Professional Documents
Culture Documents
0 ratings0% found this document useful (0 votes)
15 viewsSharath2013 PDF
Sharath2013 PDF
Uploaded by
Jessica DukeDiabetic Retinopathy (DR) is the major cause of blindness caused by the damage to the blood vessels in the retina from diabetes. Exudates are fatty deposits on the retina which appear as yellowish regions in fundus image. To evaluate the new method, we examined 158 fundus images, including 84 abnormal images and 74 normal images.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
You might also like
- Reinhold Ebertin - Transits - What Days Favor You - American Federation of Astrologers (2015)Document128 pagesReinhold Ebertin - Transits - What Days Favor You - American Federation of Astrologers (2015)ukoszapavlinje100% (24)
- Nugroho2017 PDFDocument4 pagesNugroho2017 PDFglaretsubinNo ratings yet
- Asilomar Bhalerao Anand SaravananDocument3 pagesAsilomar Bhalerao Anand SaravanansosoNo ratings yet
- Orthodontic Application of Color Image Addition To Visualize Differences Between Sequential RadiographsDocument6 pagesOrthodontic Application of Color Image Addition To Visualize Differences Between Sequential RadiographsAmal AbdelwahedNo ratings yet
- Diabetic Retinopathy Detection Using Machine LearningDocument8 pagesDiabetic Retinopathy Detection Using Machine LearningGanavi R PatelNo ratings yet
- Diabetic Retinopathy Detection-IRO-Journals-4 1 3Document8 pagesDiabetic Retinopathy Detection-IRO-Journals-4 1 3Rizkya Putri MaulanaNo ratings yet
- A Skin Tone Detection Algorithm For An Adaptive Approach To SteganographyDocument23 pagesA Skin Tone Detection Algorithm For An Adaptive Approach To SteganographyLeela PavanNo ratings yet
- Review On Brain Tumour Detection Using Digital Image ProcessingDocument7 pagesReview On Brain Tumour Detection Using Digital Image ProcessingShivangi33No ratings yet
- Sopharak ExudateFeaturesDocument4 pagesSopharak ExudateFeaturesHassan FaouziNo ratings yet
- EMBC2017 - Exudate Detection For Diabetic Retinopathy With Convolutional Neural NetworksDocument4 pagesEMBC2017 - Exudate Detection For Diabetic Retinopathy With Convolutional Neural Networks1DA19CS156 Shubha SNo ratings yet
- Adaptive Neuro-Fuzzy Inference System Approach For The Automatic Screening of Diabetic Retinopathy in Fundus ImagesDocument7 pagesAdaptive Neuro-Fuzzy Inference System Approach For The Automatic Screening of Diabetic Retinopathy in Fundus ImagesadaneNo ratings yet
- Detection of Diabetic Retinopathy WordDocument6 pagesDetection of Diabetic Retinopathy WordThe MinisterNo ratings yet
- Automatic Vessel Extraction With Combined Bottom Hat and Match FilterDocument5 pagesAutomatic Vessel Extraction With Combined Bottom Hat and Match FilterBahareh Bozorg ChamiNo ratings yet
- Skin Color SegDocument6 pagesSkin Color SegSachin MoreNo ratings yet
- Iris Diagnosis - A Quantitative Non-Invasive Tool Fordiabetes DetectionDocument7 pagesIris Diagnosis - A Quantitative Non-Invasive Tool Fordiabetes Detectionkrishna mohan pandeyNo ratings yet
- Automated Skin Lesion Segmentation Using K-Means Clustering From Digital Dermoscopic ImagesDocument6 pagesAutomated Skin Lesion Segmentation Using K-Means Clustering From Digital Dermoscopic ImagesArpit GuptaNo ratings yet
- 635 A Stand-Alone MATLAB Application For The Detection of Optic Disc and MaculaDocument5 pages635 A Stand-Alone MATLAB Application For The Detection of Optic Disc and MaculaRama SubramanianNo ratings yet
- 1207 1551 (1) 68Document9 pages1207 1551 (1) 68Mohamad SannanNo ratings yet
- Recognition of Polyps in The Images of Wireless Capsule EndosDocument4 pagesRecognition of Polyps in The Images of Wireless Capsule EndosInternational Journal of Application or Innovation in Engineering & ManagementNo ratings yet
- Madooei2013 Chapter AutomaticDetectionOfBlue-WhiteDocument8 pagesMadooei2013 Chapter AutomaticDetectionOfBlue-WhitemaryNo ratings yet
- Intensity Features Based Classification of Hard Exudates in Retinal ImagesDocument5 pagesIntensity Features Based Classification of Hard Exudates in Retinal ImagesAleenaNo ratings yet
- Bin Ar Ization RetinalDocument12 pagesBin Ar Ization RetinalMohammad Asmat Ullah KhanNo ratings yet
- Color Constancy For Improving Skin DetectionDocument18 pagesColor Constancy For Improving Skin DetectionAI Coordinator - CSC JournalsNo ratings yet
- A Novel and Efficient Feature Extraction Method For Iris RecognitionDocument3 pagesA Novel and Efficient Feature Extraction Method For Iris RecognitionkaleysureshNo ratings yet
- Dokumen Pendukung Karya IlmiahDocument7 pagesDokumen Pendukung Karya IlmiahyaniNo ratings yet
- Diabetic Retinopathy-Early Detection Using Im-Age Processing TechniquesDocument5 pagesDiabetic Retinopathy-Early Detection Using Im-Age Processing Techniqueskumar_9583No ratings yet
- Comparative Study of Segmentation Techniques in Diabetic Retinopathy DetectionDocument4 pagesComparative Study of Segmentation Techniques in Diabetic Retinopathy DetectionAnam FarooqNo ratings yet
- An Intelligent Black Widow Optimization On Image Enhancement With Deep Learning Based Ovarian Tumor Diagnosis ModelDocument16 pagesAn Intelligent Black Widow Optimization On Image Enhancement With Deep Learning Based Ovarian Tumor Diagnosis ModelAgnesNo ratings yet
- An Automatic Segmentation & Detection of Blood Vessels and Optic Disc in Retinal ImagesDocument5 pagesAn Automatic Segmentation & Detection of Blood Vessels and Optic Disc in Retinal ImagesMia AmaliaNo ratings yet
- A New Method For Eye Detection in Color Images: Mohamad Ali Azimi Mohammad RahmatiDocument8 pagesA New Method For Eye Detection in Color Images: Mohamad Ali Azimi Mohammad RahmatiOscar Duque SuarezNo ratings yet
- ObjectiveDocument22 pagesObjectiveKoratala HarshaNo ratings yet
- Detection of Forgeries in Digital Color ImageDocument5 pagesDetection of Forgeries in Digital Color ImagegangasagarjadhavNo ratings yet
- Adaptive Neuro Fuzzy Inference System Assisted Diagnosis of Diabetic Retinopathy From Fundus ImageDocument5 pagesAdaptive Neuro Fuzzy Inference System Assisted Diagnosis of Diabetic Retinopathy From Fundus ImageadaneNo ratings yet
