Professional Documents
Culture Documents
Name Dosage Form (S) Adult Dose Adverse Events Nucleoside Reverse Transcriptase Inhibitors (Nrtis)
Name Dosage Form (S) Adult Dose Adverse Events Nucleoside Reverse Transcriptase Inhibitors (Nrtis)
Uploaded by
Andreea NanuOriginal Description:
Original Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Name Dosage Form (S) Adult Dose Adverse Events Nucleoside Reverse Transcriptase Inhibitors (Nrtis)
Name Dosage Form (S) Adult Dose Adverse Events Nucleoside Reverse Transcriptase Inhibitors (Nrtis)
Uploaded by
Andreea NanuCopyright:
Available Formats
Name
Dosage Adult Dose Form(s) Nucleoside reverse transcriptase inhibitors (NRTIs) Abacavir (Ziagen) 300-mg tablet; 600 mg PO q 20-mg/mL oral or solution 300 mg PO bi 'i anosine ((i e)) *2+-mg% 200mg% 2+0-mg% ,00-mg entericcoate ca#sule; *0-mg/mL sus#ension 1mtricitabine (1mtriva) 200-mg ca#sule; *0-mg/mL oral solution Lamivu ine (1#ivir)
Adverse Events
!"#ersensitivit" reaction (ma" inclu e $ever% ras&% nausea% vomiting% iarr&ea% malaise% s&ortness o$ breat&% coug&% #&ar"ngitis) -60 .g/ ,00 mg Peri#&eral neuro#at&"% #ancreatitis% PO q nausea% lactic aci osis 060 .g/ 2+0 mg PO q 2inimal to)icit"% &"#er#igmentation
3tavu ine (Zerit)
4eno$ovir ((irea ) Zalcitabine (!ivi )
200 mg PO q or 2,0 mg (2, mL) oral solution PO q *+0-mg% 300-mg 300 mg PO q tablet; or *0-mg/mL oral *+0 mg PO bi solution *+-mg% 20-mg% -60 .g/ ,0 mg 30-mg% ,0-mg PO bi ca#sule; *-mg/mL oral 060 .g/ 30 mg solution PO bi 300-mg tablet 300 mg PO q
2inimal to)icit"
Peri#&eral neuro#at&"% #ancreatitis% lactic aci osis% li#oatro#&"% &"#erli#i emia 5ausea% vomiting% iarr&ea% &ea ac&e% ast&enia% renal insu$$icienc" Peri#&eral neuro#at&"% #ancreatitis% lactic aci osis% stomatitis 5ausea% vomiting% &ea ac&e% ast&enia% anemia% neutro#enia
0637+-mg% 067+- 067+ mg PO ti mg tablet Zi ovu ine (8etrovir) 300-mg tablet% 300 mg PO bi *00-mg ca#sule; or *0-mg/mL oral 200 mg PO ti solution; *0-mg/mL intravenous solution Nonnucleoside reverse transcriptase inhibitors (NNRTIs) 'elavir ine (8escri#tor) *00-mg% 200-mg ,00 mg PO ti 8as&% &ea ac&e tablets 1$aviren9 (3ustiva) 600-mg tablet; 600 mg PO q 8as&% :53 (eg% somnolence% vivi 200-mg ca#sule reams% con$usion% visual &allucinations) 1travirine (;ntelence) *00 mg tablet 200 mg PO bi 8as&% nausea 5evira#ine ((iramune) 200-mg tablet; 200 mg PO 8as&% &e#atitis *0-mg/mL bi a
sus#ension Protease inhibitors (PIs) Ata9anavir (8e"ata9) *+0-mg% 200mg% 300-mg ca#sules
