Professional Documents
Culture Documents
Cell Culture and Transfection
Cell Culture and Transfection
Uploaded by
Firjon N Ika0 ratings0% found this document useful (0 votes)
41 views10 pagesThis document provides supplementary methods and results for a study examining how Leu607Phe alleles of the DISC1 gene differentially affect centrosomal PCM1 localization and neurotransmitter release. It describes the cell culture and transfection methods used to express tagged Leu607 or Phe607 DISC1 variants. It also details the protocols for studying co-localization with PCM1, western blotting, and a noradrenaline release assay to analyze the effects on neurotransmitter levels. Statistical analyses are outlined and pilot studies suggest high KCl-stimulated noradrenaline release in the assay is due to reversal of the noradrenaline transporter.
Original Description:
serba serbi
Copyright
© © All Rights Reserved
Available Formats
DOC, PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentThis document provides supplementary methods and results for a study examining how Leu607Phe alleles of the DISC1 gene differentially affect centrosomal PCM1 localization and neurotransmitter release. It describes the cell culture and transfection methods used to express tagged Leu607 or Phe607 DISC1 variants. It also details the protocols for studying co-localization with PCM1, western blotting, and a noradrenaline release assay to analyze the effects on neurotransmitter levels. Statistical analyses are outlined and pilot studies suggest high KCl-stimulated noradrenaline release in the assay is due to reversal of the noradrenaline transporter.
Copyright:
© All Rights Reserved
Available Formats
Download as DOC, PDF, TXT or read online from Scribd
Download as doc, pdf, or txt
0 ratings0% found this document useful (0 votes)
41 views10 pagesCell Culture and Transfection
Cell Culture and Transfection
Uploaded by
Firjon N IkaThis document provides supplementary methods and results for a study examining how Leu607Phe alleles of the DISC1 gene differentially affect centrosomal PCM1 localization and neurotransmitter release. It describes the cell culture and transfection methods used to express tagged Leu607 or Phe607 DISC1 variants. It also details the protocols for studying co-localization with PCM1, western blotting, and a noradrenaline release assay to analyze the effects on neurotransmitter levels. Statistical analyses are outlined and pilot studies suggest high KCl-stimulated noradrenaline release in the assay is due to reversal of the noradrenaline transporter.
Copyright:
© All Rights Reserved
Available Formats
Download as DOC, PDF, TXT or read online from Scribd
Download as doc, pdf, or txt
You are on page 1of 10
SUPPLEMENTARY MATERIAL
DISC-1 Leu607Phe alleles differentially affect centrosomal PCM1
localization and neurotransmitter release
Eastwood et al.
Acno!led"ments
Supported by the UK Medical Research Council (Research grant #G000!"0# and
a Centre $ward %ro& the Stanley Medical Research 'nstitute. (e than) $)ira
Sawa %or his contributions.
1
Su##lementary Methods and Materials
Cell Culture and $ransfection !ith %&-ta""ed Leu607 or Phe607 DISC-1'
S*+S,, cells (European Collection o% Cell Cultures- .orton /own- UK# were
cultured in /ulbecco0s Modi%ied Eagle Mediu& (/MEM1 Sig&a- .oole- UK#
supple&ented with !02 %oetal cal% seru& (Sig&a#- 3&M 4+gluta&ine (Sig&a#
and !2 non+essential a&ino acids (Sig&a#. Cells were &aintained in a
hu&idi%ied incubator at 567C and 2 C8
3
. Constructs containing either 9+
tagged 4eu:06 or .he:06 /'SC+! were transiently trans%ected into cells using
;urbo%ect (<er&entas- ,or)- UK# in accordance with &anu%acturer0s instructions.
<or western blotting and =
5
*> noradrenaline neurotrans&itter release assays-
cells were sub+cultured into si?+well culture plates at a density o% 3. ? !0
cells
per well. 63 hours a%ter plating- 3 @g per well o% plas&id was &i?ed with
trans%ection reagent and applied to 5 wells- with trans%ection reagent alone
applied to the re&aining 5 wells as e?peri&ental controls. Cells were grown %or a
%urther 3A hours be%ore utiliBation. Each e?peri&ental run consisted o% cells at a
di%%erent passage nu&ber in which two culture plates per Cariant were run in
parallel- one plate %or the neurotrans&itter assay- with cells %ro& the other being
harCested %or western blotting studies.
