Professional Documents
Culture Documents
Module # Module #38 38: High Temperature Deformation of Crystalline Materials
Module # Module #38 38: High Temperature Deformation of Crystalline Materials
Uploaded by
Frederico FernandesOriginal Description:
Original Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Module # Module #38 38: High Temperature Deformation of Crystalline Materials
Module # Module #38 38: High Temperature Deformation of Crystalline Materials
Uploaded by
Frederico FernandesCopyright:
Available Formats
2/13/2012
Module #38
#38
[Excerpt from Deformation and Fracture of Crystalline and Non-crystalline
Non crystalline Solids Course Notes]
Part I: Creep and Superplasticity
Learning Objectives
Prof. M.L. Weaver
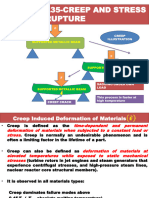
High Temperature Deformation
of Crystalline Materials
T < 0.5 Tmp
T > 0.5 Tmp
HIGH
TEMPERATURE
DEFORMATION
MODES
design so < ys
design so < ys
We must also consider
permanent deformation
occurring by CREEP over
long periods of time.
CREEP = time-dependent plastic deformation
YIELDING = time-independent plastic deformation
Prof. M.L. Weaver
2/13/2012
We can incorporate these
things into Deformation
Mechanism Maps (DMM)
[Frost and Ashby]
Ashby]. Well
We ll
address this a bit later.
Stress
YIELDING
glide
/G
creep
They are useful in design
applications have DMM
for each alloy but to really
know/predict
p
high
g T useful
life of alloy, still must do
testing usually
accelerated!
elastic
creep
Temperature
T/Tmp
Prof. M.L. Weaver
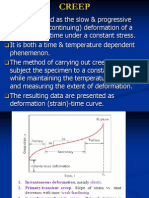
Phenomenological
Description of Creep
STRAIN
CREEP
TIME DEPENDENT
Elastic
CONSTANT
LOAD!
YIELDING
TIME INDEPENDENT
Elastic
Plastic
t1
time
When stress is removed
Prof. M.L. Weaver
2/13/2012
Constant Load Creep Test
III
d
dt
III
I
INC. ,T
II
II
INC. ,T
III
II
II
III
0
tf
time
time
tf
Stage II: constant AND minimum, II
There are 3 stages of creep
Prof. M.L. Weaver
Difference between constant load and
constant stress creep tests
Strain
Constant
Load
SS
d
dt
Fracture
X
Constant
Stress
dt
II
III
Secondary (steady state)
creep
Tertiary creep
time
Primary (transient)
creep
Constant stress testing delays the onset of
tertiary creep
Prof. M.L. Weaver
2/13/2012
STAGES OF CREEP
STAGE 0:
Initial Strain on Loading (Elastic, Plastic ???)
STAGE I:
Transient (Primary) Creep
with
i h time
i & strain
i
and subgrains form TEM
to strength of material Analyses
Cause
STAGE II:
Steady State Creep
microstructure constant
rate of due
Cause
to deformation
Constant
minimum
creep rate,
t II
rate of
due to recovery
Prof. M.L. Weaver
STAGES OF CREEP contd
STAGE III: Tertiary Creep
with time & strain
fracture is soon to occur after Stage III begins
recrystallization
Cause coarsening of 2nd phase particles
formation of internal cracks & voids
Prof. M.L. Weaver
2/13/2012
CREEP
Design Considerations
Usually either II or t f
This parameter is usually addressed in
scientific analyses as it is most readily
correlated with microstructures and
creep mechanisms.
tf:
Use when failure must not occur but
deformation/creep is tolerable.
Ex., Casing on rocket combustion chamber
II :
Use when long life is necessary and dimensional
tolerances are critical.
