Professional Documents
Culture Documents
0 ratings0% found this document useful (0 votes)
17 views1 s2.0 S0040609096087494 Main
1 s2.0 S0040609096087494 Main
Uploaded by
Luis Alejandro TafurThe electrical resistivity of TiN prepared at 400 degC with 8% of nitrogen flow was 24 / z / 2 cm, equivalent to that of bulk TiN. Films showed low resistivity at higher nitrogen flow ( 10% and 12%)
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
You might also like
- Report of Infrared Plastic Solar CellDocument22 pagesReport of Infrared Plastic Solar CellAjay Mv85% (54)
- Coatings: Microstructure and Mechanical Properties of Tan Thin Films Prepared by Reactive Magnetron SputteringDocument16 pagesCoatings: Microstructure and Mechanical Properties of Tan Thin Films Prepared by Reactive Magnetron SputteringTrinh ThinhNo ratings yet
- Stress and Oxidation Behaviours of R.f.-Sputtered (Ti, A1) N FilmsDocument8 pagesStress and Oxidation Behaviours of R.f.-Sputtered (Ti, A1) N FilmsIgor Oliveira NascimentoNo ratings yet
- Effects of Nitrogen Flow Rates On The Growth Morphology of TiAlN Films Prepared by An Rf-Reactive Sputtering TechniqueDocument5 pagesEffects of Nitrogen Flow Rates On The Growth Morphology of TiAlN Films Prepared by An Rf-Reactive Sputtering TechniquePhysics MPSTMENo ratings yet
- Structural and Electrochemical Impedance Spectroscopic Studies On Reactive Magnetron Sputtered Titanium Oxynitride (Tion) Thin FilmsDocument6 pagesStructural and Electrochemical Impedance Spectroscopic Studies On Reactive Magnetron Sputtered Titanium Oxynitride (Tion) Thin FilmsJussier VitorianoNo ratings yet
- 5ungureanu PDFDocument5 pages5ungureanu PDFsuganyaNo ratings yet
- Art 2009Document6 pagesArt 2009Carito CastilloNo ratings yet
- Hot Filament Assisted CVD of Titanium Nitride FilmsDocument6 pagesHot Filament Assisted CVD of Titanium Nitride FilmsMagui Ros MonNo ratings yet
- 1.deposition of Tantalum Nitride Thin Films by D.C. Magnetron SputteringDocument6 pages1.deposition of Tantalum Nitride Thin Films by D.C. Magnetron SputteringNishat ArshiNo ratings yet
- Preparation and Characterization of TiO2 Nanotube Arrays Via Anodization of Titanium Films Deposited On FTO Conducting Glass at Room TemperatureDocument7 pagesPreparation and Characterization of TiO2 Nanotube Arrays Via Anodization of Titanium Films Deposited On FTO Conducting Glass at Room TemperatureAruna RajanNo ratings yet
- Nambara 2007Document6 pagesNambara 2007Nassar Al-EssawiNo ratings yet
- Characteristics of Arc Plasma Deposited TiAlZrCN CoatingsDocument7 pagesCharacteristics of Arc Plasma Deposited TiAlZrCN CoatingsПавел МисуноNo ratings yet
- Atomic Layer Deposition of TiO2 From Tetrakis (Dimethylamino) Titanium and H2ODocument7 pagesAtomic Layer Deposition of TiO2 From Tetrakis (Dimethylamino) Titanium and H2OAilinJlsNo ratings yet
- Magnetic Conductive Outer Layer in Oxygen-Deficient Tio Single CrystalsDocument7 pagesMagnetic Conductive Outer Layer in Oxygen-Deficient Tio Single CrystalsSahil AkramNo ratings yet
- Journal Pre-Proofs: Electrochemistry CommunicationsDocument14 pagesJournal Pre-Proofs: Electrochemistry CommunicationsSandouda Bettaieb Ep RouatbiNo ratings yet
- Flash Evaporation1Document11 pagesFlash Evaporation1Sudha AsNo ratings yet
- Eis Analysis On Low Temperature Fabrication of Tio Porous Films For Dye-Sensitized Solar CellsDocument36 pagesEis Analysis On Low Temperature Fabrication of Tio Porous Films For Dye-Sensitized Solar CellsUdai SinghNo ratings yet
- Velten Et Al-2002-Journal of Biomedical Materials ResearchDocument11 pagesVelten Et Al-2002-Journal of Biomedical Materials ResearchjeffreylopezNo ratings yet
- Thin Solid Films: A.S. Daryapurkar, J.T. Kolte, P. GopalanDocument6 pagesThin Solid Films: A.S. Daryapurkar, J.T. Kolte, P. GopalanTuhinNo ratings yet
- Characterization of Sputter-Deposited Chromium Nitride Thin FilmsDocument5 pagesCharacterization of Sputter-Deposited Chromium Nitride Thin FilmsNassima JagharNo ratings yet
- Anodization P - TiO2555Document9 pagesAnodization P - TiO2555Sudip SinhaNo ratings yet
- Scripta NiTi TEM 04Document5 pagesScripta NiTi TEM 041985krNo ratings yet
- Effect of Nitrogen Flow Rate On Structure and Properties of Nanocrystalline Tin Thin Films Produced by Unbalanced Magnetron SputteringDocument8 pagesEffect of Nitrogen Flow Rate On Structure and Properties of Nanocrystalline Tin Thin Films Produced by Unbalanced Magnetron SputteringEngels OchoaNo ratings yet
- Des Feux 2004Document6 pagesDes Feux 2004kraya.sahla.mahlaNo ratings yet
- Effects of Rapid Thermal Annealing On The Local EnvironmentDocument11 pagesEffects of Rapid Thermal Annealing On The Local EnvironmentAhmedNo ratings yet
- Electrochemical Corrosion and Materials Properties of Ananthakumar2012Document9 pagesElectrochemical Corrosion and Materials Properties of Ananthakumar2012Chercheurs Permanents UrmmNo ratings yet
- Phase Transition and Properties of Ti Al N Thin ®lms Prepared by R.f.-Plasma Assisted Magnetron SputteringDocument6 pagesPhase Transition and Properties of Ti Al N Thin ®lms Prepared by R.f.-Plasma Assisted Magnetron SputteringjamesNo ratings yet
- TEM Study of TiO2Document5 pagesTEM Study of TiO2CristianAtanasiuNo ratings yet
- Effects of Tin Coating On The Corrosion of Nanostructured Ti-30Ta-Xzr Alloys For Dental ImplantsDocument6 pagesEffects of Tin Coating On The Corrosion of Nanostructured Ti-30Ta-Xzr Alloys For Dental ImplantsAtria PradityanaNo ratings yet
- Unique TiO2 Paste For High Efficiency Dye-Sensitized Solar CellsDocument3 pagesUnique TiO2 Paste For High Efficiency Dye-Sensitized Solar CellsasadNo ratings yet
- ManuscriptDocument17 pagesManuscriptfarshad davoodianNo ratings yet
- Crystal Phase Transition and Properties of Titanium Oxide Nanotube Arrays Prepared by AnodizationDocument4 pagesCrystal Phase Transition and Properties of Titanium Oxide Nanotube Arrays Prepared by AnodizationEduardo BadelNo ratings yet
