Professional Documents
Culture Documents
0 ratings0% found this document useful (0 votes)
341 viewsChem Cheat Sheet
Chem Cheat Sheet
Uploaded by
ryan26480This document summarizes key concepts from several chemistry chapters:
1. It introduces atomic theory and the three states of matter. It also defines density and discusses significant figures.
2. It covers the laws of definite and multiple proportions regarding chemical composition. It also summarizes some classic experiments in atomic theory.
3. It discusses ionic and covalent bonding, distinguishing between their properties and bond formations. It also covers determining chemical formulas.
Copyright:
Attribution Non-Commercial (BY-NC)
Available Formats
Download as PDF, TXT or read online from Scribd
You might also like
- Principles of Electronic Materials and Devices 4th Edition Kasap Solutions ManualDocument26 pagesPrinciples of Electronic Materials and Devices 4th Edition Kasap Solutions ManualJeremySmithwrok98% (45)
- Molecule Shapes Student HandoutDocument5 pagesMolecule Shapes Student Handoutapi-2982478730% (1)
- Simulation BondingDocument12 pagesSimulation BondingCristina Nicomedes Aguinaldo0% (2)
- Electric Fields & Potential WorksheetDocument4 pagesElectric Fields & Potential WorksheetFandy Ardy50% (2)
- IX. Waves, Sounds and FluidsDocument66 pagesIX. Waves, Sounds and FluidsMa Angelica Shane Navarro0% (1)
- Basic and Biochemistry Notes Website VersionDocument14 pagesBasic and Biochemistry Notes Website Versionapi-292966101No ratings yet
- Ionization RangeDocument6 pagesIonization RangeMichael Bowles100% (1)
- PES SheetDocument4 pagesPES SheetColin Shin100% (1)
- Electron Configuration and Quantum NumbersDocument50 pagesElectron Configuration and Quantum NumbersGustavo LarrazabalNo ratings yet
- Chemistry of Hydrocarbon Experiment 16: ObjectiveDocument10 pagesChemistry of Hydrocarbon Experiment 16: ObjectiveWena Sta Maria100% (1)
- LoraineDocument29 pagesLoraineR I R INo ratings yet
- Modern Physics Notes PDFDocument64 pagesModern Physics Notes PDFBernie EffieNo ratings yet
- Chemistry Module 3Document14 pagesChemistry Module 3MASHNo ratings yet
- Formation of Ionic BondDocument14 pagesFormation of Ionic BondZaza MawarNo ratings yet
- 131279806192793304Chemistry-Engineering Chemistry Lab Manuals (Revised)Document41 pages131279806192793304Chemistry-Engineering Chemistry Lab Manuals (Revised)kaushal guptaNo ratings yet
- PHYS 1100 Electric Fields SolutionsDocument18 pagesPHYS 1100 Electric Fields SolutionsCLester MadShadowNo ratings yet
- Scientific Notation: A Quick Way To WriteDocument15 pagesScientific Notation: A Quick Way To WriteJoan May de LumenNo ratings yet
- Lewis Theory of BondingDocument8 pagesLewis Theory of BondingJeto SantosNo ratings yet
- Modern PhysicsDocument74 pagesModern PhysicsLàXsun ShrèsthàNo ratings yet
- Scientific MeasurementsDocument47 pagesScientific MeasurementsArvin Corpuz DiamanteNo ratings yet
- Balloons and Static Electricity ScriptedDocument44 pagesBalloons and Static Electricity Scriptedluqman_HNo ratings yet
- Atomic Structure WorksheetDocument2 pagesAtomic Structure Worksheetrangerblue94% (17)
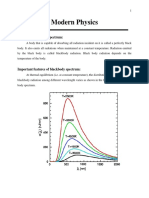
- Some Important Point: Modern PhysicsDocument5 pagesSome Important Point: Modern PhysicsBhanuNo ratings yet
- Solved Problems AustraliaDocument12 pagesSolved Problems AustraliaIsidoro VaquilaNo ratings yet
- Importance of Physics in Everyday LifeDocument2 pagesImportance of Physics in Everyday LifeAllen Mae Flores AdoresNo ratings yet
- Electrochemical CellDocument30 pagesElectrochemical CellSubhu100% (1)
