Professional Documents
Culture Documents
Insulin and Incretins
Insulin and Incretins
Uploaded by
pykkoCopyright:
Available Formats
You might also like
- David Dent - Insect Pest ManagementDocument425 pagesDavid Dent - Insect Pest Managementsaphito83% (6)
- Effect of Sunlight Against Green Beans GrowthDocument4 pagesEffect of Sunlight Against Green Beans GrowthDanny Ramadhan0% (1)
- History of Psychiatry: Hypnosis, False Memory and Multiple Personality: A Trinity of AffinityDocument10 pagesHistory of Psychiatry: Hypnosis, False Memory and Multiple Personality: A Trinity of AffinityJulianna BartalusNo ratings yet
- Amess Management of Hyper Triglyceride Mia in The Diabetic PatientDocument5 pagesAmess Management of Hyper Triglyceride Mia in The Diabetic Patientjimrod1950No ratings yet
- Effect of Diuretics On The Plasma Lipid Profile: KEY WORDS: Hypertension, Diuretics, Dyslipidaemia, Glucose IntoleranceDocument7 pagesEffect of Diuretics On The Plasma Lipid Profile: KEY WORDS: Hypertension, Diuretics, Dyslipidaemia, Glucose IntoleranceVims BatchNo ratings yet
- NursingresearchreportDocument12 pagesNursingresearchreportapi-300699057No ratings yet
- Markel 2011 - The Resurgence of Niacin, From Nicotinic Acid To NiaspanlaropiprantDocument7 pagesMarkel 2011 - The Resurgence of Niacin, From Nicotinic Acid To NiaspanlaropiprantAlbert CalvetNo ratings yet
- Diretrizes de Prática Clínica para o Tratamento de HipertrigliceriemiaDocument24 pagesDiretrizes de Prática Clínica para o Tratamento de Hipertrigliceriemiaqmatheusq wsantoswNo ratings yet
- Cardiovascular Outcomes StudiesDocument5 pagesCardiovascular Outcomes StudiesAnonymous LOeICSOqNo ratings yet
- Optimizing The Care of Patients With Type 2 Diabetes Using Incretin-Based Therapy: Focus On GLP-1 Receptor AgonistsDocument12 pagesOptimizing The Care of Patients With Type 2 Diabetes Using Incretin-Based Therapy: Focus On GLP-1 Receptor AgonistsRidha Surya NugrahaNo ratings yet
- Lipid Effects of Antihypertensive Medications: Nonstatin Drugs (W Borden, Section Editor)Document8 pagesLipid Effects of Antihypertensive Medications: Nonstatin Drugs (W Borden, Section Editor)Septi Fadhilah SPNo ratings yet
- Secondary Causes of DyslipidemiaDocument3 pagesSecondary Causes of DyslipidemiaAnghelo Aldair Velásquez CarrilloNo ratings yet
- Heart Views-Cardio GlicemicDocument24 pagesHeart Views-Cardio Glicemicsupit1No ratings yet
- Metabolic Syndrome in SaudisDocument4 pagesMetabolic Syndrome in SaudisOnon EssayedNo ratings yet
- Srikant H 2016Document10 pagesSrikant H 2016aditya sekarNo ratings yet
- Lipids Module 4Document16 pagesLipids Module 4angelmedurNo ratings yet
- ECBCM - Oral Meds T2 Diab - Clinician06282011Document8 pagesECBCM - Oral Meds T2 Diab - Clinician06282011Ridha Surya NugrahaNo ratings yet
- ProjectDocument32 pagesProjectNithya sudevNo ratings yet
- Precision MedicineDocument23 pagesPrecision MedicineLavina Jain100% (1)
- Linagliptin - Farmacology, Efficacy and SafetyDocument7 pagesLinagliptin - Farmacology, Efficacy and SafetyMisaelDominguezNo ratings yet
- Reduction of Plasma Triglycerides by Diet in Subjects With Chronic Renal FailureDocument15 pagesReduction of Plasma Triglycerides by Diet in Subjects With Chronic Renal FailureDina sayunaNo ratings yet
- Type 2 Diabetes Mellitus (Article) Author United States Department of Veterans AffairsDocument9 pagesType 2 Diabetes Mellitus (Article) Author United States Department of Veterans Affairsnaljnaby9No ratings yet
- Hyperlipidemia in Diabetes-Etiology, Consequences and Treatment Edward Shahady MD Athero-Thrombosis in DiabetesDocument6 pagesHyperlipidemia in Diabetes-Etiology, Consequences and Treatment Edward Shahady MD Athero-Thrombosis in DiabetesindahkurNo ratings yet
- Endocrinology and Metabolism Clinics-Lípidos 2009Document234 pagesEndocrinology and Metabolism Clinics-Lípidos 2009Tony Miguel Saba SabaNo ratings yet