Ex., Jet engine turbine blade
Prof. M.L. Weaver
Empirical Law for Stage I and Stage II creep
Sttrain
Constant
Load
SS
d
dt
Fracture
X
Constant
Stress
dt
Secondary (steady state)
creep
Tertiary creep
Gives a
good fit to
experimental
data
time
Primary (transient)
creep
Garofalos relationship
creep o t 1 e rt sst
o instantaneous strain on loading
t limit for transient creep
r ratio of transient creep rate to transient creep strain
ss steady-state creep rate
F. Garofalo, Fundamentals of Creep and Creep-Rupture in Metals, (Macmillan, New York, 1965) p. 16.
Prof. M.L. Weaver
2/13/2012
Decomposition of
strain into components
STRAIN
Primary
creep
Secondary
creep
creep
sst
t 1 e rt
time
creep o t 1 e rt sst
Prof. M.L. Weaver
Empirical Relationships for Stages I and II
There are of course many other relationships (i.e., time
functions).
Each applies over a specific range of temperature and
stress for particular alloys.
I refer you to the following texts for more detail:
F. Garofalo, Fundamentals of Creep and Creep-Rupture in Metals
(Macmillan, New York, 1965),
F.R.N.
FRN N
Nabarro
b
and
dH
H.L.
L d
de Villi
Villiers, The
Th Ph
Physics
i off C
Creep (Taylor
(T l &
Francis, London, 1995),
M.E. Kassner, Fundamentals of Creep in Metals and Alloys, 2nd Edition
(Elsevier, 2009),
J-P. Poirier, Creep of Crystals (Cambridge University Press, 1985).
J-S. Zhang, High temperature Deformation and Fracture of Materials
(Woodhead Publishing, Oxford, 2010).
Prof. M.L. Weaver
2/13/2012
STAGE II
Steady-State Creep Rate (Sherby-Dorn Relationship)
n
Q
Q
II ss A exp c o exp c
RT
RT
o
n
A & n are material constants
Qc is the creep activation energy
Vary with
&T
due to changes in the
creep mechanism
This equation also holds for many materials and establishes
the temperature and stress dependence of the creep rate.
Correlates with the activation energy for
the mechanisms causing creep. Well
address the mechanisms shortly (in great
detail).
Qc R
ln
1 1
T T
1
2
Prof. M.L. Weaver
Activatio
on Energy for Self Diffusion, QSDD (MJ/mole)
For many materials:
When T 0.3 0.5 Tmp
Qc activation energy for self diffusion of elements/ions
100
Nb
-Fe
Fe UO2
MgO
NaCl
Ni
Al O
-Fe 2 3
-Co
AgBr
Cu
Al
Zn Mg
-Ti
10-1
In
Ca
Au
Pb
Ag
-Ti
Sn
Li
Na
K
Qc = QSD
10-2
10-2
10-1
100
Activation Energy for Creep, Qc (MJ/mole)
Activation energies for steady state creep and self diffusion for a select group of crystalline materials.
Adapted from O.D. Sherby and A.K. Miller, J. Eng. Mater. Technol., v. 101 (1979) p. 387.
Prof. M.L. Weaver
2/13/2012
Several deformation
mechanisms are
operative
Slip
Climb
Cross slip
Twinning
Diffusion???
etc.
Single deformation
mechanism is
operative
(Diffusional flow)
Q = variable
Q = constant
Qc
Hsd
Variation in apparent activation energy for creep in aluminum with temperature.
Adapted from O.D. Sherby et al., Acta Metall., v. 5, p. 219 (1957)
Prof. M.L. Weaver
In general, Hc (or Qc) becomes constant above T/Tm = 0.3 0.5.
This indicates that there is a single operative creep mechanism
1.2
COPPER
1.0
Qc
ALUMINUM
0.8
Q sd 0.6
TUNGSTEN
0.4
SODIUM CHLORIDE
0.2
0
0
02
0.2
04
0.4
06
0.6
08
0.8
10
1.0
HOMOLOGOUS TEMPERATURE, T/Tm
Figure. Ratio between activation energy for steady state creep and activation energy for
self/bulk diffusion as a function of temperature. Data from O.D. Sherby and A.K. Miller, J.
Eng. Mater. Technol., 101 (1979) 387. Figure adapted from Meyers and Chawla,
Mechanical Behavior of Materials, 1st edition, Prentice Hall, Upper Saddle River, NJ, 1999,
p. 554.