<or co+localiBation studies- undi%%erentiated S*+S,, cells were plated at a
density o% ? !0
cells per cha&ber into a A well cha&ber slide (9(R
'nternational- 4utterworth- UK#. $%ter 3A hours- 0.@g per well o% plas&id was
&i?ed with trans%ection reagent and constructs containing either Cariant applied
to 3 wells- with trans%ection reagent alone applied to the re&aining 3 wells as
e?peri&ental controls.
2
DISC-1 and PCM1 Co-localization Studies
3A hours a%ter trans%ection- S*+S,, cells were %i?ed with ðanol at +307C %or
: &in- giCen 3 D !0 &in washes with phosphate bu%%ered saline (.ES# and
bloc)ed %or ! hour at R; with !02 nor&al goat seru& (Sig&a# diluted in .ES
containing 0.!2 ;riton D+!00 (.ES+;# and 32 boCine seru& albu&in (ES$1
Sig&a#. Cells were then incubated %or ! hour at R; with pri&ary antibodies
diluted in .ES+; containing !2 nor&al goat seru& and 32 ES$ (rabbit anti+
pericentriolar &aterial+! (.CM+!# at !F300- Santa CruB- *eidelberg- Ger&any1
&ouse anti 9 at !F!000- Sig&a#. $%ter 5 D !0 &in washes in .ES- cells were
incubated %or ! hour at R; with secondary antibodies (goat anti+rabbit $le?a
<luor :"- goat anti &ouse $le?a <luor A""- both at !F30001 'nCitrogen# diluted
in .ES+; containing !2 nor&al goat seru& and 32 ES$. Cells were giCen 5 D!0
&in washes in .ES- dipped into distilled water- and coCer slipped using
9ectashield (9ector 4aboratories 4td- .eterborough- UK#. Cell staining was
e?a&ined using a Gi)on Eclipse 5:00 &icroscope and i&ages captured using a
MC'/ Elite Cersion 6.0 i&age analysis syste& ('nter%ocus- *aCerhill- UK#. .CM!
centroso&al i&&unoreactiCe area was &easured in a total o% :0 cells %or each
Cariant and HI0 untrans%ected cells (in three separate e?peri&ents#.
Geurotrans&itter and western blotting assays were per%or&ed in I replicate
e?peri&ents.
(estern )lottin"
'&&ediately be%ore cell harCesting- to each !0 &l o% suspension bu%%er (0.!M
GaCl- 0.0!M ;ris+*Cl- 0.00!M E/;$- !2 sodiu& dodecyl sulphate# one co&plete
&ini protease inhibitor coc)tail tablet (Roche /iagnostics 4td- Eurgess *ill- UK#
was added. Cells were harCested using 300 @l per well o% 0.32 ;rypsin+E/;$
3
(Sig&a#. $%ter incubating at 567C %or 3 &in- to each well "00 @l o% .ES was
added- and the cells trans%erred to ! &l eppendor% tubes. Cells were pelleted by
centri%uging at !3-000g %or &in- and the cell pellet resuspended in !00 @l o%
suspension bu%%er. Sa&ples were then boiled %or !0 &in- centri%uged at !3-000g
%or !0 &in and the supernatant collected. ;he protein content o% each sa&ple
was assessed using the Erad%ord assay (Sig&a#.
.ilot studies established that loading 3 @g o% protein was in the linear range o%
detection %or all proteins e?a&ined. '&&ediately be%ore loading sa&ples were
&i?ed with D S/S gel+loading bu%%er (0.M ;ris+*Cl- !02 sodiu& dodecyl
sulphate- 02 glycerol- 2 J+&ercaptoethanol- 0.!2 bro&ophenol blue# and
boiled %or !0 &in. Sa&ples were %ractionated by electrophoresis through a !32
polyacryla&ide gel together with precision plus protein standards (Eio+Rad-
*e&el *e&pstead- UK#. Separated proteins were trans%erred onto polyCinyl
di%luoride (.9/<# &e&branes using an iElot dry blotting syste& (.rogra& 5- 6
&in1 'nCitrogen- .aisely- UK#.