- Classification of Matter: General and Organic Chemistry Prof. Rowena H. AragonDocument41 pagesClassification of Matter: General and Organic Chemistry Prof. Rowena H. AragonJeremy TulaganNo ratings yet
- 2 CH241 Polar Covalent BondsDocument94 pages2 CH241 Polar Covalent Bondsalyssa_marie_keNo ratings yet
- QuantumDocument255 pagesQuantummuditNo ratings yet
- General Chemistry ModuleDocument367 pagesGeneral Chemistry ModuleBirhaanu TolesaNo ratings yet
- ES Module 3 - Quarter 1 - Types of SolidsDocument13 pagesES Module 3 - Quarter 1 - Types of SolidsAnalynAsuncionAtaydeNo ratings yet
- Modern Physics Notes 2020 PDFDocument16 pagesModern Physics Notes 2020 PDFNorthrop B2spiritNo ratings yet
- Physics 101 Chapter 8 RotationDocument52 pagesPhysics 101 Chapter 8 RotationAndrew GoolsbyNo ratings yet
- Torque and Rotation PhysicsDocument20 pagesTorque and Rotation PhysicsSaf Tanggo DiampuanNo ratings yet
- Uniform and Non - Uniform Circular MotionDocument4 pagesUniform and Non - Uniform Circular MotionExpertsmindEduNo ratings yet
- Engineering Physics - 104: Photoelectric Effect & Postulates of Quantum MechanicsDocument22 pagesEngineering Physics - 104: Photoelectric Effect & Postulates of Quantum MechanicsSalaar Khan100% (1)
- Chemistry 11 Concept ReviewDocument31 pagesChemistry 11 Concept ReviewJames Ernest Baker-taylor100% (1)
- Chemical Bonding and Molecular StructureDocument14 pagesChemical Bonding and Molecular StructureRoshita G PillaiNo ratings yet
- General Chemistry 1: Quarter 1 - Module 2Document17 pagesGeneral Chemistry 1: Quarter 1 - Module 2CODILLO, CARLO R.No ratings yet
- Biology Unit Science 9Document9 pagesBiology Unit Science 9api-373530490No ratings yet
- Butterfly Life Cycle Lesson PlanDocument4 pagesButterfly Life Cycle Lesson PlanPeter GeorgeNo ratings yet
- Atoms:: Particle Electron Proton Neutron Discovery Nature of Charge Negative Amount of Charge MassDocument6 pagesAtoms:: Particle Electron Proton Neutron Discovery Nature of Charge Negative Amount of Charge MassNasser SsennogaNo ratings yet
- Chapter 12 - NucleusDocument23 pagesChapter 12 - NucleusSherena Peter Govind67% (3)
- Models of Molecular Compounds Lab (Ms. Possible)Document5 pagesModels of Molecular Compounds Lab (Ms. Possible)Steven GomescoelloNo ratings yet
- KinematicsDocument16 pagesKinematicsSovaid Soroush KhanNo ratings yet
- CHAPTER 01 The Foundation of ChemistryDocument105 pagesCHAPTER 01 The Foundation of ChemistryazulaNo ratings yet
- CH 7 Ionic BondsDocument27 pagesCH 7 Ionic Bondsapi-239855791No ratings yet
- PropertiesDocument41 pagesPropertiesMarielle LabradoresNo ratings yet
- Worksheet - Cell Membrane - Review Worksheet KeyDocument3 pagesWorksheet - Cell Membrane - Review Worksheet Keytyler guyNo ratings yet
- Kinetic Mastering PhysicsDocument16 pagesKinetic Mastering PhysicsKyle DonagheyNo ratings yet
- CHEM20024 Lecture Notes 01 (Review On Chem Bonding)Document17 pagesCHEM20024 Lecture Notes 01 (Review On Chem Bonding)Lorielle OlivaNo ratings yet
- Very Short Answer QuestionDocument35 pagesVery Short Answer QuestionAyush SigdelNo ratings yet
- ElectrochemDocument12 pagesElectrochemKatrin Nicole AbelardoNo ratings yet
- Practice Problems Density and Specific GravityDocument2 pagesPractice Problems Density and Specific GravityNOVASTAR222100% (1)
- Stoichiometric CalculationsDocument33 pagesStoichiometric CalculationsHazrati Ummi100% (1)
- Chemical BondingDocument51 pagesChemical BondingDaniel MaglalangNo ratings yet
- Topic 1-Osmosity and Tonicity - EditedDocument6 pagesTopic 1-Osmosity and Tonicity - Editedsalve joy villanuevaNo ratings yet
- Created By: Heri Reksa S.Pd. Chemistry Teacher SMAN 1 BogorDocument45 pagesCreated By: Heri Reksa S.Pd. Chemistry Teacher SMAN 1 BogorHeri ReksaNo ratings yet