Prof. M.L. Weaver
2/13/2012
Generalized form of the Dorn Equation
n
ADGb b
, where D Do exp(Q / kT )
kT G d
D = diffusion coefficient
d = grain size
b = Burgers vector
k = Boltzmanns constant
T = the absolute temperature (degrees Kelvin)
G = the shear modulus
= applied stress
n = stress exponent
p = inverse grain size exponent
A = a dimensionless constant.
This form of the Dorn equation applies for all creep mechanisms.
Prof. M.L. Weaver
Creep Mechanisms
Diffusion Creep
Nabarro-Herring
Coble
Dislocation (Power-Law) Creep
All involve
some form of
diffusion
Solute
S
drag
Dislocation climb-glide
Prof. M.L. Weaver
2/13/2012
CREEP MECHANISMS
T
It is convenient to discuss
deformation (and creep)
mechanisms in terms of
temperature and/or
applied stress.
/G
Deformation mechanism
maps (DMMs) *
* Adapted from: M.F. Ashby, Acta
Metall., v. 20 (1972) p. 887-897.
Ideal shear strength
Dislocation glide
(slip)
Yield strength
(low temp)
Dislocation creep
(Power law creep)
(high temp)
or
/G
Coble (g.b.)
Diffusion creep
NH (lattice)
Elastic
El
i
regime
T/Tmp
Prof. M.L. Weaver
WELL ADDRESS THE DETAILS IN A LITTLE WHILE
DISLOCATION GLIDE AT LOW TEMPERATURES
Low T or High :
Dislocation Glide (DG) Creep
As we noted previously, dislocation () motion can be assisted by
thermal activation.
Thermal activation influences the lattice resistance to dislocation motion
(recall the Peierls barrier).
With sufficient thermal activation, a dislocation can overcome barriers to
their motion at lower applied stresses.
This can involve diffusive processes, as we will discuss later, but does
not necessarily involve them.
them
For the moment, let us consider thermally assisted dislocation glide
without diffusion.
This is not really creep in the truest sense of the word. Rather it is
simply slip assisted by heating.
Prof. M.L. Weaver
10
2/13/2012
DISLOCATION GLIDE AT LOW TEMPERATURES
Enerrgy
The figures below show the lattice energy before and after a bypasses an
obstacle on its slip plane.
Enerrgy
No stress
applied
With stress
applied
Eo
Eo
E
Obstacle to motion
Obstacle to motion
Position
Position
Eo is the activation energy that must be overcome by the dislocation to
cause it to move. Can be applied thermally (magnitude on the order of kT).
Applied stresses and thermal energy alters the energy-distance profile and
tends to drive the dislocation past the barrier.
Using principles of kinetics, we can derive a deformation rate.
Prof. M.L. Weaver
Rate at which s bypass obstacle (forward in stress-aided dir.):
(E E)
Forward rate exp o
kT
Thermal energy
Reverse reaction rate:
E
[Slower than in forward dir
dir.]]
Reverse rate exp o
kT
Net flow rate strain rate, scales with the difference betw.:
Eo
E 1
exp
kT
kT
dg o exp
Materials
parameter based
on freq. atomic
vibration
E
E
o exp o exp
kT
kT
Prof. M.L. Weaver
Work done by stress
to overcome barrier
Eo
bAs
exp
kT
kT
dg o exp
depends on ratio
bAs
kT
E
exp
1 at low T
kT
Work done by stress
to overcome barrier
Area of slip plane
This derivation is based on
slip in absence of diffusion.
Eqn. similar to creep eqn.
involving diffusion.
11
2/13/2012
Diffusional Flow Creep Mechanisms
Nabarro-Herring (NH) Creep
Occurs solely by bulk diffusional mass transport
((i.e., bulk diffusion).
)
Is important for at higher Ts and lower s than
was the case for dislocation glide creep.
Can
C occur in
i crystalline
lli andd amorphous
h
materials.
i l
Prof. M.L. Weaver
Diffusional Flow Creep Mechanisms contd
Nabarro-Herring (NH) Creep
Grain boundaries are sources and sinks for vacancies.
