Professional Documents
Culture Documents
Mole Concept Solution Practice Set Objective by S.K.sinha See Chemistry Animations at
Mole Concept Solution Practice Set Objective by S.K.sinha See Chemistry Animations at
Uploaded by
myiitchemistryOriginal Description:
Original Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Mole Concept Solution Practice Set Objective by S.K.sinha See Chemistry Animations at
Mole Concept Solution Practice Set Objective by S.K.sinha See Chemistry Animations at
Uploaded by
myiitchemistryCopyright:
Available Formats
SINHA IIT CHEMISTRY
mail to sinha@kotatimes.com
sinhaiitchemistry.com
Mole Concept (Solution-2)
1. 342 gm of 20 % by mas of Ba(OH)2 solution (sp.gr. 0.57) is reacted with 1200 mL of 2
M HNO3. If the final density is same as pure water then molarity of the ion in resulting
solution by nature of the above solution is identified, is:
(a) 0.25 (b) 0.5 M (c) 0.888 M (d) None
2. What volume of HCl solution of density 1.2 g/cm3 and containing 36.5 % by weight HCl,
must be allowed to react with zinc (Zn) in order to liberate 4.0 g of hydrogen.
(a) 333.33 mL (b) 500 mL (c) 614.66 mL (d) None
3. A bottle of an aqueous H2O2 solution is labelled as ‘28 V’ H2O2 and the density of the
solution in g/mL is 1.25. Choose the correct option.
(a) Molality of H2O2 solution is 2 (b) Molarity of H2O2 solution is 5
(c) Molality of H2O2 solution is 2.15 (d) None of these
4. The impure 6 gm of NaCl is dissolved in water and then treated with excess of silver
nitrate solution. The weight of precipitate of silver chloride is found to be 14 g. The %
purity of NaCl solution would be:
(a) 95 % (b) 85 % (c) 75% (d) 65 %
5. 10 L of hard water required 5.6 g of lime for removing hardness. Hence temporary
hardness in ppm of CaCO3.
(a) 1000 (b) 2000 (c) 100 (d) 1
6. A sample of peanut oil weighing 2 g is added to 25 ml of 0.40 M KOH. After saponifica
tion is complete, 8.5 ml of 0.28 M H2SO4 is needed to neutralize excess of KOH. The
saponification number of peanut oil is:
(a) 146.75 (b) 223.44 (c) 98.9 (d) None
7. Al2(SO4)3 solution of 1 molal concentration is present in 1 litre solution of 2.684 g/cc.
How many moles of BaSO4 would be precipitated on adding BaCl2 in excess?
(a) 2 moles (b) 3 moles (c) 6 moles (d) 12 moles
8. A certain public water supply contains 0.10 ppb (part per billion ) of chloroform (CHCl3).
How many molecules of CHCl3 would be obtained in 0.478 mL drop of this water?
(a) 4 × 10-3 × NA (b) 10-3 × NA (c) 4 × 10-10 × NA (d) None
9. One litre of a sample of hard water contain 4.44 mg CaCl2 and 1.9 mg of MgCl2. What
is the total hardness in terms of ppm of CaCO3?
(a) 2 ppm (b) 3 ppm (c) 4 ppm (d) 6 ppm
10. 0.1 mole H3POx is completely neutralized by 5.6 g KOH then the true statement is:
(a) x = 3 and give acid is dibasic
(b) x = 4 and given acid has no P-H linkage
(c) x = 2 and given acid does not form acid salt
(d) all of these
Mole Concept (Solution-2) Answers
1. (c) 2. (a) 3. (c) 4. (a) 5. (a) 6. (a) 7.(c) 8. (a) 9. (d) 10.(c)
1-D-10, Talwandi, Commerce College Road , Kota Mo. 9314905055 , Ph. 0744-2422383
You might also like
- Bridgewater Associates Presentation (Updated)Document17 pagesBridgewater Associates Presentation (Updated)GHaj100% (3)
- 6th Central Pay Commission Salary CalculatorDocument15 pages6th Central Pay Commission Salary Calculatorrakhonde100% (436)
- Marginal Costing in Managerial Decision MakingDocument16 pagesMarginal Costing in Managerial Decision Makingyashashree_taori4443100% (4)