- Ionic and Covalent Bonding Comics GuidelinesDocument1 pageIonic and Covalent Bonding Comics GuidelinesArch Broqueza33% (3)
- Reference Material I - Unit-I-Introduction To Modern PhysicsDocument58 pagesReference Material I - Unit-I-Introduction To Modern Physicssaugat100% (1)
- General and Inorganic Chemistry For Pharmacy PDFDocument91 pagesGeneral and Inorganic Chemistry For Pharmacy PDFthucinorNo ratings yet
- Week 1module StudentDocument12 pagesWeek 1module Studentkylie jonesNo ratings yet
- Quantum Physics of GodDocument37 pagesQuantum Physics of GodSarleneNo ratings yet
- Answer and Solutions - JEE - Main 2023 - PH 1 - 25 01 2023 - Morning - Shift 1 PDFDocument25 pagesAnswer and Solutions - JEE - Main 2023 - PH 1 - 25 01 2023 - Morning - Shift 1 PDFinduja c mNo ratings yet
- Multiwfn 3.6 (Dev) PDFDocument644 pagesMultiwfn 3.6 (Dev) PDFdirka betancourtNo ratings yet
- Detailed Lesson Plan (DLP) FormatDocument2 pagesDetailed Lesson Plan (DLP) FormatJim Alesther LapinaNo ratings yet
- 02 Quarks and LeptonsDocument104 pages02 Quarks and LeptonsAnish KarnaNo ratings yet
- Module 5 Physical PharmacyDocument7 pagesModule 5 Physical Pharmacycrayon100% (2)
- Julia Martins - Modeling Periodic Trends Project AolDocument8 pagesJulia Martins - Modeling Periodic Trends Project Aolapi-550916827No ratings yet
- CBSE Class 9 Science Revision Notes Chapter - 4 Structure of AtomDocument7 pagesCBSE Class 9 Science Revision Notes Chapter - 4 Structure of Atomnajaf_shaanNo ratings yet
- Phy Assignment NITDocument1 pagePhy Assignment NITKartik GvrNo ratings yet
- Entry Test Preparation Test No.2 (ECAT)Document3 pagesEntry Test Preparation Test No.2 (ECAT)NAEEM UR REHMANNo ratings yet
- Physicalscience SyllbusDocument3 pagesPhysicalscience Syllbusvincy sundara rajNo ratings yet
- PhysicsBowl 2013Document11 pagesPhysicsBowl 2013Ajay NegiNo ratings yet
- Chapter 4 - Systems in HospitalsDocument40 pagesChapter 4 - Systems in Hospitalsmarye agegnNo ratings yet
- KEITH BREWER High PH Therapy For Cancer Tests On Mice and Humans 12PDocument12 pagesKEITH BREWER High PH Therapy For Cancer Tests On Mice and Humans 12PAna LuNo ratings yet
- Fluorescence Spectroscopy and Imaging: Basic Principles and Sources of ContrastDocument78 pagesFluorescence Spectroscopy and Imaging: Basic Principles and Sources of Contrastraul reyesNo ratings yet
- Question Bank For Summative Assesment-IIDocument19 pagesQuestion Bank For Summative Assesment-IIApex InstituteNo ratings yet
- Purposes and Methods of Research in Mathematics EdDocument10 pagesPurposes and Methods of Research in Mathematics Edhassankanj134No ratings yet
- Electrostatic Forces, Charges and FieldsDocument9 pagesElectrostatic Forces, Charges and FieldsDeepak PachauriNo ratings yet
- Lesson 1 To 4 IcDocument66 pagesLesson 1 To 4 IcscientistgenerosoNo ratings yet
- Ch12 SolutionsDocument12 pagesCh12 SolutionsJoe ReynoldsNo ratings yet
- CH 6Document16 pagesCH 6Iratechaos100% (2)
- Feb. 2022 Ptu PaperDocument2 pagesFeb. 2022 Ptu Paperthakralshivam21No ratings yet
- U-2StructureofAtom1 89626Document23 pagesU-2StructureofAtom1 89626Anirudh AshrithNo ratings yet
- 9J Quick QuizDocument2 pages9J Quick QuizOneth RajapakseNo ratings yet
- NMR and EsrDocument52 pagesNMR and EsrSenthil Sethu FvfcNo ratings yet
- Special RelativityDocument17 pagesSpecial RelativityDiego M GranziolNo ratings yet
Chem Cheat Sheet
Chem Cheat Sheet
Uploaded by
ryan264800 ratings0% found this document useful (0 votes)
341 views2 pagesThis document summarizes key concepts from several chemistry chapters:
1. It introduces atomic theory and the three states of matter. It also defines density and discusses significant figures.