Under an applied stress vacancies will move to grain boundaries
with compressive stresses. Atoms will be drawn to grain
boundaries with tensile stresses.
This diffusion leads to shape change.
Applied stress
Arrows denote
vacancy flux
Vacancy
current
After flux
Contraction
Poisson effect
Original
shape
Vacancy sink
Vacancy source
Prof. M.L. Weaver
12
2/13/2012
At equilibrium, with no applied stress, the atomic fraction of vacancies at a
given temperature (T) is:
# vacancies
# atoms
Qf
nV
NV exp
no
kT
Qf = vacancy formation energy
The equilibrium concentration is thus:
Co
Qf
NV 1
exp
kT
= atomic volume
When a vacancy is created at a face under tension , it is assisted by a
contribution to the free energy. The opposite occurs on compressed
faces The local equilibrium concentrations at both faces becomes:
faces.
tension
compression
Ctensile Co exp
kT
Ccompressive Co exp
kT
Obviously the
concentration is higher
on the tensile side
Prof. M.L. Weaver
There will be a net flux of vacancies from the higher concentration (i.e.,
tensile) to the lower concentration (i.e., compressive) areas. From Ficks
law, this is:
C
J DV c DV
The vacancy diffusivity, DV, can be expressed as:
Q
DV DVo exp m
kT
Qm = vacancy migration energy
This allows us to re-write the flux as:
C
J DV
assuming that the diffusion distance (x) is equivalent to the grain size (d)
and that is a numerical constant.
Prof. M.L. Weaver
13
2/13/2012
Multiplication of the flux J by the diffusion area (~d2) gives the volumetric
flow rate which is the number of vacancies passing through an area per
second.
V
Jd 2
t
From this, the volume of vacancies arriving per second is . If we divide
by the diffusion area, we get the elongation per second, which can be
divided by the diffusion length (d) to yield the strain rate:
d
3 Jd 2 3
dt
d
d
or
C DV Co
exp
DV
exp
2
d
d
kT
kT
d
Prof. M.L. Weaver
exp
1
kT
kT
Since << kT:
Therefore:
C DV Co 2 2 DV Co
DV
1 kT 1 kT
d
d
d2
kTd 2
From physical metallurgy, the lattice self-diffusion coefficient DL is
given by:
DL DV Co
Thus, the NH creep rate is:
NH
2 DL
ANH
kTd 2
DL
2
d kT
Prof. M.L. Weaver
14
2/13/2012
The NH creep equation can be expressed more generally as
2
NH
ANH DLGb b
kT
d G
ANH 10 16
DL lattice diffusion coefficient
NH creep is competitive with glide creep
NH creep with T and
(where glide is not competitive)
More important in ceramics than in metals b/c glide is
more difficult in ceramics
Prof. M.L. Weaver
15
You might also like
- Media MBBR (BIOCIP)Document25 pagesMedia MBBR (BIOCIP)mektan0% (1)
- Design Against CreepDocument112 pagesDesign Against CreepRishabh TripathiNo ratings yet
- Publication 4 29484 1037Document9 pagesPublication 4 29484 1037peterNo ratings yet
- Thermal Cycling Effects On The Fatigue Behaviour of Low Carbon SteelDocument13 pagesThermal Cycling Effects On The Fatigue Behaviour of Low Carbon SteelShubhom PaulNo ratings yet
- Irradiation Creep of Nuclear Graphite: B. RandDocument35 pagesIrradiation Creep of Nuclear Graphite: B. RandWaleed ShakilNo ratings yet
- Fatigue and FractureDocument69 pagesFatigue and FractureBilel AydiNo ratings yet
- Creep Lab ManualDocument11 pagesCreep Lab ManualapproachtheprobNo ratings yet
- Deformation Mechanism Maps - Oct27Document41 pagesDeformation Mechanism Maps - Oct27Fame Boy SamNo ratings yet
