Professional Documents
Culture Documents
Smith 4th Ed Chap 9 Alcohols, Ethers and Epoxides (Fall 2013)
Smith 4th Ed Chap 9 Alcohols, Ethers and Epoxides (Fall 2013)
Uploaded by
Clifford Philogene0 ratings0% found this document useful (0 votes)
121 views47 pages1. The document summarizes key concepts about alcohols from Chapter 9 including their structure, nomenclature, physical properties, preparation from alkenes, and common reactions.
2. Alcohols can be prepared through acid-catalyzed hydration of alkenes or hydroboration-oxidation, and they undergo important reactions like dehydration back to alkenes, and conversion to alkyl halides and alkyl sulfonates.
3. Carbocation intermediates play a key role in the mechanisms of alcohol reactions like dehydration and conversion to halides, and carbocation rearrangements must sometimes be considered.
Original Description:
Organic chemistry
Copyright
© © All Rights Reserved
Available Formats
PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this Document1. The document summarizes key concepts about alcohols from Chapter 9 including their structure, nomenclature, physical properties, preparation from alkenes, and common reactions.
2. Alcohols can be prepared through acid-catalyzed hydration of alkenes or hydroboration-oxidation, and they undergo important reactions like dehydration back to alkenes, and conversion to alkyl halides and alkyl sulfonates.
3. Carbocation intermediates play a key role in the mechanisms of alcohol reactions like dehydration and conversion to halides, and carbocation rearrangements must sometimes be considered.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
Download as pdf or txt
0 ratings0% found this document useful (0 votes)
121 views47 pagesSmith 4th Ed Chap 9 Alcohols, Ethers and Epoxides (Fall 2013)
Smith 4th Ed Chap 9 Alcohols, Ethers and Epoxides (Fall 2013)
Uploaded by
Clifford Philogene1. The document summarizes key concepts about alcohols from Chapter 9 including their structure, nomenclature, physical properties, preparation from alkenes, and common reactions.
2. Alcohols can be prepared through acid-catalyzed hydration of alkenes or hydroboration-oxidation, and they undergo important reactions like dehydration back to alkenes, and conversion to alkyl halides and alkyl sulfonates.
3. Carbocation intermediates play a key role in the mechanisms of alcohol reactions like dehydration and conversion to halides, and carbocation rearrangements must sometimes be considered.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
Download as pdf or txt
You are on page 1of 47
1 1
(Chap. 9) ALCOHOLS, ETHERS & EPOXIDES
ALCOHOLS
structure, nomenclature, & some physical properties (9.1, 9.2, 9.3A, 9.5)
preparation (9.6 & review of chemistry from Ch. 10 [alkene reactions])
(A) acid-catalyzed hydration of C=C's (10.12)
(B) hydroboration-oxidation of C=C's (10.16)
reactions (9.7A)
(A) dehydration to afford C=Cs (9.8, 9.9, 9.10)
(B) conversion to alkyl halides (9.11, 9.12) & alkyl sulfonates (9.13)
R = alkyl, aryl
(refer to textbook)
common abbrev. = ROH
2
primary secondary tertiary
OH
this
hydroxyl is
part of a
hemiacetal
FG,
described in
aldehyde &
ketone
chapter
Note that
hydroxyl
groups can
be part of
larger FGs
(e.g., as in
carboxylic
acids)
Classify the alcohols indicated below
Chap. 9
phenol FG; NOT ROH FG
3
Chap. 9
4
very weak acids very strong acids
Chap. 9
5
Write the balanced equation for this reaction and provide a correct mechanism
balanced equation
a correct mechanism:
Chap. 9
IN ORGANIC RXN MECHANISMS
YOUVE SEEN THUS FAR,
CARBOCATION INTERMEDIATES
CAN FORM FROM C=C, R-X, R-OH:
carbocations (formation) - mini review
Clearly show three reactions that lead to formation
of this carbocation mechanistic intermediate:
one from a C=C
one from an R-X
one from an R-OH
IN ORGANIC RXN MECHANISMS,
CARBOCATION INTERMEDIATES HAVE
THREE POSSIBLE FATES:
(1) REACTION WITH A NUCLEOPHILE:
anionic (:X
-
) or neutral (e.g., H
2
O:)
(2) LOSS OF AN a-PROTON TO FORM A C=C
(3) REARRANGEMENT TO ANOTHER CARBOCATION
OF SIMILAR OR GREATER STABILITY
:
unsymm.