2. It covers the laws of definite and multiple proportions regarding chemical composition. It also summarizes some classic experiments in atomic theory.
3. It discusses ionic and covalent bonding, distinguishing between their properties and bond formations. It also covers determining chemical formulas.
Original Description:
Basic formulas and equations for a chemistry student.
Copyright
© Attribution Non-Commercial (BY-NC)
Available Formats
PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentThis document summarizes key concepts from several chemistry chapters:
1. It introduces atomic theory and the three states of matter. It also defines density and discusses significant figures.
2. It covers the laws of definite and multiple proportions regarding chemical composition. It also summarizes some classic experiments in atomic theory.
3. It discusses ionic and covalent bonding, distinguishing between their properties and bond formations. It also covers determining chemical formulas.
Copyright:
Attribution Non-Commercial (BY-NC)
Available Formats
Download as PDF, TXT or read online from Scribd
Download as pdf or txt
0 ratings0% found this document useful (0 votes)
341 views2 pagesChem Cheat Sheet
Chem Cheat Sheet
Uploaded by
ryan26480This document summarizes key concepts from several chemistry chapters:
1. It introduces atomic theory and the three states of matter. It also defines density and discusses significant figures.
2. It covers the laws of definite and multiple proportions regarding chemical composition. It also summarizes some classic experiments in atomic theory.
3. It discusses ionic and covalent bonding, distinguishing between their properties and bond formations. It also covers determining chemical formulas.
Copyright:
Attribution Non-Commercial (BY-NC)
Available Formats
Download as PDF, TXT or read online from Scribd
Download as pdf or txt
You are on page 1of 2
Chapter One
Atomic Theory- Proposed by John Dalton, matter is
composed of small, scientic particles
Classications of Matter
-Solids-Denite shape, compressed molecules
-Crystalline-Arranged in a pattern
-Amorphous-No unique, long term pattern
-Liquids-No denite shape, compressed molecules
-Takes shape of container
-Gas- No denite shape, spaced out molecules
-Takes shape of container
Density D=M/V
Accuracy-Near Expected Range
Precision-Near Other Figures
Sig Fig Rules
-All Non-Zeros are sig.
-Interior Zeros (between 2 non-zeros) are sig.
-Trailing Zeros (after decimal) are sig.
-Zeros before Decimal need to go into Scientic
Notation
Addition/Sub.- Smallest Decimal Spot
Multiplication/Div.- Smallest # sig gs.
Chapter Two
Law of Denite Proportions: All samples of a given
compound, regardless of preparation, have the
same properties and ratios of the same compound
(i.e. HO=18g=16g H/2g O= 8.0:1 ratio)
Law of Multiple Proportions: When their are two
elements (i.e. A+B) the masses of element B
combine with 1 g of element A can be expressed as
a ratio of small #s
(i.e. CO= 1g C to 2.67g of O)
Experiments
-Cathode Ray Tube-Led to discovery of electrons by
sending them through a ray tube. This help
determine the mass per g of electrons
-Done by JJ Thompson
-Oil Drop Experiment- Calculated charge per
electron
-Done by Robert Milken
-Gold Foil Experiment- Determined structure of
atoms because alpha particles reected back,
disproving plum pudding model
-Done by Rutherford
Properties of Matter
Metals: Usually shiny, malleability, ductility, good
conductors of heat and electricity.
Non-Metals: Poor Conductors, typically gain
electrons
Metalloids: Mixed properties
Avagadros #= 6.022 X 10!"
Chapter Three
Ionic Bonds-Donates !e, between metal and non-
metal
Covalent Bonds-occurs between 2 non-metals,
involves the sharing of electrons
Differences: Matter properties, creation of lattices,
bonding type.
1 PPM= 1g per 10#g
Determining chemical formula based off weight
1. convert g to moles
2. divide by lowest # of moles
3. round out or multiply
Chapter Four
Molarity of solutions= amount of solute (in mol)/
volume of solution (per L)
Dilution Equation= MV= MV
Electrolytes=Solutions that conduct electricity well,
generally ions are the best.