- Preparation and Characterization of Peroxo Titanic Acid Solution Using TiclDocument6 pagesPreparation and Characterization of Peroxo Titanic Acid Solution Using TiclThanh ThủyNo ratings yet
- Current Applied Physics: Sciverse SciencedirectDocument4 pagesCurrent Applied Physics: Sciverse Sciencedirectslct88No ratings yet
- 10.1016@j.apsusc.2016.12.102Document5 pages10.1016@j.apsusc.2016.12.102TaryoNo ratings yet
- SYNTHESIS AND APPLICATIONS OF TiO2 NANOPARTICLESDocument10 pagesSYNTHESIS AND APPLICATIONS OF TiO2 NANOPARTICLESSoheil MirtalebiNo ratings yet
- 2015 Article 814Document6 pages2015 Article 814hrishikesh.friends4everNo ratings yet
- Enhancing The Performance of Dye-Sensitized Solar Cells Based On An Organic Dye by Incorporating TiO2 Nanotube in A TiO2 Nanoparticle FilmDocument8 pagesEnhancing The Performance of Dye-Sensitized Solar Cells Based On An Organic Dye by Incorporating TiO2 Nanotube in A TiO2 Nanoparticle FilmMuhammad Bilal QadirNo ratings yet
- Corrosion NanotubosDocument14 pagesCorrosion NanotubosjeffreylopezNo ratings yet
- International Refereed Journal of Engineering and Science (IRJES)Document5 pagesInternational Refereed Journal of Engineering and Science (IRJES)www.irjes.comNo ratings yet
- 2 - Fabrication and Photocatalytic Activity of TiO2 Nano Ber Membrane 2009Document3 pages2 - Fabrication and Photocatalytic Activity of TiO2 Nano Ber Membrane 2009Yavuz GençNo ratings yet
- Effects of Ni Doping On Photocatalytic Activity of Tio Thin Films Prepared by Liquid Phase Deposition TechniqueDocument5 pagesEffects of Ni Doping On Photocatalytic Activity of Tio Thin Films Prepared by Liquid Phase Deposition TechniqueRudolf JasonNo ratings yet
- DM TratamentDocument5 pagesDM TratamentLiliana GhiorghitaNo ratings yet
- Band Gap Studies of Anatase Tio2 NanoparticlesDocument7 pagesBand Gap Studies of Anatase Tio2 Nanoparticlesnarayanan1701_318170No ratings yet
- Materials Letters: Fangfang Wang, Zhi Zheng, Falong JiaDocument4 pagesMaterials Letters: Fangfang Wang, Zhi Zheng, Falong JiaPawan Kumar DubeyNo ratings yet
- Experimental DetailsDocument13 pagesExperimental DetailsMuhammad KafillahNo ratings yet
- Daniel KOTTFER, Milan Ferdinandy, Pavol HVIZDOŠ, Lukasz KaczmarekDocument8 pagesDaniel KOTTFER, Milan Ferdinandy, Pavol HVIZDOŠ, Lukasz KaczmarekJigar M. UpadhyayNo ratings yet
- Physica E: A.F. Lotus, S.N. Tacastacas, M.J. Pinti, L.A. Britton, N. Stojilovic, R.D. Ramsier, G.G. ChaseDocument5 pagesPhysica E: A.F. Lotus, S.N. Tacastacas, M.J. Pinti, L.A. Britton, N. Stojilovic, R.D. Ramsier, G.G. ChaseEstudiante2346No ratings yet
- Antibacterial Properties and Corrosion Resistance of Nitrogen-Doped Tio Coatings On Stainless SteelDocument8 pagesAntibacterial Properties and Corrosion Resistance of Nitrogen-Doped Tio Coatings On Stainless SteelGabriel Vazquez VegaNo ratings yet
- Physica C: K. Kim, D.P. Norton, D.K. Christen, C. Cantoni, T. Aytug, A. GoyalDocument7 pagesPhysica C: K. Kim, D.P. Norton, D.K. Christen, C. Cantoni, T. Aytug, A. GoyalzddnkhanNo ratings yet
- Microstructure and Mechanical Properties of Tin/Tialn Multilayer Coatings Deposited by Arc Ion Plating With Separate TargetsDocument6 pagesMicrostructure and Mechanical Properties of Tin/Tialn Multilayer Coatings Deposited by Arc Ion Plating With Separate TargetsTurgay KıvakNo ratings yet
- Structure and Properties of Ti Si N Films Prepared by ICP Assisted Magnetron SputteringDocument5 pagesStructure and Properties of Ti Si N Films Prepared by ICP Assisted Magnetron SputteringEngels OchoaNo ratings yet
- 1 s2.0 S096014819900097X MainDocument11 pages1 s2.0 S096014819900097X MaincimavNo ratings yet
- Wunderlich PowderMet ConfSerDocument7 pagesWunderlich PowderMet ConfSertsaqif putraNo ratings yet
- Photoelectrochemical Studies of Nanocrystalline Tio Film ElectrodesDocument8 pagesPhotoelectrochemical Studies of Nanocrystalline Tio Film ElectrodesFayeen K ShariarNo ratings yet
- Kondo ScatteringDocument8 pagesKondo ScatteringPRIYA MANDALNo ratings yet
- Materials Letters: C.K. Chung, W.T. Chang, C.F. Chen, M.W. LiaoDocument4 pagesMaterials Letters: C.K. Chung, W.T. Chang, C.F. Chen, M.W. LiaoAsandulesa AndreeaNo ratings yet
- Paper MS-PDocument7 pagesPaper MS-PkapilNo ratings yet
- Materials Letters: Yuwen Liu, Yongping Pu, Zixiong SunDocument4 pagesMaterials Letters: Yuwen Liu, Yongping Pu, Zixiong SunSamah SamahNo ratings yet
- Ap A 2014Document9 pagesAp A 2014Ramanathaswamy PandianNo ratings yet
- Lecture Notes of Engineering MaterialsDocument7 pagesLecture Notes of Engineering Materialsahsan888100% (1)
- MM406: Semiconductor Devices and Processing Tutorial 1Document4 pagesMM406: Semiconductor Devices and Processing Tutorial 1Vasit AliNo ratings yet
- Introduction of Electron ConfigurationDocument26 pagesIntroduction of Electron ConfigurationKram M RazatlabNo ratings yet
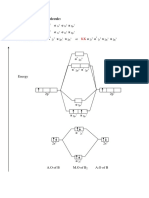
- Chemical BondingDocument28 pagesChemical BondingPrince DigvijayNo ratings yet
- Molecular Orbital Diagram For B, C, N, O, F and NeDocument12 pagesMolecular Orbital Diagram For B, C, N, O, F and NeGods ArmyNo ratings yet
- Unit 4 PDFDocument17 pagesUnit 4 PDFsoumengoswami10No ratings yet
- Colloids and Surfaces A: Ravi Shankar Kumar, Tushar SharmaDocument13 pagesColloids and Surfaces A: Ravi Shankar Kumar, Tushar SharmaRavi ShankarNo ratings yet
- Residual Stresses in Titanium Nitride Thin Films Deposited - Benegra - R.M.souza - 2006Document5 pagesResidual Stresses in Titanium Nitride Thin Films Deposited - Benegra - R.M.souza - 2006Enriqe PuentesNo ratings yet
- Improvement of Green Antisolvent-Isopropanol and Additive-Thiorea On Carbon Based CsPbiBr2 Perovskite Solar CellsDocument7 pagesImprovement of Green Antisolvent-Isopropanol and Additive-Thiorea On Carbon Based CsPbiBr2 Perovskite Solar CellsSoham AshNo ratings yet
- Quantum Numbers and Electron Configuration: Group 8 11-St. Camill Us de LellisDocument10 pagesQuantum Numbers and Electron Configuration: Group 8 11-St. Camill Us de LellisRyzellNo ratings yet
