Professional Documents
Culture Documents
Halo Alkanes and Arenes
Halo Alkanes and Arenes
Uploaded by
Rahul Sharma0 ratings0% found this document useful (0 votes)
155 views8 pages1. The document discusses the classification, nomenclature, preparation, properties and reactions of haloalkanes and haloarenes.
2. It describes the nucleophilic substitution reactions of haloalkanes which can proceed by either SN1 or SN2 mechanisms. SN1 reactions give racemic mixtures while SN2 reactions invert configuration.
3. The stereochemistry of nucleophilic substitution reactions is also discussed including terms like chiral centers, enantiomers and retention or inversion of configuration.
Original Description:
class 12th organic chapter 1st notes
Original Title
Halo Alkanes and arenes
Copyright
© © All Rights Reserved
Available Formats
PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this Document1. The document discusses the classification, nomenclature, preparation, properties and reactions of haloalkanes and haloarenes.
2. It describes the nucleophilic substitution reactions of haloalkanes which can proceed by either SN1 or SN2 mechanisms. SN1 reactions give racemic mixtures while SN2 reactions invert configuration.
3. The stereochemistry of nucleophilic substitution reactions is also discussed including terms like chiral centers, enantiomers and retention or inversion of configuration.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
Download as pdf or txt
0 ratings0% found this document useful (0 votes)
155 views8 pagesHalo Alkanes and Arenes
Halo Alkanes and Arenes
Uploaded by
Rahul Sharma1. The document discusses the classification, nomenclature, preparation, properties and reactions of haloalkanes and haloarenes.
2. It describes the nucleophilic substitution reactions of haloalkanes which can proceed by either SN1 or SN2 mechanisms. SN1 reactions give racemic mixtures while SN2 reactions invert configuration.
3. The stereochemistry of nucleophilic substitution reactions is also discussed including terms like chiral centers, enantiomers and retention or inversion of configuration.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
Download as pdf or txt
You are on page 1of 8
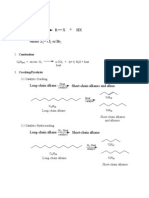
Halo alkanes and Halo aerenes
1. Classification based on no of halogen atom
Mono: - RX, di - , tri 1, 2, 3 tri halo propane
2. Classification of mono substituted haloalkane sp
3
-> 1
0
, 2
0
, 3
0
Allylic -> CH
2
=CH-CH
2
X, Benzylic Benzylic chloride 1-phenyl
Chloro methane
3. sp
2
Vinylic CH
3
CH=CX , arylic C
6
H
5
X
4. Nomenclature:-
o-, m-, p- dichloro Benzene
5. Preparation of alkyl halide: - (i) from alcohol
(ii) From Hydrocarbon: - Free radicle/ Electrophilic substitution/ sandmeyers
reaction/ alkene
(a) Free radicle
(b)Electrophilic Substitution
(c)Sandmeyers reaction
(d) Form alkene Markonikovs rule CH
3
CH=CH
2
+ HI -----> CH
3
-CHI-CH
3
(iii) Halogen exchange method Finkelstein
Swarts RCl/Br + AgF/H
2
F
2
/CoF
2
/SbF
3
-------> RF + - - - -
6. Properties:- (i) RCl Colourless & RBr/RI coloured when exposed to light
(ii) b.p. RI > RBr > RCl > RF (due to molar mass) b.p. decreases on branching
m.p increases on branching m.p of p- > ortho due to crystal lattice
(iii) Insoluble in water due to no hydrogen bonding.
(iv) Nucleophilic substitution reaction: - When strong nucleophile replaces weaker
nucleophile is called nucleophilic substitution reaction.
RX + NaOH/HOH/NaOR/NaI/NH
3
-------> ROH/ROH/R-O-R/RI/RNH
2
RX + KCN/AgCN/KNO/AgNO
2
/RCOOAg/LiAlH
4
---------> RCN/RNC/R-ONO/RNO
2
/
RCOOR/RH
KCN form RCN while AgCN form RNC since KCN is ionic which provide CN but
AgCN is covalent, therefore e
-
pair on N easily donated.