- Automated Quantification of Clinically SignificantDocument5 pagesAutomated Quantification of Clinically SignificantHaleema ahsanNo ratings yet
- Sanders - Samei - 2016 - Patient-Specific Quantification of Image Quality - Method For Measuring Resolution in Clinical CT Images - Medical - PhysicsDocument10 pagesSanders - Samei - 2016 - Patient-Specific Quantification of Image Quality - Method For Measuring Resolution in Clinical CT Images - Medical - PhysicsmartuflashNo ratings yet
- DSA Image Registration Based On Multiscale Gabor Filters and Mutual InformationDocument6 pagesDSA Image Registration Based On Multiscale Gabor Filters and Mutual InformationerdoganaaaNo ratings yet
- A New Hybrid Method For TheDocument8 pagesA New Hybrid Method For ThesipijNo ratings yet
- Automated Tumor Segmentation in MR Brain Image Using Fuzzy C-Means Clustering and Seeded Region MethodologyDocument7 pagesAutomated Tumor Segmentation in MR Brain Image Using Fuzzy C-Means Clustering and Seeded Region MethodologyIAES IJAINo ratings yet
- Sujeet Bhagwat (04) - Aditya Gujar (27) - Swaraj Govindwar (26) - Krishna GatlewarDocument25 pagesSujeet Bhagwat (04) - Aditya Gujar (27) - Swaraj Govindwar (26) - Krishna GatlewarSwaraj GovindwarNo ratings yet
- Robust and Fast Iris Localization Using Contrast Stretching and Leading Edge DetectionDocument7 pagesRobust and Fast Iris Localization Using Contrast Stretching and Leading Edge DetectionInternational Journal of Application or Innovation in Engineering & ManagementNo ratings yet
- Brain Tumer Extraction From Mri Image Using K-Means Clustring TecniqueDocument7 pagesBrain Tumer Extraction From Mri Image Using K-Means Clustring TecniqueRohit AryaNo ratings yet
- Segmentation of Retinal Blood Vessels Using A Novel Clustering Algorithm (RACAL) With A Partial Supervision StrategyDocument13 pagesSegmentation of Retinal Blood Vessels Using A Novel Clustering Algorithm (RACAL) With A Partial Supervision StrategySathiya Moorthy C ANo ratings yet
- Azienda Sanitaria Firenze, Fisica Sanitaria, Firenze, Italy Azienda Sanitaria Firenze, Radioterapia, Firenze, ItalyDocument2 pagesAzienda Sanitaria Firenze, Fisica Sanitaria, Firenze, Italy Azienda Sanitaria Firenze, Radioterapia, Firenze, ItalyLarolina KhoirunnisaaNo ratings yet
- Exudates Detection at Premature Stage in Diabetic RetinopathyDocument6 pagesExudates Detection at Premature Stage in Diabetic RetinopathylambanaveenNo ratings yet
- 10 1 1 85 5278 PDFDocument14 pages10 1 1 85 5278 PDFStaurt AttkinsNo ratings yet
- Conglomeration For Color Image Segmentation of Otsu Method, Median Filter and Adaptive Median FilterDocument8 pagesConglomeration For Color Image Segmentation of Otsu Method, Median Filter and Adaptive Median FilterInternational Journal of Application or Innovation in Engineering & ManagementNo ratings yet
- 2011 - Multi Channel Film Dosimetry With Non Uniformity CorrectionDocument12 pages2011 - Multi Channel Film Dosimetry With Non Uniformity Correctioncnsouza2000No ratings yet
- MIA PresentationDocument8 pagesMIA Presentationahsan puriNo ratings yet
- Design If Iris Based Medical Diagnosis SystemDocument4 pagesDesign If Iris Based Medical Diagnosis SystemDr Vaishali MathapatiNo ratings yet
- A Simple Approach For Relatively Automated Hippocampus Segmentation From Sagittal View of Brain MriDocument8 pagesA Simple Approach For Relatively Automated Hippocampus Segmentation From Sagittal View of Brain MriijbbjournalNo ratings yet
- Identification of Diabetic Retinopathy Using Fuzzy Logic and Back Propagation Neural NetworkDocument8 pagesIdentification of Diabetic Retinopathy Using Fuzzy Logic and Back Propagation Neural Networkpurushothaman sinivasanNo ratings yet
- E7732068519 PDFDocument6 pagesE7732068519 PDFAleenaNo ratings yet
- Estimating Velocity of Nailfold Vessels Flow Using Improvised Deconvolution AlgorithmDocument6 pagesEstimating Velocity of Nailfold Vessels Flow Using Improvised Deconvolution AlgorithmIJERDNo ratings yet
- Spatial Domain Enhancement Techniques For Detection of Lung Tumor From Chest X-Ray ImageDocument10 pagesSpatial Domain Enhancement Techniques For Detection of Lung Tumor From Chest X-Ray ImageInternational Journal of Application or Innovation in Engineering & ManagementNo ratings yet
- Danne WitzDocument6 pagesDanne WitzKhairniNo ratings yet
- A New Technique For Hand Gesture RecognitionDocument4 pagesA New Technique For Hand Gesture RecognitionchfakhtNo ratings yet
- Qno1: Define Spectrophotometer?: WorkingDocument3 pagesQno1: Define Spectrophotometer?: WorkingHussnain MughalNo ratings yet
- Blood Vessel Segmentation in Fundus ImagesDocument5 pagesBlood Vessel Segmentation in Fundus ImagesNaoufal NaoufalNo ratings yet
- Standard and Super-Resolution Bioimaging Data Analysis: A PrimerFrom EverandStandard and Super-Resolution Bioimaging Data Analysis: A PrimerNo ratings yet
- Multi-modality Cardiac Imaging: Processing and AnalysisFrom EverandMulti-modality Cardiac Imaging: Processing and AnalysisPatrick ClarysseNo ratings yet
- Computer Stereo Vision: Exploring Depth Perception in Computer VisionFrom EverandComputer Stereo Vision: Exploring Depth Perception in Computer VisionNo ratings yet
- Circular Building Process Reuse of Insulators From Construction and Demolition Waste To Produce Lime MortarsBuildings PDFDocument25 pagesCircular Building Process Reuse of Insulators From Construction and Demolition Waste To Produce Lime MortarsBuildings PDFGonzalo Fuentes GuimetNo ratings yet
- Comptia A+ 220-601 Practice Test QuestionsDocument32 pagesComptia A+ 220-601 Practice Test Questionsmidnight sun fanNo ratings yet
- TNT PH 2 PLDT ROSARIO - As-Built PlanDocument3 pagesTNT PH 2 PLDT ROSARIO - As-Built PlanDebussy PanganibanNo ratings yet
- MathsDocument2 pagesMathsJoshua S JedheeshNo ratings yet
- Tips and Tricks For The Lab: Column Troubleshooting and AlternativesDocument17 pagesTips and Tricks For The Lab: Column Troubleshooting and AlternativesgiritejaNo ratings yet
- Kitab TayarDocument50 pagesKitab TayarAhmad Adzha100% (1)
- Physics 7E: Practice MidtermDocument5 pagesPhysics 7E: Practice MidtermFUSION AcademicsNo ratings yet
- Scentroid SS400-Operation Manual-V2.0Document21 pagesScentroid SS400-Operation Manual-V2.0delboni delboniNo ratings yet
