By: Hafiza Asfa Shafique Microbiology BS Biotechnology VDocument13 pagesStandard Plate Count Method: By: Hafiza Asfa Shafique Microbiology BS Biotechnology VJawadNo ratings yet
- Isolation of Microorganism and EnumerationDocument13 pagesIsolation of Microorganism and EnumerationkrbiotechNo ratings yet
- Bacteria EnumerationDocument27 pagesBacteria EnumerationDaisy Arora KhuranaNo ratings yet
- Bacteriological Water AnalysisDocument8 pagesBacteriological Water AnalysisCasio Je-urNo ratings yet
- Formal Report Quantitative Analysis of Microbial Populations Through Standard Viable Plate Count Methods MicrobiologyDocument4 pagesFormal Report Quantitative Analysis of Microbial Populations Through Standard Viable Plate Count Methods MicrobiologyGino100% (1)
- Food Microbiology (CFD 20203) Unikl Lab Manual Micet: Malaysian Institute of Chemical and Bioengineering TechnologyDocument11 pagesFood Microbiology (CFD 20203) Unikl Lab Manual Micet: Malaysian Institute of Chemical and Bioengineering TechnologyLiyana HalimNo ratings yet
- Laboratory Seven: Enumeration of BacteriaDocument6 pagesLaboratory Seven: Enumeration of BacteriaSulav Acharya0% (1)
- Serial Dilutions and PlatingDocument17 pagesSerial Dilutions and PlatingVon Valentine MhuteNo ratings yet
- Food Lab 1Document13 pagesFood Lab 1Nurul Syaheerah100% (1)
- Effects of Nutrition and Temperature On The Population Growth of Bacteria 2011Document9 pagesEffects of Nutrition and Temperature On The Population Growth of Bacteria 2011si_miaomiaoNo ratings yet
- CPB 30103 Biochemical Engineering UniKL MICET Experiment 4: Determination of Bacterial Loads Viable Cell Counts Full Lab ReportDocument12 pagesCPB 30103 Biochemical Engineering UniKL MICET Experiment 4: Determination of Bacterial Loads Viable Cell Counts Full Lab ReportSiti Hajar Mohamed100% (2)
- Lab # 7 - Serial DilutionDocument2 pagesLab # 7 - Serial DilutionKESHAWN TYRONE SUTHERLANDNo ratings yet
- Laboratory 4-6Document3 pagesLaboratory 4-6JOSENo ratings yet
- Count of Colony Forming UnitDocument2 pagesCount of Colony Forming UnitTejas DoshiNo ratings yet
- Laboratory 8 Enumeration: E. Coli Using Three Different Methods: Optical Density, Direct Counts and Plate Counts. YouDocument6 pagesLaboratory 8 Enumeration: E. Coli Using Three Different Methods: Optical Density, Direct Counts and Plate Counts. YouskgohNo ratings yet
- Growth CurveDocument4 pagesGrowth Curvedinchus_106No ratings yet
- Practical - Turbidimetric MethodsDocument9 pagesPractical - Turbidimetric MethodsProbioticsAnywhere100% (1)
- Week 8 Aerobic Plate CountDocument7 pagesWeek 8 Aerobic Plate CountSam 0505No ratings yet
- Outlines of dairy bacteriology A concise manual for the use of students in dairyingFrom EverandOutlines of dairy bacteriology A concise manual for the use of students in dairyingNo ratings yet
- Front Mission 3 - 1Document31 pagesFront Mission 3 - 1Arief Happy RachmadhiNo ratings yet
- Front Mission 3 - 4Document110 pagesFront Mission 3 - 4Arief Happy Rachmadhi0% (1)
- Graywater Guide For Your LandscapeDocument12 pagesGraywater Guide For Your LandscapeU8x58No ratings yet
- FAO Fisheries & Aquaculture - Species Fact Sheets - Penaeus Merguiensis (De Man, 1888)Document4 pagesFAO Fisheries & Aquaculture - Species Fact Sheets - Penaeus Merguiensis (De Man, 1888)Arief Happy RachmadhiNo ratings yet
Microbateri in Food and Water
Microbateri in Food and Water
Uploaded by
Arief Happy RachmadhiCopyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Microbateri in Food and Water
Microbateri in Food and Water
Uploaded by
Arief Happy RachmadhiCopyright:
Available Formats
Food and Water 1
THE MICROBIOLOGICAL EXAMINATION
OF FOODS & WATER
Foods and water contaminated with pathogenic microorganisms are major avenues for the spread
of infectious diseases. In this country, strict state and federal guidelines regulate food and water
processing industries, even so, many outbreaks of diseases linked to contaminated foods or water
occur every year.