(v) Elimination reaction: - When RX react with alc. KOH form alkene
CH
3
CH
2
Cl + alc.KOH ------> CH
2
=CH
2
+ KCl
Saytzeff rule: - When assymetric alkylhalide react with alc.KOH form alkene
major product is (which has more alkyl gp)
(vi) Reaction with metal: - Alkyl halide reacts with metal to form organo-metallic
compound. I
st
such compound was formed by Victor Grignard called Grignard
reagent.
RX + Mg ----dry ether-------> RMgX which is very reactive. React with
moisture to form alkane
RMgX + H
2
O ---> RH + Mg(OH)X
(vii) Wurtz reaction: - 2RX + Na ----dry ether----> R-R + 2NaX
Fittig reaction 2C
6
H
5
X + 2Na ----> C
6
H
5
-C
6
H
5
+ 2NaX
Wurtz fittig reaction: - C
6
H
5
X + RX + 2Na -----> C
6
H
5
R + 2NaX
8. Chemical reactions of Haloarenes: - (i) Nucleophilic substitution reaction
(ii) Electrophilic substitution reaction
(iii) Reaction with metal
(i) Nucleophilic substitution reaction: - Halo arenes doesnot give since
(a) C-Cl is strong partial = bond (169pm but in RX C-Cl = 177pm)
(b) phenyl cation is unstable due to resonance
But under drastic condition (or in presence of e
-
withdrawing gp NO
2
) give
(ii) Electrophilic substitution reaction: - give O, -P compound
(a) Halogenation
(b) Nitration
(c) Sulphonation
(d) Friedel Craft alkylation/ acylation
(iii) Reaction with metal Fittig/ Wurtz Fittig reaction
9. Polyhalogen compounds
(a) Dichloromethane CH
2
Cl:- used as solvent in paint remover/metal cleaning
drugs but harmful to central nervous system.
(b) Chloroform CHCl
3
:- used as solvent for fats, alkaloids, I
2
in Freon R-22,
anaesthatics. Damage liver, react with O
2
to form poisonous gas phosgene
CHCl
3
+ O
2
-----light-------> 2COCl
2
+ 2HCl (Carbonylchloride)
Therefore, stored in dark, full bottle (Since no air)
(c) Iodoform CHI
3
:- used as anaesthatic but give foul odour of I
2
(d) Carbon tetrachloride CCl
4
:- used as solvent, refrigerant, propellant, fire
extinguisher but exposure causes liver cancer, nausea, vomiting, coma.
(e) Freons chlorofluoro carbon CF
2
Cl
2
:- Used as propellents refrigerator, air
conditioning, upset ozone layer.
(f) D.D.T p,p-dichloro,diphenyl trichloro ethane
Paul muller was awarded noble prize on it
Used against mosquito and lice
Highly toxic to fish
10. Mechanism of nucleophilic substitution reaction:- Stronger nucleophile
replace weaker nucleophile in alkyl halide is called nucleophilic substitution
reaction. Reaction proceed through two mechanism
(a) S
N
2 mechanism : - (i) rate depend on conc. Of two reactant (2 order)
(ii) Reaction proceed through single step. Breaking of bond and incoming
nucleophile take place simultaneously.
(iii) Due to steric effect reactivity 1
0
>2
0
>3
0
/ RI > RBr > RCl > RF
(iv) Product has inverted configuration as compared to reactant
:Nu attach on opposite side of halide
(b) S
N
1 mechanism (i) Rate depend on conc. of one reactant (Ist order)
(ii) Reaction proceed in two step formation of cation then cation react with
nucleophile
(iii) Reactivity 3
0
>2
0
>1
0
/ RI > RBr > RCl > RF
(iv) Give Product as recemic mixture. Since carbocation formed in slow step
Therefore, :Nu may attach to either side ( may same configuration or inverted to
reactant)
(v) Require polar protic solvent (H
2
O, ROH, RCOOH)
(vi) (CH
3
)
3
-X ----slow---> (CH
3
)
3
C
+
+ X
-
(CH
3
)
3
C
+
+ :Nu --> (CH
3
)
3
C-Nu:
(Produce E)
(vii) Allylic (CH
2
=CH-CH
2
X) & benzylic show high reactivity in S
N
1
11. Stereochemical aspect of nucleophilic substitution reaction
(i) Plane polarised light Light obtained through nicol prism (Calcite ore CaCO
3
) is
called plane polarised light.