,00 mg PO q or 300 mg < ritonavir *00 mg PO q 'arunavir (Pre9ista) ,00-mg% 600-mg =00 mg q < tablets ritonavir *00 mg PO q b or 600 mg bi < ritonavir *00 mg PO bi >osam#renavir (Le)iva) 700-mg tablet; 700 mg bi < +0-mg/mL oral ritonavir *00 sus#ension mg PO bi or *,00 mg PO bi or *,00 mg < ritonavir *00200 mg PO q b ;n inavir (:ri)ivan) *00-mg% 200=00 mg PO q=& mg% 333-mg% =00 mg PO bi ,00-mg ca#sules < ritonavir *00200 mg PO bi Lo#inavir/ritonavir 200-mg/+0-mg ,00 mg/*00 mg (?aletra) tablet; PO bi or =0-mg/20-mg =00 mg/200 mg #er mL oral PO q b solution 5el$inavir ((irace#t) 2+0-mg% 62+-mg *2+0 mg PO tablets bi or 7+0 mg PO ti (5el$inavir cannot be booste 6) 8itonavir (5orvir) *00-mg so$t @oosting ose gelatin ca#sule $or ot&er =0-mg/mL oral #rotease solution in&ibitors/ *00,00 mg/ (re$er to ot&er #rotease in&ibitors $or s#eci$ic ose) 5onboosting ose (8itonavir use as sole #rotease in&ibitor)/ 600
;n irect &"#erbilirubinemia% #rolonge P8 interval% &"#ergl"cemia 8as&% nausea% iarr&ea% &"#erli#i emia% &"#ergl"cemia
8as&% nausea% vomiting% iarr&ea% &"#erli#i emia% &"#ergl"cemia
5e#&rolit&iasis% nausea% in irect &"#erbilirubinemia% &"#erli#i emia% &"#ergl"cemia 5ausea% vomiting% iarr&ea% ast&enia% &"#erli#i emia% &"#ergl"cemia 'iarr&ea% &"#erli#i emia% &"#ergl"cemia
5ausea% vomiting% iarr&ea% ast&enia% &"#erli#i emia% oral #arest&esias% &"#ergl"cemia
3aquinavir (;nvirase)
+00-mg tablet; 200-mg &ar gelatin ca#sule
4i#ranavir (A#tivus)
2+0-mg so$t gelatin ca#sule
mg bi c *000 mg < ritonavir *00 mg PO bi Anbooste saquinavir is not recommen e +00 mg < ritonavir 200 mg PO bi Anbooste ti#ranavir is not recommen e ,00 mg PO bi
5ausea% iarr&ea% &ea ac&e% &"#erli#i emia% &"#ergl"cemia
!e#atoto)icit"% ras&% &"#erli#i emia% &"#ergl"cemia% intracranial &emorr&age (rare cases re#orte )
Integrase inhibitor (II) 8altegravir (;sentress) ,00-mg tablet
5ausea% iarr&ea% &ea ac&e% :? elevations% m"o#at&"/r&ab om"ol"sis (rare)
Chemo ine receptor antagonist (CCR! antagonist) 2araviroc (3el9entr") *+0-mg% 300-mg 300 mg PO bi :onsti#ation% i99iness% in$ection% tablets *+0 mg PO bi ras& (:BP3A, in&ibitors C in ucers) 600 mg PO bi (:BP3A, in ucers) Fusion inhibitor (FI) 1n$uvirti e (>u9eon) D0-mg/mL D0 mg 3: bi ;nFection-site reactions (eg% #ain% #oE er $or er"t&ema% in uration% no ules) inFection Combination "ormulations 1#9icom - Abacavir (600 mg) < lamivu ine (300 mg) q 4ri9ivir - Abacavir (300 mg) < lamivu ine (*+0 mg) < 9i ovu ine (300 mg) bi 4ruva a - 4eno$ovir (300 mg) < emtricitabine (200 mg) q Atri#la - 4eno$ovir (300 mg) < emtricitabine (200 mg) < e$aviren9 (600 mg) q :ombivir - Zi ovu ine (300 mg) < lamivu ine (*+0 mg) bi Name Dosage Adult Dose Adverse Events Form(s) Nucleoside reverse transcriptase inhibitors (NRTIs) Abacavir (Ziagen) 300-mg tablet; 600 mg PO q !"#ersensitivit" reaction (ma" 20-mg/mL oral or inclu e $ever% ras&% nausea% solution 300 mg PO bi vomiting% iarr&ea% malaise% s&ortness o$ breat&% coug&% #&ar"ngitis) 'i anosine ((i e)) *2+-mg% 200-60 .g/ ,00 mg Peri#&eral neuro#at&"% #ancreatitis% mg% 2+0-mg% PO q nausea% lactic aci osis ,00-mg entericcoate ca#sule; 060 .g/ 2+0 mg 3