Municipal water and food products are routinely monitored for both the total number of
microorganisms and presence of pathogens, but there are many challenges to doing so. Even
nonpathogens, if present in large enough numbers, can cause deterioration of food products and
distaste in water. And testing for all types of pathogens is not practical since we do not know which
may be present.
The alternative to testing for many different types of pathogens is to test for bacteria that
indicate the potential presence of pathogens. The most common source of pathogens in food and
water is fecal contamination, of either animal or human origin. Feces also contain innumerable
nonpathogenic bacteria, and the presence of these other bacteria in food or water indicates that fecal
contamination has occurred (undesirable enough) and the potential presence of pathogens. The
most commonly used indicator bacteria are fecal coliform and fecal enterococci.
Coliform Bacteria
Coliform bacteria can serve as indicators of fecal contamination. They are not themselves
pathogenic but are denizens of the digestive systems of animals, and thus abundant in feces.
Coliform are defined as Gram-negative aerobic or facultative anaerobes, nonspore-forming, rod
shaped bacteria that ferment lactose with acid and gas production. E. coli, the most abundant
bacterium of the human colon, is the most important indicator of human fecal contamination.
However, some coliform bacteria, such as Enterobacter aerogenes, are of non-fecal origin and may
be present in uncontaminated samples. Thus testing for coliform bacteria involves several tests to
minimize the possibility of false positive results. Bear in mind that the presence of indicator bacteria
does not mean that human pathogens are definitely present, but their presence suggests that the fecal
contamination has occurred, and that pathogens may be present.
Methods of counting bacteria
There are two standard methods of counting bacteria: the standard plate count (SPC) and the
most probable number (MPN) methods. Either technique can be used with selective or nonselective technique the method of choice depends largely upon the number of bacteria to be
counted in the sample. The SPC is routinely used for samples that have a relatively large number
of bacteria, which can be diluted down and grown as a countable number of colonies in a petri plate.
The MPN method is used when the number of bacteria to be counted is so low that the cells could
not be detected if a sample were applied to a plate, which is often the case when counting coliform
bacteria.
Food and Water 2
Summary of exercise
1. You will count the total number of bacteria in a sample of spoiled milk using the Standard Plate
Count technique.
2. You will perform a coliform count of a water sample using the Most Probable Number (MPN)
technique.
I. Standard Plate Count of Bacteria in Food Products
The Standard Plate Count is the most common method used to quantify bacteria in foods. To
perform a standard plate count, the food to be tested is suspended in liquid and a sample is then
spread over the surface of a solid medium in a petri plate. Bacterial cells present will form colonies
that can be counted to determine the number of cells in the original sample. When the objective is
to estimate the total number of bacteria, a complex medium called Plate Count Agar is commonly
used since it will support growth of many different types of bacteria. We call the results the number
of Colony Forming Units (PFU), not total bacteria. This is because no single culture medium will
support all different types of bacteria, we can only count those that do grow to form a visible colony.
Serial Dilution of samples
When performing a bacteria count, between 30 and 300 bacterial colonies need to be on the
plate. A minimum of 30 assures that the data is statistically reliable; however, if more than 300
colonies are present, competition for nutrients can suppress growth of colonies. For example, if a
sample were to contain 106 cells\ml, a 1 ml sample would contain 106 bacteria far more than the
300 cell limit of a standard plate count and the sample must be serially diluted:
The standard way to dilute a sample in microbiology is through Serial Dilution. As shown in
Figure 1, a sample is diluted step-wise in a series of tubes containing sterile diluent. Each step yields
a particular dilution factor:
Dilution factor for each step
Final volume in the tube
Vol of the sample added to the tube
from which the total dilution of the sample can be calculated:
Total dilution of a sample plated =
Product of all dilution steps
Vol of the sample plated
Since the original concentration of bacteria in the milk sample is unknown, we will plate several
dilutions, and hope that at least one will yield between 30- 300 colonies on the plate.