(ii) Optical activity :- When plane plorised light is passed through certain
compound it rotated to right direction called dextro (d,+) and if rotated to left
direction called leavo(l,-). These compounds are called optically active and the
phenomenon is called optical activity.
(iii) Asymmetric centre :- If all four substituent attached with C-atom are different
then it is called asymmetric C-atom.
Assymetry is responsible for optical activity.
(iv) Chirality :- The objects which form non superimposable image on their mirror
image is called chiral and the property is called chirality. Eg p, q, butane 2-ol, 2,3
dihydroxy propanal
(v) Achirality :- Objects which form superimposable image on their mirror image is
called achiral and the property is called achirality. Eg. Propan-2-ol
(vi) Enantiomers: - Optical active isomers are called enantiomers (d and l)
(vii) Recemic mixture: - Mixture of two enantiomers (d and l) in equal proportion
have zero optical rotation is called racemic mixture (dl of +-) and the process is
called recemisation.
(viii) Retention: - Retention of configuration is, preservation of integrity of spatial
arrangement of bond to asymmetric centre, during chemical reaction i.e product
have same configuration.
(ix) Inversion: - If bond broken and configuration change during reaction is called
inversion.
You might also like
- Simulation of Chlorobenzene Plant by Using Aspen PlusDocument29 pagesSimulation of Chlorobenzene Plant by Using Aspen PlusAshish Mishra100% (3)
- SET-NET Pericyclic ReactionsDocument61 pagesSET-NET Pericyclic ReactionsBapu ThoratNo ratings yet
- Ch16 Benzene ChemistryDocument16 pagesCh16 Benzene ChemistrySherlHolmesNo ratings yet
- CBSE Class 12 Chem Notes Question Bank Haloalkanes and Haloarenes PDFDocument18 pagesCBSE Class 12 Chem Notes Question Bank Haloalkanes and Haloarenes PDFhehe11No ratings yet
- Organometallics and Catalysis - IVDocument36 pagesOrganometallics and Catalysis - IVankit guptaNo ratings yet
- Ch6 HALOALKANES AND HALOARENES HHW WORKSHEETDocument4 pagesCh6 HALOALKANES AND HALOARENES HHW WORKSHEETAaditya SharmaNo ratings yet
- Hsslive-Xii-Chem-10. Alkyhalides and Aryl HalidesDocument13 pagesHsslive-Xii-Chem-10. Alkyhalides and Aryl HalidesHakim AbbasNo ratings yet
- Worksheet-Haloalkanes and HaloarenesDocument5 pagesWorksheet-Haloalkanes and HaloarenesAslamNo ratings yet
- Alcohol Phenol & EtherDocument13 pagesAlcohol Phenol & EtherAbir DuttaNo ratings yet
- 11 Chemistry Notes Chapter 3Document13 pages11 Chemistry Notes Chapter 3Gyani ChachaNo ratings yet
- Molecular RearrangementsDocument158 pagesMolecular RearrangementsRamesh Katkam75% (4)
- Formulae For: Haloalkane & Hal OarenesDocument9 pagesFormulae For: Haloalkane & Hal OarenesSâmïr Kumar MundariNo ratings yet
- CBSE Class-12 Chemistry Quick Revision Notes Chapter-11: Alcohols, Phenols and Ethers Structure of AlcoholsDocument8 pagesCBSE Class-12 Chemistry Quick Revision Notes Chapter-11: Alcohols, Phenols and Ethers Structure of AlcoholsSAKET TYAGI100% (1)
- Sni, Nighbouring GP Participation & E1cbDocument19 pagesSni, Nighbouring GP Participation & E1cbsaheedvkNo ratings yet
- HYDROCARBONS Plusone HssliveDocument13 pagesHYDROCARBONS Plusone HssliveAthulRKrishnanNo ratings yet
- Protection Groups in Organic PDFDocument67 pagesProtection Groups in Organic PDFToàn MinhNo ratings yet
- Unit - 1 Lesson - 1Document271 pagesUnit - 1 Lesson - 1Rakesh SharmaNo ratings yet