substituted
C=C
2
o
carbocation
3
o
carbocation (favored!)
hydride/alkyl migration (shift)
to form another cation of equal
or greater (not lesser) stability
a different 3
o
carbocation
=
=
overall: an alkene migration
(1)
(2)
(3)
(1)
(2)
(3)
and/or
and/or
X
X
carbocations (reactions) - mini review
8
ALCOHOLS preparation: (a) acid-catalyzed hydration of ALKENES
(b) hydroboration-oxidation of ALKENES
Markovnikov
regioselectivity
non-Markovnikov
regioselectivity
complementary
methods
sample reactions
overall transformation = HYDRATION
elements of water added to C=C
What exactly is hydration...
and how does it differ from
hydrolysis I always get
those two confused!
(2 reactions; chap. 10)
Chap. 9
NOTE: appreciate the difference between the terms hydration & hydrolysis
Hydration = combining with the elements of H
2
O (an addition rxn):
Hydrolysis = cleavage by H
2
O, w/the elements of H
2
O shared between
the fragments (a substitution rxn):
H and OH DONT literally need to come from a single molecule of H-O-H; they often come from
different sources (e.g., as youll see when examining rxn mechanisms)
In alkene hydration, the overall transformation is addition of the elements of water (H & OH) to C=C
In hydrolysis, the overall transformation involves the substitution of the elements of water (H & OH)
Youll encounter many
examples of
hydrolysis reactions
in orgo II
9
Note:
10
ALCOHOLS preparation: (a) acid-catalyzed hydration of ALKENES
(b) hydroboration-oxidation of ALKENES
Markovnikov
regioselectivity
non-Markovnikov
regioselectivity
complementary
methods
sample reactions
overall transformation = HYDRATION
elements of water added to C=C
H
+
(E
+
)
In both cases,
electrophile ends up
on least substituted
carbon
alcohol
regioisomers
( E
+
)
(2 reactions; chap 10)
Chap. 9
(review) mechanistic
explanation of
regioselectivity:
11
ALCOHOLS reactions: (a) dehydration to ALKENES
(b) conversion to ALKYL HALIDES
& ALKYL SULFONATES
(a) acid-catalyzed dehydration
The mechanism of alcohol DEHYDRATION is the exact reverse of the mechanism of alkene HYDRATION - same mechanistic
intermediates but arrows flow in opposite direction!...this is true for equilibrium reactions; e.g.:
Typical acids used for alcohol
dehydration are H
2
SO
4
or
p-toluenesulfonic acid (TsOH)
Use Le Chteliers principle to drive equilibrium to left or right; e.g., remove alkene by distillation as it is formed, thus driving
the equilibrium to the right
(1 reaction)
(4 reactions)
Chap. 9
12
Often, a less stable carbocation will be converted into either a more stable or equally
stable carbocation by a shift of a hydrogen or an alkyl group (called a rearrangement)
Because the migrating group in a 1,2-shift moves with two bonding electrons, the
carbon it leaves behind now has only three bonds (six electrons), giving it a net
positive (+) charge
Recall: carbocation rearrangements
Note: carbocation rearrangements lead to carbocations of equal or greater
stability NOT the other way around!
2
o
3
o
2
o
2
o
Chap. 9
complete conversion
partial conversion
13
Be aware that unintentional cationic rearrangements (alkyl & hydride shifts) can occur during acid-cat. dehydration of alcohols
Example
(practice
mechanism
problem):
Q: how can you tell that a rearr. has occurred? A: focus on carbon skeleton & look at the position of alkyl groups on longest chain
ALCOHOLS reactions:
Chap. 9
14
Dehydration of 1
o
alcohols cant occur via an E1 mech. since 1 carbocations are highly unstable;
thus, 1 alcohols undergo dehydration via an E2 mech.
E2 Dehydration of 1
o
Alcohols
ALCOHOLS reactions:
a 1
o
carbon
E2 mech.
(avoids a
high-energy
1
o
carbocation
formed via an E1
mechanism!)
NO!! NO!!
_____
Chap. 9
15
Some organic compounds decompose in the presence of strong acid, so other methods have been developed to
convert alcohols to alkenes.