- Config - Multiple - T24 - Envir - Using - JBoss 6.XX EAPDocument12 pagesConfig - Multiple - T24 - Envir - Using - JBoss 6.XX EAPFin Technet100% (1)
- Falcon Technology - Joe Zeck, DCI ChinaDocument8 pagesFalcon Technology - Joe Zeck, DCI ChinaZAMIRNo ratings yet
- CMD-105 - Migrating Your CONTROL-M Environment To The Latest VersionDocument37 pagesCMD-105 - Migrating Your CONTROL-M Environment To The Latest Versionsam33961No ratings yet
- Critical Appraisal: DR Joshna RajbaranDocument36 pagesCritical Appraisal: DR Joshna RajbaranqwertyasdfggmuNo ratings yet
- 202 - EE8601, EE6601 Solid State Drives - NotesDocument140 pages202 - EE8601, EE6601 Solid State Drives - NotesUdayNo ratings yet
- Introduction To Internal Combustion Engines: Asst. Prof. Vishnu SankarDocument57 pagesIntroduction To Internal Combustion Engines: Asst. Prof. Vishnu SankarMohammad Ishfaq BhatNo ratings yet
- 02.2 - PMI-RMP Exam Content Outline - FinalDocument5 pages02.2 - PMI-RMP Exam Content Outline - FinalbravevnNo ratings yet
- Neutralization Scrubbers - EN PDFDocument3 pagesNeutralization Scrubbers - EN PDFThe EngineerNo ratings yet
- Tibco Spotfire Sample Resume 1Document4 pagesTibco Spotfire Sample Resume 1Srinivas CharyNo ratings yet
- ML Ass 4Document7 pagesML Ass 4Santhosh Kumar PNo ratings yet
- Cache Memory SchemeDocument7 pagesCache Memory SchemejainvidishaNo ratings yet
- Fiery Color Test Page: PMS 640CDocument3 pagesFiery Color Test Page: PMS 640CElvÏs ElvÏs100% (1)
- TheProfessionalNurseSelf AssessmentScaleDocument14 pagesTheProfessionalNurseSelf AssessmentScaleNabeeha Fazeel100% (1)
- PL/SQL User Defined Types Record and Table: Please Use Speaker Notes For Additional Information!Document15 pagesPL/SQL User Defined Types Record and Table: Please Use Speaker Notes For Additional Information!sekharmcpNo ratings yet
- Python Exercises Documentation: Release 1.0Document15 pagesPython Exercises Documentation: Release 1.0Debi Prasad SinhaNo ratings yet
- Student - Live Session Planing 17 - 4-2023 To 22-04-2023Document7 pagesStudent - Live Session Planing 17 - 4-2023 To 22-04-2023nothing noNo ratings yet
- Chapter 1 HomeworkDocument3 pagesChapter 1 HomeworkRAMAH W MASNo ratings yet
- HazLoc PosterDocument1 pageHazLoc PosterSteven MarksNo ratings yet
- 515 - 550 WP DatasheetDocument2 pages515 - 550 WP DatasheetDaniel GnanaselvamNo ratings yet
- Linde C-Matic AutobonicsDocument6 pagesLinde C-Matic AutobonicsJONHHY NGUYEN DANGNo ratings yet
- 2.3 - The Factor Theorem Math 30-1Document14 pages2.3 - The Factor Theorem Math 30-1Math 30-1 EDGE Study Guide Workbook - by RTD LearningNo ratings yet
Sharath2013 PDF
Sharath2013 PDF
Uploaded by
Jessica Duke0 ratings0% found this document useful (0 votes)
15 views5 pagesDiabetic Retinopathy (DR) is the major cause of blindness caused by the damage to the blood vessels in the retina from diabetes. Exudates are fatty deposits on the retina which appear as yellowish regions in fundus image. To evaluate the new method, we examined 158 fundus images, including 84 abnormal images and 74 normal images.
Original Description:
Original Title
sharath2013.pdf
Copyright
© © All Rights Reserved
Available Formats
PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentDiabetic Retinopathy (DR) is the major cause of blindness caused by the damage to the blood vessels in the retina from diabetes. Exudates are fatty deposits on the retina which appear as yellowish regions in fundus image. To evaluate the new method, we examined 158 fundus images, including 84 abnormal images and 74 normal images.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
Download as pdf or txt
0 ratings0% found this document useful (0 votes)
15 views5 pagesSharath2013 PDF
Sharath2013 PDF
Uploaded by
Jessica DukeDiabetic Retinopathy (DR) is the major cause of blindness caused by the damage to the blood vessels in the retina from diabetes. Exudates are fatty deposits on the retina which appear as yellowish regions in fundus image. To evaluate the new method, we examined 158 fundus images, including 84 abnormal images and 74 normal images.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
Download as pdf or txt
You are on page 1of 5
Automatic Detection of Exudates in Retinal Images
Using Histogram Analysis
Sharath Kumar P N , Rajesh Kumar R
Health Informatics and Software Technology Group
Centre for Development of Advanced Computing
Thiruvananthapuram, India
sharath_kumar_pn@yahoo.co.in
Dr. Anuja Sathar , Dr. Sahasranamam V
Department of Ophthalmology
Regional Institute of Ophthalmology
Thiruvananthapuram, India
anujasathar@gmail.com
Abstract Diabetic Retinopathy (DR) is the major cause of
blindness caused by the damage to the blood vessels in the retina
from diabetes. It cannot be prevented but early detection through
fundus imaging by an ophthalmologist can prevent further vision
loss. Presence of microaneurysms, hemorrhages, cotton-wool
spots and exudates are the symptoms of mild DR. Of these, the
detection of exudates is one of the important factors in the early
diagnosis of DR. Exudates are fatty deposits on the retina which
appear as yellowish regions in fundus image. Fundus images
show considerable variation in brightness which makes
automatic detection of exudates difficult. In this study, we are
proposing a new method for preprocessing and false positive
elimination towards the reliable detection of exudates. The
brightness of the fundus image was changed by the nonlinear
curve with brightness values of the hue saturation value (HSV)
space. To emphasize brighter yellow regions (exudates), gamma
correction was performed on each red and green components of
the image. Subsequently, the histograms of each red and green
component were extended. After that, the exudates candidates
were detected using histogram analysis. Finally, false positives
were removed by using multi-channel histogram analysis. To
evaluate the new method for the detection of exudates, we
examined 158 fundus images, including 84 abnormal images with
exudates and 74 normal images. The sensitivity and specificity for
the detection of abnormal and normal cases were 88.45% and
95.5% respectively.