***Do the practice problems at the end of this lab exercise; these must be
turned in with the results of this lab exercise.***
Food and Water 3
Figure 1. Example dilution series for a sample.
Your dilution series will be longer, and you will plate the 4
highest dilutions.
Supplies
sterile 1 ml pipets
milk sample - on front bench
7 tubes containing 9 ml of sterile distilled water (yellow caps)
4 plates containing Plate Count bottom agar.
4 tubes containing 4 ml of overlay agar held @ 50EC, clear cap
Procedures
First, some helpful hints . . .
When performing your dilutions:
1. Be sure to observe aseptic technique, and use a new sterile pipet for each dilution step.
Why?
2. Thoroughly mix each tube before preceding with the next dilution step. However, do not
mix so violently that the contents splash near the cap.
Food and Water 4
1. Preparing the serial dilutions
As previously noted, some guesswork must be performed to estimate the number of bacteria
in food samples. In this case we will estimate that the spoiled milk contains 105 - 109
bacteria/ml.
1. Plan your serial dilution clearly and copy them neatly in the space provided on the Results
pages.
2. You will plate samples from samples diluted from 104 to 107. (Note that 30 colonies on a
plate at 104 dilution represents an original concentration of 3 x 105, and 300 colonies on a
plate at 107 dilution represents an original concentration of 3 x 109.)
2. Plating the diluted samples
A 1 ml sample from the selected tubes will be plated using the Agar Overlay Method. This
technique helps to distribute the cells evenly over the plate surface.
1. Label along the edge of the bottom of 4 bottom agar plates with:
a. your name(s) and date
b. the dilution factor of the sample plated
Prepare one overlay at a time following this procedure.
2. Working near one of the 50EC water baths, aseptically pipet 1 ml into a tube of soft agar.
3. Mix the overlay thoroughly by rotating the tube between the palms of your hands.
4. Pour the overlay onto the bottom agar, reposition the lid, and gently swirl the plate to
spread the overlay.
5. Return the plate to your bench and allow the overlay to solidify.
6. Incubate all of the plates at 37EC for 24 to 48 hours.
3. Counting the colonies
The colonies can be easily counted with the bacterial colony counters present in the laboratory.
Most of the colonies that form will occur within the agar overlay (i.e., below the surface); however,
you will also see some colonies growing on the surface of the agar.
1. Place the plate upside down on the colony counter. Turn on the power switch; this will
illuminate the plate from below.
2. Count the colonies while viewing through plate through the magnifying glass mounted
above it.
3. Mark the petri plate over each colony with a marking pen to indicate which colonies have
been counted. Use the Hand tally provided to record the number of colonies counted.
4. When you are done, record your results in Table 1, and properly discard the plates.
4. Calculating the original cell concentration
The concentration of bacteria in the original samples is calculated by multiplying the colony
counts by the total dilution factor. Only plates that contain between 30 and 300 colonies should be
used to calculate the original concentration. Why? Counts less than 30 are not considered to be
statistically reliable, and if the number exceeds 300 colonies, it is likely that colony formation by
some bacteria will be suppressed, leading to an underestimation of the actual bacterial numbers.
Sometimes two plates may have colony counts between 30 - 300, and if so, the concentrations
calculated for each plate should be averaged.
Food and Water 5
II. Using the Most Probable Number Technique to Count Coliform in Water
In water, only a few coliform per liter can represent a potential health hazard. In this situation,
the concentration of cells is too low to count with a standard plate count (no cells may be present
within any particular 1 ml sample). The most probable number (MPN) technique is one solution
to this problem.
Theory of Most Probable Number counts
The MPN technique is a statistical method of estimating the concentration of bacteria. Imagine
a hypothetical situation where a sample of water contains 10 bacteria evenly dispersed in 100 ml
of water. If different sample volumes were analyzed, each 10 ml sample withdrawn would contain
1 cell, but only 1 out of 10 1 ml samples would contain a cell. If the samples were added to culture
media, the samples containing a cell would show growth. Conversely, knowing the volume of the
samples and the number of tubes showing growth, we could know the concentration of bacteria in
an unknown original sample.