A common method uses phosphorus oxychloride (POCl
3
) and pyridine (an amine base) in place of H
2
SO
4
or TsOH
POCl
3
converts OH into a good leaving group
Dehydration then proceeds by an E2 mechanism
Dehydration of Alcohols Using POCl
3
: same result as strong acid but MILDER conditions
ALCOHOLS reactions:
OH
H
H
2
O
Chap. 9
16
ALCOHOLS reactions:
E2
S
N
2
Chap. 9
17
(b) conversion of ALCOHOLS to ALKYL HALIDES
ALCOHOLS reactions:
poor LG in S
N
reactions: OH
-
is conj. base of a weak acid (HOH);
thus, must convert hydroxyl (OH) group into a good LG before an
S
N
reaction can occur!
3
o
ROH
2
o
ROH
1
o
ROH
H
+
X
-
H
+
X
-
H
+
X
-
Againin mechanisms involving carbocations (S
N
1/E1),
unintended rearrangements can occur:
see p 348 in your textbook for mechanism
Chap. 9
use of HX (X = Cl, Br, I) (review, chap. 7)
18
ALCOHOLS reactions:
What alkyl halide products would be afforded in the following reactions? Include all stereoisomers where appropriate,
and mechanistically explain your answers.
(a)
(c)
(b)
(d)
Show how to convert 1-propanol to each of the synthetic targets shown below. Use any necessary reactants or
reagents, show the product of each synthetic step, and indicate any necessary reaction conditions.
(i)
(ii)
(iii)
(iv) (v)
(vi)
Practice synthesis/reaction problems
(S)
(R)
Chap. 9
19
ALCOHOLS reactions:
Propose reasonable mechanisms for the following reactions. Use arrows correctly to indicate bond making and bond breaking (i.e.,
movement of electrons), show all mechanistic intermediates, and indicate any formal charges on atoms.
(a) (b)
(c)
(d)
(e) (f)
(g)
(h)
(i)
OMG!... sequential cationic
rearrangements must be involved
in this problem!!
Practice mechanism problems
Chap. 9
challenge problem
20
1
o
& 2 R-OH are converted to R-Cl using SOCl
2
(thionyl chloride) &
to R-Br using PBr
3
(phosphorus tribromide)
both reagents convert OH into a good leaving group in situ (directly in the
reaction mixture) and provide the nucleophile (either Cl or Br) to displace the
leaving group via an S
N
2 reaction
since NO strong acid (HX) is present, use of SOCl
2
& PBr
3
avoids:
unwanted cationic rearrangements of the carbon skeleton
unwanted reaction of other acid-reactive FGs in the substrate molecule
conversion of alcohols to ALKYL HALIDES with SOCl
2
and PBr
3
ALCOHOLS reactions:
Chap. 9
R-OH R-X
SOCl
2
or PBr
3
X = Cl (SOCl
2
)
Br (PBr
3
)
(1
o
& 2) (1
o
& 2)
(S
N
2 reaction)
Conversion of 1
o
or 2
o
alcohols to alkyl chlorides with SOCl
2
1
o
alcohol
2
o
alcohol
NOTE: SAME result is obtained with HCl, but SOCl
2
avoids use of HCl & is S
N
2 rxn
S
N
2
S
N
2
21
Chap. 9
22
Conversion of 1
o
or 2
o
alcohols to alkyl bromides with PBr
3
ALCOHOLS reactions:
1
o
alcohol
2
o
alcohol
S
N
2
S
N
2
Chap. 9
23
Indicate an appropriate reagent for each of the following transformations:
(a)
(b)
(c)
(d)
(e)
ALCOHOLS reactions: Practice synthesis/reaction problems
Chap. 9
24
use for chemoselective &
stereoselective conversion
of ROH RX
use for chemoselective &
stereoselective conversion
of ROH RX
ALCOHOLS reactions:
HCl
HBr
Hl
SOCl
2
PBr
3
Chap. 9
25
Alcohols can be converted into ALKYL TOSYLATES: R-OTs acts like R-X; X = halide
An alkyl tosylate is composed of two parts:
1. alkyl group R, derived from R-OH
2. the tosylate group (short for p-toluenesulfonate), which is a v. good LG
A tosyl group, CH
3
C
6
H
4
SO
2
, is abbreviated Ts, so an alkyl tosylate becomes R-OTs
formation & reaction of ALKYL TOSYLATES;
tosylate (OTs
-
) as a great leaving group (LG)
ALCOHOLS reactions:
O O
Chap. 9
Why?