$ll washes and incubations were per%or&ed with .ES containing 0.!2 ;ween+30
(.ES+;w#. Gon+speci%ic binding sites were bloc)ed by incubating &e&branes %or
! hour at R; with 32 non+%at &il). <or Ceri%ication o% trans%ection with 9+
tagged .he:06+ and 4eu:06 /'SC+!- blots were then incubated with horse+
radish pero?idase (*R.# labelled anti+9 (!F000- 'nCitrogen# in 2 non+%at &il)
%or 3 hours at R;- a%ter which they were giCen 5 D ! &in washed in .ES+;w- and
deCeloped using an EC4+plus western blotting detection syste& (GE *ealthcare-
$&ersha&- UK#. <or western blotting o% tyrosinated and detyrosinated alpha+
tubulin- blots were incubated %or ! hour at R; with pri&ary antibodies (&ouse
4
anti+tryosinated alpha tubulin at !F0-000-clone ;UE+!$3- Sig&a1 rabbit anti+
detryosinated alpha tubulin at !F!0-000- Millipore- (at%ord- UK# diluted in !2
ES$. $%ter 5 D ! &in washes- blots were incubated %or ! hour at R; with
secondary antibodies diluted in 32 non+%at &il) (*R. labelled goat anti+&ouse
and *R. labelled goat anti+rabbit- both at !F000- Eio+Rad#- and a%ter 5 %inal
washes in .ES+;w- &e&branes were deCeloped as aboCe. Me&branes were
placed against EC4 %il& ($&ersha& *yper%il& EC4- GE *eathcare# %or the
%ollowing ti&es (anti+9F 5 &in1 anti+tryosinated alpha tubulinF ! sec1 anti+
detyrosinated alpha tubulinF 3 &in#. $ll KuantitatiCe assays were per%or&ed in
duplicate in nine replicate e?peri&ents- and tyrosinated and de+tyrosinated
alpha tubulin protein was &easured using an $lpha'&ager 5A00 (Gentic
Research 'nstru&ents 4td- Eraintree- UK# i&age analysis syste&.
*
+
,- .oradrenaline /elease Assay
3A hours a%ter trans%ection with either 9+tagged 4eu:06 or .he:06 /'SC+!- =
5
*>
noradrenaline neurotrans&itter release was e?a&ined using an established
assay.
S!
;he culture &edia was re&oCed and cells were giCen two brie% washes
in *epes bu%%ered saline (*ESF 30&M *epes- !A0&M GaCl- &M KCl- 3.&M
CaCl
3
- !.3&M K*
3
.8
A
- !&M MgCl
3-
0.3&M ascorbic acid- 0.3&M pargyline and
:&M /+glucose#. Cells were then loaded with =
5
*> noradrenaline (0. @CiLwell1
.er)in El&er- Eeacons%ield- UK# diluted in *ES %or ! hour in a hu&idi%ied
incubator at 567C and 2 C8
3
. $%ter A D ! &in washes in *ES cells at roo&
te&perature (R;#- cells were incubated with *ES %or &in at R; and the
per%usate collected (&M =K
M
>#. Cells were then incubated %or &in at R; with
*ES containing !00&M KCl
(with the concentration o% GaCl reduced to A0&M to
&aintain os&olarity#. $%ter collecting the per%usate (!00&M =K
M
>#- adherent cells
5
were lysed and detached %ro& the culture plate by the addition o% d*
3
0 and
collected (re&aining noradrenaline#. ;he Kuantities o% released and re&aining
=
5
*> noradrenaline was deter&ined by liKuid scintillation counting- and e?pressed
as a percentage o% total =
5
*> noradrenaline collected. Geurotrans&itter assays
were per%or&ed in I replicate e?peri&ents.
Geurotrans&itters can be released by both Cesicular e?ocytosis and by
&e&brane carriers responsible %or their upta)e (carrier &ediated release#.
S3
;o
better characterise which &echanis& &ay underlie =
5
*> noradrenaline release at
and !00&M =K
M
> under our e?peri&ental conditions- pilot studies were
conducted. !0@& desipra&ine has been shown to inhibit the noradrenaline
transporter in both directions-
S5
and was applied during post incubation washes
and during release.