Keywordsdiabetic retinopathy; fundus images; exudates;
histogram analysis; preprocessing; false positive elimination.
I. INTRODUCTION
In India, there are approximately 31.7 million patients with
diabetes and every year 1.7 million new patients with diabetes
are recorded [1]. Approximately 5.6 million are thought to
suffer from diabetic retinopathy (DR). This disease can be
detected and prevented from developing into blindness if it is
treated at an early stage. However, it has been recorded that
people losing their vision following the onset of DR are
increasing year by year. Retinal photographs obtained by the
fundus camera are used to diagnose DR. Ophthalmologists
examine the presence of hemorrhages, microaneurysms, and
exudates in order to diagnose DR. By using fluorescein
angiograms, ophthalmologist can detect changes in blood
vessels. However, it is difficult to use fluorescein as a contrast
medium for diagnosing all the medical examinees subjected to
mass screening.
In past, many studies have been reported on the use of
fundus images in the detection of DR [27]. Hatanaka et al.
proposed a CAD scheme for the detection of hemorrhages and
exudates in ocular fundus images [2]. Niemeijer et al. proposed
a method to describe and evaluate a machine learning-based
automated system to detect exudates and cotton-wool spots in
retinal photographs and differentiate them from drusen for
early diagnosis of DR [3]. Sopharak et al. presented a method
for detecting exudates using fuzzy C-means clustering [4].
Grisan et al. proposed a method for the segmentation of dark
lesions in fundus images based on local thresholding and pixel
density [5]. They reported a mean detection rate of 94% of the
lesions present in an image based on a dataset of 60 annotated
images. Yazid et al. proposed a method to sharpen the edge to
simplify the segmentation process for cotton wool spots and
exudates through ramp width reduction [6]. Xiaohui and Opas
have presented a two way strategy method for the detection of
lesions [7]. Based on different properties of bright lesions and
dark lesions, bottom-up and top-down strategies are applied
respectively to cope with the main difficulties in lesions
detection such as inhomogeneous illumination. In bright lesion
detection, a three-stage, bottom-up approach is applied. After
local contrast enhancement preprocessing stage, two step
Improved Fuzzy C-Means is applied in Luv color space to
segment candidate bright lesion areas. Finally, a hierarchical
SVM classification structure is applied to classify bright non-
lesion areas, exudates and cotton wool spots.
In the above methods, authors have also mentioned about
two problems along with the detection of exudates. One is the
wide variation in the brightness and image resolution other one
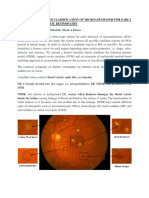
is the high percentage of false positives (FPs). Fig. 1 (a) and (b)
shows the typical fundus images without and with exudates
respectively. In this study, we also address these by developing
methods for fundus image normalization and FPs elimination.
Fig. 1. Typical fundus images. (a) Normal retina. (b) Retina having exudates.
2013 IEEE Recent Advances in Intelligent Computational Systems (RAICS)
978-1-4799-2178-2/13/$31.00 2013 IEEE 277
II. METHODS
A. Overall Scheme
Our overall detection scheme consists
(1) image acquisition, (2) image normalizati
of optic disc, (4) detection of exudates
elimination of FPs using multi-channel hi
Further details are described as follows.
Fig. 2. Flowchart for detecting exudates in fundus imag
B. Image acquisition
A dataset of 158 fundus images were
fundus camera. 89 fundus images were obta
size of 1,500 1,152 pixels and 24-bit color,
were obtained with an array size of 2,144
24-bit color and 29 fundus images were obta
size of 4,288 2,848 pixels with 24-bit
images were annotated by an ophthalmolog
89 fundus images were taken from the D
which was already annotated [8]. An example
image is shown in Fig. 3 (a). Subsequently
matrix was first standardized to 640 x 480 p
detailed subsamples from the original imag
processing efficiency.
C. Image normalization
Due to the flash light used in obtaining t
there is an atypical change in the color of th
To bring uniform change in the color of the f
applied a scheme of brightness correction us
value (HSV) space. First, the brightness va
space were calculated. Second, the calculate
was then smoothened using 9 x 9 median
global correction was performed on the smoo
value of HSV space and the finally ob
correction value Bc(i, j) is given by the follow
Bc(i, ]) = 1 - {I(i, ]) - 1
Image Acquisition
Image Normalization
Extraction of optic
disc
Detection of Exudates
Elimination of false
positives in optic disc
Finally detected
exudates
s of five stages:
ion, (3) extraction
s candidates, (5)
istogram analysis.
ges.
captured using a
ined with an array
, 40 fundus images
1,424 pixels with
ained with an array
color. 69 fundus
gist and remaining
Diaretdb1 database
e of a color fundus
y, the scale of the
pixels by obtaining
e data to improve
the fundus images,
he fundus images.
fundus images, we
sing hue saturation
alues of the HSV
d brightness value
n filtering. Lastly,
othened brightness
btained brightness
wing equation:
1]
2
Where V(i, j) is the smoo
HSV space.
Next, the red value R(i, j)
value B(i, j) were changed b
equally corrected from the ce
fundus image (as shown in Fig.
corrected by Bc(i, j) were then p
The gamma value was experim
Fig. 3 (c)). Finally, the histogra
image were extended (as show
color images were then conve
selecting only the green comp
which will have better contra
fundus images were standardize
(a)
(c)
Fig. 3. Color contrast enhancement.
Brightness of fundus image was c
brightness values of HSV space. (c
adjusted in the dynamic range. (d) G
green component.
D. Detection of optic disc and
The grayscale image obtain
to detect optic disc and exuda
exudates were then extracted
where the threshold values corr
disc were obtained. This is been
Before the exudates candid
understand the distribution of
lesions viz. hemorrhages, exuda
vessels and optic disc which oc
study analysis, intensity profile
hemorrhages being the darkest
vessels which is not as dark
exudates which appear more in
color and finally the brightest
intensity distribution of a fundu
corresponding to each one of th
hemorrhages and last peak bein
4 (a,d). This study knowledge
while looking for the intensity r
othened brightness value of the
), green value G(i, j), and blue
by Bc(i, j). The brightness was
enter to the skirt region of the
3 (b)). The color fundus images
processed by gamma correction.
mentally set to 0.5 (as shown in
ams of each red, green and blue
wn in Fig. 3 (c)). The corrected
erted into grayscale images by
ponent (as shown in Fig. 3 (d))
ast of the vascular details. The
ed by using these processes.