26
Tosylate is a good LG because its conjugate acid, p-toluenesulfonic acid (CH
3
C
6
H
4
SO
3
H, TsOH), is a strong
acid (pK
a
= ~ 3)thus, its conjugate base (CH
3
C
6
H
4
SO
3
-
, TsO
-
) is a weak base (i.e., happy anion, good LG)
formation and use of tosylates
ALCOHOLS reactions:
Stereochemistry of tosylate formation
(2S)-2-Butanol is converted to its tosylate with retention of configuration at the stereogenic center
since the C-O bond of the alcohol is not broken in the process
Chap. 9
27
formation and use of tosylates
ALCOHOLS reactions:
Step [1], formation of tosylate, proceeds with retention of configuration at a
stereogenic center
Step [2] is an S
N
2 reaction, so it proceeds with inversion of configuration
because the nucleophile attacks from the backside
Overall there is a net inversion of configuration at a stereogenic center
Overall Inversion of Stereochemistry in REACTION of alkyl tosylates
Br:
-
Br
the HX-free R-OH R-X conversion shown above can also be done by direct rxn with PBr
3
or SOCl
2
however, alkyl tosylates are useful since they can react with OTHER nucleophiles besides halides!...
Chap. 9
Nu:
-
= halide, CN
-
, N
3
-
, etc.
28
Because alkyl tosylates have good leaving groups, they undergo both
nucleophilic substitution and elimination, exactly as alkyl halides do
Generally, alkyl tosylates are treated with strong nucleophiles and bases,
in which case the mechanism of substitution is S
N
2,
and the mechanism of elimination is E2
formation and use of tosylates
ALCOHOLS reactions:
Chap. 9
29
Note: since R-OTs behaves like R-X, S
N
1 & E1 rxns are also possible.
Provide a mechanism to explain the following rxn:
formation and use of tosylates
ALCOHOLS reactions:
Chap. 9
30
formation and use of tosylates
ALCOHOLS reactions:
(a) Provide the products of the following reactions.
(b) Propose reasonable mechanisms for the following reactions.
(i)
(ii)
Practice problems
Chap. 9
31
Summary of ALCOHOL preparation & reactions discussed in lecture
I recommend that you come up with your own
FG preparation/reaction summaries (like the one above)
for each new FG discussed in lecture
its one good way to (i) collect your thoughts,
(ii) organize the material for review & for USE IN SOLVING PROBLEMS
(iii) learn how FGs can be converted to one another
(important for solving synthesis problems)
Chap. 9
32
R = alkyl, aryl
ETHERS
structure & nomenclature (9.1, 9.2, 9.3B, 9.5)
preparation (9.6)
(A) Williamson ether synthesis (p 334)
(B) acid-cat. add'n of ROH's to C=C's (10.12; p 395)
reactions cleavage by conc. HX (9.14)
(Chap. 9) ALCOHOLS, ETHERS & EPOXIDES
Chap. 9
(refer to textbook)
common abbrev. = ROR
33
ETHERS preparation:
complementary
methods
(a) Williamson ether synthesis (S
N
2 rxn)
(b) acid-cat. addition of ROH to alkenes (C=C elec. addn rxn)
our synthetic
target molecule
(a) Williamson ether synth. (S
N
2 rxn): used to prepare O-C(1
o
) bond: must use 1
o
R-X (X = halide, OTs) as E
+
1
o
2
o
(2 reactions)
Chap. 9
34
ETHERS preparation:
complementary
methods
(a) Williamson ether synthesis (S
N
2 rxn)
(b) acid-cat. addition of ROH to alkenes (C=C elec. addn rxn)
our synthetic
target molecule
(b) acid-cat. addn of ROH to C=C: used to prepare O-C(2
o
or 3
o
) bond; its an alkene electrophilic addn rxn
analogous to alkene hydration!...use ROH instead of HOH in the rxn.