Statistical Analyses
$ll datasets &et criteria %or nor&ality using the Kolo&ogoroC+S&irnoC one+
sa&ple test- and para&etric tests were there%ore applied. .aired sa&ple t+tests
were used in the statistical analyses o% noradrenaline release and the tyrosinated
to detyrosinated alpha+tubulin western blot studies. <or each Cariant and
e?peri&ental condition- Calues obtained a%ter trans%ection were co&pared with
those %ro& their own e?peri&ental control (untrans%ected cells#. <or the .CM!
i&&unoreactiCity studies di%%erences between the three groups (untrans%ected-
phe:06 /'SC+!- 4eu:06 /'SC+!# was assessed by analysis o% Cariance with
planned co&parisons between groups e?plored using least signi%icant di%%erence
(4S/# test.
6
Su##lementary /esults and Discussion
.eurotransmitter Assay
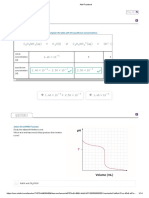
$lthough release at &M =K
M
> was una%%ected by !0@& desipra&ine- release
sti&ulated by !00 &M =K
M
> was reduced to &M =K
M
> leCels (Supple&entary
<igure !#- indicating that release at high =K
M
>in our assay conditions is due to
reCersal o% the noradrenaline transporter. ;he %inding that at &M =K
M
>
noradrenaline release was una%%ected by !0@& desipra&ine indicates that
reCersal o% the noradrenaline transporter is unli)ely to be the &echanis&
underlying this %or& o% release- and instead re%lects spontaneous Cesicle %usion.
SA
Microtu)ules and syna#tic function
/ata %ro& seCeral sources suggest that &icrotubules play a role in synaptic
%or&ation- &aintenance and %unction. <or e?a&ple- &icrotubules haCe been
de&onstrated to be i&portant in dendritic spine deCelop&ent and &orphology-
S+
:
whilst growth cone turning-
S6+"
a?on path %inding-
SI+!0
and a?on branching
S!!+!3
are all dependent on interactions between &icrotubules and &icro%ila&ents.
<urther&ore- &icrotubules %unction as rails along which &olecular &otors
including )inesin super%a&ily proteins and dyneins transport intracellular cargoes
such as &RG$s- protein co&ple?es and organelles-
S!5
including precursors o%
synaptic Cesicles to a?on ter&inals.
S!A+!:
'n this respect- &icrotubules haCe been
de&onstrated to be i&portant in the transport and %unction o% receptors- both
during synaptogenesis and in adult synaptic plasiticity.
S!6+30
Su##lementary /eferences
7
S!. (ebster GN- 9aughan .<;- .eers C. Mol. Brain Res 300!1 89F 0+6.
S3. 4eCi G- Raiteri M. Trends Neurosci !II51 16F A!+A!I.
S5. 'rie K- (urt&an RN. Brain Res !I"61 423F 5I!+5IA.
SA. Glitsch M/. Cell Calcium 300"1 43F I+!.
S. Gu N- <irestein E4- Oheng NP. J Neurosci 300"1 28F !3!30+!3!3A.
S:. *u D- 9iessel&ann C- Ga& S- Merra& E- /ent E(. J Neurosci 300"1 28:
!50IA+!5!0.
S6. ;ana)a E- Kirschner M(. J Cell Biol!II1 128: !36+!56.
S". (illia&son ;- Gordon (ee)s .R- Schachner M- ;aylor N. Proc Natl Acad Sci
USA !II: 93: !33!+!33:.
SI. Suter /M- <orscher .. J Neurobiol 30001 44: I6+!!5.
S!0 Schae%er $(- Kabir G- <orscher .. J Cell Biol 30031 158: !5I+!3.
S!!. /ent E(- Earnes $M- ;ang <- Kalil K. J Neurosci 300A1 24: 5003+50!3.
S!3. Kalil K- /ent E(. Curr Opin Neurobiol 3001 15: 3!+3:.
S!5. *iro)awa G- ;a)e&ura R. Exp Cell Res 300A1 301: 0+I.