*0-mg/mL sus#ension 1mtricitabine (1mtriva) 200-mg ca#sule; *0-mg/mL oral solution Lamivu ine (1#ivir)
PO q 2inimal to)icit"% &"#er#igmentation
3tavu ine (Zerit)
4eno$ovir ((irea ) Zalcitabine (!ivi )
200 mg PO q or 2,0 mg (2, mL) oral solution PO q *+0-mg% 300-mg 300 mg PO q tablet; or *0-mg/mL oral *+0 mg PO bi solution *+-mg% 20-mg% -60 .g/ ,0 mg 30-mg% ,0-mg PO bi ca#sule; *-mg/mL oral 060 .g/ 30 mg solution PO bi 300-mg tablet 300 mg PO q
2inimal to)icit"
Peri#&eral neuro#at&"% #ancreatitis% lactic aci osis% li#oatro#&"% &"#erli#i emia 5ausea% vomiting% iarr&ea% &ea ac&e% ast&enia% renal insu$$icienc" Peri#&eral neuro#at&"% #ancreatitis% lactic aci osis% stomatitis 5ausea% vomiting% &ea ac&e% ast&enia% anemia% neutro#enia
0637+-mg% 067+- 067+ mg PO ti mg tablet Zi ovu ine (8etrovir) 300-mg tablet% 300 mg PO bi *00-mg ca#sule; or *0-mg/mL oral 200 mg PO ti solution; *0-mg/mL intravenous solution Nonnucleoside reverse transcriptase inhibitors (NNRTIs) 'elavir ine (8escri#tor) *00-mg% 200-mg ,00 mg PO ti 8as&% &ea ac&e tablets 1$aviren9 (3ustiva) 600-mg tablet; 600 mg PO q 8as&% :53 (eg% somnolence% vivi 200-mg ca#sule reams% con$usion% visual &allucinations) 1travirine (;ntelence) *00 mg tablet 200 mg PO bi 8as&% nausea 5evira#ine ((iramune) 200-mg tablet; 200 mg PO 8as&% &e#atitis a *0-mg/mL bi sus#ension Protease inhibitors (PIs) Ata9anavir (8e"ata9) *+0-mg% 200,00 mg PO q ;n irect &"#erbilirubinemia% mg% 300-mg or #rolonge P8 interval% ca#sules 300 mg < &"#ergl"cemia ritonavir *00 mg PO q 'arunavir (Pre9ista) ,00-mg% 600-mg =00 mg q < 8as&% nausea% iarr&ea% tablets ritonavir *00 &"#erli#i emia% &"#ergl"cemia mg PO q b or 600 mg bi < ritonavir *00
mg PO bi >osam#renavir (Le)iva) 700-mg tablet; 700 mg bi < +0-mg/mL oral ritonavir *00 sus#ension mg PO bi or *,00 mg PO bi or *,00 mg < ritonavir *00200 mg PO q b ;n inavir (:ri)ivan) *00-mg% 200=00 mg PO q=& mg% 333-mg% =00 mg PO bi ,00-mg ca#sules < ritonavir *00200 mg PO bi Lo#inavir/ritonavir 200-mg/+0-mg ,00 mg/*00 mg (?aletra) tablet; PO bi or =0-mg/20-mg =00 mg/200 mg #er mL oral PO q b solution 5el$inavir ((irace#t) 2+0-mg% 62+-mg *2+0 mg PO tablets bi or 7+0 mg PO ti (5el$inavir cannot be booste 6) 8itonavir (5orvir) *00-mg so$t @oosting ose gelatin ca#sule $or ot&er =0-mg/mL oral #rotease solution in&ibitors/ *00,00 mg/ (re$er to ot&er #rotease in&ibitors $or s#eci$ic ose) 5onboosting ose (8itonavir use as sole #rotease in&ibitor)/ 600 mg bi c 3aquinavir (;nvirase) +00-mg tablet; *000 mg < 200-mg &ar ritonavir *00 gelatin ca#sule mg PO bi Anbooste saquinavir is not recommen e 4i#ranavir (A#tivus) 2+0-mg so$t +00 mg < gelatin ca#sule ritonavir 200 mg PO bi Anbooste