(b)
(d)
(a) Original color fundus image. (b)
hanged by the nonlinear curve with
c) Image after gamma correction and
Grayscale image obtained by selecting
exudates candidates
ned in the previous step was used
ates. Accordingly optic disc and
d using the histogram analysis
responding to exudates and optic
n described as follows.
dates are extracted, one should
intensity values of the different
ates and other objects viz. blood
ccur in a fundus image. From our
e of a fundus image starts with
lesions, second being the blood
k as hemorrhages, next is the
number having yellowish bright
being the optic disc. So, in the
us image one can find four peaks
he four objects with first peak for
ng for optic disc as shown in Fig.
is being utilized in our system
range of exudates and optic disc.
278
(a)
(b)
(c)
Fig. 4. Exudates detection approach. (a) Histogra
(c) Portions of optic disc d
(e) Optic disc candidate extraction from
(
(
(
am from green channel. (b) Exudate candidates along w
etected from green channel. (d) Histog
red channel. (f) Localization of optic disc
(d)
(e)
(f)
with optic disc candidate detection.
gram from red channel.
using binary filling operation.
279
(a)
(b)
Fig. 5. Elimination of false positives by combining the exudates detection from green channel and optic disc extraction from red channel. (a) Binary image
showing the finally detected exudates after eliminating false positives. (b) Detected Exudates outlined in the fundus image.
The exudates candidates were extracted with the help of the
threshold value chosen from the peaks in the histogram
obtained from the green component (as shown in Fig. 4 (a)). As
discussed earlier, we choose the third peak to extract exudates
candidates. With the chosen value, simple binary thresholding
is applied to the green component and the possible exudates
candidates were detected. Detected exudates are outlined as
shown is Fig. 4 (b).
Similarly, optic disc localization was performed by
thresholding the green component of the RGB color image.
Here, we choose the fourth peak to extract optic disc and the
threshold value corresponding to the fourth peak was obtained
from the histogram of the green component as shown in Fig. 4
(a). Due to the blood vessels present inside the optic disc
region, optic disc is not extracted in full but in small portions as
shown in Fig. 4 (c). The method to overcome this is explained
in the next stage.
E. Elimination of false positives using multi-channel
histogram analysis
While extracting exudates using automatically obtained
threshold value from histogram of the green component, few
candidates in the optic disc region will also be detected as
shown in Fig. 4 (b). In order to remove those falsely detected
exudates candidates, we use multi-channel histogram analysis
method.
Firstly, we obtain a threshold value from the peaks
corresponding to optic disc which is found from the histogram
of the red component as shown in Fig. 4 (d). Using this
threshold value, optic disc candidate is fully extracted as shown
in Fig. 4 (e). This is done by thresholding the red component to
the selected threshold value. The shape of the extracted optic
disc region is then approximated to a circle followed by a
binary filling operation. Here, approximation was done by
considering the average size of the optic disc obtained from our
image dataset. The fully extracted and outlined optic disc is as
shown in Fig. 4 (f). The idea of using the red component is
because optic disc being the brightest and dense object in the
fundus images. Also, from the section II.D we saw that
thresholding applied to the green component resulted in multi-
object optic disc detection as shown in Fig. 4 (c) instead of
single-object identification from the red component as shown
in Fig. 4 (f). This is due to the blood vessels present in the optic
disc. In nature, blood vessels are dark which cant be found
from green component. But with the red component, optic disc
can be found fully as the intensities of optic disc and blood
vessels will be more saturated. While extracting we made sure
that only optic disc candidates will be detected. This is done
with the help of binary opening operation.
Secondly, we make use of the exudates detected by
thresholding the green component as obtained in section II.D.
Finally, using the optic disc and exudates (including
candidates in optic disc) pixel informations obtained from red
and green component respectively, candidates in the optic disc
which presented as false exudates are removed. This is done by
binary filtering operation considering the fully extracted optic
disc as a mask. Fig. 5 (a) shows the finally detected exudates
from the fundus image and Fig. 5 (b) shows the outlining of
finally detected exudates.
III. RESULTS
The contrast of exudates in the images was enhanced. The
color of the processed image was standardized by the
histogram normalization method. However, if the fundus image
has some laser marks, the marks are changed brighter and more
obscure than the original color pixel.
A dataset of 158 fundus images having three different
resolutions was used for the experiment. The images were
obtained from two different sources and have sufficient
variations in color, illumination and quality. These included 69
images acquired in a diagnostic examination at the Regional
Institute of Ophthalmology (RIO), Thiruvananthapuram, India
and 89 images taken from the Diaretdb1 database [8].
The images in the dataset were classified by an
ophthalmologist as abnormal or normal image based on the
280
presence or absence of exudates respectively. Of the 69 images
obtained from RIO, 22 were normal and 47 were abnormal.
The Diaretdb1 database contained 37 normal and 52 abnormal
images.
Here, to evaluate our method of detecting exudates, we
examined 158 fundus images. Exudates were detected in 74 out
of 84 images with actual exudates and only 4 false positives
were detected in the 74 images without exudates. By using our
scheme, we succeeded in obtaining satisfactory results with a
sensitivity of 88.45% (74/84) and specificity of 95.5% (70/74).
Our method could remove as many FPs. However, our method
could not detect approximately 10% of exudates. The main
reason was that exudates that were not well focused became
undetected.
Table I summarises the results of our method for three data
sets captured under three different resolutions. Table I also
gives sensitivity (SN) and Specificity (SP) individually for the
three data sets. Elimination of incorrectly detected exudates
was done based on the method of finding the thresholds from
the histograms of red and green channels.
In Table II, the sensitivity and specificity results of our
exudates detection algorithm are presented along with other
published results from in the literature. Here, the comparison
has been made with the results claimed by the respective
authors and the image datasets used for their studies which are
different from ours and from one another. Even though the
result (Specificity) claimed by Sopharak et al [4] are higher
than our proposed method; their method was validated for only
10 images.
TABLE I. ACCURACY DETAILS OF OUR SYSTEM
Data set
Type of
Image
Diagnosed
Images
Correctly
Identified
Images
Accuracy
(%)
1
(1500 X 1152)
DR
Normal
37
52
32
49
SN = 86.48
SP = 94.23
2
(2144 X 1424)
DR
Normal
27
13
24
12
SN = 88.88
SP = 92.3
3
(4288 X 2848)
DR
Normal
20
9
18
9
SN = 90
SP = 100
TABLE II. COMPARISON OF RESULTS FOR EXUDATES DETECTION
Author No. of
Images
No. of Images
with exudates
SN
(%)
SP
(%)
Hatanaka et al. [2] 109 16 77 83
Niemeijer et al. [3] 300 105 95 86
Sopharak et al. [4] 10 DNP* 86.86 99.55
Our Method(Single
channel)
158 84 88.45 91
Our Method(Mutli
channel)
158 84 88.45 95.5
DNP* - data not provided in the paper
IV. CONCLUSION
In this study, a new method for automatic detection of
exudates in the digitized fundus images using multi-channel
histogram analysis is presented. This method can be applied to
the computer-aided diagnosis (CAD) system for diagnosing
eye diseases. The accuracy of the method was evaluated
against the ground-truth annotated by the expert
ophthalmologists. It was demonstrated that the algorithm can
detect abnormalities related to the DR with high accuracy and
reliability. The result of this work on exudates detection in the
fundus images demonstrated that the efficiency and accuracy of
the diagnosis of the DR can be considerably improved using
computer aided detection methods. In the future, the integrated
fundus image analysis scheme will be further improved and
more clinical cases will be reported for evaluating its accuracy.