- English Translation Maktubat Imam Rabbani by Sufi Irshad AlamDocument59 pagesEnglish Translation Maktubat Imam Rabbani by Sufi Irshad AlamAA10No ratings yet
- Concentration Terms and Eudiometry: (Physical Chemistry) Exercise (O-I) Introduction of Concentration Terms 1Document59 pagesConcentration Terms and Eudiometry: (Physical Chemistry) Exercise (O-I) Introduction of Concentration Terms 1Jayarj singh100% (1)
- Assignment (Stoichiometry) - 1 PDFDocument4 pagesAssignment (Stoichiometry) - 1 PDFJatin SinglaNo ratings yet
- GRiGNARD REAGENT!!Document22 pagesGRiGNARD REAGENT!!GazalNo ratings yet
- Hydrocarbons DPPsDocument37 pagesHydrocarbons DPPsAsif HodaNo ratings yet
- Quantitative and QualitativeDocument15 pagesQuantitative and QualitativesquadralsupremeNo ratings yet
- DPP 04 Ionic Equilibrium JH Sir-4295Document4 pagesDPP 04 Ionic Equilibrium JH Sir-4295T sidharth100% (1)
- Prince Singh: Physical & Inorganic ChemistryDocument5 pagesPrince Singh: Physical & Inorganic ChemistryJatin Singla100% (1)
- Liquid SolutionsDocument9 pagesLiquid SolutionsrockNo ratings yet
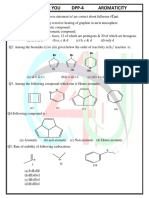
- Aromaticity DPP 4Document4 pagesAromaticity DPP 4SubhadeepNo ratings yet
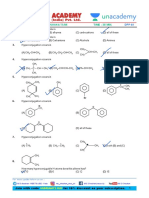
- Electrophilic Aromatic Substitution DPPDocument35 pagesElectrophilic Aromatic Substitution DPPAsif Hoda0% (1)
- Jitendra Hirwani: Daily Practice Problem OF Physical Chemistry For NeetDocument8 pagesJitendra Hirwani: Daily Practice Problem OF Physical Chemistry For NeetabhishekNo ratings yet
- Complete Course in Organic Chemistry by Vineet Khatri Sir: Class: Xi Time: 35 Min. DPP. NO.17Document4 pagesComplete Course in Organic Chemistry by Vineet Khatri Sir: Class: Xi Time: 35 Min. DPP. NO.17Arnab KumarNo ratings yet
- Jitendra Hirwani: Daily Practice Problem OF Physical Chemistry For NeetDocument5 pagesJitendra Hirwani: Daily Practice Problem OF Physical Chemistry For NeetabhishekNo ratings yet
- CLS Aipmt-19-20 XIII Che Study-Package-3 Level-1 Chapter-15 PDFDocument40 pagesCLS Aipmt-19-20 XIII Che Study-Package-3 Level-1 Chapter-15 PDFThavasimariselvam N100% (1)
- P-Block (Group 13 To 14) NM Solution (-1) Chem PDFDocument31 pagesP-Block (Group 13 To 14) NM Solution (-1) Chem PDFChauhan RonakNo ratings yet
- PRACTICE SHEET - 03 (Chemistry) : CH - CH - CH CN CN CNDocument3 pagesPRACTICE SHEET - 03 (Chemistry) : CH - CH - CH CN CN CNABD 17No ratings yet
- Prince Singh: Physical & Inorganic ChemistryDocument5 pagesPrince Singh: Physical & Inorganic ChemistryJatin SinglaNo ratings yet
- SN DPPDocument34 pagesSN DPPtyagiabhishek145No ratings yet
- C - 2Y - Dilute Solution and Colligative Properties - Assignment 1Document5 pagesC - 2Y - Dilute Solution and Colligative Properties - Assignment 1Phani PadmasriNo ratings yet
- DPP Quantum Number JH Sir-3574Document5 pagesDPP Quantum Number JH Sir-3574shiva royNo ratings yet
- Goc & Eas Test-IiDocument7 pagesGoc & Eas Test-IiAniket GuptaNo ratings yet
- Prince Singh Atomic Structure Chemical Kinetics: Physical & Inorganic ChemistryDocument6 pagesPrince Singh Atomic Structure Chemical Kinetics: Physical & Inorganic ChemistryJatin SinglaNo ratings yet
- Bakliwal Tutorials: Topic: Inductive Effect Part - A: SubjectiveDocument3 pagesBakliwal Tutorials: Topic: Inductive Effect Part - A: SubjectiveRitesh SonawaneNo ratings yet
- D and F Block DPPDocument4 pagesD and F Block DPPKalyan ReddtNo ratings yet