8as&% nausea% vomiting% iarr&ea% &"#erli#i emia% &"#ergl"cemia
5e#&rolit&iasis% nausea% in irect &"#erbilirubinemia% &"#erli#i emia% &"#ergl"cemia 5ausea% vomiting% iarr&ea% ast&enia% &"#erli#i emia% &"#ergl"cemia 'iarr&ea% &"#erli#i emia% &"#ergl"cemia
5ausea% vomiting% iarr&ea% ast&enia% &"#erli#i emia% oral #arest&esias% &"#ergl"cemia
5ausea% iarr&ea% &ea ac&e% &"#erli#i emia% &"#ergl"cemia
!e#atoto)icit"% ras&% &"#erli#i emia% &"#ergl"cemia% intracranial &emorr&age (rare cases re#orte )
ti#ranavir is not recommen e Integrase inhibitor (II) 8altegravir (;sentress) ,00-mg tablet ,00 mg PO bi 5ausea% iarr&ea% &ea ac&e% :? elevations% m"o#at&"/r&ab om"ol"sis (rare)
Chemo ine receptor antagonist (CCR! antagonist) 2araviroc (3el9entr") *+0-mg% 300-mg 300 mg PO bi :onsti#ation% i99iness% in$ection% tablets *+0 mg PO bi ras& (:BP3A, in&ibitors C in ucers) 600 mg PO bi (:BP3A, in ucers) Fusion inhibitor (FI) 1n$uvirti e (>u9eon) D0-mg/mL D0 mg 3: bi ;nFection-site reactions (eg% #ain% #oE er $or er"t&ema% in uration% no ules) inFection Combination "ormulations 1#9icom - Abacavir (600 mg) < lamivu ine (300 mg) q 4ri9ivir - Abacavir (300 mg) < lamivu ine (*+0 mg) < 9i ovu ine (300 mg) bi 4ruva a - 4eno$ovir (300 mg) < emtricitabine (200 mg) q Atri#la - 4eno$ovir (300 mg) < emtricitabine (200 mg) < e$aviren9 (600 mg) q :ombivir - Zi ovu ine (300 mg) < lamivu ine (*+0 mg) bi
G'osing gui es assume an absence o$ rug- rug interactions (e)ce#t ritonavir) an normal renal an &e#atic $unction6 a A minister 200 mg q $or 2 Eee.s% t&en increase to 200 mg bi 6 b A##rove onl" $or antiretroviral treatmentHnaIve #atients c 4itrate ose over *, a"s% beginning Eit& 300 mg bi on a"s *-2% ,00 mg bi on a"s 3-+% an +00 mg bi on a"s 6-*36 A##rove onl" $or antiretroviral treatmentHe)#erience #atients Eit& rug resistance
You might also like
- Aristopharma Product ListDocument16 pagesAristopharma Product ListShahidullah Kaisar Shovon91% (11)
- Mcneil & Argus Pharmaceuticals Limited: Product List of Antiretroviral / Anti Viral DrugsDocument6 pagesMcneil & Argus Pharmaceuticals Limited: Product List of Antiretroviral / Anti Viral DrugsMcneil ArgusNo ratings yet
- Catalogos EneroDocument109 pagesCatalogos EnerosebastianNo ratings yet
- Anexos Finales 5 - 6 - 7 - 8 - 9Document403 pagesAnexos Finales 5 - 6 - 7 - 8 - 9Mile Castillo VillarealNo ratings yet
- NovellDocument4 pagesNovellFerry AndrianNo ratings yet
- Stok Opname & Pengajuan 09 NovemberDocument24 pagesStok Opname & Pengajuan 09 NovemberAnnisa duaNo ratings yet
- Daftar Kebutuhan Obat 2021 (Emergency)Document6 pagesDaftar Kebutuhan Obat 2021 (Emergency)dini mahdianiNo ratings yet