For example, suppose a water sample were analyzed with the following results:
Volume
Number of
Number of cultures
cultured
samples cultured
showing bacterial growth
10
10
10
1.0
20
2
0.1
20
0
Since the smallest sample size that yielded growth was 1.0 ml, and growth occurred in 2 out of the
20 samples tested, the number of bacteria in the original could be calculated as 2 cells per 20 ml (i.e.,
5 cells per /100ml).
In the real world bacteria are randomly but not evenly distributed in a water sample. Thus, in
our example above, if twenty 1 ml samples were cultured, growth might occur in fewer than 20
tubes. And, some tubes in which growth occurs might actually start with 2 or more cells. If growth
occurs, all we know is that at least one viable bacterial cell was present. What a bummer!
However, because the cells are randomly distributed, statistical techniques can be used to
estimate the number of bacteria in the original sample, i.e., the "most probable number!" A
statistical estimate of the number of bacteria can be obtained from an appropriate statistical table,
such as the one shown on next page.
Table I. MPN Index when five 10 ml samples, one 1 ml sample, and one 0.1 ml sample are
Food and Water 6
tested.
Number of tubes giving
Positive Reaction (acid + gas)*
10 ml
1 ml
0.1 ml
95% confidence limits
MPN cells
per 100 ml
Lower
Upper
0
0
0
<2
0
5.9
0
1
0
2
0.050
13
1
0
0
2.2
0.050
13
1
1
0
4.4
0.52
14
2
0
0
5.0
0.54
19
2
1
0
7.6
1.5
19
3
0
0
8.8
1.6
29
3
1
0
12
3.1
30
4
0
0
15
3.3
46
4
0
1
20
5.9
48
4
1
0
21
6.0
53
5
0
0
38
6.4
330
5
0
1
96
12
370
5
1
0
240
12
3700
5
1
1
88
*This table only includes those combinations of positive tubes that occur with a significant
frequency. If the other nine possible combinations occur with a greater frequency than 1%, than
faulty technique should be suspected.
Counting coliform bacteria involves three steps
The MPN technique is used to estimate coliform bacteria (not total bacteria), and therefore
selective and differential media must be used. There are 3 stages to this method (see Figure 2):
1. The Presumptive test. The samples are first inoculated into lactose broth containing a Durham
tube. In a positive culture, growth will occur with the production of both acid and gas. Because few
other types of bacteria can ferment lactose and yield acid and gas, a positive presumptive test is a
very good indication of the presence of coliform.
2. The Confirmed test. Some non-coliform bacteria can yield a false positive in the presumptive
test. Thus, all positive lactose broth cultures are subjected to the confirmed test: a loopful of each
culture is streaked onto Eosine Methylene Blue agar (EMB) and inoculated into Brilliant Green
Lactose Bile Broth (BGLBB). EMB is a differential medium; eosine and methylene blue combine
to yield a distinctive coloration pattern for coliform bacteria. The presence of bile and brilliant
green in BGLBB make this medium selective for Gram-negative bacteria. A confirmed test is
positive when colonies with a green metallic sheen form on EMB and gas occurs in BGLBB.
3. The Completed test. Cells from an isolated colony on EMB are transferred to an agar slant and
again transferred to lactose broth. If acid and gas are again observed, and Gram-negative rods are
found in the slant culture, then the identification of coliform is considered positive.
Food and Water 7
Figure 2. Steps of the MPN procedure
Typical Appearance of
Selected Bacteria on EMB
Agar
E. coli &
Klebsiella pneumoniae:
colonies with a dark center
and green metallic sheen
Enterobacter aerogenes:
pink colonies with no sheen
Proteus mirabilis &
Salmonella typhimurium:
colorless colonies
Composition of media used in MPN (g/L)
Lactose Broth
Brilliant Green Bile Broth
Peptone . . . . . . 10
Beef extct . . . . . 1
Lactose . . . . . . . 5
NaCl. . . . . . . . . . 5
Phenol red . . . . . 0.018
Peptone . . . . . . 10 g
Lactose . . . . . . . 10
Oxgall (bile) . . . . 20
Brilliant green . . . 0.0133
Plate Count Agar
Eosine methylene Blue Agar
Casein Digest . . . . 5
Yeast Extract . . . . 2.5
Glucose . . . . . . . . 1
Agar . . . . . . . . . 15
Peptone . . . . . . .