- Goc and Isomerism Notes - PMD - 1 PDFDocument46 pagesGoc and Isomerism Notes - PMD - 1 PDFrutvik bhoraniyaNo ratings yet
- Classification of Organometallic CompoundsDocument28 pagesClassification of Organometallic CompoundsDingetegna GodanaNo ratings yet
- 12 Chemistry Impq CH10 Haloalkanes and Haloarenes 01Document10 pages12 Chemistry Impq CH10 Haloalkanes and Haloarenes 01Swaroop Surendra0% (1)
- Reactions of AlkanesDocument1 pageReactions of AlkanesnofacejackNo ratings yet
- Electron Spectra PDFDocument42 pagesElectron Spectra PDFFaizanAli100% (1)
- Chemical Synthesis of Chalcones by Claisen-Schmidt Condensation Reaction and Its CharacterizationDocument5 pagesChemical Synthesis of Chalcones by Claisen-Schmidt Condensation Reaction and Its CharacterizationIJRASETPublicationsNo ratings yet
- Free Download Here: Advanced Inorganic Chemistry P L Soni PDFDocument2 pagesFree Download Here: Advanced Inorganic Chemistry P L Soni PDFNirbhay KumarNo ratings yet
- Name ReactionsDocument10 pagesName ReactionsParam SoniNo ratings yet
- Organic Give Reasons (2023-24)Document13 pagesOrganic Give Reasons (2023-24)xefayo4337No ratings yet
- Chapter 6: Organohalogens: Alkyl Halide Vinyl Halide Aryl HalideDocument13 pagesChapter 6: Organohalogens: Alkyl Halide Vinyl Halide Aryl HalidecikguhafidzuddinNo ratings yet
- The Oxidation States of Tin and Relative Stabilities of Tin and LeadDocument3 pagesThe Oxidation States of Tin and Relative Stabilities of Tin and LeadDaniel Rodman100% (1)
- Possible electrolytes for SOFC: synthesis and characterization of Bi3-xMexTaO7-δ and others fluorite-related phasesDocument95 pagesPossible electrolytes for SOFC: synthesis and characterization of Bi3-xMexTaO7-δ and others fluorite-related phasesnicheltetracarbonileNo ratings yet
- MGOM 1 Introduction To OrganometallicsDocument70 pagesMGOM 1 Introduction To OrganometallicsIyan Maulana100% (1)
- 11.alcohol, Phenol & Ethers Colour Booklet PDFDocument59 pages11.alcohol, Phenol & Ethers Colour Booklet PDFMridu BhandariNo ratings yet
- Reaction IntermediatesDocument5 pagesReaction Intermediatescybercp100% (1)
- Shapiro Reaction and BamfordDocument13 pagesShapiro Reaction and BamfordharishNo ratings yet
- 1.aldehydes, Ketones and Carboxylic AcidsDocument117 pages1.aldehydes, Ketones and Carboxylic AcidsKRISHNARJUNA NNo ratings yet
- 12 Chemistry Notes Ch12 Aldehydes Ketones and CarboxylicacidDocument11 pages12 Chemistry Notes Ch12 Aldehydes Ketones and Carboxylicacidankajkumar100% (1)
- Electrolytic Conduction NotesDocument94 pagesElectrolytic Conduction NotesPaolo Peps50% (2)
- Carboxylic Acids ..Document27 pagesCarboxylic Acids ..Mariam HamedNo ratings yet
- Metal Nitrosyl ComplexDocument6 pagesMetal Nitrosyl Complexsusanenulfah100% (2)
- Organic Synthesis Via Enolates BSC III CH IVDocument10 pagesOrganic Synthesis Via Enolates BSC III CH IVSanjay ShirodkarNo ratings yet
- Mechanism and Structure in Organic Chemistry-NGP-Gould PDFDocument60 pagesMechanism and Structure in Organic Chemistry-NGP-Gould PDFAJIT CHAUDHARINo ratings yet
- UmpolungDocument28 pagesUmpolungmeauna100% (1)
- (2099) Lecture Notes P Block 15 16 E.pdf - TMPDocument39 pages(2099) Lecture Notes P Block 15 16 E.pdf - TMPRamJiPandeyNo ratings yet
- Named ReactionsDocument72 pagesNamed ReactionsVenkatesh Chukkala0% (1)