1
o
2
o
(2 reactions)
Chap. 9
35
cleavage of ethers with strong acids such as conc. HBr and conc. HI
via successive S
N
rxns
ETHERS reactions:
example:
a proposed mechanism:
(1 reaction)
Chap. 9
36
R = alkyl, aryl
EPOXIDES
structure, nomenclature & general reactivity (9.1, 9.2, 9.3C, 9.5)
preparation (9.6; 12.8)
(A) intramolecular Williamson ether synthesis using halohydrins (10.15)
(later well see a more straightforward & common method described in chap. 12.8)
reactions (9.15)
(A) nucleophilic ring opening with strong nucleophiles under basic/neutral conditions
(B) nucleophilic ring opening under acidic conditions
(Chap. 9) ALCOHOLS, ETHERS & EPOXIDES
Chap. 9
(refer to textbook)
37
(intramolecular Williamson ether synthesis)
(A) Intramolecular Williamson ether synthesis using VICINAL HALOHYDRINS
EPOXIDES preparation:
Chap. 9
38
Epoxide structure affects its chemical reactivity
Chap. 9
Nu: nuc. attack leads
to ring opening
(A) NEUTRAL/BASIC rxn conditions (w/strong Nuc.)
(B) ACIDIC rxn conditions
Note:
all epoxide rxns you will see involve ring opening
but rxn outcomes (& mechanisms) change under diff. rxn conditions (A) vs. (B)
39
EPOXIDES reactions:
epoxide as E
+
Well examine nucleophilic ring opening under:
Chap. 9
think of the oxygen as
a LG in an S
N
rxn
EPOXIDES reactions:
please note:
rxn of unsymmetrically substituted epoxides (e.g., monosubstituted epoxides)
with nucleophiles under any conditions can potentially afford one or both
constitutional isomers (regioisomers) of alcohol product shown below:
new
bond
new
bond
40
Chap. 9
a monosubstituted
epoxide
constitutional isomers
(regioisomers)
of alcohol products
41
(A) Nucleophilic ring opening under NEUTRAL/BASIC conditions w/strong nucleophiles:
S
N
2 mechanism (least hindered carbon is attacked preferentially)
*
*
= asymm. (stereogenic )center:
optically active epoxide would afford
optically active product w/retention of stereochem. at asymm. center since rxn occurs at other epoxide carbon
*
think of the oxygen as a
LG in an S
N
rxn
*
Chap. 9
EPOXIDES reactions:
42
Note: achiral (optically inactive) reactants yield optically inactive products even if products are chiral
Mechanistic explanation for racemic mixture:
at equal rates
50% A : 50% B
Chap. 9
43
Chap. 9
(a) Provide the major organic products of the following reactions.
EPOXIDES reactions:
(i)
(ii)
44
Chap. 9
(b) Provide a reasonable mechanism for the following reaction. Use arrows correctly to indicate
bond making and bond breaking, show all mechanistic intermediates and clearly indicate any
charged atoms with appropriate symbols.
EPOXIDES reactions:
new bond
45
(B) Nucleophilic ring opening under
ACIDIC conditions nuc. attack @ MOST substituted carbon (S
N
1-like)
inversion of configuration @ carbon being attacked (S
N
2-like)
rxn proceeds via a borderline S
N
2 mech.
Mechanism proposed to explain the above results:
Chap. 9
46
Regioselectivity of epoxide ring opening w/nucleophiles - SUMMARY
EPOXIDES reactions:
examples from the chemical literature:
ACIDIC
conditions
BASIC
conditions
Long, F. A., Pritchard, J. G. J. Am. Chem. Soc. 1956, 78, 2663
BASIC/NEUTRAL
conditions
ACIDIC conditions
Chap. 9
47
(1) Prepare compounds (i)-(vi) from 4-methyl-1-pentene as your only organic starting reactant. Use any
necessary reagents & as many molecules of 4-methyl-1-pentene as you need. Show the product of each
synthetic step (do NOT include any mechanistic details; i.e., arrow pushing, cationic/anionic intermediates, etc.).
(i)
(ii)
(iii) (iv)
(vi) (v)
(2) The epoxide ring can also open by an acid-catalyzed rearrangement reaction, like the one shown below.
Propose a reasonable mechanism for the following rearrangement reaction. Use arrows correctly to indicate
bond making and bond breaking (i.e., movement of electrons), show all mechanistic intermediates, and indicate
any formal charges on atoms.