Lactose . . . . . . . .
K Phosphate . . . .
Eosine-Y. . . . . . .
Methylene Blue .
Agar . . . . . . . . .
10
10
2.0
0.4
0.065
15
Food and Water 8
Supplies:
5 Tubes of double-strength lactose broth (green cap)
3 Tubes of single strength lactose broth (Red cap; one of these is for Completed Test)
1 sterile 16mm test tube (Black cap; for collecting water sample)
1 nutrient agar slant, yellow cap
1 ten ml pipet,
2 one ml pipets
1 plate of EMB agar
media bottle containing 99 ml of sterile water
1 tube of Brilliant Green Bile Lactose Broth (Blue cap)
Procedures
1. Presumptive Test
We will be continuing the yearly microbiological study of "Goose Run, the oasis of water
purity that runs through campus. Collect water from the creek behind Marietta Hall.
1. Collect a water sample into the sterile 16mm tube. Return the sample to the lab.
2. Transfer 1 ml of the sample to the bottle containing 99 ml of sterile H2O, and thoroughly
mix the sample. (Previous studies have found such high concentrations of coliform that
we need to dilute the sample first.)
3. Using a 10 ml pipet, aseptically inoculate each tube of double strength lactose broth with
10 ml of the water sample.
4. Using a 1 ml pipet, inoculate one tube containing single strength lactose broth with a 1 ml
volume of the water sample. Using another 1 ml pipet, inoculate a second tube of single
strength lactose broth with a 0.1 ml volume of the diluted water sample.
5. Incubate the tubes at 37EC for 48 hours.
6. A positive presumptive test is indicated by the formation of both acid and gas. Lactose
broth is red initially and turns yellow under acidic conditions. Record your results in
Table 2, and determine the MPN of coliform in the water sample from the MPN table.
Multiply this value by 100, the dilution of the original water sample.
2. Confirmed Test
Normally confirmed and completed tests would be run for all positive presumptive tests.
You will run a confirmed test for only one tube showing a positive presumptive test.
1. From a tube containing presumptive coliform, inoculate a tube of Brilliant Green Lactose
Bile Broth (BGLBB) and streak a plate containing Eosin Methylene Blue (EMB) Agar.
Both media are selective for Gram-negative bacteria.
2. Incubate at 37EC for 48 hours.
3. A positive confirmed test will show the presence of dark centered colonies with a metallic
green sheen. Other non-coliform colonies have different appearances (see table on next
page). Gas formation in BGLBB is also positive. Both tests must be positive for a
positive confirmed test.
4. Record your results in Table 2.
Food and Water 9
3. Completed Test
1. From an isolated colony with a typical coliform appearance from your EMB PLATE,
reinoculate a tube of lactose broth and a TSA slant.
2. Incubate both tubes at 37EC for 24 - 48 hours.
3. A positive completed test is indicated by the formation of acid and gas in the lactose broth,
and Gram-negative rods on the nutrient agar.
4. Record your results in Table 2.
Food and Water 10
Food and Water 11
NAME: _________________________
In a fashion similar to Figure 1, diagram (neatly) the dilution series you used for your Standard
Plate Count, and identify the dilution factor for each tube and the total dilution factor for each
plate.
Food and Water 12
Do the practice problems on pages 14 & 15 first.
wwIndicate where your results do not agree with your expectations, and write a brief explanation
of the discrepancyww
NOTE: The subsurface colonies are very small; they look like little dots in the medium; count
these and any surface colonies.
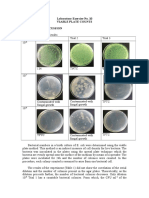
Table 1. Standard plate count of milk sample
Plate1
Plate 2
Plate 3
Plate 4
Dilution Factor
Colony Count
Cell concentration*
(viable cells / ml)
Note: express values using appropriate scientific notation (e.g., 2.3 x 105)
Which plates had a countable (between 30 - 300) number of colonies?