S!A. 8)ada ,- ,a&aBa)i *- Se)ine $iBawa ,- *iro)awa G. Cell !II1 81: 6:I+
6"0.
S! ,one)awa ,- *arada $- 8)ada ,- <una)oshi ;- Kanai ,- ;a)ei , et al. J Cell
Biol !II"1 141: A5!+AA!.
S!: Ohao C- ;a)ita N- ;ana)a ,- Setou M- Ga)agawa ;- ;a)eda S et al. Cell 300!1
105: "6+I6.
S!6. Serge $- <ourgeaud 4- *e&ar $- ChoKuet /. J Cell Sci 30051 116: 0!+
033.
S!". (ashbourne .- Eennett NE- Mc$llister $K. Nat. Neurosci 3003 5: 6!+6I.
S!I. (ashbourne .- 4iu DE- Nones EG- Mc$llister $K. J Neurosci 300A1 24: "35+
"3:A.
8
S30. ,uen E,- Niang P- <eng N- ,an O. J Biol C!em 3001 280: 3IA30+3IA36.
Su##lementary 0i"ure 1' =
5
*> noradrenaline release at !00&M =K
M
> is
attenuated by incubation with !0@M desipra&ine. (9alues are &ean M S/- nQ5#.
9
5mM [K
+
] 100mM [K
+
]
0.0
2.5
5.0
7.5
10.0
12.5
Control
10M Desipramine
%
T
o
t
a
l
[
3
H
]
N
o
r
a
r
e
n
a
l
i
n
e
10
You might also like
- Histology NBME Review and AnswersDocument25 pagesHistology NBME Review and Answerspyogenius50% (4)
- Octogon DegusDocument6 pagesOctogon DegusKaren Esteves ZúñigaNo ratings yet
- 938 FullDocument5 pages938 FullDiana ElamNo ratings yet
- Chromosome Aberrations Penyimpangan Kromosom: (Dok) Kelainan Atau Perubahan Kromosom, Baik Dalam Jumlah Maupun BentuknyaDocument25 pagesChromosome Aberrations Penyimpangan Kromosom: (Dok) Kelainan Atau Perubahan Kromosom, Baik Dalam Jumlah Maupun BentuknyaDiah Dewi AnggraeniNo ratings yet
- SHP Revisi IIIDocument12 pagesSHP Revisi IIIzie_luph_taz13No ratings yet
- Supplemental Material (Detailed Methods) : in Vivo Virus Transduction To Express HuaDocument10 pagesSupplemental Material (Detailed Methods) : in Vivo Virus Transduction To Express HuaCLPHtheoryNo ratings yet
- Dna MicroarrayDocument14 pagesDna MicroarraymaniiiiiiiiNo ratings yet
- BME 5 Final Study GuideDocument4 pagesBME 5 Final Study Guidevanarp123No ratings yet
- CerebrolysinDocument3 pagesCerebrolysinMelissa Fatima Laurente Dosdos100% (2)
- Food Control: Min Hwa Lee, Doo-Sung Cheon, Changsun ChoiDocument4 pagesFood Control: Min Hwa Lee, Doo-Sung Cheon, Changsun ChoivafeasNo ratings yet
- Complete OATP1B1 and OATP1B3 Deficiency Causes Human Rotor Syndrome by Interrupting Conjugated Bilirubin Reuptake Into The LiverDocument18 pagesComplete OATP1B1 and OATP1B3 Deficiency Causes Human Rotor Syndrome by Interrupting Conjugated Bilirubin Reuptake Into The LiveramraovcinaNo ratings yet
- Homework 8 - Final Project: Andres Garcia-ArceDocument28 pagesHomework 8 - Final Project: Andres Garcia-ArceAndrés García ArceNo ratings yet
- IV Ciclo Parasitologia Lectura #1-1-20180816114149Document3 pagesIV Ciclo Parasitologia Lectura #1-1-20180816114149DJose SCNo ratings yet
- Citosol (Thiamylal Sodium) Triggers Apoptosis and Affects Gene Expressions of Murine Leukemia RAW 264.7 Cells - RS-C Wu, C-S YuDocument1 pageCitosol (Thiamylal Sodium) Triggers Apoptosis and Affects Gene Expressions of Murine Leukemia RAW 264.7 Cells - RS-C Wu, C-S YuAlondra MaldonadoNo ratings yet
- Characterization Ribdnucleoprotein AutoantigenDocument9 pagesCharacterization Ribdnucleoprotein AutoantigenLe DungNo ratings yet
- Paper - Pleurotus CultivationDocument7 pagesPaper - Pleurotus CultivationSudhir NawathyeNo ratings yet