Ether & epoxide practice problems:
Chap. 9
You might also like
- Functional Group Transformation NotebookDocument78 pagesFunctional Group Transformation NotebookchedhedNo ratings yet
- Experiment No: 6 Expirement Title: Preparation of 2-Hexanol From 1-HexeneDocument5 pagesExperiment No: 6 Expirement Title: Preparation of 2-Hexanol From 1-HexeneMoemedi Moshakga40% (5)
- ch11 PDFDocument96 pagesch11 PDFMau BaraquelNo ratings yet
- Production of Ethanol by MolassesDocument154 pagesProduction of Ethanol by MolassesSAIMA ZAMEERNo ratings yet
- DAT Bootcamp - Reaction Summary Sheet PDFDocument28 pagesDAT Bootcamp - Reaction Summary Sheet PDFbbNo ratings yet
- Web - Chem.ucsb - Edu - Zhang - ZLM-chapter 10-AlcoholDocument4 pagesWeb - Chem.ucsb - Edu - Zhang - ZLM-chapter 10-AlcoholKrisna PamungkasNo ratings yet
- Preparation and Reaction of Carboxylic AcidsDocument6 pagesPreparation and Reaction of Carboxylic AcidsIndhumathiNo ratings yet
- 02b PDFDocument19 pages02b PDFSyed Ali Akbar BokhariNo ratings yet
- CH 18Document32 pagesCH 18Dimas MitraNo ratings yet
- CH22Document23 pagesCH22David LiNo ratings yet
- Pdf-Haloalkanes and HaloarenesDocument159 pagesPdf-Haloalkanes and HaloarenesOmkar Singh Shekhawat100% (2)
- Haloalkanes & HaloarenesDocument10 pagesHaloalkanes & Haloarenesakshatshukla2021No ratings yet
- HaloalkanesDocument13 pagesHaloalkanesChingYan TanNo ratings yet
- Alkene Alkyne PDFDocument45 pagesAlkene Alkyne PDFKartikeya AryaNo ratings yet
- Alcohols, Ethers and PhenolsDocument45 pagesAlcohols, Ethers and Phenolsshivam08No ratings yet
- ALKANOLSDocument25 pagesALKANOLSKoki KingNo ratings yet
- Chemistry of The Alcohols Alcohols: Monohydric C H OHDocument23 pagesChemistry of The Alcohols Alcohols: Monohydric C H OHAyodele AdeyonuNo ratings yet
- Chem 331Document11 pagesChem 331Satya KamNo ratings yet
- 10.4 Halogenoalkanes 10.4.1 Introduction To Halogenoalkane ReactivityDocument16 pages10.4 Halogenoalkanes 10.4.1 Introduction To Halogenoalkane Reactivitydiyaray100% (1)
- Useful Reactions PDFDocument8 pagesUseful Reactions PDFagusrimbombanteNo ratings yet
- Catalytic Carbon-Carbon Bond FormationDocument30 pagesCatalytic Carbon-Carbon Bond Formationjshine868100% (1)
- 3 Functional Groups Alcohols:: Lecture Notes Chem 51B S. KingDocument27 pages3 Functional Groups Alcohols:: Lecture Notes Chem 51B S. KingHuấnĐìnhNguyễnNo ratings yet
- C10K Carbonyl Chemistry EmailDocument37 pagesC10K Carbonyl Chemistry EmailMatthew яeject'z BennettNo ratings yet
- Xii Ch10 Haloalkane and HaloarenesDocument4 pagesXii Ch10 Haloalkane and HaloarenesYash RajNo ratings yet
- Organic Synthesis Via Enolates BSC III CH IVDocument10 pagesOrganic Synthesis Via Enolates BSC III CH IVSanjay ShirodkarNo ratings yet
- Expt 5 - Reactions and SolubilityDocument15 pagesExpt 5 - Reactions and SolubilitySangeeth George0% (2)
- Alcohols, Diols, TriolsDocument32 pagesAlcohols, Diols, TriolsShivam GuptaNo ratings yet
- Nucleophilic Substitution and EliminationDocument8 pagesNucleophilic Substitution and EliminationNurul HidayahNo ratings yet
- Halogen Derivative of AlkaneDocument29 pagesHalogen Derivative of AlkaneDeepti Kaskar60% (5)
- Alcohols Contain A Hydroxyl Group (OH Group) Bonded To An SPDocument2 pagesAlcohols Contain A Hydroxyl Group (OH Group) Bonded To An SPbaskhemNo ratings yet