- Meds DiabetesDocument5 pagesMeds DiabetesAnjangsari 'aRie' WijayantiNo ratings yet
- New Practice in Semaglutide On Type-2 Diabetes and Obesity: Clinical Evidence and ExpectationDocument8 pagesNew Practice in Semaglutide On Type-2 Diabetes and Obesity: Clinical Evidence and ExpectationBoris ThielmannNo ratings yet
- Green Cardamom... Improves Serum Irisin, Glucose Indices, and Lipid Profiles In... Non-Alcoholic Fatty Liver Disease Patients ...Document11 pagesGreen Cardamom... Improves Serum Irisin, Glucose Indices, and Lipid Profiles In... Non-Alcoholic Fatty Liver Disease Patients ...Daniel GilbertoNo ratings yet
- F 2433 CMED Saxagliptin A Selective DPP 4 Inhibitor For The Treatment of Type 2 D.PDF 3311Document12 pagesF 2433 CMED Saxagliptin A Selective DPP 4 Inhibitor For The Treatment of Type 2 D.PDF 3311Ridha Surya NugrahaNo ratings yet
- Beyond Blood Lipids: Phytosterols, Statins and Omega-3 Polyunsaturated Fatty Acid Therapy For HyperlipidemiaDocument13 pagesBeyond Blood Lipids: Phytosterols, Statins and Omega-3 Polyunsaturated Fatty Acid Therapy For HyperlipidemiaChristian Ivan SantosoNo ratings yet
- Management of Persistent Hyperglycemia in Type 2 Diabetes MellitusDocument47 pagesManagement of Persistent Hyperglycemia in Type 2 Diabetes MellitusEslam khedrNo ratings yet
- Bays 2013Document26 pagesBays 2013Lukman Al-HafizNo ratings yet
- Incretin Mimetics and Dipeptidyl Peptidase-4 Inhibitors: Innovative Treatment Therapies For Type 2 DiabetesDocument11 pagesIncretin Mimetics and Dipeptidyl Peptidase-4 Inhibitors: Innovative Treatment Therapies For Type 2 DiabetesRidha Surya NugrahaNo ratings yet
- Practical Laboratory Medicine: Subashini C. Thambiah, Leslie Charles LaiDocument21 pagesPractical Laboratory Medicine: Subashini C. Thambiah, Leslie Charles LaiAdina BrahaNo ratings yet
- Pathology of Type 2 DiabetesDocument4 pagesPathology of Type 2 DiabetesJenny SembranoNo ratings yet
- Profil Lipid Dan Risiko Penyakit Jantung Koroner Andi Surya AmalDocument5 pagesProfil Lipid Dan Risiko Penyakit Jantung Koroner Andi Surya AmalAyu Dewi SuwantiNo ratings yet
- DM2 and Thiazide 2018Document13 pagesDM2 and Thiazide 2018Osvaldo VillarNo ratings yet
- Cardio Metabolic Risk in DiabetesDocument29 pagesCardio Metabolic Risk in DiabetesoctoindradjajaNo ratings yet
- 2017 Article 551Document11 pages2017 Article 551Albert EdoNo ratings yet
- AntioksidanDocument5 pagesAntioksidanRakasiwi GalihNo ratings yet
- 2 - Metabolic Syndrome-Role of Dietary Fat TypeDocument7 pages2 - Metabolic Syndrome-Role of Dietary Fat TypeJuliana ZapataNo ratings yet
- Does Metformin Raise Cholesterol Levels - DIQDocument2 pagesDoes Metformin Raise Cholesterol Levels - DIQFathima Sheik KatherNo ratings yet
- Clinical Efficacy Evaluation and Longterm Prognosis of Glucagonlike Peptide1 Combined With Sodium Glucose CotransportersDocument9 pagesClinical Efficacy Evaluation and Longterm Prognosis of Glucagonlike Peptide1 Combined With Sodium Glucose CotransportersvinayNo ratings yet
- Original Article: Journal of Clinical Lipidology (2017)Document12 pagesOriginal Article: Journal of Clinical Lipidology (2017)IrhamNo ratings yet
- Glucagon-Like Peptide 1 수용체작용제 최신지견Document8 pagesGlucagon-Like Peptide 1 수용체작용제 최신지견이호원No ratings yet
- Retrospective Case Series of Patients With Diabetes or Prediabetes Who Were Switched From Omega-3-Acid Ethyl Esters To Icosapent EthylDocument11 pagesRetrospective Case Series of Patients With Diabetes or Prediabetes Who Were Switched From Omega-3-Acid Ethyl Esters To Icosapent EthylAgus SetiawanNo ratings yet
- j3 2010Document12 pagesj3 2010Taha FransNo ratings yet
- Diabetes and Hyperlipidemia: A Direct Quantitative AnalysisDocument6 pagesDiabetes and Hyperlipidemia: A Direct Quantitative AnalysisAnonymous Kc2tM8No ratings yet