Strength of acids= Ability to dissociate completely
Dissolved acids make great electrolytes
Titrations are not dilutions!
Convert amounts to moles then nd ratio then nd
appropriate amount of neutralization substance then
convert to grams
Redox- Ability to transfer electrons from one
substance to another
Reduction-gaining electrons
Oxidation-losing elections OILRIG
Oxidizing agent is, itself, reduced and vise-versa
Chapter Six
1st Law of Thermodynamics- All energy in the
universe is constant
reactants have high E than products
E`` > 0 E` < 0
reactants have lower E than products
Chapter Seven
Photon is a packet of light
Wavelength is the length of a wave
Amplitude is its height (relates to intensity)
The shortest possible wavelength is a gamma ray
Humans typically can see between 750 and 400 nm
Photoelectric effect- Chance for a photon to be
emitted when the proper frequency is shined on a
metal.
h= Plancks Constant= 6.626x10!34 JxS
c= speed of light= 3.00x10 m/s
m of an electron=9.11x10!31kg
V= c/I (frequency/speed of travel/Hz)
hv=1 (binding energy/energy to release a photon)
KEemitted photon= hv-1
emissions spectra= same for each particular
element, aids in identifying elements
de Broglie relation= I= h/mv
Ephoton=hc/I
E=hv
Quantum #s
principle=n (level electrons are at) (also # of sub-
levels)
angular momentum=l (species which orbit)(n-1)
magnetic number=ml (+,-, and 0 of l value)
Photon Calculations
En= -2.18x10!18(1/n)
AE=Enal-Einital
Chapter Five
Boyles Law PV=PV
Charles Law V/T=V/T
Avagadros Law V/n=V/n
Ideal Gas Law PV=nRT
R= .08206 L x atm/mol x K
DLPP=P+P+P...=Ptotal
Pa=XaPtotal (X=# moles in a substance)
P=Ftotal/A
Fcollision= m x 2V/AT
Grahms Law of Effusion rate a/rate b= vM b/ M a
Common
Diatomic or
Polyatomic
Molecules
H
N
Cl
P
S
SI Base Units and
Prex Multipliers
Length-M, Mass-Kg,
Time-Sec, Temp-K,Amt.-
Mol.
SI Prexes
Kilo (10!)
Deci (10!)
Centi (10!)
Milli (10!)
Micro (10!)
Nano (10!)
Pico (10!)
Acid/Base Strengths
Red denotes a weakness
Acids Bases
HCl NaOH
HBr LiOh
HI KOH
HNO Ca(OH)
HSO Ba(OH)
HClO NH
HCHO
HF
Redox Rules
1.Oxidation state of an atom in a free element is 0
2. Oxidation state of a monoatomic atom is equal
to its charge (i.e Na' is +1)
3. Neutral molecules have a charge of 0
-ion is equal to charge of ion
4. Metals always have positive oxidation states
Group 1A= +1
Group 2A= +2
5.In compounds
- O is -2
- H is +1
- F is -1
-Group 7A is -1 Entries at top take
-Group 6A is -2 precedence
-Group 5A is -3 those below
**Oxidation states of compounds must = 0
matter
variable composition?
pure substance mixture
separate into simpler substances? uniform?
element compound heterogeneous homogenous
pure substances
element compound
atomic molecular molecular Ionic
Chapter Ten
Valence Shell Electron Pair Repulsion
-Theory that lone pairs and bonds repel each other,
resulting a maximization of separation.
Electron Geometry-Lewis Structure Geometry
Molecular Geometry-Actual Geometry
Valance Bond Theory
-Electrons reside in quantum mechanical orbitals
-Hybridized orbitals are when two orbitals smosh
together
-This requires energy and central atoms are most
likely to hybridize
Sigma Only Bonds- sp3 (typically have only single
bonds)
Pi Bonds- sp2 (typically have a double bond)
Dual Pi Bonds- sp (typically in triple bonds)
Chapter Eleven
IM Forces
Dispersion-Temporary uctuation creating a
weak attraction
Dipole-Dipole- Molecules have a permanent
polarity allowing two polar parts to attract
-Hydrogen Bonding- With O,F,N. The H atoms
are strongly attracted to the O, F, N of other
molecules
Ion-Dipole- Extremely strong attraction of a
dipole to an ion.
Surface Tension-Tendency of Liquids to
minimize surface area by pushing and pulling.
Stronger IM forces result in higher ST.