- L.S.T. Leung Chik Wai Memorial School F.6 Chemistry Chapter 34: Hydroxy-Compounds 羥基化合物 chapt. 34: p.1Document21 pagesL.S.T. Leung Chik Wai Memorial School F.6 Chemistry Chapter 34: Hydroxy-Compounds 羥基化合物 chapt. 34: p.1IEyra ShaHeraNo ratings yet
- The Reaction Gives Pure Alkyl HalidesDocument8 pagesThe Reaction Gives Pure Alkyl HalidesMohammed IliasNo ratings yet
- 12 Chemistry Keypoints Revision Questions Chapter 10 PDFDocument13 pages12 Chemistry Keypoints Revision Questions Chapter 10 PDFSahil Kalra100% (1)
- Chapter 5 Alkyl HalidesDocument32 pagesChapter 5 Alkyl HalidesMohd HanafiahNo ratings yet
- Carbonyl CompoundsDocument29 pagesCarbonyl CompoundsKarthik SharmaNo ratings yet
- Carbonyl CompoundsDocument29 pagesCarbonyl CompoundsKarthik SharmaNo ratings yet
- Note HaloalkanesDocument7 pagesNote HaloalkanesNabin JoshiNo ratings yet
- Hydrocarbons (Hints)Document2 pagesHydrocarbons (Hints)hchawla421No ratings yet
- 133 AcylationDocument4 pages133 AcylationAnonymous vRpzQ2BLNo ratings yet
- Aldehydes and KetonesDocument9 pagesAldehydes and KetonesCamille AdleNo ratings yet
- Aldehydes and Ketones For IitjeeDocument65 pagesAldehydes and Ketones For Iitjeevarundhall1994No ratings yet
- Reaction of Aldehydes & Ketones-2Document30 pagesReaction of Aldehydes & Ketones-2MGoyalNo ratings yet
- Hand-Out: Chemistry Chapter 4: Haloalkanes & HaloarenesDocument11 pagesHand-Out: Chemistry Chapter 4: Haloalkanes & HaloarenesLuisgarciaBerlangaNo ratings yet
- Revised Organic ChemistryDocument90 pagesRevised Organic ChemistryMinh TieuNo ratings yet
- Aldehyde and KetonesDocument45 pagesAldehyde and KetonesShivam GuptaNo ratings yet
- Li 2014Document4 pagesLi 2014Khaidir WijayaNo ratings yet
- Halo Alkanes & HaloarenesDocument30 pagesHalo Alkanes & HaloarenesEarNo ratings yet
- Lecture CycloalkanesDocument21 pagesLecture CycloalkanesDain GutierrezNo ratings yet
- Alcoholes 3Document47 pagesAlcoholes 3Дана ЧилибаеваNo ratings yet
- Carboxylic AcidsDocument3 pagesCarboxylic AcidsDian Agus SetyawatiNo ratings yet
- F334 - What's in A Medicine?Document11 pagesF334 - What's in A Medicine?Becky Tenney100% (1)
- Organic Chemistry Ii (Och221T) : Chemical Engineering Class 2017BDocument61 pagesOrganic Chemistry Ii (Och221T) : Chemical Engineering Class 2017BSiphelele SimelaneNo ratings yet
- SXHS XII (CHEM) P.T-2 Imp Questions 2023Document7 pagesSXHS XII (CHEM) P.T-2 Imp Questions 2023sampritmodiNo ratings yet
- CarbonylsDocument7 pagesCarbonylsThanadet PhongchompornNo ratings yet
- Jee-Aldehyde Ketone & Carboxylic Acid Chem XiiDocument72 pagesJee-Aldehyde Ketone & Carboxylic Acid Chem Xiiswastik aroraNo ratings yet
- Organic Chemistry H2 Questions MCQDocument15 pagesOrganic Chemistry H2 Questions MCQkitonium100% (1)
- SCHR Au Zer 1967Document9 pagesSCHR Au Zer 1967Stephanie AvilaNo ratings yet
- Reactions of Alkenes&Alkynes - SummaryDocument37 pagesReactions of Alkenes&Alkynes - SummaryJerome JavierNo ratings yet
- Organic ReactionsDocument21 pagesOrganic Reactionschiomamoronu08No ratings yet
- Reactions of AlcoholDocument20 pagesReactions of AlcoholHaslimi HassanNo ratings yet
- Ligand Platforms in Homogenous Catalytic Reactions with Metals: Practice and Applications for Green Organic TransformationsFrom EverandLigand Platforms in Homogenous Catalytic Reactions with Metals: Practice and Applications for Green Organic TransformationsNo ratings yet