What is the concentration of bacteria in the original milk sample? (average plates if appropriate)
If you did not get 30 - 300 colonies on any of your plates, what dilutions do you think might
have yielded an appropriate number of colonies?
Food and Water 13
Results of MPN tests
Table 2. Presumptive Test
Tube 1
Tubes receiving:
ACID
GAS
Tube 2
ACID
GAS
Tube 3
ACID
GAS
Tube 4
ACID
GAS
Tube 5
ACID
GAS
10 ml samples
1.0 ml sample
O.1 ml sample
Number of positive tubes (acid + gas)
10 ml samples
________
1.0 ml sample
________
0.1 ml sample
________
MPN (from Table I) = _______ x 100 (dilution factor) = _______ cells/100 ml
RESULTS OF CONFIRMED TEST
RESULTS OF COMPLETED TEST
Did any colonies on EMB
have a green metallic sheen?
yes / no
Were acid and gas produced in
the lactose broth?
yes / no
Was gas produced in BGLBB?
Appearance of cell on the agar slant:
cell shape: __________
Gram-reaction: __________
yes / no
Was your confirmed test positive?
yes / no
Was your completed test positive?
yes / no
Also turn in an answer to these questions:
1. What are the challenges to identifying pathogens in food and water samples? What are the
characteristics of coliform bacteria that make them good indicator bacteria?
2. Suppose that you wanted to determine the number of fecal coliform in food sample by
adapting the media used for identifying coliform to the SPC technique. Thinking out of the
box, explain how you could do this?
Food and Water 14
Dilution Practice problems turn in with lab results
Note: numbers $1000 and # 0.01 should be expressed in scientific notation
1. What is the equation used to calculate
... an individual dilution factor:
... a total dilution factor:
2. When ___ of a sample is combined
with ____ of media the
dilution factor is ____
1 ml
9 ml
______
4 ml
4 ml
______
5 ml
15 ml
______
2 ml
8 ml
______
If the above dilutions were performed and 0.2 ml of the final tube were plated, the total
dilution factor would be _______________.
3. Based upon the dilution series shown here:
A. The total dilution factors are: (show calculation)
Plate A: ______________
Plate B: ______________
B. Which plate has a countable
number of colonies? ____
C. What would have been the original
concentration of bacteria?
_________________ (Show calculation)
Food and Water 15
4. Based upon the dilution series shown here:
A. Write in the dilution factor for each tube.
B. What is the total dilution factor for each
plate? (show calculations)
Plate A: ______________
Plate B: ______________
C. What is the expected number of colonies on each plate?
(show calculations) .
Plate A: ______________
Plate B: ______________
Which plate has a countable number of colonies? ______
5. Based upon the dilution series
shown here:
A. Write in the dilution factor for each
tube.
B. What is the total dilution factor for
the plate? (show calculation)
C. What would be the bacteria concentration in the original tube? _________________
(show calculation)
Food and Water 16
You might also like
- Lab Report. Serial DilutionDocument5 pagesLab Report. Serial Dilutionanon_747148947100% (2)
- Report BacteriaDocument11 pagesReport BacteriaSuzeanni JalilNo ratings yet
- SPREad PlateDocument7 pagesSPREad PlateJermeLou BaoNo ratings yet
- 9-Food and WaterTHE MICROBIOLOGICAL EXAMINATION OF FOODS & WATERDocument16 pages9-Food and WaterTHE MICROBIOLOGICAL EXAMINATION OF FOODS & WATERArri KurniawanNo ratings yet
- EnumerationDocument6 pagesEnumerationJITHIN VNo ratings yet