- Chemistry Reviewer Part 1Document3 pagesChemistry Reviewer Part 1kurtbusbus1No ratings yet
- Sch3u - Complete Class NotesDocument124 pagesSch3u - Complete Class NotesSafe SideNo ratings yet
- Generation of Superoxide Ions at Oxide SurfacesDocument10 pagesGeneration of Superoxide Ions at Oxide Surfacesbinir88610No ratings yet
- EE206 Material ScienceDocument2 pagesEE206 Material Sciencevpzfaris50% (2)
- Spectroscopy 04 Electronic Spectroscopy 23 24Document58 pagesSpectroscopy 04 Electronic Spectroscopy 23 24ramonrubio2004No ratings yet
- Shapes of MoleculesDocument17 pagesShapes of Moleculesbasaallen566No ratings yet
- Lesson Exemplar On Physical Changes of Solid, Liquid and GasDocument3 pagesLesson Exemplar On Physical Changes of Solid, Liquid and GasJimielle Shane Simon100% (1)
- PH3256 - Unit 01 - Question BankDocument2 pagesPH3256 - Unit 01 - Question Bankmathuammu6805No ratings yet
- 15 Semi Conductor ElectronicsDocument2 pages15 Semi Conductor Electronicstirumalaraoa7890No ratings yet
- Material Science MotivationDocument18 pagesMaterial Science MotivationPrateek BayalNo ratings yet
- Chapter 27 Homework Solution On University Physics IIDocument8 pagesChapter 27 Homework Solution On University Physics IIFrancisco JaramilloNo ratings yet
- Lab Report OnDocument10 pagesLab Report Onbhaski88No ratings yet
- Chemical Bonding Class11th by PS Sir IIT JEEDocument44 pagesChemical Bonding Class11th by PS Sir IIT JEEMahendra PandaNo ratings yet
- I-V Characterization of Tunnel Diodes and Multojunction Solar CellsDocument7 pagesI-V Characterization of Tunnel Diodes and Multojunction Solar CellsMaura MusioNo ratings yet
- Pyl 100 - Tutorial Sheet 5Document2 pagesPyl 100 - Tutorial Sheet 5HardeepSinghNo ratings yet
- Clausius-Mossotti Relation - Knowino PDFDocument3 pagesClausius-Mossotti Relation - Knowino PDFSurajNo ratings yet
- ITPO 2016 ProblemsDocument2 pagesITPO 2016 Problemsfreefall014100% (1)
- Electrical Properties of Materials: Chap 1Document31 pagesElectrical Properties of Materials: Chap 1Shuvro Sankar SenNo ratings yet
- 1984givord - SSC - Magnetic Properties of Y2Fe14B and Nd2Fe14B1 Single CrystalsDocument4 pages1984givord - SSC - Magnetic Properties of Y2Fe14B and Nd2Fe14B1 Single CrystalsPatrícia CanazartNo ratings yet
1 s2.0 S0040609096087494 Main
1 s2.0 S0040609096087494 Main
Uploaded by
Luis Alejandro Tafur0 ratings0% found this document useful (0 votes)
17 views5 pagesThe electrical resistivity of TiN prepared at 400 degC with 8% of nitrogen flow was 24 / z / 2 cm, equivalent to that of bulk TiN. Films showed low resistivity at higher nitrogen flow ( 10% and 12%)
Original Description:
Original Title
1-s2.0-S0040609096087494-main
Copyright
© © All Rights Reserved
Available Formats
PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentThe electrical resistivity of TiN prepared at 400 degC with 8% of nitrogen flow was 24 / z / 2 cm, equivalent to that of bulk TiN. Films showed low resistivity at higher nitrogen flow ( 10% and 12%)
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
Download as pdf or txt
0 ratings0% found this document useful (0 votes)
17 views5 pages1 s2.0 S0040609096087494 Main
1 s2.0 S0040609096087494 Main
Uploaded by
Luis Alejandro TafurThe electrical resistivity of TiN prepared at 400 degC with 8% of nitrogen flow was 24 / z / 2 cm, equivalent to that of bulk TiN. Films showed low resistivity at higher nitrogen flow ( 10% and 12%)
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
Download as pdf or txt
You are on page 1of 5
E L S E V I E R Thin Solid Films 287 (1996) 115-119
Characterization of TiN films prepared by a conventional magnetron
sputtering system: influence of nitrogen flow percentage and
electrical properties
M. Ka wa mur a a, y . Abe a, H. Yanagisawa b, K. Sa sa k i"
"Department of Materials Science, Faculty of Engineering, Kitami Institute of Technology, Kitami 090, Japan
b Department of Electrical and Electronic Engineering, Faculty of Engineering, gitami Institute of Technology. Kitumi 090, Japan
Received 19 September 1995; accepted 17 January 1996
Abstract
TiN films were prepared by reactive sputtering using a radio-frequency magnetron sputtering system. Ti films containing nitrogen were
also formed, depending on nitrogen flow. For all films the crystal structure, chemical binding state and composition were investigated. An
abrupt transition occurred in the narrow region between 7% and 8% nitrogen flow. Other phases like Ti2N or amorphous state were not
observed in the transition process. The electrical resistivity of TiN prepared at 400 C with 8% of nitrogen flow was 24/z/2 cm, equivalent to
that of bulk TiN. Films showed low resistivity at higher nitrogen flow ( 10% and 12%). Films kept low resistivity (30/z/2 cm) at 50 nm
thickness, and the continuity of the film was confirmed even at 5 nm thickness.
Keywords: Electrical properties and measurements; Sputtering; Titanium nitrid; X-ray diffraction
1. I ntroducti on
TiN thin films have been widely used for various applica-
tions; decorative coating making use of the golden color, hard
coatings on tools, diffusion barriers and electrodes for chem-
ical and thermal stability and low resistivity. Many research-
ers have studied the formation process of TiN films by
reactive sputtering using various systems [ 1-6] . Other prep-
aration methods have also been reported, such as reactive
evaporation, chemical vapor deposition (CVD) [ 7-9] . For
the reactive sputtering method, an influence of nitrogen con-
tent in the reactive gas mixture has been commonly men-
tioned [ 6, 10] . As a formation mechanism of TiN and some
other transition metal nitrides (e. g. Zr N), it has been sup-
posed that a gradual increase of nitrogen contents in metal
films led to transition to nitride films. Changes in crystal
structure and chemical binding state have been shown
[ 11-141.