- Petrochemical IndustryDocument30 pagesPetrochemical Industrybuhayche14No ratings yet
- Akiya2002 - Roles of Water For Chemical Reactions in High-Temperature WaterDocument26 pagesAkiya2002 - Roles of Water For Chemical Reactions in High-Temperature Waterfarida_crNo ratings yet
- 2.10 HWDocument5 pages2.10 HWChong BengNo ratings yet
- SYBSc Paper I, Unit-III, AlcoholDocument74 pagesSYBSc Paper I, Unit-III, Alcoholmyebooks.pramodNo ratings yet
- Chapter 08 MergedDocument38 pagesChapter 08 MergedreemNo ratings yet
- Ethanol ProductionDocument13 pagesEthanol ProductionRhea MambaNo ratings yet
- 4 Alkenes Addition ReactionsDocument44 pages4 Alkenes Addition ReactionsDyrus WongNo ratings yet
- Sim, Y - Dehydration of CyclohexanolDocument6 pagesSim, Y - Dehydration of CyclohexanolYura SimNo ratings yet
- Sem 2 Unit 2Document81 pagesSem 2 Unit 2ArvnabhNo ratings yet
- Hydration of A Terminal AlkyneDocument8 pagesHydration of A Terminal AlkyneMunna PatelNo ratings yet
- Norma Astm - PVRDocument72 pagesNorma Astm - PVRHenrique SouzaNo ratings yet
- EtanolDocument57 pagesEtanolexe241293No ratings yet
- Class 12th Chemistry Chapter 11 (Alcohols, Phenols and Ethers) Important Solved QuestionsDocument27 pagesClass 12th Chemistry Chapter 11 (Alcohols, Phenols and Ethers) Important Solved QuestionsSC VARMANo ratings yet
- Module 9: Alcohols & Phenols: Pharmaceutical Organic Chemistry (Lecture)Document6 pagesModule 9: Alcohols & Phenols: Pharmaceutical Organic Chemistry (Lecture)Abigail P. ARANGGANo ratings yet
- McMurry Chapter 9Document46 pagesMcMurry Chapter 9Christine TapawanNo ratings yet
- Sample Paper Chemistry Theory (043) MM:70 Time: 3 HoursDocument10 pagesSample Paper Chemistry Theory (043) MM:70 Time: 3 HoursJabez Jeena100% (1)
- Alkynes: An Introduction To Organic Synthesis: (Ref: Mcmurry'S Organic Chemistry, 7 Edition)Document24 pagesAlkynes: An Introduction To Organic Synthesis: (Ref: Mcmurry'S Organic Chemistry, 7 Edition)Tristan BadillaNo ratings yet
- Chapter 11 - AlkynesDocument48 pagesChapter 11 - AlkynesTempuraNo ratings yet
- Drying Agent Selection Guide: (Back To The Chemistry Archive)Document2 pagesDrying Agent Selection Guide: (Back To The Chemistry Archive)ŠĭlệncěIšmyPŕIdệNo ratings yet
- 4.2 Hydration of AlkenesDocument33 pages4.2 Hydration of AlkenesDawit BirhanuNo ratings yet
- Alkenes ReactionsDocument69 pagesAlkenes ReactionsAhmad SayyedahmadNo ratings yet
- Alcohols Phenols and EthersDocument81 pagesAlcohols Phenols and Ethersjjprakash82chemNo ratings yet
- Lec 6C Alkyne Structure and ReactivityDocument37 pagesLec 6C Alkyne Structure and Reactivityseanroi.villas.lawNo ratings yet
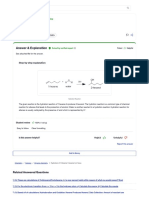
- (Solved) Hydration of 1-Hexene 1-Hexene To 2 Hexanol Equation Write The Equation of The Reaction - Course HeroDocument2 pages(Solved) Hydration of 1-Hexene 1-Hexene To 2 Hexanol Equation Write The Equation of The Reaction - Course HeroJdkrkejNo ratings yet
- Lecture 3 - Cement HydrationDocument16 pagesLecture 3 - Cement HydrationAshnaBeeslallNo ratings yet
- Chapter 10 - Electrophilic Additions To AlkenesDocument30 pagesChapter 10 - Electrophilic Additions To AlkenesBianca-Rebeca PetreNo ratings yet