- 34-Creep Deformation-31-10-2023Document22 pages34-Creep Deformation-31-10-2023NandiniNo ratings yet
- Yang EncyclopediaofMaterialComposites 2020Document14 pagesYang EncyclopediaofMaterialComposites 2020AnilNo ratings yet
- Creep ExperimentalDocument8 pagesCreep ExperimentalPanji Haryono AzisNo ratings yet
- Minerals Engineering: K.W. Chu, B. Wang, A.B. Yu, A. VinceDocument13 pagesMinerals Engineering: K.W. Chu, B. Wang, A.B. Yu, A. VinceNopparutNo ratings yet
- A True-Stress Creep Model Based On Deformation Mechanisms For Polycrystalline MaterialsDocument8 pagesA True-Stress Creep Model Based On Deformation Mechanisms For Polycrystalline Materialshassan ghorbaniNo ratings yet
- Instrumentation Training Tutorial1 PDFDocument6 pagesInstrumentation Training Tutorial1 PDFMOHAMED SHARKAWINo ratings yet
- Tutorial 1 - Basic Engineering Science: Instrumentation and ControlDocument6 pagesTutorial 1 - Basic Engineering Science: Instrumentation and Control@@ngNo ratings yet
- Tutorial 1 - Basic Engineering Science: Instrumentation and ControlDocument6 pagesTutorial 1 - Basic Engineering Science: Instrumentation and ControlMohd A IshakNo ratings yet
- Instrumentation Training Tutorial Part1Document6 pagesInstrumentation Training Tutorial Part1Gary8100% (1)
- Tutorial 1 - Basic Engineering Science: Instrumentation and ControlDocument6 pagesTutorial 1 - Basic Engineering Science: Instrumentation and Controls_waqarNo ratings yet
- Module # Module #39 39: Fatigue of MaterialsDocument26 pagesModule # Module #39 39: Fatigue of MaterialsSon TranNo ratings yet
- Binder CreepDocument29 pagesBinder CreepettypasewangNo ratings yet
- Obect:-Apparatus Required:: Practical No.1Document5 pagesObect:-Apparatus Required:: Practical No.1mohammad_nizamaniNo ratings yet
- Urvival at High Temperatures: Sub-Topics High Temperature Materials CreepDocument33 pagesUrvival at High Temperatures: Sub-Topics High Temperature Materials CreepessamNo ratings yet
- 2012.maroufi - Cond Rad BoltzmanDocument11 pages2012.maroufi - Cond Rad BoltzmanAT8iNo ratings yet
- Conflong CreepDocument31 pagesConflong CreepAnonymous LMfIjpENo ratings yet
- Time-Temperature Behavior of Carbon - Epoxy Laminates PDFDocument15 pagesTime-Temperature Behavior of Carbon - Epoxy Laminates PDFShabi HadashNo ratings yet
- Creep Analysis of Plastically Deformed Shrink-Fitted Joints: Researcharticle-MechanicalengineeringDocument12 pagesCreep Analysis of Plastically Deformed Shrink-Fitted Joints: Researcharticle-MechanicalengineeringshyamNo ratings yet
- Mse2 1Document55 pagesMse2 1binoNo ratings yet
- Celentano 2002Document13 pagesCelentano 2002fernando_iteNo ratings yet
- A0materials Science and EngineeringDocument58 pagesA0materials Science and EngineeringalfajNo ratings yet
- Circulation Rate Modelling of Mill ChargDocument8 pagesCirculation Rate Modelling of Mill ChargrodrigoNo ratings yet
- Case Study CreepDocument9 pagesCase Study CreepKevinSatriaNo ratings yet
- Creep and High Temperature Failure: OutlineDocument6 pagesCreep and High Temperature Failure: OutlineAravind PhoenixNo ratings yet
- Reactive Potentials and Applications (Cont'd) : Markus J. BuehlerDocument56 pagesReactive Potentials and Applications (Cont'd) : Markus J. Buehlerchethugowda7No ratings yet
- What About Polymer Melts?: Green-Tobolsky Temporary Network ModelDocument7 pagesWhat About Polymer Melts?: Green-Tobolsky Temporary Network ModelShawn WaltzNo ratings yet