In spite of these efforts, the physical properties of the
prepared TiN films deviate from the bulk TiN value. To solve
the problem, depositions applying bias to substrate were car-
ried out [ 4, 5] . However , it is still unclear to what extent we
can prepare the high-quality TiN thin films by conventional
reactive sputtering. The focus of this paper is to clear up this
issue systematically. In the present work, TiN films were
0040-6090/96/$15.00 1996 Elsevier Science S.A. All rights reserved
PI! S0040-6090 ( 96) 08749-4
prepared by reactive sputtering with a radio-frequency (r. f. )
magnetron sputtering system under various nitrogen flows
and substrate temperatures. The electrical resistivity of the
films obtained was examined and its thickness dependency
was clarified. The formation mechanism of TiN films was
also discussed by investigating the crystal structure, chemical
composition and chemical binding state of films of both Ti
and TiN.
2, Expe r ime nta l
2.1. Prcparation of films
Ti and TiN thin films were prepared by reactively sputter-
ing titanium in a mixed N2-Ar gas in an r.f. magnetr on sput-
tering system. High purity sputtering gases with various
nitrogen flow percentages (0-12% ) were introduced into the
system with the total gas flow controlled at 5 sccra. Total gas
pressure was measured by a capacitance manometer and fixed
at 6.7 10-m Pa. A 2 inch diameter disk of 99. 99% pure
titanium was used as targe). Coming # 70 59 glasses
(30 40 1.1 mm 3) cleaned with detergent, ethanol and ace-
tone in ultrasonic vibration were used as substrates. The sub-
strates were generally heated to 200 C, 300 C or 400 C
! 16 M. Kawamura et al. / Thin Solid Films 287 (1996) 115-119
during deposition, but deposition without heating was also
done for comparison.
For cleaning the target surface and achieving high-quality
vacuum conditions, 15 min ofpresputtering in Ar atmosphere
was done. The system was then pumped down below
6. 7x 10 -5 Pa. Before the film deposition, 15 rain of pres-
puttering was carried out in the gas mixture used for deposi-
tion. The r.f. power was 100 W, and the distance between
substrate and target was fixed at 55 mm.
2.2. Characteriz ation
a) N2 flow 6%
. . / . ' " . " " . /
30 40 50 60 70 80
20 1 degree
The electrical resistivities of the thin films prepared on
glasses were measured by the four-point probe method. For
some films, the temperature coefficients of resistivity (TCR)
were calculated using resistivity values measured at 30 C
and 60 (2. The film thicknesses were estimated by multiple
beam interferometry (MBI). The crystal structure of the films
was examined by the X-ray diffraction technique (XRD)
with Cu Ka radiation.
The chemical composition of the films was determined
from Auger transition signal intensities of Ti (LMM) and N
(KLL), comparing them with those of a bulk TiN standard
sample of 99% purity. It is known that the complete separa-
tion of the overlapped N(KL2L3) peak and Ti(L3M2.3M2.3)
peak is quite difficult. Here, the N/ Ti atomic ratio was deter-
mined from the positive excursion (at 377 eV) of the N
KILL2 line and the negative excursion of L3M2.3M4.s Ti peak
at 418 eV, according to the procedure of Johansson et al. [ 15 ]
The chemical binding state of the films was investigated
from their N Is and Ti 2p peaks by X-ray photoelectron
spectroscopy (XPS) using monochromatic AI Ka radiation.
Calibration was based on the Ti 2p~/2 electron peak (454.1
eV) in pure Ti thin film [ 16]. Before the analyses by Auger
electron spectroscopy (AES) and X-ray photoelectron spec-
troscopy (XPS), the thin films and the standard sample were
etched by an Ar ion gun to removo any contaminated layer
at the surface. Because the nitrogen bonding is weak in some
samples, the etching time was only 30 s for AES analysis so
as not to affect the film composition.
3. Results a n d discussion
The obtained thin films showed different appearances
depending on nitrogen flow during sputtering. Silver films
were obtained at 0, 4 and 6% nitrogen flow. Thin films in
gold, on the other hand, were formed at higher percentages
(8, 10 and 12%) at all substrate temperatures. The two kinds
of samples were presumed to be Ti containing nitrogen and
TiN, respectively, from their colors, and this was confirmed
by XRD measurements. Fig. I shows XRD patterns for films
prepared at nitrogen flows of 6% (a) and 8% (b). At all the
substrate temperatures, a single-phase Ti (hop structure) is
formed at 6% and that of TiN (NaCl-type structure) is formed
at 8%. The same result was obtained without substrate heating
] ~ N2 flow 8% b)
z .~
t
.[" ."" /" , . / " 4,ooc
30 40 50 60 70 80
20 1 degree
Fig. i. XRD patterns of the films prepared at substmte temperatures of 200,
300 and 400 C. (a) 6% of nitrogen flow and (b) 8% of nitrogen flow.
(not shown here). The product phases were dominated only
by the nitrogen flow percent of the reaction gas, and no
dependency on the substrate temperature was observed.
A preferential orientation on the substrate temperature is
also observed in Fig. 1; for films deposited at 400 C, the
main diffraction lines are Ti (011) and TiN (200), which
accords with JCPDS cards [ 17,18]. For films deposited at
lower temperatures, however, they are Ti (002) and TiN
( 1 ! I ), respectively. By comparison of the interplanar spac-
ings, no difference was recognized at all the temperatures for
both Ti and TiN phases,
The change of XRD patterns under various nitrogen flow
percentage: at 400 (2 is shown in Fig. 2. The thin film pre-
pared at 7% showed a Ti metal phase and silver color, which
means the transition from Ti to TiN occurs between 7% and
8%. With the increase of nitrogen flow, peaks of the Ti phase
shift to lower diffraction angles as a result of nitrogen incor-
poration, especially at the nitrogen flows of 6% and 7%. The
enlargement of the (011) interplanar spacing was 0.016 A, at
7% compared to 0% (pure Ti). An amorphous state or
another phase like TiaN did not appear in the process of the
transition in the present work. When the nitrogen flow per-
centage becomes high and reaches a limit of accommodation,
the transition to TiN phase occurred. Once the TiN phase was
formed at 8%, the XRD pattern did not change at 10% and
12%.
For the films deposited at 400 (2, deposition rate and nitro-
gen content were investigated as a function of nitrogen flow
during sputtering. As shown in Fig. 3, deposition rate drops
at the region between 4% and 8%. It is naturally understood
M. Kawamura et ai. /Thin Solid Films 287 (1996) 115-119 117
N2 flow' ' 0%
o~
4%
=i
-C
30 ' ' 4' 0 50
201 degree
Fig. 2, X R D patterns of t he fi lms prepared at 400 C under the various
ni t rogen fl ow percentages.
1 , , . , ~' : . ' . ,, . ,,,,. ~ , , . . . , , . ,. ; , . . , 25
~' (1,4
0 2 4 6 8 10 12
nftrogen flow / %
20
s ~
0
14
Fig. 3. Change of deposition rate and film composition at the substrate
temperature of 400 C as a function of nitrogen flow percent.
that the decrease of the deposition rate was caused by nitri-
darien of the surface of the Ti target, decreasing the sputtering
rate.