- Synthesis and Application of Eosin: Kabeer Fatima, Sofia Nosheen, Humera and Munazza AzharDocument7 pagesSynthesis and Application of Eosin: Kabeer Fatima, Sofia Nosheen, Humera and Munazza AzharPetr Svoboda67% (3)
- Transeffect of Coordination CompoundsDocument38 pagesTranseffect of Coordination CompoundsAnish KumarNo ratings yet
- Organometallic Reactions and Catalysis - 2Document34 pagesOrganometallic Reactions and Catalysis - 2Irma AlfaBetNo ratings yet
- Introduction To Lattice EnergyDocument16 pagesIntroduction To Lattice EnergyRohan AhmedNo ratings yet
- Hydrides and DihydrogenDocument33 pagesHydrides and DihydrogenAnonymous lClX1bNo ratings yet
- S Block ElementsDocument8 pagesS Block ElementsSwati Jadhav100% (3)
- Mcmurry 12Document62 pagesMcmurry 12Ngurah MahasviraNo ratings yet
- 861523122971Document33 pages861523122971Hamzah AbuadmahNo ratings yet
- Thermodynamic Versus Kinetic Reaction Control, Diffusion ControlDocument7 pagesThermodynamic Versus Kinetic Reaction Control, Diffusion ControlenvirocompNo ratings yet
- Multiple Choice Questions: Question Bank Class: Xii, Chemistry Unit 4: Haloalknaes & HaloarenesDocument27 pagesMultiple Choice Questions: Question Bank Class: Xii, Chemistry Unit 4: Haloalknaes & HaloarenesAkshita BoroNo ratings yet
- Important Questions For CBSE Class 10 Science Chapter 2Document32 pagesImportant Questions For CBSE Class 10 Science Chapter 2Shaurya 5116jNo ratings yet
- Aldehydes, Ketones and Carboxylic Acids-Anil-hssliveDocument16 pagesAldehydes, Ketones and Carboxylic Acids-Anil-hssliveRanit Mukherjee100% (1)
- Organic ReactionsDocument44 pagesOrganic ReactionsJanhavi PatilNo ratings yet
- Organic Chemistry For USTH Students Lecture 2: Electrophilic Addition To C CDocument107 pagesOrganic Chemistry For USTH Students Lecture 2: Electrophilic Addition To C CminhminhNo ratings yet
- Chap. 4: Non-Aqueous SolventsDocument9 pagesChap. 4: Non-Aqueous Solventsmd mithu100% (1)
- Chemistry Study Material XII - Part 2 - 0Document91 pagesChemistry Study Material XII - Part 2 - 0krishnakumar01928374No ratings yet
- Xii Ch10 Haloalkane and HaloarenesDocument4 pagesXii Ch10 Haloalkane and HaloarenesYash RajNo ratings yet
- 12 Chemistry Keypoints Revision Questions Chapter 10 PDFDocument13 pages12 Chemistry Keypoints Revision Questions Chapter 10 PDFSahil Kalra100% (1)
- C10 PDFDocument9 pagesC10 PDFRahul SharmaNo ratings yet
- Some Special Functions: Dr. Suresh Kumar, BITS PilaniDocument14 pagesSome Special Functions: Dr. Suresh Kumar, BITS PilaniRahul SharmaNo ratings yet
- Boundary Value Problems: Dr. Suresh Kumar, BITS PilaniDocument11 pagesBoundary Value Problems: Dr. Suresh Kumar, BITS PilaniRahul SharmaNo ratings yet
- Second Order Differential Equations: Dr. Suresh Kumar, BITS PilaniDocument16 pagesSecond Order Differential Equations: Dr. Suresh Kumar, BITS PilaniRahul SharmaNo ratings yet
- Aldehyde, Ketone and Carboxylic Acid Class 12 CbseDocument8 pagesAldehyde, Ketone and Carboxylic Acid Class 12 CbseRahul SharmaNo ratings yet
- First Order Differential Equations: Dr. Suresh Kumar, BITS PilaniDocument11 pagesFirst Order Differential Equations: Dr. Suresh Kumar, BITS PilaniRahul SharmaNo ratings yet
- Xam Idea ChemDocument331 pagesXam Idea ChemLokesh Badpanda86% (7)
- The RC CircuitDocument2 pagesThe RC CircuitRahul SharmaNo ratings yet
- Redox Reactions1Document18 pagesRedox Reactions1Rahul SharmaNo ratings yet