The techniques employed in our study will help in improving
diagnostic accuracy as well as in improving the workflow
efficiency of the DR screening. Our next focus will be to
develop a full fledged automated system to detect other
symptoms of mild DR viz. microaneurysms, hemorrhages and
cotton-wool spots which will help in assisting
ophthalmologists.
ACKNOWLEDGEMENT
The Department of ophthalmology, Regional Institute of
Ophthalmology (RIO), Thiruvananthapuram provided us with
the required set of fundus images. The ophthalmologists and
domain experts from RIO were consulted for gaining domain
knowledge and for the verification and establishment of ground
truth for the experiment.
REFERENCES
[1] Diabetic retinopathy: An Indian perspective, M. Rema & R. Pradeepa
Madras Diabetes Research Foundation & Dr Mohans Diabetes
Specialities Centre, Chennai, India, Indian J Med Res 125, March 2007,
pp 297-310.
[2] Y. Hatanaka, T. Nakagawa, Y. Hayash, A. Fujita, Y. Mizukusa, M.
Kakogawa, K. Kawase, T. Hara, and H. Fujita, CAD scheme for
detection of hemorrhages and exudates in ocular fundus images, in
Proc. SPIE Medical Imaging 2007: Computer-aided Diagnosis, San
Diego, 2007, vol. 6514, pp. 65142M-1-65142M-8.
[3] Niemeijer M, van Ginneken B, Russell SR, Suttorp-Schulten MS,
Abrmoff MD, Automated detection and differentiation of drusen,
exudates, and cotton-wool spots in digital color fundus photographs for
diabetic retinopathy diagnosis, Invest Ophthalmol Vis Sci. 2007
May;48(5):2260-7.
[4] Sopharak A, Uyyanonvara B, Barman S, Automatic Exudate Detection
from Non-dilated Diabetic Retinopathy Retinal Images Using Fuzzy C-
means Clustering, Sensors. 2009; 9(3):2148-2161.
[5] Grisan E, Ruggeri A, Segmentation of candidate dark lesions in fundus
images based on local thresholding and pixel density, Conf Proc IEEE
Eng Med Biol Soc. 2007;2007:6736-9.
[6] Yazid, H. ; Arof, H. ; Mokhtar, N, Edge Sharpening for Diabetic
Retinopathy Detection, IEEE Conference on Cybernetics and
Intelligent Systems (CIS), pp. 41 44, June 2010.
[7] Xiaohui Zhang ; Chutatape, O., Top-down and Bottom-up Strategies in
Lesion Detection of Background Diabetic Retinopathy, IEEE Computer
Society Conference on Computer Vision and Pattern Recognition
(CVPR), vol. 2, pp. 422-428, June 2005.
[8] Standard Diabetic Retinopathy Database, School of Industrial
Engineering and Management, Lappeenranta University of Technology,
Finland. http://www2.it.lut.fi/project/imageret/diaretdb1/
281
You might also like
- Reinhold Ebertin - Transits - What Days Favor You - American Federation of Astrologers (2015)Document128 pagesReinhold Ebertin - Transits - What Days Favor You - American Federation of Astrologers (2015)ukoszapavlinje100% (24)
- Nugroho2017 PDFDocument4 pagesNugroho2017 PDFglaretsubinNo ratings yet
- Asilomar Bhalerao Anand SaravananDocument3 pagesAsilomar Bhalerao Anand SaravanansosoNo ratings yet
- Orthodontic Application of Color Image Addition To Visualize Differences Between Sequential RadiographsDocument6 pagesOrthodontic Application of Color Image Addition To Visualize Differences Between Sequential RadiographsAmal AbdelwahedNo ratings yet
- Diabetic Retinopathy Detection Using Machine LearningDocument8 pagesDiabetic Retinopathy Detection Using Machine LearningGanavi R PatelNo ratings yet
- Diabetic Retinopathy Detection-IRO-Journals-4 1 3Document8 pagesDiabetic Retinopathy Detection-IRO-Journals-4 1 3Rizkya Putri MaulanaNo ratings yet
- A Skin Tone Detection Algorithm For An Adaptive Approach To SteganographyDocument23 pagesA Skin Tone Detection Algorithm For An Adaptive Approach To SteganographyLeela PavanNo ratings yet
- Review On Brain Tumour Detection Using Digital Image ProcessingDocument7 pagesReview On Brain Tumour Detection Using Digital Image ProcessingShivangi33No ratings yet
- Sopharak ExudateFeaturesDocument4 pagesSopharak ExudateFeaturesHassan FaouziNo ratings yet
- EMBC2017 - Exudate Detection For Diabetic Retinopathy With Convolutional Neural NetworksDocument4 pagesEMBC2017 - Exudate Detection For Diabetic Retinopathy With Convolutional Neural Networks1DA19CS156 Shubha SNo ratings yet
- Adaptive Neuro-Fuzzy Inference System Approach For The Automatic Screening of Diabetic Retinopathy in Fundus ImagesDocument7 pagesAdaptive Neuro-Fuzzy Inference System Approach For The Automatic Screening of Diabetic Retinopathy in Fundus ImagesadaneNo ratings yet
- Detection of Diabetic Retinopathy WordDocument6 pagesDetection of Diabetic Retinopathy WordThe MinisterNo ratings yet
- Automatic Vessel Extraction With Combined Bottom Hat and Match FilterDocument5 pagesAutomatic Vessel Extraction With Combined Bottom Hat and Match FilterBahareh Bozorg ChamiNo ratings yet
- Skin Color SegDocument6 pagesSkin Color SegSachin MoreNo ratings yet
- Iris Diagnosis - A Quantitative Non-Invasive Tool Fordiabetes DetectionDocument7 pagesIris Diagnosis - A Quantitative Non-Invasive Tool Fordiabetes Detectionkrishna mohan pandeyNo ratings yet
- Automated Skin Lesion Segmentation Using K-Means Clustering From Digital Dermoscopic ImagesDocument6 pagesAutomated Skin Lesion Segmentation Using K-Means Clustering From Digital Dermoscopic ImagesArpit GuptaNo ratings yet
- 635 A Stand-Alone MATLAB Application For The Detection of Optic Disc and MaculaDocument5 pages635 A Stand-Alone MATLAB Application For The Detection of Optic Disc and MaculaRama SubramanianNo ratings yet
- 1207 1551 (1) 68Document9 pages1207 1551 (1) 68Mohamad SannanNo ratings yet
- Recognition of Polyps in The Images of Wireless Capsule EndosDocument4 pagesRecognition of Polyps in The Images of Wireless Capsule EndosInternational Journal of Application or Innovation in Engineering & ManagementNo ratings yet