- Catálogo G.I. Clientes 14-03-2018Document91 pagesCatálogo G.I. Clientes 14-03-2018sebastian0% (1)
- Lap or AnDocument420 pagesLap or Annelle73No ratings yet
- Kelompok 6 Prak FRSDocument2 pagesKelompok 6 Prak FRSNINA SHINTA YULIARTI 2021No ratings yet
- Pharmacy Medicine-AdditionDocument15 pagesPharmacy Medicine-AdditionDr MWAYINGANo ratings yet
- Price List of Zubit Life Care - A Pharma Company, Franchise in Pharma and PCD Pharma Company.Document6 pagesPrice List of Zubit Life Care - A Pharma Company, Franchise in Pharma and PCD Pharma Company.Zubit Life Care, PCD Pharma Company, PCD Pharma Franchise, Pharma Franchise CompanyNo ratings yet
- Mechanisms of Action of Anticonvulsants: USMLE Step 1 Web Prep - AnticonvulsantsDocument2 pagesMechanisms of Action of Anticonvulsants: USMLE Step 1 Web Prep - AnticonvulsantsfrabziNo ratings yet
- Laporan GusngDocument1,070 pagesLaporan GusngAmyNo ratings yet
- Generic Name Dosage Price: For CoughDocument6 pagesGeneric Name Dosage Price: For CoughRaffy PangilinanNo ratings yet
- Rekap Perubahan Harga Obat 2016-2017-2018-2019 1Document2 pagesRekap Perubahan Harga Obat 2016-2017-2018-2019 1nimal hayyiNo ratings yet
- Inventario 21 Nov 2023Document44 pagesInventario 21 Nov 2023eags900115No ratings yet
- Hitungan Hutan Hujan Tropis: Hemicircus ConcretusDocument8 pagesHitungan Hutan Hujan Tropis: Hemicircus ConcretusPuttyPuspitaNo ratings yet
- Melon HerbaZestDocument1 pageMelon HerbaZestCla RenceNo ratings yet
- Product List As Per DivisionDocument68 pagesProduct List As Per DivisionAshish MahaleNo ratings yet
- Retail - Price07 10 23 3Document6 pagesRetail - Price07 10 23 3Paul TuveraNo ratings yet
- ObatDocument102 pagesObatrs hj bunda halimahNo ratings yet
- Pha 1Document60 pagesPha 1Selvy WardhanyNo ratings yet
- S.No. Type Medicine NameDocument6 pagesS.No. Type Medicine Namered petalsNo ratings yet
- Price ListDocument72 pagesPrice Listfarmasi pkmwonokertoNo ratings yet
- Prenate EliteDocument3 pagesPrenate EliteManish PrakashNo ratings yet
- PfizerDocument11 pagesPfizerParth BavadiaNo ratings yet
- Laporan Harian Pengeluaran Obat Puskesmas Tiban Baru Nama ObatDocument13 pagesLaporan Harian Pengeluaran Obat Puskesmas Tiban Baru Nama ObatfatimaNo ratings yet
- 20Document1 page20hannahNo ratings yet
- Nama Obat Shift Pagi Shift Siang Shift Malam Total Awal Masuk Keluar Awal Keluar Awal Keluar Akhir Keluar AkhirDocument2 pagesNama Obat Shift Pagi Shift Siang Shift Malam Total Awal Masuk Keluar Awal Keluar Awal Keluar Akhir Keluar AkhirisyeabdullahNo ratings yet
- Injectable Drug DataDocument57 pagesInjectable Drug DataKhunJiew WannabhornNo ratings yet
- Hikmah c1Document4 pagesHikmah c1Hikmah KimaNo ratings yet
- Price List MpiDocument126 pagesPrice List MpiAnggaPermanaZulNo ratings yet