By AES analysis, the TiN film prepared at 8% of nitrogen
flow was found to be stoichiometric. At higher nitrogen per-
cent (10 and 12%), the films remained stoichiometric, and
did not become nitrogen-rich TiN. The films were found to
be very pure, with the incorporated oxygen estimated to be
below 1%. For the films obtained at 4% and 6%, the nitrogen
content was estimated as 19% and 22%, respectively. This
confirms that the film with the Ti phase contains a certain
amount of nitrogen, supporting the enlargement ofinterplanar
spacing on XRD patterns (Fig. 2).
Binding states of Ti and N were investigated by XPS, and
the spectra of Ti 2p and N ls are shown in Fig. 4 and Fig. 5,
I - " i " - - 1 . . . . . . . -
112p3~
/ 8 % - - . - ! / ~ I
/ "1"1 , p l ~ / / ~ [
i
466 461 456 451
Binding energy I oV
Fi g. 4. XPS spectr~ of T i 2p f or t he fi l ms prepared at 400 C and ni trogen
f l ow 0, 6, 8, 12%, and the T i N bul k standard sample.
:3
z,
12%- - I ~( ~
8 / ~ [ / ~
s t d~K t t 1
402 398
Nl e
~ 4
Binding energy / eV
Fig. 5. XPS spectra of N I s f or the fi hns prepared at 400 C and ni trogen
flow 4, 6, 8, 12%, trod the TiN bulk standard sample.
respectively. Spectra for standard TiN, 8 and 12% show the
Ti 2p3/2 level binding energy at 455.5 eV. The chemical shift
from the pure Ti metal was 1.4 eV, in accord with Refs. [ 12-
14,16]. For the films prepared under the conditions of 4%
and 6%, the values of Ti 2p3/2 were 454.4 and 454.5 eV,
respectively. They are very close to the value for pure Ti
(454.1 eV). Moreover, the shapes of the spectra are also very
similar to pure Ti. Characteristic shoulder peaks observed
around 455--458 eV on the spectra of TiN do riot exist on 4%
and 6% films.
As shown in Fig. 5, the N l s peaks for the films prepared
at 4% and 6% were observed at 397.4 eV, which showed a
small difference with those for the TiN films prepared at 8%
and 12% (397.8 eV). The nitrogen atoms in Ti-N solid
solution are known to occupy the interstitial Ti lattice sites
[ 19]. So the slight difference in N ls values between Ti-N
118 M. Kawamura et at. I Thin Solid Films 287 (1996) IL5-119
140
6O
20
i i i -
1/
I " " " I " " " I " ~" "
~ o . . . ,. ,. o. -
0 . . . i . , . i . . i . . i . . . i . . . i l ' I
0 2 4 6 8 10 12
Nitrogen flow 1%
14
Fig. 6. Change of the electrical resistivity of the films prepared at 400 C
under various nitrogen flow percent.
solid solution and TiN is caused by the change of effective
charge of nitrogen atoms and crystal lattice field.
Fig. 6 shows electrical resistivity of the Ti and TiN films
prepared under various nitrogen flows at 400 C. Here, the
thickness of the films was about 300 nm. The resistivity shows
its maximum at 7% followed by an abrupt decrease and a
minimum at 8%. At 0%, the value was 71 IX[l cm, which is
somewhat larger than that of bulk Ti (40 IXl~ cm). The
increase of resistivity in the Ti metal region with the increase
of nitrogen content could be explained by impurity scattering
due to incorporated nitrogen atoms. As mentioned previously
[ 10], the minimum value of resistivity was achieved near the
transition region. In our work, the value of 24 I~1~ cm, which
is equivalent to that of bulk TiN was obtained at 8% nitrogen
flow. At higher nitrogen percents ( 10~ and 12%), the values
were still as low as 30 Ixl~ cm and did not show a steep ascent.
Consequently, high quality TiN films were prepared at rela-
tively low temperature in a conventional magnetron sputter-
ing system without any modification, such as substrate bias.
The magnetron sputtering system is generally known to make
high density plasma condition at relatively lower pressure,
which is useful for the prep,~xation of high quality films.
However, we insist on the importance choosing the appro-
priate nitrogen flow from the present result. The low oxygen
content in the films also contributes to the very low resistivity.
When the substrate temperature becomes lower, the resis-
tivity is higher, as shown in Fig. 7. It is clearly understood
that the substrate heating even at 200 C improves the prop-
erties of the film remarkably. The resistivity of the film at
200 C is low and similar to the value at 400 (2. Because the
curve seems to be saturated at 400 (2, further heating of the
substrate is not needed. Result of TCR measurements shows
all positive values because of metallic conduction of elec-
trons. TCR increases with increase of substrate temperature
and decrease of resistivity. The reason for the increase of
resistivity with decreasing substrate temperature was not
clear from the XRD measurements: no noticeable variation
of the interplanar spacings or half-widths of diffraction peaks
was observed. However, the TCR change means that the
increase of the substrate temperature reduces microdefects in
the thin films and improves the crystallinity.
100
80
so
40
20
. . . . i . . . . i . . . . i . . . . i . . . .
Q ~ N2 flow 8%
1400
1200
1000
2OO
0
0 100 200 300 400 500
Substrate temperature I C
Fig. 7. Change of electrical resistivity and the TCR with subsrate tempera-
turc of the films deposition at 8% nitrogen flow,
For the recent demand, thinner TiN films than examined
above are required. TiN fihns prepared at 400 C under the
8% nitrogen flow were found to keep a low resistivity in a
wide thickness range. Fig. 8 shows the change of the resistiv-
ity with film thicknesses. The low resistivity of 30 IXl'l cm
was kept to 50 nm in thickness. The value then increased
gr~dually and was 65 i~1"1 cm at 5 nm thick. These low values
indicate that the TiN film is still continuous even at 5 nm in
thickness.
According to the Fuchs-Sondheimer theory [ 20], the ratio
of the resistivity of bulk metal p to that of the thin metal film
pf is expressed as:
_ . p: o ' f : 1 _ 3 ( 1 _ p) i ( ~_ ~_ ~3) l - e x p ( - K~)
pf o" 2K 1 - p exp( - x~)
I
where K = d/l, d is the film thickness, I is the mean free path
of electrons and P is the elastic scattering factor at the film
surface and the interface between film and substratc. ~ is an
integral variable. In the polycrystai TiN film, conduction elec-
trons arc considered to be scattered diffusely (P = 0). When
the bulk resistivity p is chosen to be 25 ~l'I cm, the result of
the calculation is also shown in Fig. 8. The calculative values
using a fitting parameter ! = 20 nm agree very well with meas-
lOO
00
6o
20
O :measured values
v e v n l u e 8
o ~ 0
. . . , , . i . . , , , . i , . | . . . . .
10 100 1000
Thickness I nm
Fig. 8. The relation of electrical resistivity and thickness of the film prepared
at 400 C with 8% nitrogen flow. The result of measurement is shown by
circles and that of calculation based on the ;Zuchs-Sondheimer theory is
shown by the solid line.