Viscosity-Long molecules are sluggish
Capillary Action-Cohesive forces pull things up
tube.Only if stronger than adhesive forces.
Lewis Structures
-Write skeletal structure (H is terminal, more
electronegative is terminal) add single bonds
-Calculate # of electrons by adding valance
electrons
-Create Octets (or duet for H)
-Add double and triple bonds
-FC: # valance electrons-lone pair-
[1/2#bonds])
-Check for resonance structures (different
types)
Energy Orbitals
1s->2s->2p->3s->3p->4s->3d->4p->4s->4d
Gas Reactions
Suldes (S1)=H2S
Carbonates and Bicarbonates (CO3)=CO2
Sultes and Bisulte (SO2) + (SO2)2= SO2
Ammonium (NH4)=NH3
You might also like
- Principles of Electronic Materials and Devices 4th Edition Kasap Solutions ManualDocument26 pagesPrinciples of Electronic Materials and Devices 4th Edition Kasap Solutions ManualJeremySmithwrok98% (45)
- Molecule Shapes Student HandoutDocument5 pagesMolecule Shapes Student Handoutapi-2982478730% (1)
- Simulation BondingDocument12 pagesSimulation BondingCristina Nicomedes Aguinaldo0% (2)
- Electric Fields & Potential WorksheetDocument4 pagesElectric Fields & Potential WorksheetFandy Ardy50% (2)
- IX. Waves, Sounds and FluidsDocument66 pagesIX. Waves, Sounds and FluidsMa Angelica Shane Navarro0% (1)
- Basic and Biochemistry Notes Website VersionDocument14 pagesBasic and Biochemistry Notes Website Versionapi-292966101No ratings yet
- Ionization RangeDocument6 pagesIonization RangeMichael Bowles100% (1)
- PES SheetDocument4 pagesPES SheetColin Shin100% (1)
- Electron Configuration and Quantum NumbersDocument50 pagesElectron Configuration and Quantum NumbersGustavo LarrazabalNo ratings yet
- Chemistry of Hydrocarbon Experiment 16: ObjectiveDocument10 pagesChemistry of Hydrocarbon Experiment 16: ObjectiveWena Sta Maria100% (1)
- LoraineDocument29 pagesLoraineR I R INo ratings yet
- Modern Physics Notes PDFDocument64 pagesModern Physics Notes PDFBernie EffieNo ratings yet
- Chemistry Module 3Document14 pagesChemistry Module 3MASHNo ratings yet
- Formation of Ionic BondDocument14 pagesFormation of Ionic BondZaza MawarNo ratings yet
- 131279806192793304Chemistry-Engineering Chemistry Lab Manuals (Revised)Document41 pages131279806192793304Chemistry-Engineering Chemistry Lab Manuals (Revised)kaushal guptaNo ratings yet
- PHYS 1100 Electric Fields SolutionsDocument18 pagesPHYS 1100 Electric Fields SolutionsCLester MadShadowNo ratings yet
- Scientific Notation: A Quick Way To WriteDocument15 pagesScientific Notation: A Quick Way To WriteJoan May de LumenNo ratings yet
- Lewis Theory of BondingDocument8 pagesLewis Theory of BondingJeto SantosNo ratings yet
- Modern PhysicsDocument74 pagesModern PhysicsLàXsun ShrèsthàNo ratings yet
- Scientific MeasurementsDocument47 pagesScientific MeasurementsArvin Corpuz DiamanteNo ratings yet
- Balloons and Static Electricity ScriptedDocument44 pagesBalloons and Static Electricity Scriptedluqman_HNo ratings yet
- Atomic Structure WorksheetDocument2 pagesAtomic Structure Worksheetrangerblue94% (17)
- Some Important Point: Modern PhysicsDocument5 pagesSome Important Point: Modern PhysicsBhanuNo ratings yet
- Solved Problems AustraliaDocument12 pagesSolved Problems AustraliaIsidoro VaquilaNo ratings yet
- Importance of Physics in Everyday LifeDocument2 pagesImportance of Physics in Everyday LifeAllen Mae Flores AdoresNo ratings yet
- Electrochemical CellDocument30 pagesElectrochemical CellSubhu100% (1)