- Centri TemplateDocument4 pagesCentri TemplateKENNETH FUENTESNo ratings yet
- Daftar ObatDocument34 pagesDaftar ObatdumaNo ratings yet
- Laporan Persediaan 1sd30okt2023Document28 pagesLaporan Persediaan 1sd30okt2023ARIEF YUNIARDINo ratings yet
- Utk Diisi Oleh PrincipalDocument5 pagesUtk Diisi Oleh PrincipalIrwan TaufiqNo ratings yet
- Tabela Importados e NootsDocument1 pageTabela Importados e Nootsrafael donatoNo ratings yet
- ProductDocument4 pagesProductAdi Fahmi AnsharyNo ratings yet
- Item Price: MAX Bunos 10 PC 50 PC 100 PCDocument1 pageItem Price: MAX Bunos 10 PC 50 PC 100 PCbashar mohammedNo ratings yet
- Med Ex 1Document117 pagesMed Ex 1alex_paskaNo ratings yet
- Treatment GuidelineDocument28 pagesTreatment Guidelinev_vijayakanth7656No ratings yet
- Hin - PH JUNE 2023 SOADocument1 pageHin - PH JUNE 2023 SOAApril LingatNo ratings yet
- Stock31dic2010 AlmFarDocument76 pagesStock31dic2010 AlmFarSergio RomeroNo ratings yet
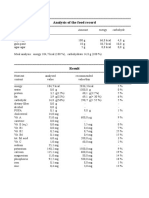
- Analysis of The Food RecordDocument2 pagesAnalysis of The Food RecordPutri MariethaNo ratings yet
- Entrega de Turno Tableteria 20200727 0534Document4 pagesEntrega de Turno Tableteria 20200727 0534mayerliNo ratings yet
- 1712 - Perhitungan Fix Tugas Ujian ABC Ven Kelompok 2-1Document6 pages1712 - Perhitungan Fix Tugas Ujian ABC Ven Kelompok 2-1SeptyNo ratings yet
- 100mg Omega 3's: DHA & Epa Folic Acid Vitamin B6 L-Arginine Vitamin E Vitamin D 3 Zinc Iron Choline Calcium Coenzyme Q10 Vitamin CDocument9 pages100mg Omega 3's: DHA & Epa Folic Acid Vitamin B6 L-Arginine Vitamin E Vitamin D 3 Zinc Iron Choline Calcium Coenzyme Q10 Vitamin CLaarni RellamaNo ratings yet
- Lista de Precios Europa QualityDocument2 pagesLista de Precios Europa QualitylapiamaNo ratings yet
- Nota Kirim PEMBAYARAN BELANJA BEKKES DAN OBAT-OBATAN POLKES PONOROGO (AJ)Document2 pagesNota Kirim PEMBAYARAN BELANJA BEKKES DAN OBAT-OBATAN POLKES PONOROGO (AJ)Prameswari Wed DingNo ratings yet
- Rekap Obat New..-2Document473 pagesRekap Obat New..-2ana siregarNo ratings yet
- Neraca Triwulan 2Document8 pagesNeraca Triwulan 2kasina kasinaNo ratings yet
- Pt. Bernofarm Ogb: NO Nama ProdukDocument10 pagesPt. Bernofarm Ogb: NO Nama ProdukamandaNo ratings yet
- Berkas Rejec InhealthDocument3 pagesBerkas Rejec InhealthRoffy OktavianNo ratings yet
- Daftar ObatDocument4 pagesDaftar Obattiara_dewinthaNo ratings yet
- MB Lab - Exam Urgent RequirementsssDocument2 pagesMB Lab - Exam Urgent RequirementssssujanupamNo ratings yet
- ScriptDocument32 pagesScriptMailyn OlmedoNo ratings yet
- FoodlogtemplateDocument4 pagesFoodlogtemplateapi-397122400No ratings yet
- HIV Medication Chart - August 2022Document2 pagesHIV Medication Chart - August 2022Manuel S PlasenciaNo ratings yet