- Clinical Trial 2016Document11 pagesClinical Trial 2016guugle gogleNo ratings yet
- "Dyslipidemia" : Dyslipidemia Caused by Excessive Hepatic Secretion of VLDLDocument5 pages"Dyslipidemia" : Dyslipidemia Caused by Excessive Hepatic Secretion of VLDLDANEKKA TANNo ratings yet
- Lipid Lowering DrugsDocument38 pagesLipid Lowering Drugsmannona200357No ratings yet
- Bipin 2023Document10 pagesBipin 2023Rajendra KulyeNo ratings yet
- Terapia Farmacologica Dell'obesità, 2008Document11 pagesTerapia Farmacologica Dell'obesità, 2008todino32No ratings yet
- Review Inhibition of Dipeptidyl Peptidase-4 With Vildagliptin A Potential New Treatment For Type 2 DiabetesDocument8 pagesReview Inhibition of Dipeptidyl Peptidase-4 With Vildagliptin A Potential New Treatment For Type 2 DiabetesAndhi FahrurrojiNo ratings yet
- Type 2 Diabetes TugaskuDocument4 pagesType 2 Diabetes Tugaskuzuhaela iqbalNo ratings yet
- Alessandro MantovaniDocument16 pagesAlessandro Mantovaniasma.almhsnNo ratings yet
- Hba1C: Predictor of Dyslipidemia and Atherogenicity in Diabetes MellitusDocument3 pagesHba1C: Predictor of Dyslipidemia and Atherogenicity in Diabetes MellitusPramod Reddy EvuriNo ratings yet
- SGLT2 Inhibitors in The Management of Type 2 Diabetes With CardiovascularRenal ComorbiditiDocument5 pagesSGLT2 Inhibitors in The Management of Type 2 Diabetes With CardiovascularRenal ComorbiditiShekharNo ratings yet
- Biomedicines 10 01090 v2Document18 pagesBiomedicines 10 01090 v2irvinpereda1997No ratings yet
- Effect of Insulin Analog Initiation Therapy On LDL/ HDL Subfraction Profile and HDL Associated Enzymes in Type 2 Diabetic PatientsDocument11 pagesEffect of Insulin Analog Initiation Therapy On LDL/ HDL Subfraction Profile and HDL Associated Enzymes in Type 2 Diabetic PatientsAan AlawiyahNo ratings yet
- Dyson LineDocument10 pagesDyson LineSharly DwijayantiNo ratings yet
- Objective:: BackgroundDocument23 pagesObjective:: BackgroundJanine DimaangayNo ratings yet
- Diabetic 2 Update Review !!Document82 pagesDiabetic 2 Update Review !!Patrick CommettantNo ratings yet
- Semaglutide Unveiled: A Comprehensive Guide to Diabetes Treatment and Weight ManagementFrom EverandSemaglutide Unveiled: A Comprehensive Guide to Diabetes Treatment and Weight ManagementNo ratings yet
- Vulovic - Brskovo Final PDFDocument27 pagesVulovic - Brskovo Final PDFDraganaNo ratings yet
- Kanker Ovarium 2 PDFDocument6 pagesKanker Ovarium 2 PDFhestiNo ratings yet
- Poster Session v3Document11 pagesPoster Session v3Fiola FinandakasihNo ratings yet
- WJ Ms Paper Sir 2013Document13 pagesWJ Ms Paper Sir 2013Rohan sharmaNo ratings yet
- High Risk NeonateDocument102 pagesHigh Risk NeonateJaya Prabha100% (2)
- Virology SummaryDocument3 pagesVirology SummaryvictorNo ratings yet
- (Download PDF) Essentials of Anatomy Physiology Kenneth Saladin Online Ebook All Chapter PDFDocument42 pages(Download PDF) Essentials of Anatomy Physiology Kenneth Saladin Online Ebook All Chapter PDFnigel.allen933100% (12)
- A Whole-Of-Nation Approach To COVID-19 Taiwan's National Epidemic Prevention TeamDocument16 pagesA Whole-Of-Nation Approach To COVID-19 Taiwan's National Epidemic Prevention Teamgrahma96No ratings yet
- Pneumonia: Nur Yahya, T 05/1087561/KU/11522Document19 pagesPneumonia: Nur Yahya, T 05/1087561/KU/11522Taufik Nur YahyaNo ratings yet
- Jurnal 5Document14 pagesJurnal 5mellvinNo ratings yet
- Selected Instructional Course LecturesDocument27 pagesSelected Instructional Course LecturesAhmad ShakirNo ratings yet
- Holoxan Pi PDFDocument18 pagesHoloxan Pi PDFKarol IonasNo ratings yet
- Human Biology 13Th Edition Mader Test Bank Full Chapter PDFDocument56 pagesHuman Biology 13Th Edition Mader Test Bank Full Chapter PDFninhdermotc1u100% (11)