- Chemical Kinetics CBSE Class XII NotesDocument5 pagesChemical Kinetics CBSE Class XII NotesRahul SharmaNo ratings yet
- Mathematics Complex Number MCQDocument7 pagesMathematics Complex Number MCQRahul SharmaNo ratings yet
- Chemistry - Classification and NomenclatureDocument20 pagesChemistry - Classification and NomenclatureDevesh AgrawalNo ratings yet
- Modern Synthesis Processes and Reactivity of Fluorinated Compounds 1St Edition Edition Groult Henri Ed Full ChapterDocument67 pagesModern Synthesis Processes and Reactivity of Fluorinated Compounds 1St Edition Edition Groult Henri Ed Full Chapterraymond.cobb946100% (15)
- OrchemDocument63 pagesOrchemQuerryflurrNo ratings yet
- Free Radical Substitution and Electrophilic AdditionDocument17 pagesFree Radical Substitution and Electrophilic Additionchicko33No ratings yet
- Phenanthrene Notes 1Document12 pagesPhenanthrene Notes 1SMART STUDY Maharashtra boardNo ratings yet
- Alcohols and PhenolsDocument72 pagesAlcohols and PhenolsChandra ReddyNo ratings yet
- Organic Chemistry I SyllabusDocument4 pagesOrganic Chemistry I Syllabuspaleoman8No ratings yet
- Chem ImpDocument34 pagesChem ImpHasanNo ratings yet
- Chap 2Document108 pagesChap 2Irfan AzaharNo ratings yet
- CHEMISTRYDocument131 pagesCHEMISTRYhamidNo ratings yet
- 11 Key Carbonyl MechanismsDocument1 page11 Key Carbonyl Mechanismsfali92100% (1)
- Reaction of AlkenesDocument27 pagesReaction of AlkenesHui JingNo ratings yet
- Alkene and Alkyne - by Resonance PDFDocument45 pagesAlkene and Alkyne - by Resonance PDFPrasad Yarra100% (1)
- L2 - Aromatic Electrophilic Substitution PDFDocument32 pagesL2 - Aromatic Electrophilic Substitution PDFPadamNo ratings yet
- Borrowing HydrogenDocument7 pagesBorrowing HydrogenVinayak KhairnarNo ratings yet
- 4.true False-Haloalknaes & HaloarenesDocument7 pages4.true False-Haloalknaes & HaloarenesrajeshwariNo ratings yet
- 24 Organic Review 2023Document37 pages24 Organic Review 2023Elyssa En YingNo ratings yet
- Textbook Organic Chemistry Principles and Mechanisms 1St Edition Joel Karty Ebook All Chapter PDFDocument53 pagesTextbook Organic Chemistry Principles and Mechanisms 1St Edition Joel Karty Ebook All Chapter PDFrachel.watson265100% (9)
- IAL - Chemistry - SB2 - Mark Scheme - T18Document2 pagesIAL - Chemistry - SB2 - Mark Scheme - T18salmaNo ratings yet
- Haloalkanes and Haloarenes - MCQSDocument3 pagesHaloalkanes and Haloarenes - MCQSDivyam GargNo ratings yet
- Hydrocarbon MAIN NotesDocument25 pagesHydrocarbon MAIN NotesHamad FarooqueNo ratings yet
- Chem Paper Thermo and Reaction Mechanism.Document12 pagesChem Paper Thermo and Reaction Mechanism.Harshvardhan GavankarNo ratings yet
- Reasoning Ques in Organic ChemistryDocument14 pagesReasoning Ques in Organic ChemistryRIHINBHATNAGAR50% (2)
- 12th Class Chemistry Chapter 12Document17 pages12th Class Chemistry Chapter 12shahmeerbhullar642No ratings yet
- CBSE Class 12 Alcohol Phenol and Ether Study NotesDocument378 pagesCBSE Class 12 Alcohol Phenol and Ether Study NotesV T PRIYANKANo ratings yet
- Assignment 2 - Task 3Document63 pagesAssignment 2 - Task 3peterNo ratings yet
- Review Questions: Medicinal Chemistry 300550Document49 pagesReview Questions: Medicinal Chemistry 300550vanyarufusNo ratings yet
- An Overview of Organic ReactionsDocument80 pagesAn Overview of Organic Reactions110003551 110ANo ratings yet
- Bpo C Chapter 10Document55 pagesBpo C Chapter 10DewiSugiartiNo ratings yet