- Antiviral Drug: Classification and Mechanism of ActionDocument1 pageAntiviral Drug: Classification and Mechanism of ActionNikita sharmaNo ratings yet
- ZGH ART Pharmacy InventoryDocument8 pagesZGH ART Pharmacy InventorywondwossenNo ratings yet
- Formularium RSBD 2022 Fix EditedDocument152 pagesFormularium RSBD 2022 Fix EditedInstalasi Farmasi RSBDNo ratings yet
- 627 1196 1 PBDocument7 pages627 1196 1 PBannisa dian zizianiNo ratings yet
- HIV Drug Chart (2021)Document1 pageHIV Drug Chart (2021)savNo ratings yet
- SHC Antiretroviral FormularyDocument1 pageSHC Antiretroviral FormularyMario BulaciosNo ratings yet
- 2019 HIV Drug ChartDocument5 pages2019 HIV Drug ChartETNo ratings yet
- HIV Drug ChartDocument5 pagesHIV Drug Chartalejandroo_26No ratings yet
- Lab Requisition SampleDocument1 pageLab Requisition SampleUser SmuggNo ratings yet
- POZ 2020 HIV Drug Chart HighDocument5 pagesPOZ 2020 HIV Drug Chart HighReni BaliNo ratings yet
- HIV Medication Chart - Jan 2019Document2 pagesHIV Medication Chart - Jan 2019Stephany SoutoNo ratings yet
- Hiv TreatmentDocument12 pagesHiv Treatmentshivaj0009No ratings yet
- Exp Ifk 2022Document2 pagesExp Ifk 2022ngawi farmasiNo ratings yet
- 4 +ok+ (11) +Filya+Aulia+Hunou+29-39+doiDocument11 pages4 +ok+ (11) +Filya+Aulia+Hunou+29-39+doiNovy SandhyanaNo ratings yet
- Generic Name Brand Name Abbreviation: © 2017 Find Your Script HIV Medications - Drug Names Quick SheetDocument1 pageGeneric Name Brand Name Abbreviation: © 2017 Find Your Script HIV Medications - Drug Names Quick SheetMonica J Ortiz PereiraNo ratings yet
- Antiviral - Classification and ImagesDocument2 pagesAntiviral - Classification and ImagesNuwaira Baloch100% (3)
- Format Rko Program 2020Document18 pagesFormat Rko Program 2020Anonymous ItXYBoaC1CNo ratings yet
- Antiretroviral Presentation - LamivudineDocument14 pagesAntiretroviral Presentation - LamivudineTehreem KhanNo ratings yet
- Farmakologi AntivirusDocument18 pagesFarmakologi AntivirusTia TiaraNo ratings yet
- Anti Viral Agents Carewell PharmaDocument5 pagesAnti Viral Agents Carewell PharmaHritik ChaubeyNo ratings yet
- FDA - HIV and AIDS Activities - Antiretroviral Drugs Used in The Treatment of HIV InfectionDocument5 pagesFDA - HIV and AIDS Activities - Antiretroviral Drugs Used in The Treatment of HIV InfectionMade YudanaNo ratings yet
- 3 Konsep Terapi ARVDocument65 pages3 Konsep Terapi ARVkristinaNo ratings yet
- Data Logistik Untuk SO SIHA 2.1 KabKotaDocument34 pagesData Logistik Untuk SO SIHA 2.1 KabKotaTeti HerawatiNo ratings yet
- Marking Scheme Sample PharmacologyDocument2 pagesMarking Scheme Sample PharmacologyObaroNo ratings yet
- Aurbindo Pharma LTDDocument2 pagesAurbindo Pharma LTDAaditya Reborn ZeusNo ratings yet