- Creep ViscoelastDocument12 pagesCreep Viscoelastlrodriguez_892566No ratings yet
- Chap06 Mechanical BehaviorDocument14 pagesChap06 Mechanical BehaviorAli khan7No ratings yet
- Intro To Fracture Mechanics, Fatigue and CreepDocument40 pagesIntro To Fracture Mechanics, Fatigue and CreepM.USMAN BIN AHMED100% (1)
- Tuned Liquid DampersDocument13 pagesTuned Liquid DampersMahaveer JanapalaNo ratings yet
- Implications of Fault Constitutive Properties For Earthquake PredictionDocument8 pagesImplications of Fault Constitutive Properties For Earthquake PredictionJose Francisco AguilarNo ratings yet
- Creep Strength: Definitions: Creep Is The Tendency of A Solid Material To Move Slowly or Deform Permanently UnderDocument13 pagesCreep Strength: Definitions: Creep Is The Tendency of A Solid Material To Move Slowly or Deform Permanently UnderupenderNo ratings yet
- Failure in MaterialDocument22 pagesFailure in Materialnur afifahNo ratings yet
- Instrumentation Training Tutorial1 PDFDocument6 pagesInstrumentation Training Tutorial1 PDFchdiNo ratings yet
- Instrumentation BasicsTrainingDocument59 pagesInstrumentation BasicsTrainingSurya SenthilnathanNo ratings yet
- Microstructure Based Creep Constitutive Model For Precipitation Strengthened Alloys: Theory and ApplicationDocument8 pagesMicrostructure Based Creep Constitutive Model For Precipitation Strengthened Alloys: Theory and Applicationhassan ghorbaniNo ratings yet
- Chapter 8 Phase TransformationsDocument102 pagesChapter 8 Phase TransformationsAnup DalalNo ratings yet
- Background and Usage: CreepDocument8 pagesBackground and Usage: CreepharicoolestNo ratings yet
- Solid State PhysiscsDocument8 pagesSolid State Physiscssoumendra ghoraiNo ratings yet
- ViscoelasticidadDocument60 pagesViscoelasticidadMisael NavarroNo ratings yet
- Creepof CeramicsDocument14 pagesCreepof CeramicsTejinder SinghNo ratings yet
- 6.1 Objective:: Experiment No:06Document5 pages6.1 Objective:: Experiment No:06General ShiekhNo ratings yet
- Creep & FatigueDocument20 pagesCreep & Fatiguegaspardo123No ratings yet
- CREEPPAPERDocument8 pagesCREEPPAPERErick CamachoNo ratings yet
- Characterisation and Study of The Creep Behaviour of Polypropylene/ Calcium Carbonate NanocompositesDocument7 pagesCharacterisation and Study of The Creep Behaviour of Polypropylene/ Calcium Carbonate NanocompositesChinedum MgbemenaNo ratings yet
- Creep and FatigueDocument20 pagesCreep and Fatiguecheveresan123100% (2)
- 17.creep & FatigueDocument20 pages17.creep & FatigueaderancNo ratings yet
- Restlife AssessmentDocument12 pagesRestlife AssessmentsachzenNo ratings yet
- Treatise on Irreversible and Statistical Thermodynamics: An Introduction to Nonclassical ThermodynamicsFrom EverandTreatise on Irreversible and Statistical Thermodynamics: An Introduction to Nonclassical ThermodynamicsRating: 1 out of 5 stars1/5 (1)
- Barron's Physics Practice Plus: 400+ Online Questions and Quick Study ReviewFrom EverandBarron's Physics Practice Plus: 400+ Online Questions and Quick Study ReviewNo ratings yet
- Building Safe Lithium Ion Batteries For Electric Vehicles A ReviewDocument42 pagesBuilding Safe Lithium Ion Batteries For Electric Vehicles A ReviewWELENTON PANISSONNo ratings yet