- Aldol Reaction - ChemistryDocument7 pagesAldol Reaction - ChemistryGamer HelperNo ratings yet
- DPP 01 Gaseous State JH Sir-3583Document11 pagesDPP 01 Gaseous State JH Sir-3583Shivam Kumar75% (4)
- Amines PDFDocument34 pagesAmines PDFRam KhannaNo ratings yet
- Chem Academy: Chemical BondingDocument4 pagesChem Academy: Chemical BondingEmraan EmmiNo ratings yet
- Reaction Mechanism Jeemain - GuruDocument12 pagesReaction Mechanism Jeemain - Guruprexa indiaNo ratings yet
- CLS Aipmt 19 20 XII Che Study Package 4 Level 1 Chapter 10Document36 pagesCLS Aipmt 19 20 XII Che Study Package 4 Level 1 Chapter 10Utkarsh KumarNo ratings yet
- Ms Chouhan & Team: Organic ChemistryDocument16 pagesMs Chouhan & Team: Organic ChemistryPrithviraj GhoshNo ratings yet
- Carbonyl Compound WorksheetDocument25 pagesCarbonyl Compound WorksheetOmendra SinghNo ratings yet
- Ummeed: Isomerism NEET Organic Chemistry Crash CourseDocument7 pagesUmmeed: Isomerism NEET Organic Chemistry Crash CourseAman PandeyNo ratings yet
- Index: Hydrocarbons (Alkanes, Alkenes & Alkynes)Document31 pagesIndex: Hydrocarbons (Alkanes, Alkenes & Alkynes)Harsh VardhanNo ratings yet
- Redox Reactions NEET PYQ SOLUTIONDocument6 pagesRedox Reactions NEET PYQ SOLUTIONsomeone nooneNo ratings yet
- Solutions - Practice Sheet - Sarthak KCET PDFDocument6 pagesSolutions - Practice Sheet - Sarthak KCET PDFAkanksh KNo ratings yet
- Halogen Derivatives PDFDocument32 pagesHalogen Derivatives PDFRaju Singh100% (1)
- Practice TestDocument14 pagesPractice TestHimanshu JindalNo ratings yet
- Alkene DPPDocument20 pagesAlkene DPPKalyan ReddtNo ratings yet
- Chemistry Advanced Level Problem Solving (ALPS-6) - PaperDocument14 pagesChemistry Advanced Level Problem Solving (ALPS-6) - PaperAnanmay ChauhanNo ratings yet
- Etoos 9 PDFDocument24 pagesEtoos 9 PDFB. P. A Music INDIA100% (1)
- ADV. I 57 - 64 (Exercise 3)Document8 pagesADV. I 57 - 64 (Exercise 3)Aditya ShahNo ratings yet
- Stereoisomerism VKP SirDocument49 pagesStereoisomerism VKP SirSandeep ReddyNo ratings yet
- Physics DPPDocument6 pagesPhysics DPPHarshika KatariyaNo ratings yet
- DPP - 01 - Reaction MechanismDocument5 pagesDPP - 01 - Reaction Mechanismbaibhav singhNo ratings yet
- 5 DPP - 56to81 - FinalDocument44 pages5 DPP - 56to81 - FinalArnab KumarNo ratings yet
- 13 DPP 03g Goc Excel Acid+BaseDocument5 pages13 DPP 03g Goc Excel Acid+BasekljNo ratings yet
- Organic Chemistry - GRDocument52 pagesOrganic Chemistry - GRPRIYANSHU KUMARNo ratings yet
- Reduction, Oxidation - Hydrolysis APSP PDFDocument24 pagesReduction, Oxidation - Hydrolysis APSP PDFGOURISH AGRAWALNo ratings yet
- Organic Chemistry: Daily Practice ProblemsDocument35 pagesOrganic Chemistry: Daily Practice ProblemsRaju SinghNo ratings yet
- DPP Module-1 01JA To 06JA OrganicDocument21 pagesDPP Module-1 01JA To 06JA OrganicAkkaldevi Saivinayak CRNo ratings yet
- DPP 2Document2 pagesDPP 2ashutosh99878No ratings yet
- Alcohol, Ether & Phenol - QuestionDocument3 pagesAlcohol, Ether & Phenol - Questionbest badmintonNo ratings yet
- Hydrocarbon DPPDocument24 pagesHydrocarbon DPPDhruv Jyot SinghNo ratings yet
- Chem Academy: ThermodynamicsDocument5 pagesChem Academy: ThermodynamicsHamit RanaNo ratings yet
- Iupac 1Document37 pagesIupac 1shodhan shettyNo ratings yet
- DPP 1 Optical Isomerism VKP Sir-3706Document3 pagesDPP 1 Optical Isomerism VKP Sir-3706Sanjay Mani TripathiNo ratings yet
- DPP GoccccDocument10 pagesDPP GoccccMayur Khichi0% (1)