- Madooei2013 Chapter AutomaticDetectionOfBlue-WhiteDocument8 pagesMadooei2013 Chapter AutomaticDetectionOfBlue-WhitemaryNo ratings yet
- Intensity Features Based Classification of Hard Exudates in Retinal ImagesDocument5 pagesIntensity Features Based Classification of Hard Exudates in Retinal ImagesAleenaNo ratings yet
- Bin Ar Ization RetinalDocument12 pagesBin Ar Ization RetinalMohammad Asmat Ullah KhanNo ratings yet
- Color Constancy For Improving Skin DetectionDocument18 pagesColor Constancy For Improving Skin DetectionAI Coordinator - CSC JournalsNo ratings yet
- A Novel and Efficient Feature Extraction Method For Iris RecognitionDocument3 pagesA Novel and Efficient Feature Extraction Method For Iris RecognitionkaleysureshNo ratings yet
- Dokumen Pendukung Karya IlmiahDocument7 pagesDokumen Pendukung Karya IlmiahyaniNo ratings yet
- Diabetic Retinopathy-Early Detection Using Im-Age Processing TechniquesDocument5 pagesDiabetic Retinopathy-Early Detection Using Im-Age Processing Techniqueskumar_9583No ratings yet
- Comparative Study of Segmentation Techniques in Diabetic Retinopathy DetectionDocument4 pagesComparative Study of Segmentation Techniques in Diabetic Retinopathy DetectionAnam FarooqNo ratings yet
- An Intelligent Black Widow Optimization On Image Enhancement With Deep Learning Based Ovarian Tumor Diagnosis ModelDocument16 pagesAn Intelligent Black Widow Optimization On Image Enhancement With Deep Learning Based Ovarian Tumor Diagnosis ModelAgnesNo ratings yet
- An Automatic Segmentation & Detection of Blood Vessels and Optic Disc in Retinal ImagesDocument5 pagesAn Automatic Segmentation & Detection of Blood Vessels and Optic Disc in Retinal ImagesMia AmaliaNo ratings yet
- A New Method For Eye Detection in Color Images: Mohamad Ali Azimi Mohammad RahmatiDocument8 pagesA New Method For Eye Detection in Color Images: Mohamad Ali Azimi Mohammad RahmatiOscar Duque SuarezNo ratings yet
- ObjectiveDocument22 pagesObjectiveKoratala HarshaNo ratings yet
- Detection of Forgeries in Digital Color ImageDocument5 pagesDetection of Forgeries in Digital Color ImagegangasagarjadhavNo ratings yet
- Adaptive Neuro Fuzzy Inference System Assisted Diagnosis of Diabetic Retinopathy From Fundus ImageDocument5 pagesAdaptive Neuro Fuzzy Inference System Assisted Diagnosis of Diabetic Retinopathy From Fundus ImageadaneNo ratings yet
- Automated Quantification of Clinically SignificantDocument5 pagesAutomated Quantification of Clinically SignificantHaleema ahsanNo ratings yet
- Sanders - Samei - 2016 - Patient-Specific Quantification of Image Quality - Method For Measuring Resolution in Clinical CT Images - Medical - PhysicsDocument10 pagesSanders - Samei - 2016 - Patient-Specific Quantification of Image Quality - Method For Measuring Resolution in Clinical CT Images - Medical - PhysicsmartuflashNo ratings yet
- DSA Image Registration Based On Multiscale Gabor Filters and Mutual InformationDocument6 pagesDSA Image Registration Based On Multiscale Gabor Filters and Mutual InformationerdoganaaaNo ratings yet
- A New Hybrid Method For TheDocument8 pagesA New Hybrid Method For ThesipijNo ratings yet
- Automated Tumor Segmentation in MR Brain Image Using Fuzzy C-Means Clustering and Seeded Region MethodologyDocument7 pagesAutomated Tumor Segmentation in MR Brain Image Using Fuzzy C-Means Clustering and Seeded Region MethodologyIAES IJAINo ratings yet
- Sujeet Bhagwat (04) - Aditya Gujar (27) - Swaraj Govindwar (26) - Krishna GatlewarDocument25 pagesSujeet Bhagwat (04) - Aditya Gujar (27) - Swaraj Govindwar (26) - Krishna GatlewarSwaraj GovindwarNo ratings yet
- Robust and Fast Iris Localization Using Contrast Stretching and Leading Edge DetectionDocument7 pagesRobust and Fast Iris Localization Using Contrast Stretching and Leading Edge DetectionInternational Journal of Application or Innovation in Engineering & ManagementNo ratings yet
- Brain Tumer Extraction From Mri Image Using K-Means Clustring TecniqueDocument7 pagesBrain Tumer Extraction From Mri Image Using K-Means Clustring TecniqueRohit AryaNo ratings yet
- Segmentation of Retinal Blood Vessels Using A Novel Clustering Algorithm (RACAL) With A Partial Supervision StrategyDocument13 pagesSegmentation of Retinal Blood Vessels Using A Novel Clustering Algorithm (RACAL) With A Partial Supervision StrategySathiya Moorthy C ANo ratings yet
- Azienda Sanitaria Firenze, Fisica Sanitaria, Firenze, Italy Azienda Sanitaria Firenze, Radioterapia, Firenze, ItalyDocument2 pagesAzienda Sanitaria Firenze, Fisica Sanitaria, Firenze, Italy Azienda Sanitaria Firenze, Radioterapia, Firenze, ItalyLarolina KhoirunnisaaNo ratings yet
- Exudates Detection at Premature Stage in Diabetic RetinopathyDocument6 pagesExudates Detection at Premature Stage in Diabetic RetinopathylambanaveenNo ratings yet
- 10 1 1 85 5278 PDFDocument14 pages10 1 1 85 5278 PDFStaurt AttkinsNo ratings yet
- Conglomeration For Color Image Segmentation of Otsu Method, Median Filter and Adaptive Median FilterDocument8 pagesConglomeration For Color Image Segmentation of Otsu Method, Median Filter and Adaptive Median FilterInternational Journal of Application or Innovation in Engineering & ManagementNo ratings yet
- 2011 - Multi Channel Film Dosimetry With Non Uniformity CorrectionDocument12 pages2011 - Multi Channel Film Dosimetry With Non Uniformity Correctioncnsouza2000No ratings yet
- MIA PresentationDocument8 pagesMIA Presentationahsan puriNo ratings yet
- Design If Iris Based Medical Diagnosis SystemDocument4 pagesDesign If Iris Based Medical Diagnosis SystemDr Vaishali MathapatiNo ratings yet