- Plant Rarity Lab ReportDocument15 pagesPlant Rarity Lab Reportapi-529961637No ratings yet
- Quantitative Lignan: Identification and Estimation BovineDocument5 pagesQuantitative Lignan: Identification and Estimation BovineSaeed KhazayelNo ratings yet
- Ibsc22 WP90Document13 pagesIbsc22 WP90rjgomes79No ratings yet
- Balasubramanian 2013 Cloning and Molecular Analysis of The Aspartic Protease Sc-ASP110 GeneDocument8 pagesBalasubramanian 2013 Cloning and Molecular Analysis of The Aspartic Protease Sc-ASP110 GeneIliana Zazueta OjedaNo ratings yet
- Koo Ken 2014Document7 pagesKoo Ken 2014Perla Andrea Urriola AraosNo ratings yet
- Full TextDocument11 pagesFull TextNatasha KaurNo ratings yet
- Article 9 Fatma PDFDocument8 pagesArticle 9 Fatma PDFComan GigiNo ratings yet
- ProQuestDocuments 2013 05 30Document8 pagesProQuestDocuments 2013 05 30limfositNo ratings yet
- Thyroid Hormone SummaryDocument11 pagesThyroid Hormone SummaryAishwarya SinghNo ratings yet
- Full-Process Radiosensitization Based On Nanoscale Metal-Organic FrameworksDocument29 pagesFull-Process Radiosensitization Based On Nanoscale Metal-Organic FrameworksIin Tirta SunartaNo ratings yet
- JMM5207 569Document4 pagesJMM5207 569Olivia SuritnoNo ratings yet
- A New Approach To Immunological Sexing of Sperm Blecher 1999Document13 pagesA New Approach To Immunological Sexing of Sperm Blecher 1999Sergio L.No ratings yet
- Comparison of Antisense Oligonucleotides and Sirnas in Cell Culture and in VivoDocument5 pagesComparison of Antisense Oligonucleotides and Sirnas in Cell Culture and in VivoDeeksha Baliyan MalikNo ratings yet
- Flow CytometryDocument10 pagesFlow CytometryDaniel BarrigaNo ratings yet
- Flowcytometry PrinciplesDocument189 pagesFlowcytometry PrinciplescandiddreamsNo ratings yet
- Heterochromatin On The Inactive X Chromosome Delays Replication Timing Without Affecting Origin UsageDocument6 pagesHeterochromatin On The Inactive X Chromosome Delays Replication Timing Without Affecting Origin Usageapi-3700537No ratings yet
- Fundamentals Week 6 TutoringDocument38 pagesFundamentals Week 6 Tutoringsas345sas345No ratings yet
- An Inexpensive High-Throughput MethodDocument9 pagesAn Inexpensive High-Throughput MethodjeanlouiskouakouNo ratings yet
- Jas 28 3 JAN0280030369Document6 pagesJas 28 3 JAN0280030369Roxana MariaNo ratings yet
- Calorie RestrictionDocument18 pagesCalorie RestrictionJuan Felipe QuinteroNo ratings yet
- Gujral FCM 2Document128 pagesGujral FCM 2candiddreamsNo ratings yet
- DNA Repair Protein Involved in Heart and Blood DevelopmentDocument12 pagesDNA Repair Protein Involved in Heart and Blood DevelopmentSol Jumaide WerbleNo ratings yet
- Cell-Mediated Immunity To Liver Antigen in Toxic Liver InjuryDocument8 pagesCell-Mediated Immunity To Liver Antigen in Toxic Liver InjurySurya PrakashNo ratings yet
- The Distribution and Behaviour of Intravenously inDocument10 pagesThe Distribution and Behaviour of Intravenously inBruce RoserNo ratings yet
- Differential Protein Expression During Tail Regeneration of Anolis CarolinensisDocument11 pagesDifferential Protein Expression During Tail Regeneration of Anolis Carolinensisbill reevesNo ratings yet