- Microbiology Practical 3Document7 pagesMicrobiology Practical 3Kah Jun100% (1)
- Practical 3 Lab Report MicrobiologyDocument14 pagesPractical 3 Lab Report MicrobiologynurulakhmalNo ratings yet
- Chapter 5 - Basic Techniques in Food MicrobiologyDocument54 pagesChapter 5 - Basic Techniques in Food MicrobiologyNurhayati Hasanah100% (1)
- Direct MeasurementDocument33 pagesDirect MeasurementAira VillarinNo ratings yet
- Methods For Microbiological Examination of of FoodDocument5 pagesMethods For Microbiological Examination of of Food9qdd95pw9kNo ratings yet
- Counting BacteriaDocument30 pagesCounting Bacteriayuppie_raj2175100% (1)
- Experiment 3 - Air, Water and Soil PollutionDocument9 pagesExperiment 3 - Air, Water and Soil PollutionTama KalongaNo ratings yet
- Enumeration of Microorganisms: Microbiology BIOL 275Document10 pagesEnumeration of Microorganisms: Microbiology BIOL 275Diya GhosalNo ratings yet
- C5 Mic 254Document64 pagesC5 Mic 254Aqilah NajwaNo ratings yet
- Exp4 Total Count BacteriaDocument6 pagesExp4 Total Count Bacterialzero93100% (2)
- Full Report Bacterial CountDocument10 pagesFull Report Bacterial CountSuzeanni Jalil100% (1)
- Bacterial TitreDocument3 pagesBacterial TitreManjunatha Bukkambudi KrishnaswamyNo ratings yet
- Enumeration of Microorganisms: Microbiology BIOL 275Document10 pagesEnumeration of Microorganisms: Microbiology BIOL 275yudhieNo ratings yet
- EnumerationDocument7 pagesEnumerationSikin Sikin100% (1)
- Introduction To Lab Ex. 19: Enumeration of Bacteria - Pour Plate TechniqueDocument7 pagesIntroduction To Lab Ex. 19: Enumeration of Bacteria - Pour Plate TechniqueRenan RodriguesNo ratings yet
- Practical 12Document7 pagesPractical 125408 ManshaNo ratings yet
- Experiment 5Document5 pagesExperiment 5Nipuni Saubhagya RathnayakeNo ratings yet
- Exercise 17 Microbial Analysis of WaterDocument6 pagesExercise 17 Microbial Analysis of WaterNecka AmoloNo ratings yet
- Microbial Load: Pasteurized vs. Raw Milk Kathy Ziuzianska Biology 240, Section 301 October 19 2011 Robert Bellas, PHDDocument7 pagesMicrobial Load: Pasteurized vs. Raw Milk Kathy Ziuzianska Biology 240, Section 301 October 19 2011 Robert Bellas, PHDkasiapitrasiaNo ratings yet
- Measurement of GrowthDocument6 pagesMeasurement of GrowthPrerna NegiNo ratings yet
- Food Microbiology FullDocument6 pagesFood Microbiology FullhasifNo ratings yet
- Direct Measurements of Microbial Growth: Viable Counts: Experiment 4Document3 pagesDirect Measurements of Microbial Growth: Viable Counts: Experiment 4gsharkzNo ratings yet
- Food Micro Lab 1Document6 pagesFood Micro Lab 1Elcanah Joy BernardoNo ratings yet
- Laboratory Exercise No. 10 Viable Plate Counts Results and DiscussionDocument3 pagesLaboratory Exercise No. 10 Viable Plate Counts Results and Discussionvanessa olga100% (1)
- Bacterial CountsDocument5 pagesBacterial CountsAndrew LeeNo ratings yet
- Bacteriological Water AnalysisDocument6 pagesBacteriological Water Analysisramkrishna01No ratings yet
- UntitledDocument30 pagesUntitledLEE JIA XINNo ratings yet
- Microbial Analysis of FoodDocument77 pagesMicrobial Analysis of FoodAbdurrazaq YakubuNo ratings yet
- Lab 17 Serial DilutionDocument15 pagesLab 17 Serial DilutionCedricDiehlNo ratings yet
- Virology - Isolation & Cultivation of BacgteriophageDocument12 pagesVirology - Isolation & Cultivation of BacgteriophageProbioticsAnywhereNo ratings yet
- Total Bacterial CounrDocument10 pagesTotal Bacterial CounrIrish OrleansNo ratings yet
- Serial Dilution ProtocolsDocument5 pagesSerial Dilution ProtocolsYugendra Babu K100% (1)