- Mole Concept Solution Practice Set ObjectiveDocument3 pagesMole Concept Solution Practice Set ObjectiveGagandeep SinghNo ratings yet
- JEE - Chemistry - Mole ConceptDocument26 pagesJEE - Chemistry - Mole Conceptofficial.archit234No ratings yet
- Solid State / Crystalline State ChemistryDocument26 pagesSolid State / Crystalline State ChemistrymyiitchemistryNo ratings yet
- Geometrical Isomerism (Animated)Document114 pagesGeometrical Isomerism (Animated)myiitchemistry60% (5)
- Unique Approach of Problem Solving in Isomerism by S.K.sinha HTTP://WWW - Openchemistry.inDocument6 pagesUnique Approach of Problem Solving in Isomerism by S.K.sinha HTTP://WWW - Openchemistry.inmyiitchemistry100% (3)
- Set of 50 Obj in General Organic Chemistry by S.K.sinha HTTP://WWW - Openchemistry.inDocument6 pagesSet of 50 Obj in General Organic Chemistry by S.K.sinha HTTP://WWW - Openchemistry.inmyiitchemistry50% (4)
- Mole Concept Solution Practice Set 1 Objective by S.K.sinha See Chemistry Animations atDocument2 pagesMole Concept Solution Practice Set 1 Objective by S.K.sinha See Chemistry Animations atmyiitchemistry100% (4)
- Basic Inorganic Nomenclature FOR IIT-JEE ENTRANCE TEST by S.K.sinha See Chemistry Animations atDocument5 pagesBasic Inorganic Nomenclature FOR IIT-JEE ENTRANCE TEST by S.K.sinha See Chemistry Animations atmyiitchemistry88% (17)
- IONIC EQUILLIBRIUM FOR IIT-JEE ENTRANCE TEST by S.K.sinha See Chemistry Animations at Http://openchemistry - inDocument31 pagesIONIC EQUILLIBRIUM FOR IIT-JEE ENTRANCE TEST by S.K.sinha See Chemistry Animations at Http://openchemistry - inmyiitchemistry100% (7)
- How To Find No of Structural Isomers by S.K.sinha See Chemistry Animations at HTTP://WWW - Openchemistry.inDocument2 pagesHow To Find No of Structural Isomers by S.K.sinha See Chemistry Animations at HTTP://WWW - Openchemistry.inmyiitchemistry81% (16)
- How To Identify Geometrical Isomers by S.K.sinha See Chemistry Animations atDocument4 pagesHow To Identify Geometrical Isomers by S.K.sinha See Chemistry Animations atmyiitchemistry100% (4)
- Mole & Stoichiometry by S.K.sinha See Chemistry Animations atDocument3 pagesMole & Stoichiometry by S.K.sinha See Chemistry Animations atmyiitchemistry50% (2)
- UNSTABLE FUNCTIONAL GROUPS IN ORGANIC by S.K.sinha See Chemistry Animations atDocument1 pageUNSTABLE FUNCTIONAL GROUPS IN ORGANIC by S.K.sinha See Chemistry Animations atmyiitchemistry100% (2)
- Organic Nomenclature HTTPDocument19 pagesOrganic Nomenclature HTTPmyiitchemistry91% (11)
- PRACTICAL ORGANIC CHEMISTRY FOR IIT-JEE ENTRANCE TEST by S.K.sinha See Chemistry Animations atDocument2 pagesPRACTICAL ORGANIC CHEMISTRY FOR IIT-JEE ENTRANCE TEST by S.K.sinha See Chemistry Animations atmyiitchemistry100% (10)
- Some Important Organic Information by S.K.sinha See Chemistry Animations atDocument2 pagesSome Important Organic Information by S.K.sinha See Chemistry Animations atmyiitchemistry83% (6)
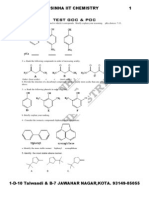
- Test1 Goc & Poc Tough by S.K.sinha See Chemistry Animations atDocument3 pagesTest1 Goc & Poc Tough by S.K.sinha See Chemistry Animations atmyiitchemistry100% (1)
- TEST 2 GOC & POC Tough by S.K.sinha See Chemistry Animations atDocument3 pagesTEST 2 GOC & POC Tough by S.K.sinha See Chemistry Animations atmyiitchemistryNo ratings yet
- TEST 2 GOC & POC Tough by S.K.sinha See Chemistry Animations atDocument3 pagesTEST 2 GOC & POC Tough by S.K.sinha See Chemistry Animations atmyiitchemistryNo ratings yet
- Biomas Terrestres y Acuatico1Document3 pagesBiomas Terrestres y Acuatico1PabloItzolNo ratings yet