- Shatalin SomDocument25 pagesShatalin Somreinafeng1No ratings yet
- Collins UpdateDocument33 pagesCollins UpdateThompson E IghaloNo ratings yet
- Suppression of Human Immunodeficiency Virus Replication by Ascorbate in Chronically and Acutely Infected Cells.Document5 pagesSuppression of Human Immunodeficiency Virus Replication by Ascorbate in Chronically and Acutely Infected Cells.Hector Javier Chavez RamirezNo ratings yet
- A Noninvasive, Direct Real Time PCR Method For Sex Determination in Multiple Avian SpeciesDocument3 pagesA Noninvasive, Direct Real Time PCR Method For Sex Determination in Multiple Avian SpeciesFatima 999No ratings yet
- Anti-Angiogenic and Antibacterial Property of Bangus Bile Research PaperDocument15 pagesAnti-Angiogenic and Antibacterial Property of Bangus Bile Research PaperRogelio PontejoNo ratings yet
- Repeat Regions in MT DnaDocument9 pagesRepeat Regions in MT DnaVeerendra UppalapatiNo ratings yet
- Research Into Cancer Signalling.20130405.131511Document2 pagesResearch Into Cancer Signalling.20130405.131511ploughdew7No ratings yet
- Zehner Et Al., 2004 - Genetic Identification of Forensically Important Flesh FliesDocument3 pagesZehner Et Al., 2004 - Genetic Identification of Forensically Important Flesh FliesJean-Bernard HuchetNo ratings yet
- NEJM appendixPDF2020Document7 pagesNEJM appendixPDF2020Marin AndreiNo ratings yet
- Insulin Secretion From Perifused Rat Pancreatic PseudoisletsDocument7 pagesInsulin Secretion From Perifused Rat Pancreatic PseudoisletskalwusaidiNo ratings yet
- Curriculum Vitae: Wimm PiDocument6 pagesCurriculum Vitae: Wimm PiJonathan RobertsNo ratings yet
- Adma202307006 Sup 0001 SuppmatDocument24 pagesAdma202307006 Sup 0001 SuppmatNica ValiNo ratings yet
- Ribosome-Inactivating Protein and Apoptosis: Abrin Causes Cell Death Via Mitochondrial Pathway in Jurkat CellsDocument8 pagesRibosome-Inactivating Protein and Apoptosis: Abrin Causes Cell Death Via Mitochondrial Pathway in Jurkat CellsKusuma WardaniNo ratings yet
- Muller Cell Expression of Gliol Fibrillory Acidic Protein Offer Genetic and Experimental Photoreceptor Degeneration in The Rat RetinaDocument8 pagesMuller Cell Expression of Gliol Fibrillory Acidic Protein Offer Genetic and Experimental Photoreceptor Degeneration in The Rat Retinaosvaldo2494No ratings yet
- Charcot-Marie-Tooth Neuropathy Type 2Document38 pagesCharcot-Marie-Tooth Neuropathy Type 2Maria Daniela VlaicuNo ratings yet
- Lab Anim-1998-Asano-214-8Document6 pagesLab Anim-1998-Asano-214-8Лилия ПоляковаNo ratings yet
- Adiliaghdam Et Al. 2022 - Sciimmunol - Abn6660 - SMDocument26 pagesAdiliaghdam Et Al. 2022 - Sciimmunol - Abn6660 - SMAchille BroggiNo ratings yet
- Morphological Variation in a Population of the Snake, Tantilla gracilis Baird and GirardFrom EverandMorphological Variation in a Population of the Snake, Tantilla gracilis Baird and GirardNo ratings yet
- Effect of Cleaners and Sanitizers On Listeria Monocytogenes Attached To Product Contact SurfacesDocument6 pagesEffect of Cleaners and Sanitizers On Listeria Monocytogenes Attached To Product Contact SurfacesKhela Pagol ManushNo ratings yet