M. Kawamura et al. / Thin Solid Films 287 (1996) 115-119 119
ured values. The mean free path o f 20 nm for Ti N is the same
order o f magni tude as that reported for Au, Ag and Cu thin
fi l ms [ 2 1 , 2 2 ] . These results also support the continuity of
the Ti N films.
4. Concl us i ons
1. Ti N thin fi l ms were prepared by reactive sputtering above
8% nitrogen fl ow. Be l ow this percentage, Ti films con-
taining nitrogen were formed, whi ch was confirmed by
XRD, XPS and AES analyses.
2. The electrical resistivity o f a Ti N film prepared at 40 0 C
and 8% o f nitrogen fl ow, was 24 ix12 cm, bei ng equivalent
to that of bulk Ti N. At higher percentage ( 1 2 %) , the value
was still as l ow as 30 Ixl~ cm. Improvement o f resistivity
by heating at 40 0 C was recognized, but resistivity of the
film at 200 C was al so l ow.
3. The Ti N film was f ound to be conti nuous even at 5 nm.
The result o f measurement was in accordance with Fuchs -
Sondhei mer theory.
Ac k nowl e dge me nt s
This work was partly supported by Sasakawa Scientific
Research Grant from The Japan Sci ence Soci ety.
Ref erences
[ ! ] M. Wittmer and H. Melchior, Thin Solid Films, 93 (1982)
397.
[2]
[3]
[4]
[5]
[6]
[7]
[8]
[91
[10]
[11]
[12]
[13]
[14]
[15]
[16]
[171
[18]
[19]
[20]
[21]
I22]
H. Joswig, A. Kohlhase and P. Kucher, Thin Solid Films, 175 (1989)
17.
M.K. Hibbs, J.-E. Sundgrcn, B.E. Jaccobson and B.-O. Johansson,
Thin Solid Films, 107 (1983) 149.
Y. Igasaki and H. Mitsuhashi, Thin Solid Films, 70 (1980) 17.
N. Kumar, K. Pourrezaei, M. Fi'ssel, T. Begley, B. Lee and E.C.
Douglas, J. Vac. Sci. Technol., A5 (1987) 1778.
W. Tsai, J. Fedr and D. Hodul, J. Electrochem. Soc., 139 (1992)
2004.
B.E. Jacobson, R. Nimmagadda and R.F. Bunshan, Thin Solid Films,
63 (1979) 333.
H.E. Cheng, M.J. Chiang and M.H. Hon, J. Electrochem. Soc., 142
(1995) 1573.
T. Ohba, T. Suzuki, H. Yagi, Y. Furumura and H. Hatano, J.
Electrochem. Soc., 142 (1995) 934.
H.-O. Blom, S. Berg and T. Larsson, Thin Solid Films, 130 (1985)
307.
H. Jiang, K. Tao and H. Li, Thin Solid Films, 258 (1995) 5 I.
M.V. Kuznetsov, MN. Zhuravlev, E.V. Shalayeva and V.A. Gubanov,
Thin Solid Films, 215 (1992) 1.
L. Porte, L. Roux and J. Hanus, Phys. Rev. B, 28 (1983) 3214.
H. Hochst, R.D. Bringans, P. Steiner and T. Wolf, Phys. Rev. B, 25
(1982) 7183.
B.O. Johansson, L-E. Sundgren, J.E. Greene, A. Rockett and S.A.
Barnett, J. Vac. Sci. Technol., A3 (1985) 303.
J.F. Moulder, W.F. SticHe, P.E. Sobol and K.D. Bomben, Handbook
of X-ray Photoelectron Spectroscopy, Perkin-Elmer, Minnesota, 1992,
p. 73.
JCPDS, File No. 5-0682.
JCPDS, File No. 38-1420.
L.E. Toth, Transition Metal Carbides and Nitrides, Academic Press,
New York, 1971, p. 87.
E.H. Sondheimer, Adv. Phys., 1 (1952) 1.
K.L. Chopra, L.C. Bobb and M.H. Francombe, J. Appl. Phys., 34
(1963) 1699.
I. Estermann and T. Schlesinger, J. Appl. Phys., 41 (1970) 2802.
You might also like
- Report of Infrared Plastic Solar CellDocument22 pagesReport of Infrared Plastic Solar CellAjay Mv85% (54)
- Coatings: Microstructure and Mechanical Properties of Tan Thin Films Prepared by Reactive Magnetron SputteringDocument16 pagesCoatings: Microstructure and Mechanical Properties of Tan Thin Films Prepared by Reactive Magnetron SputteringTrinh ThinhNo ratings yet
- Stress and Oxidation Behaviours of R.f.-Sputtered (Ti, A1) N FilmsDocument8 pagesStress and Oxidation Behaviours of R.f.-Sputtered (Ti, A1) N FilmsIgor Oliveira NascimentoNo ratings yet
- Effects of Nitrogen Flow Rates On The Growth Morphology of TiAlN Films Prepared by An Rf-Reactive Sputtering TechniqueDocument5 pagesEffects of Nitrogen Flow Rates On The Growth Morphology of TiAlN Films Prepared by An Rf-Reactive Sputtering TechniquePhysics MPSTMENo ratings yet
- Structural and Electrochemical Impedance Spectroscopic Studies On Reactive Magnetron Sputtered Titanium Oxynitride (Tion) Thin FilmsDocument6 pagesStructural and Electrochemical Impedance Spectroscopic Studies On Reactive Magnetron Sputtered Titanium Oxynitride (Tion) Thin FilmsJussier VitorianoNo ratings yet
- 5ungureanu PDFDocument5 pages5ungureanu PDFsuganyaNo ratings yet
- Art 2009Document6 pagesArt 2009Carito CastilloNo ratings yet
- Hot Filament Assisted CVD of Titanium Nitride FilmsDocument6 pagesHot Filament Assisted CVD of Titanium Nitride FilmsMagui Ros MonNo ratings yet
- 1.deposition of Tantalum Nitride Thin Films by D.C. Magnetron SputteringDocument6 pages1.deposition of Tantalum Nitride Thin Films by D.C. Magnetron SputteringNishat ArshiNo ratings yet
- Preparation and Characterization of TiO2 Nanotube Arrays Via Anodization of Titanium Films Deposited On FTO Conducting Glass at Room TemperatureDocument7 pagesPreparation and Characterization of TiO2 Nanotube Arrays Via Anodization of Titanium Films Deposited On FTO Conducting Glass at Room TemperatureAruna RajanNo ratings yet
- Nambara 2007Document6 pagesNambara 2007Nassar Al-EssawiNo ratings yet
- Characteristics of Arc Plasma Deposited TiAlZrCN CoatingsDocument7 pagesCharacteristics of Arc Plasma Deposited TiAlZrCN CoatingsПавел МисуноNo ratings yet
- Atomic Layer Deposition of TiO2 From Tetrakis (Dimethylamino) Titanium and H2ODocument7 pagesAtomic Layer Deposition of TiO2 From Tetrakis (Dimethylamino) Titanium and H2OAilinJlsNo ratings yet
- Magnetic Conductive Outer Layer in Oxygen-Deficient Tio Single CrystalsDocument7 pagesMagnetic Conductive Outer Layer in Oxygen-Deficient Tio Single CrystalsSahil AkramNo ratings yet