- Food Microbiology LR 3 & 4Document17 pagesFood Microbiology LR 3 & 4rishabh bhardwajNo ratings yet
- Standard Plate Count Method: By: Hafiza Asfa Shafique Microbiology BS Biotechnology VDocument13 pagesStandard Plate Count Method: By: Hafiza Asfa Shafique Microbiology BS Biotechnology VJawadNo ratings yet
- Isolation of Microorganism and EnumerationDocument13 pagesIsolation of Microorganism and EnumerationkrbiotechNo ratings yet
- Bacteria EnumerationDocument27 pagesBacteria EnumerationDaisy Arora KhuranaNo ratings yet
- Bacteriological Water AnalysisDocument8 pagesBacteriological Water AnalysisCasio Je-urNo ratings yet
- Formal Report Quantitative Analysis of Microbial Populations Through Standard Viable Plate Count Methods MicrobiologyDocument4 pagesFormal Report Quantitative Analysis of Microbial Populations Through Standard Viable Plate Count Methods MicrobiologyGino100% (1)
- Food Microbiology (CFD 20203) Unikl Lab Manual Micet: Malaysian Institute of Chemical and Bioengineering TechnologyDocument11 pagesFood Microbiology (CFD 20203) Unikl Lab Manual Micet: Malaysian Institute of Chemical and Bioengineering TechnologyLiyana HalimNo ratings yet
- Laboratory Seven: Enumeration of BacteriaDocument6 pagesLaboratory Seven: Enumeration of BacteriaSulav Acharya0% (1)
- Serial Dilutions and PlatingDocument17 pagesSerial Dilutions and PlatingVon Valentine MhuteNo ratings yet
- Food Lab 1Document13 pagesFood Lab 1Nurul Syaheerah100% (1)
- Effects of Nutrition and Temperature On The Population Growth of Bacteria 2011Document9 pagesEffects of Nutrition and Temperature On The Population Growth of Bacteria 2011si_miaomiaoNo ratings yet
- CPB 30103 Biochemical Engineering UniKL MICET Experiment 4: Determination of Bacterial Loads Viable Cell Counts Full Lab ReportDocument12 pagesCPB 30103 Biochemical Engineering UniKL MICET Experiment 4: Determination of Bacterial Loads Viable Cell Counts Full Lab ReportSiti Hajar Mohamed100% (2)
- Lab # 7 - Serial DilutionDocument2 pagesLab # 7 - Serial DilutionKESHAWN TYRONE SUTHERLANDNo ratings yet
- Laboratory 4-6Document3 pagesLaboratory 4-6JOSENo ratings yet
- Count of Colony Forming UnitDocument2 pagesCount of Colony Forming UnitTejas DoshiNo ratings yet
- Laboratory 8 Enumeration: E. Coli Using Three Different Methods: Optical Density, Direct Counts and Plate Counts. YouDocument6 pagesLaboratory 8 Enumeration: E. Coli Using Three Different Methods: Optical Density, Direct Counts and Plate Counts. YouskgohNo ratings yet
- Growth CurveDocument4 pagesGrowth Curvedinchus_106No ratings yet
- Practical - Turbidimetric MethodsDocument9 pagesPractical - Turbidimetric MethodsProbioticsAnywhere100% (1)
- Week 8 Aerobic Plate CountDocument7 pagesWeek 8 Aerobic Plate CountSam 0505No ratings yet
- Outlines of dairy bacteriology A concise manual for the use of students in dairyingFrom EverandOutlines of dairy bacteriology A concise manual for the use of students in dairyingNo ratings yet
- Front Mission 3 - 1Document31 pagesFront Mission 3 - 1Arief Happy RachmadhiNo ratings yet
- Front Mission 3 - 4Document110 pagesFront Mission 3 - 4Arief Happy Rachmadhi0% (1)
- Graywater Guide For Your LandscapeDocument12 pagesGraywater Guide For Your LandscapeU8x58No ratings yet
- FAO Fisheries & Aquaculture - Species Fact Sheets - Penaeus Merguiensis (De Man, 1888)Document4 pagesFAO Fisheries & Aquaculture - Species Fact Sheets - Penaeus Merguiensis (De Man, 1888)Arief Happy RachmadhiNo ratings yet