- Singlet OxygenDocument62 pagesSinglet Oxygenhamid_hinnaNo ratings yet
- Hydrogen Peroxide PDFDocument20 pagesHydrogen Peroxide PDFzibaNo ratings yet
- Alkaloids 1 1Document276 pagesAlkaloids 1 1عبدو عبدوNo ratings yet
- Hard SoftDocument24 pagesHard SoftMuhammad FahmiNo ratings yet
- Module (Amino Acids and Proteins)Document18 pagesModule (Amino Acids and Proteins)Edgie JunelaNo ratings yet
- TGA Spectra by Akmal JavedDocument23 pagesTGA Spectra by Akmal Javedasad abbasNo ratings yet
- Lesson 8 - Amino AcidDocument10 pagesLesson 8 - Amino Acidchristian Jay HorseradaNo ratings yet
- 2 ScribdDocument7 pages2 ScribdROG PUBGNo ratings yet
- Lecture-10-Clous and Merox ProcessesDocument43 pagesLecture-10-Clous and Merox ProcessesAnilKumar100% (2)
- 3 LipidsDocument42 pages3 LipidsSyifaAnandaNo ratings yet
- Abaris Composite Bonding OshkoshDocument51 pagesAbaris Composite Bonding Oshkoshv1alfredNo ratings yet
- Biobased Epoxy Synthesized From A Vanillin Derivative and Its Reinforcement Using Lignin-Containing Cellulose NanofibrilsDocument10 pagesBiobased Epoxy Synthesized From A Vanillin Derivative and Its Reinforcement Using Lignin-Containing Cellulose NanofibrilsMaria FilipNo ratings yet
- 2018 May TZ2 Paper 3 HL Chemistry MarkschemeDocument10 pages2018 May TZ2 Paper 3 HL Chemistry MarkschememounishadmNo ratings yet
- Epoxy Resin ManufactureDocument17 pagesEpoxy Resin Manufactureradhika khannaNo ratings yet
- Dry CleaningDocument11 pagesDry CleaningNailah KaharNo ratings yet
- VCO Standard Flyer - VCO Standard APCCDocument2 pagesVCO Standard Flyer - VCO Standard APCCRiza Muhammad100% (1)
- Parker O-Ring Division MaterialsDocument10 pagesParker O-Ring Division Materialsseeralan_1986No ratings yet
- Bioinorganic Chemistry LectureDocument28 pagesBioinorganic Chemistry LectureKamal ChoudhuryNo ratings yet
- Pengaruh PH Ekstraksi Terhadap Rendemen, Sifat Fisiko-Kimia Dan Fungsional Konsentrat Protein KACANG GUDE (Cajanus Cajan (L.) Millsp.)Document11 pagesPengaruh PH Ekstraksi Terhadap Rendemen, Sifat Fisiko-Kimia Dan Fungsional Konsentrat Protein KACANG GUDE (Cajanus Cajan (L.) Millsp.)Rezaa RezNo ratings yet
- Translation WORKSHEETDocument2 pagesTranslation WORKSHEETPol Marasigan BanzonNo ratings yet
- Abdel-Moneim (2007) PDFDocument24 pagesAbdel-Moneim (2007) PDFinsaniachusnaNo ratings yet
- Biological Activity of Essential Oils andDocument61 pagesBiological Activity of Essential Oils and4negero100% (1)
- Show My Learning 1Document14 pagesShow My Learning 1ح UAE2No ratings yet
- 3Document18 pages3Coral Cruz MadridNo ratings yet
- 11 Chemistry Notes Chapter 12Document16 pages11 Chemistry Notes Chapter 12avni AroraNo ratings yet
- Effects of PH, Temperature and Concentration On Enzyme PepsinDocument4 pagesEffects of PH, Temperature and Concentration On Enzyme PepsinDebrah DebbieNo ratings yet
- Tunnel Segmental Lining Durability (Fastening)Document10 pagesTunnel Segmental Lining Durability (Fastening)Cristián GonzálezNo ratings yet
- USP Codigo Columnas PDFDocument2 pagesUSP Codigo Columnas PDFleslieNo ratings yet
- 2016 Avanti Product GuideDocument2 pages2016 Avanti Product GuideMunicipal Sewer & Water MagazineNo ratings yet