- A Simple Approach For Relatively Automated Hippocampus Segmentation From Sagittal View of Brain MriDocument8 pagesA Simple Approach For Relatively Automated Hippocampus Segmentation From Sagittal View of Brain MriijbbjournalNo ratings yet
- Identification of Diabetic Retinopathy Using Fuzzy Logic and Back Propagation Neural NetworkDocument8 pagesIdentification of Diabetic Retinopathy Using Fuzzy Logic and Back Propagation Neural Networkpurushothaman sinivasanNo ratings yet
- E7732068519 PDFDocument6 pagesE7732068519 PDFAleenaNo ratings yet
- Estimating Velocity of Nailfold Vessels Flow Using Improvised Deconvolution AlgorithmDocument6 pagesEstimating Velocity of Nailfold Vessels Flow Using Improvised Deconvolution AlgorithmIJERDNo ratings yet
- Spatial Domain Enhancement Techniques For Detection of Lung Tumor From Chest X-Ray ImageDocument10 pagesSpatial Domain Enhancement Techniques For Detection of Lung Tumor From Chest X-Ray ImageInternational Journal of Application or Innovation in Engineering & ManagementNo ratings yet
- Danne WitzDocument6 pagesDanne WitzKhairniNo ratings yet
- A New Technique For Hand Gesture RecognitionDocument4 pagesA New Technique For Hand Gesture RecognitionchfakhtNo ratings yet
- Qno1: Define Spectrophotometer?: WorkingDocument3 pagesQno1: Define Spectrophotometer?: WorkingHussnain MughalNo ratings yet
- Blood Vessel Segmentation in Fundus ImagesDocument5 pagesBlood Vessel Segmentation in Fundus ImagesNaoufal NaoufalNo ratings yet
- Standard and Super-Resolution Bioimaging Data Analysis: A PrimerFrom EverandStandard and Super-Resolution Bioimaging Data Analysis: A PrimerNo ratings yet
- Multi-modality Cardiac Imaging: Processing and AnalysisFrom EverandMulti-modality Cardiac Imaging: Processing and AnalysisPatrick ClarysseNo ratings yet
- Computer Stereo Vision: Exploring Depth Perception in Computer VisionFrom EverandComputer Stereo Vision: Exploring Depth Perception in Computer VisionNo ratings yet
- Circular Building Process Reuse of Insulators From Construction and Demolition Waste To Produce Lime MortarsBuildings PDFDocument25 pagesCircular Building Process Reuse of Insulators From Construction and Demolition Waste To Produce Lime MortarsBuildings PDFGonzalo Fuentes GuimetNo ratings yet
- Comptia A+ 220-601 Practice Test QuestionsDocument32 pagesComptia A+ 220-601 Practice Test Questionsmidnight sun fanNo ratings yet
- TNT PH 2 PLDT ROSARIO - As-Built PlanDocument3 pagesTNT PH 2 PLDT ROSARIO - As-Built PlanDebussy PanganibanNo ratings yet
- MathsDocument2 pagesMathsJoshua S JedheeshNo ratings yet
- Tips and Tricks For The Lab: Column Troubleshooting and AlternativesDocument17 pagesTips and Tricks For The Lab: Column Troubleshooting and AlternativesgiritejaNo ratings yet
- Kitab TayarDocument50 pagesKitab TayarAhmad Adzha100% (1)
- Physics 7E: Practice MidtermDocument5 pagesPhysics 7E: Practice MidtermFUSION AcademicsNo ratings yet
- Scentroid SS400-Operation Manual-V2.0Document21 pagesScentroid SS400-Operation Manual-V2.0delboni delboniNo ratings yet
- Config - Multiple - T24 - Envir - Using - JBoss 6.XX EAPDocument12 pagesConfig - Multiple - T24 - Envir - Using - JBoss 6.XX EAPFin Technet100% (1)
- Falcon Technology - Joe Zeck, DCI ChinaDocument8 pagesFalcon Technology - Joe Zeck, DCI ChinaZAMIRNo ratings yet
- CMD-105 - Migrating Your CONTROL-M Environment To The Latest VersionDocument37 pagesCMD-105 - Migrating Your CONTROL-M Environment To The Latest Versionsam33961No ratings yet
- Critical Appraisal: DR Joshna RajbaranDocument36 pagesCritical Appraisal: DR Joshna RajbaranqwertyasdfggmuNo ratings yet
- 202 - EE8601, EE6601 Solid State Drives - NotesDocument140 pages202 - EE8601, EE6601 Solid State Drives - NotesUdayNo ratings yet
- Introduction To Internal Combustion Engines: Asst. Prof. Vishnu SankarDocument57 pagesIntroduction To Internal Combustion Engines: Asst. Prof. Vishnu SankarMohammad Ishfaq BhatNo ratings yet
- 02.2 - PMI-RMP Exam Content Outline - FinalDocument5 pages02.2 - PMI-RMP Exam Content Outline - FinalbravevnNo ratings yet
- Neutralization Scrubbers - EN PDFDocument3 pagesNeutralization Scrubbers - EN PDFThe EngineerNo ratings yet
- Tibco Spotfire Sample Resume 1Document4 pagesTibco Spotfire Sample Resume 1Srinivas CharyNo ratings yet
- ML Ass 4Document7 pagesML Ass 4Santhosh Kumar PNo ratings yet
- Cache Memory SchemeDocument7 pagesCache Memory SchemejainvidishaNo ratings yet
- Fiery Color Test Page: PMS 640CDocument3 pagesFiery Color Test Page: PMS 640CElvÏs ElvÏs100% (1)
- TheProfessionalNurseSelf AssessmentScaleDocument14 pagesTheProfessionalNurseSelf AssessmentScaleNabeeha Fazeel100% (1)
- PL/SQL User Defined Types Record and Table: Please Use Speaker Notes For Additional Information!Document15 pagesPL/SQL User Defined Types Record and Table: Please Use Speaker Notes For Additional Information!sekharmcpNo ratings yet
- Python Exercises Documentation: Release 1.0Document15 pagesPython Exercises Documentation: Release 1.0Debi Prasad SinhaNo ratings yet
- Student - Live Session Planing 17 - 4-2023 To 22-04-2023Document7 pagesStudent - Live Session Planing 17 - 4-2023 To 22-04-2023nothing noNo ratings yet
- Chapter 1 HomeworkDocument3 pagesChapter 1 HomeworkRAMAH W MASNo ratings yet
- HazLoc PosterDocument1 pageHazLoc PosterSteven MarksNo ratings yet
- 515 - 550 WP DatasheetDocument2 pages515 - 550 WP DatasheetDaniel GnanaselvamNo ratings yet
- Linde C-Matic AutobonicsDocument6 pagesLinde C-Matic AutobonicsJONHHY NGUYEN DANGNo ratings yet
- 2.3 - The Factor Theorem Math 30-1Document14 pages2.3 - The Factor Theorem Math 30-1Math 30-1 EDGE Study Guide Workbook - by RTD LearningNo ratings yet