- Journal Pre-Proofs: Electrochemistry CommunicationsDocument14 pagesJournal Pre-Proofs: Electrochemistry CommunicationsSandouda Bettaieb Ep RouatbiNo ratings yet
- Flash Evaporation1Document11 pagesFlash Evaporation1Sudha AsNo ratings yet
- Eis Analysis On Low Temperature Fabrication of Tio Porous Films For Dye-Sensitized Solar CellsDocument36 pagesEis Analysis On Low Temperature Fabrication of Tio Porous Films For Dye-Sensitized Solar CellsUdai SinghNo ratings yet
- Velten Et Al-2002-Journal of Biomedical Materials ResearchDocument11 pagesVelten Et Al-2002-Journal of Biomedical Materials ResearchjeffreylopezNo ratings yet
- Thin Solid Films: A.S. Daryapurkar, J.T. Kolte, P. GopalanDocument6 pagesThin Solid Films: A.S. Daryapurkar, J.T. Kolte, P. GopalanTuhinNo ratings yet
- Characterization of Sputter-Deposited Chromium Nitride Thin FilmsDocument5 pagesCharacterization of Sputter-Deposited Chromium Nitride Thin FilmsNassima JagharNo ratings yet
- Anodization P - TiO2555Document9 pagesAnodization P - TiO2555Sudip SinhaNo ratings yet
- Scripta NiTi TEM 04Document5 pagesScripta NiTi TEM 041985krNo ratings yet
- Effect of Nitrogen Flow Rate On Structure and Properties of Nanocrystalline Tin Thin Films Produced by Unbalanced Magnetron SputteringDocument8 pagesEffect of Nitrogen Flow Rate On Structure and Properties of Nanocrystalline Tin Thin Films Produced by Unbalanced Magnetron SputteringEngels OchoaNo ratings yet
- Des Feux 2004Document6 pagesDes Feux 2004kraya.sahla.mahlaNo ratings yet
- Effects of Rapid Thermal Annealing On The Local EnvironmentDocument11 pagesEffects of Rapid Thermal Annealing On The Local EnvironmentAhmedNo ratings yet
- Electrochemical Corrosion and Materials Properties of Ananthakumar2012Document9 pagesElectrochemical Corrosion and Materials Properties of Ananthakumar2012Chercheurs Permanents UrmmNo ratings yet
- Phase Transition and Properties of Ti Al N Thin ®lms Prepared by R.f.-Plasma Assisted Magnetron SputteringDocument6 pagesPhase Transition and Properties of Ti Al N Thin ®lms Prepared by R.f.-Plasma Assisted Magnetron SputteringjamesNo ratings yet
- TEM Study of TiO2Document5 pagesTEM Study of TiO2CristianAtanasiuNo ratings yet
- Effects of Tin Coating On The Corrosion of Nanostructured Ti-30Ta-Xzr Alloys For Dental ImplantsDocument6 pagesEffects of Tin Coating On The Corrosion of Nanostructured Ti-30Ta-Xzr Alloys For Dental ImplantsAtria PradityanaNo ratings yet
- Unique TiO2 Paste For High Efficiency Dye-Sensitized Solar CellsDocument3 pagesUnique TiO2 Paste For High Efficiency Dye-Sensitized Solar CellsasadNo ratings yet
- ManuscriptDocument17 pagesManuscriptfarshad davoodianNo ratings yet
- Crystal Phase Transition and Properties of Titanium Oxide Nanotube Arrays Prepared by AnodizationDocument4 pagesCrystal Phase Transition and Properties of Titanium Oxide Nanotube Arrays Prepared by AnodizationEduardo BadelNo ratings yet
- Preparation and Characterization of Peroxo Titanic Acid Solution Using TiclDocument6 pagesPreparation and Characterization of Peroxo Titanic Acid Solution Using TiclThanh ThủyNo ratings yet
- Current Applied Physics: Sciverse SciencedirectDocument4 pagesCurrent Applied Physics: Sciverse Sciencedirectslct88No ratings yet
- 10.1016@j.apsusc.2016.12.102Document5 pages10.1016@j.apsusc.2016.12.102TaryoNo ratings yet
- SYNTHESIS AND APPLICATIONS OF TiO2 NANOPARTICLESDocument10 pagesSYNTHESIS AND APPLICATIONS OF TiO2 NANOPARTICLESSoheil MirtalebiNo ratings yet
- 2015 Article 814Document6 pages2015 Article 814hrishikesh.friends4everNo ratings yet
- Enhancing The Performance of Dye-Sensitized Solar Cells Based On An Organic Dye by Incorporating TiO2 Nanotube in A TiO2 Nanoparticle FilmDocument8 pagesEnhancing The Performance of Dye-Sensitized Solar Cells Based On An Organic Dye by Incorporating TiO2 Nanotube in A TiO2 Nanoparticle FilmMuhammad Bilal QadirNo ratings yet
- Corrosion NanotubosDocument14 pagesCorrosion NanotubosjeffreylopezNo ratings yet
- International Refereed Journal of Engineering and Science (IRJES)Document5 pagesInternational Refereed Journal of Engineering and Science (IRJES)www.irjes.comNo ratings yet
- 2 - Fabrication and Photocatalytic Activity of TiO2 Nano Ber Membrane 2009Document3 pages2 - Fabrication and Photocatalytic Activity of TiO2 Nano Ber Membrane 2009Yavuz GençNo ratings yet
- Effects of Ni Doping On Photocatalytic Activity of Tio Thin Films Prepared by Liquid Phase Deposition TechniqueDocument5 pagesEffects of Ni Doping On Photocatalytic Activity of Tio Thin Films Prepared by Liquid Phase Deposition TechniqueRudolf JasonNo ratings yet
- DM TratamentDocument5 pagesDM TratamentLiliana GhiorghitaNo ratings yet
- Band Gap Studies of Anatase Tio2 NanoparticlesDocument7 pagesBand Gap Studies of Anatase Tio2 Nanoparticlesnarayanan1701_318170No ratings yet
- Materials Letters: Fangfang Wang, Zhi Zheng, Falong JiaDocument4 pagesMaterials Letters: Fangfang Wang, Zhi Zheng, Falong JiaPawan Kumar DubeyNo ratings yet
- Experimental DetailsDocument13 pagesExperimental DetailsMuhammad KafillahNo ratings yet
- Daniel KOTTFER, Milan Ferdinandy, Pavol HVIZDOŠ, Lukasz KaczmarekDocument8 pagesDaniel KOTTFER, Milan Ferdinandy, Pavol HVIZDOŠ, Lukasz KaczmarekJigar M. UpadhyayNo ratings yet
- Physica E: A.F. Lotus, S.N. Tacastacas, M.J. Pinti, L.A. Britton, N. Stojilovic, R.D. Ramsier, G.G. ChaseDocument5 pagesPhysica E: A.F. Lotus, S.N. Tacastacas, M.J. Pinti, L.A. Britton, N. Stojilovic, R.D. Ramsier, G.G. ChaseEstudiante2346No ratings yet