- FIP Statement of Professional Standards (Code of Ethics)Document4 pagesFIP Statement of Professional Standards (Code of Ethics)Cesly Jewel Acosta AvilesNo ratings yet
- The First Global Revolution TextDocument184 pagesThe First Global Revolution TextAuren790% (10)
- Panasonic Sa-Akx38ph Sa-Akx38pnDocument70 pagesPanasonic Sa-Akx38ph Sa-Akx38pnAndrewer100% (1)
- Review Sheet For Math Midterm Exam Revised 1Document34 pagesReview Sheet For Math Midterm Exam Revised 1api-294158678No ratings yet
- AbacusDocument97 pagesAbacusأنور مازوز أبو يوسفNo ratings yet
- Conductivity UspDocument6 pagesConductivity UspgodloveyouNo ratings yet
- Expressions of Interest & Support - Resource ExpoDocument3 pagesExpressions of Interest & Support - Resource ExpoSharon A StockerNo ratings yet
- Ahmedabad Smart City - Solar RooftopDocument1 pageAhmedabad Smart City - Solar RooftopShintia AhediaNo ratings yet
- Shopper How To Use Grainger Punchout Catalog1Document6 pagesShopper How To Use Grainger Punchout Catalog1chaab2006No ratings yet
- Rudder - Steering Gear Speed RulesDNVGL-RU-SHIP-Pt4Ch10 19Document1 pageRudder - Steering Gear Speed RulesDNVGL-RU-SHIP-Pt4Ch10 19Tolias EgwNo ratings yet
- PCS-222B I Instruction Manual en Demostic General R1.00 (En ZNKZ0100.0086.0001)Document88 pagesPCS-222B I Instruction Manual en Demostic General R1.00 (En ZNKZ0100.0086.0001)Ricchie Gotama SihiteNo ratings yet
- Chapter 1 ResearchDocument6 pagesChapter 1 ResearchNosaj TriumpsNo ratings yet
- HESI Math and Science Answers Exam 2023Document4 pagesHESI Math and Science Answers Exam 2023Nelson MandelaNo ratings yet
- Study Qs 1Document2 pagesStudy Qs 1Aditya RaoNo ratings yet
- Suspension Electromagnetic Power GenerationDocument100 pagesSuspension Electromagnetic Power GenerationMohit AggarwalNo ratings yet
- BIW Welding Fixture ProcessDocument31 pagesBIW Welding Fixture ProcessAnonymous 9q5GEfm8I50% (4)
- Message of Sec. Teresita Quintos Deles On The 11th National Peace Consciousness MonthDocument3 pagesMessage of Sec. Teresita Quintos Deles On The 11th National Peace Consciousness MonthOffice of the Presidential Adviser on the Peace ProcessNo ratings yet
- Proposed Two Storey Duplex Residential BuildingDocument4 pagesProposed Two Storey Duplex Residential Buildingjen mejiaNo ratings yet
- Hawassa University College of Social Sciences: August 2022Document3 pagesHawassa University College of Social Sciences: August 2022Eyuel Nebiyu100% (1)
- Answer Answer Any Two Full Questions, Each Carries 15 Marks MarksDocument3 pagesAnswer Answer Any Two Full Questions, Each Carries 15 Marks MarksanuNo ratings yet
- Giáo Trình Writing Level 4 ENG 267: Mã môn họcDocument76 pagesGiáo Trình Writing Level 4 ENG 267: Mã môn họcThanh HiềnNo ratings yet
- (Finance) Stock Market Development and Economic Growth Evidence From Underdeveloped Nation Nepal (5044)Document41 pages(Finance) Stock Market Development and Economic Growth Evidence From Underdeveloped Nation Nepal (5044)Surendra ManandharNo ratings yet
- Rebuild Mysql ReplicateDocument5 pagesRebuild Mysql ReplicateomyeudaihiepNo ratings yet
- Problems of Education in The 21st Century, Vol. 66, 2015Document93 pagesProblems of Education in The 21st Century, Vol. 66, 2015Scientia Socialis, Ltd.No ratings yet
- Health Safety Assignment 1Document21 pagesHealth Safety Assignment 1Harshvardhan VyasNo ratings yet
- Cognitive BiasDocument11 pagesCognitive BiasJonny SvenssonNo ratings yet