- Rheology of Complex FluidsDocument130 pagesRheology of Complex FluidsghfdlfNo ratings yet
- Dokumen - Tips - New Waterborne Technologies For The Formulation of The Formulation of Problem SolvingDocument41 pagesDokumen - Tips - New Waterborne Technologies For The Formulation of The Formulation of Problem SolvingLong An Đỗ100% (1)
- Method For Emission Spectrometric Analysis of Plain Carbon and Low Alloy Steels Point To Plane Technique (Document8 pagesMethod For Emission Spectrometric Analysis of Plain Carbon and Low Alloy Steels Point To Plane Technique (DiptiNo ratings yet
- CH 08Document42 pagesCH 08chimenNo ratings yet
- Smartcare Multi Purpose PolymerDocument2 pagesSmartcare Multi Purpose PolymerPuneet ChahalNo ratings yet
- Corbel CalculationDocument1 pageCorbel CalculationQuangKhảiNo ratings yet
- Unacademy CapacitanceDocument39 pagesUnacademy CapacitanceMayank TripathiNo ratings yet
- MTech ENGINEERINGDESIGDocument53 pagesMTech ENGINEERINGDESIGVincent LinzieNo ratings yet
- ELS Module 4 RevisedDocument27 pagesELS Module 4 RevisedJane EntunaNo ratings yet
- ASTM D 5034 Breaking Strength/Elongation Grab MethodDocument9 pagesASTM D 5034 Breaking Strength/Elongation Grab MethodsaminaNo ratings yet
- Determination of Titratable Acidity in White WineDocument17 pagesDetermination of Titratable Acidity in White WineKannan RanganathanNo ratings yet
- General Chemistry 2: Third Quarter-Module 2Document18 pagesGeneral Chemistry 2: Third Quarter-Module 2Jezysaint Ruth Del SocorroNo ratings yet
- Api 510 Q&aDocument13 pagesApi 510 Q&aShaikh Aftab100% (2)
- Luminate PDS BF 10609 USLDocument2 pagesLuminate PDS BF 10609 USLArash AbbasiNo ratings yet
- Sydney Grammar 2019 Chemistry Trial PaperDocument62 pagesSydney Grammar 2019 Chemistry Trial PaperYuanfeng WeiNo ratings yet
- Phosphonate Persulfate UV Oxidation - AP-70 - 900Document6 pagesPhosphonate Persulfate UV Oxidation - AP-70 - 900wulalan wulanNo ratings yet
- Super Critical Boiler Commissioning1Document44 pagesSuper Critical Boiler Commissioning1Vishal SinghNo ratings yet
- Instar Vibration Testing of Small Satellites Part 3Document9 pagesInstar Vibration Testing of Small Satellites Part 3Youssef wagdyNo ratings yet
- PYROLUSITEDocument10 pagesPYROLUSITEenockelisantemsangiNo ratings yet
- M Tech ProjectDocument98 pagesM Tech Projectmanoj phadtareNo ratings yet
- Reduction of Casting Defects Using Taguchi Method: Arun Basil Jacob Arunkumar O.NDocument10 pagesReduction of Casting Defects Using Taguchi Method: Arun Basil Jacob Arunkumar O.NtabibkarimNo ratings yet
- Internship ReportDocument48 pagesInternship ReportSanket KharpudeNo ratings yet
- Industrial ElectrochemDocument11 pagesIndustrial ElectrochemHassan AgNo ratings yet
- Short Piles 2Document94 pagesShort Piles 2vivek_gesNo ratings yet
- Lecture #1 Introduc - On To Electrochemistry: Kaori SugiharaDocument47 pagesLecture #1 Introduc - On To Electrochemistry: Kaori SugiharaLoga NathanNo ratings yet
- Pyrolysis Technology, Equipment and Measuring Device: Syngas PyrolyzerDocument15 pagesPyrolysis Technology, Equipment and Measuring Device: Syngas PyrolyzerShantanuNo ratings yet
- Optimization of Drilling and Blasting Operations in An Open Pit Mine-The SOMAIR ExperienceDocument4 pagesOptimization of Drilling and Blasting Operations in An Open Pit Mine-The SOMAIR ExperienceWilkhen ChuraNo ratings yet