- Classification of Matter: General and Organic Chemistry Prof. Rowena H. AragonDocument41 pagesClassification of Matter: General and Organic Chemistry Prof. Rowena H. AragonJeremy TulaganNo ratings yet
- 2 CH241 Polar Covalent BondsDocument94 pages2 CH241 Polar Covalent Bondsalyssa_marie_keNo ratings yet
- QuantumDocument255 pagesQuantummuditNo ratings yet
- General Chemistry ModuleDocument367 pagesGeneral Chemistry ModuleBirhaanu TolesaNo ratings yet
- ES Module 3 - Quarter 1 - Types of SolidsDocument13 pagesES Module 3 - Quarter 1 - Types of SolidsAnalynAsuncionAtaydeNo ratings yet
- Modern Physics Notes 2020 PDFDocument16 pagesModern Physics Notes 2020 PDFNorthrop B2spiritNo ratings yet
- Physics 101 Chapter 8 RotationDocument52 pagesPhysics 101 Chapter 8 RotationAndrew GoolsbyNo ratings yet
- Torque and Rotation PhysicsDocument20 pagesTorque and Rotation PhysicsSaf Tanggo DiampuanNo ratings yet
- Uniform and Non - Uniform Circular MotionDocument4 pagesUniform and Non - Uniform Circular MotionExpertsmindEduNo ratings yet
- Engineering Physics - 104: Photoelectric Effect & Postulates of Quantum MechanicsDocument22 pagesEngineering Physics - 104: Photoelectric Effect & Postulates of Quantum MechanicsSalaar Khan100% (1)
- Chemistry 11 Concept ReviewDocument31 pagesChemistry 11 Concept ReviewJames Ernest Baker-taylor100% (1)
- Chemical Bonding and Molecular StructureDocument14 pagesChemical Bonding and Molecular StructureRoshita G PillaiNo ratings yet
- General Chemistry 1: Quarter 1 - Module 2Document17 pagesGeneral Chemistry 1: Quarter 1 - Module 2CODILLO, CARLO R.No ratings yet
- Biology Unit Science 9Document9 pagesBiology Unit Science 9api-373530490No ratings yet
- Butterfly Life Cycle Lesson PlanDocument4 pagesButterfly Life Cycle Lesson PlanPeter GeorgeNo ratings yet
- Atoms:: Particle Electron Proton Neutron Discovery Nature of Charge Negative Amount of Charge MassDocument6 pagesAtoms:: Particle Electron Proton Neutron Discovery Nature of Charge Negative Amount of Charge MassNasser SsennogaNo ratings yet
- Chapter 12 - NucleusDocument23 pagesChapter 12 - NucleusSherena Peter Govind67% (3)
- Models of Molecular Compounds Lab (Ms. Possible)Document5 pagesModels of Molecular Compounds Lab (Ms. Possible)Steven GomescoelloNo ratings yet
- KinematicsDocument16 pagesKinematicsSovaid Soroush KhanNo ratings yet
- CHAPTER 01 The Foundation of ChemistryDocument105 pagesCHAPTER 01 The Foundation of ChemistryazulaNo ratings yet
- CH 7 Ionic BondsDocument27 pagesCH 7 Ionic Bondsapi-239855791No ratings yet
- PropertiesDocument41 pagesPropertiesMarielle LabradoresNo ratings yet
- Worksheet - Cell Membrane - Review Worksheet KeyDocument3 pagesWorksheet - Cell Membrane - Review Worksheet Keytyler guyNo ratings yet
- Kinetic Mastering PhysicsDocument16 pagesKinetic Mastering PhysicsKyle DonagheyNo ratings yet
- CHEM20024 Lecture Notes 01 (Review On Chem Bonding)Document17 pagesCHEM20024 Lecture Notes 01 (Review On Chem Bonding)Lorielle OlivaNo ratings yet
- Very Short Answer QuestionDocument35 pagesVery Short Answer QuestionAyush SigdelNo ratings yet
- ElectrochemDocument12 pagesElectrochemKatrin Nicole AbelardoNo ratings yet
- Practice Problems Density and Specific GravityDocument2 pagesPractice Problems Density and Specific GravityNOVASTAR222100% (1)
- Stoichiometric CalculationsDocument33 pagesStoichiometric CalculationsHazrati Ummi100% (1)
- Chemical BondingDocument51 pagesChemical BondingDaniel MaglalangNo ratings yet
- Topic 1-Osmosity and Tonicity - EditedDocument6 pagesTopic 1-Osmosity and Tonicity - Editedsalve joy villanuevaNo ratings yet