- Antibacterial Properties and Corrosion Resistance of Nitrogen-Doped Tio Coatings On Stainless SteelDocument8 pagesAntibacterial Properties and Corrosion Resistance of Nitrogen-Doped Tio Coatings On Stainless SteelGabriel Vazquez VegaNo ratings yet
- Physica C: K. Kim, D.P. Norton, D.K. Christen, C. Cantoni, T. Aytug, A. GoyalDocument7 pagesPhysica C: K. Kim, D.P. Norton, D.K. Christen, C. Cantoni, T. Aytug, A. GoyalzddnkhanNo ratings yet
- Microstructure and Mechanical Properties of Tin/Tialn Multilayer Coatings Deposited by Arc Ion Plating With Separate TargetsDocument6 pagesMicrostructure and Mechanical Properties of Tin/Tialn Multilayer Coatings Deposited by Arc Ion Plating With Separate TargetsTurgay KıvakNo ratings yet
- Structure and Properties of Ti Si N Films Prepared by ICP Assisted Magnetron SputteringDocument5 pagesStructure and Properties of Ti Si N Films Prepared by ICP Assisted Magnetron SputteringEngels OchoaNo ratings yet
- 1 s2.0 S096014819900097X MainDocument11 pages1 s2.0 S096014819900097X MaincimavNo ratings yet
- Wunderlich PowderMet ConfSerDocument7 pagesWunderlich PowderMet ConfSertsaqif putraNo ratings yet
- Photoelectrochemical Studies of Nanocrystalline Tio Film ElectrodesDocument8 pagesPhotoelectrochemical Studies of Nanocrystalline Tio Film ElectrodesFayeen K ShariarNo ratings yet
- Kondo ScatteringDocument8 pagesKondo ScatteringPRIYA MANDALNo ratings yet
- Materials Letters: C.K. Chung, W.T. Chang, C.F. Chen, M.W. LiaoDocument4 pagesMaterials Letters: C.K. Chung, W.T. Chang, C.F. Chen, M.W. LiaoAsandulesa AndreeaNo ratings yet
- Paper MS-PDocument7 pagesPaper MS-PkapilNo ratings yet
- Materials Letters: Yuwen Liu, Yongping Pu, Zixiong SunDocument4 pagesMaterials Letters: Yuwen Liu, Yongping Pu, Zixiong SunSamah SamahNo ratings yet
- Ap A 2014Document9 pagesAp A 2014Ramanathaswamy PandianNo ratings yet
- Lecture Notes of Engineering MaterialsDocument7 pagesLecture Notes of Engineering Materialsahsan888100% (1)
- MM406: Semiconductor Devices and Processing Tutorial 1Document4 pagesMM406: Semiconductor Devices and Processing Tutorial 1Vasit AliNo ratings yet
- Introduction of Electron ConfigurationDocument26 pagesIntroduction of Electron ConfigurationKram M RazatlabNo ratings yet
- Chemical BondingDocument28 pagesChemical BondingPrince DigvijayNo ratings yet
- Molecular Orbital Diagram For B, C, N, O, F and NeDocument12 pagesMolecular Orbital Diagram For B, C, N, O, F and NeGods ArmyNo ratings yet
- Unit 4 PDFDocument17 pagesUnit 4 PDFsoumengoswami10No ratings yet
- Colloids and Surfaces A: Ravi Shankar Kumar, Tushar SharmaDocument13 pagesColloids and Surfaces A: Ravi Shankar Kumar, Tushar SharmaRavi ShankarNo ratings yet
- Residual Stresses in Titanium Nitride Thin Films Deposited - Benegra - R.M.souza - 2006Document5 pagesResidual Stresses in Titanium Nitride Thin Films Deposited - Benegra - R.M.souza - 2006Enriqe PuentesNo ratings yet
- Improvement of Green Antisolvent-Isopropanol and Additive-Thiorea On Carbon Based CsPbiBr2 Perovskite Solar CellsDocument7 pagesImprovement of Green Antisolvent-Isopropanol and Additive-Thiorea On Carbon Based CsPbiBr2 Perovskite Solar CellsSoham AshNo ratings yet
- Quantum Numbers and Electron Configuration: Group 8 11-St. Camill Us de LellisDocument10 pagesQuantum Numbers and Electron Configuration: Group 8 11-St. Camill Us de LellisRyzellNo ratings yet
- Chemistry Reviewer Part 1Document3 pagesChemistry Reviewer Part 1kurtbusbus1No ratings yet
- Sch3u - Complete Class NotesDocument124 pagesSch3u - Complete Class NotesSafe SideNo ratings yet
- Generation of Superoxide Ions at Oxide SurfacesDocument10 pagesGeneration of Superoxide Ions at Oxide Surfacesbinir88610No ratings yet
- EE206 Material ScienceDocument2 pagesEE206 Material Sciencevpzfaris50% (2)
- Spectroscopy 04 Electronic Spectroscopy 23 24Document58 pagesSpectroscopy 04 Electronic Spectroscopy 23 24ramonrubio2004No ratings yet
- Shapes of MoleculesDocument17 pagesShapes of Moleculesbasaallen566No ratings yet
- Lesson Exemplar On Physical Changes of Solid, Liquid and GasDocument3 pagesLesson Exemplar On Physical Changes of Solid, Liquid and GasJimielle Shane Simon100% (1)
- PH3256 - Unit 01 - Question BankDocument2 pagesPH3256 - Unit 01 - Question Bankmathuammu6805No ratings yet
- 15 Semi Conductor ElectronicsDocument2 pages15 Semi Conductor Electronicstirumalaraoa7890No ratings yet
- Material Science MotivationDocument18 pagesMaterial Science MotivationPrateek BayalNo ratings yet
- Chapter 27 Homework Solution On University Physics IIDocument8 pagesChapter 27 Homework Solution On University Physics IIFrancisco JaramilloNo ratings yet
- Lab Report OnDocument10 pagesLab Report Onbhaski88No ratings yet
- Chemical Bonding Class11th by PS Sir IIT JEEDocument44 pagesChemical Bonding Class11th by PS Sir IIT JEEMahendra PandaNo ratings yet
- I-V Characterization of Tunnel Diodes and Multojunction Solar CellsDocument7 pagesI-V Characterization of Tunnel Diodes and Multojunction Solar CellsMaura MusioNo ratings yet
- Pyl 100 - Tutorial Sheet 5Document2 pagesPyl 100 - Tutorial Sheet 5HardeepSinghNo ratings yet
- Clausius-Mossotti Relation - Knowino PDFDocument3 pagesClausius-Mossotti Relation - Knowino PDFSurajNo ratings yet
- ITPO 2016 ProblemsDocument2 pagesITPO 2016 Problemsfreefall014100% (1)
- Electrical Properties of Materials: Chap 1Document31 pagesElectrical Properties of Materials: Chap 1Shuvro Sankar SenNo ratings yet
- 1984givord - SSC - Magnetic Properties of Y2Fe14B and Nd2Fe14B1 Single CrystalsDocument4 pages1984givord - SSC - Magnetic Properties of Y2Fe14B and Nd2Fe14B1 Single CrystalsPatrícia CanazartNo ratings yet