- Created By: Heri Reksa S.Pd. Chemistry Teacher SMAN 1 BogorDocument45 pagesCreated By: Heri Reksa S.Pd. Chemistry Teacher SMAN 1 BogorHeri ReksaNo ratings yet
- Ionic and Covalent Bonding Comics GuidelinesDocument1 pageIonic and Covalent Bonding Comics GuidelinesArch Broqueza33% (3)
- Reference Material I - Unit-I-Introduction To Modern PhysicsDocument58 pagesReference Material I - Unit-I-Introduction To Modern Physicssaugat100% (1)
- General and Inorganic Chemistry For Pharmacy PDFDocument91 pagesGeneral and Inorganic Chemistry For Pharmacy PDFthucinorNo ratings yet
- Week 1module StudentDocument12 pagesWeek 1module Studentkylie jonesNo ratings yet
- Quantum Physics of GodDocument37 pagesQuantum Physics of GodSarleneNo ratings yet
- Answer and Solutions - JEE - Main 2023 - PH 1 - 25 01 2023 - Morning - Shift 1 PDFDocument25 pagesAnswer and Solutions - JEE - Main 2023 - PH 1 - 25 01 2023 - Morning - Shift 1 PDFinduja c mNo ratings yet
- Multiwfn 3.6 (Dev) PDFDocument644 pagesMultiwfn 3.6 (Dev) PDFdirka betancourtNo ratings yet
- Detailed Lesson Plan (DLP) FormatDocument2 pagesDetailed Lesson Plan (DLP) FormatJim Alesther LapinaNo ratings yet
- 02 Quarks and LeptonsDocument104 pages02 Quarks and LeptonsAnish KarnaNo ratings yet
- Module 5 Physical PharmacyDocument7 pagesModule 5 Physical Pharmacycrayon100% (2)
- Julia Martins - Modeling Periodic Trends Project AolDocument8 pagesJulia Martins - Modeling Periodic Trends Project Aolapi-550916827No ratings yet
- CBSE Class 9 Science Revision Notes Chapter - 4 Structure of AtomDocument7 pagesCBSE Class 9 Science Revision Notes Chapter - 4 Structure of Atomnajaf_shaanNo ratings yet
- Phy Assignment NITDocument1 pagePhy Assignment NITKartik GvrNo ratings yet
- Entry Test Preparation Test No.2 (ECAT)Document3 pagesEntry Test Preparation Test No.2 (ECAT)NAEEM UR REHMANNo ratings yet
- Physicalscience SyllbusDocument3 pagesPhysicalscience Syllbusvincy sundara rajNo ratings yet
- PhysicsBowl 2013Document11 pagesPhysicsBowl 2013Ajay NegiNo ratings yet
- Chapter 4 - Systems in HospitalsDocument40 pagesChapter 4 - Systems in Hospitalsmarye agegnNo ratings yet
- KEITH BREWER High PH Therapy For Cancer Tests On Mice and Humans 12PDocument12 pagesKEITH BREWER High PH Therapy For Cancer Tests On Mice and Humans 12PAna LuNo ratings yet
- Fluorescence Spectroscopy and Imaging: Basic Principles and Sources of ContrastDocument78 pagesFluorescence Spectroscopy and Imaging: Basic Principles and Sources of Contrastraul reyesNo ratings yet
- Question Bank For Summative Assesment-IIDocument19 pagesQuestion Bank For Summative Assesment-IIApex InstituteNo ratings yet
- Purposes and Methods of Research in Mathematics EdDocument10 pagesPurposes and Methods of Research in Mathematics Edhassankanj134No ratings yet
- Electrostatic Forces, Charges and FieldsDocument9 pagesElectrostatic Forces, Charges and FieldsDeepak PachauriNo ratings yet
- Lesson 1 To 4 IcDocument66 pagesLesson 1 To 4 IcscientistgenerosoNo ratings yet
- Ch12 SolutionsDocument12 pagesCh12 SolutionsJoe ReynoldsNo ratings yet
- CH 6Document16 pagesCH 6Iratechaos100% (2)
- Feb. 2022 Ptu PaperDocument2 pagesFeb. 2022 Ptu Paperthakralshivam21No ratings yet
- U-2StructureofAtom1 89626Document23 pagesU-2StructureofAtom1 89626Anirudh AshrithNo ratings yet
- 9J Quick QuizDocument2 pages9J Quick QuizOneth RajapakseNo ratings yet
- NMR and EsrDocument52 pagesNMR and EsrSenthil Sethu FvfcNo ratings yet
- Special RelativityDocument17 pagesSpecial RelativityDiego M GranziolNo ratings yet