- Icd 11 Introduction ClinicalDocument50 pagesIcd 11 Introduction ClinicalMuhammad Farooq Ul HassanNo ratings yet
- DocxDocument53 pagesDocxkuro hanabusaNo ratings yet
- COVID - 19 Treatment and VaccinesDocument2 pagesCOVID - 19 Treatment and Vaccinesridwan_olawale_1No ratings yet
- Prepared By: M.Bilal BSN, Rn. MSN UhsDocument52 pagesPrepared By: M.Bilal BSN, Rn. MSN UhsXweet AdilNo ratings yet
- Uterine Anomaly, Fibroid Uterus, Ovarian Tumor, Uterine ProlapseDocument30 pagesUterine Anomaly, Fibroid Uterus, Ovarian Tumor, Uterine ProlapseVijith.V.kumar80% (5)
- Assignment and Portfolio Consent FormDocument16 pagesAssignment and Portfolio Consent Formapi-549168092No ratings yet
- What Is A Root CanalDocument5 pagesWhat Is A Root CanaljjahaddinNo ratings yet
- Discharge PlaningDocument28 pagesDischarge PlaningRuhiyat Spd100% (1)
- Definitions and Reporting Framework For Tuberculosis - 2013 RevisionDocument47 pagesDefinitions and Reporting Framework For Tuberculosis - 2013 RevisionDavien UtoyoNo ratings yet
- LRM 1 Iv Bone Loss and Patterns of Bone DestructionDocument36 pagesLRM 1 Iv Bone Loss and Patterns of Bone DestructionSamridhi SrivastavaNo ratings yet
- Richa Shrivastava Assistant Professor Department of Pharmacy BITS PilaniDocument18 pagesRicha Shrivastava Assistant Professor Department of Pharmacy BITS PilaniCHOUGULE KISHOR SURYAKANTNo ratings yet
- Pulmonary Patho PDFDocument623 pagesPulmonary Patho PDFNabeel Shahzad100% (1)
- CHN - 1Document23 pagesCHN - 1Raagul SaravanaKumarNo ratings yet
Insulin and Incretins
Insulin and Incretins
Uploaded by
pykkoCopyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Insulin and Incretins
Insulin and Incretins
Uploaded by
pykkoCopyright:
Available Formats
Send Orders of Reprints at reprints@benthamscience.
org
Cardiovascular & Hematological Agents in Medicinal Chemistry, 2012, 10, 289-294
289
Mechanisms of Incretin Effects on Plasma Lipids and Implications for the
Cardiovascular System
Changting Xiao, Satya Dash and Gary F. Lewis*
Departments of Medicine and Physiology, University of Toronto, Toronto, ON, M5G 2C4, Canada
Abstract: Dyslipidemia is a prominent feature of type 2 diabetes and insulin resistance that contributes to increased
atherosclerosis and cardiovascular disease (CVD) risks under these conditions. Incretin-based therapies (GLP-1 receptor
agonists and DPP-4 inhibitors) have recently been developed and are approved for clinical use for treatment of type 2
diabetes. Besides improved glycemic control, other benefits are being increasingly appreciated, one of which is improved
plasma lipid profile. This review aims to summarize the evidence and potential mechanism of such agents in humans in
modulating fasting and postprandial lipoprotein metabolism.
Keywords: Apolipoprotein B-100, apolipoprotein B-48, cardiovascular disease, chylomicron, dipeptidyl peptidase-4,
dyslipidemia, glucagon-like peptide-1, incretin-based therapies, triglyceride-rich lipoprotein, triglycerides, type 2 diabetes, very
low-density lipoprotein.
INTRODUCTION
Type 2 diabetes (T2D) is a complex metabolic disorder
that is associated with an increased risk of cardiovascular
complications [1]. In addition to hyperglycemia,
dyslipidemia characterized by hypertriglyceridemia, low
plasma concentrations of high-density lipoprotein (HDL) and
qualitative changes in low-density lipoprotein (LDL) are
typically seen in patients with T2D and contribute to the
increased risk of cardiovascular complications [2].
Triglyceride (TG)-rich lipoproteins (TRL), comprised of
hepatically-derived apolipoprotein (apo) B-100-containing
very low-density lipoprotein (VLDL) and intestinallyderived apoB-48-containing chylomicrons, are elevated in
T2D and insulin resistance. VLDL TG overproduction
activates cholesteryl ester transfer protein that catalyzes the
exchange of TG and cholesterol ester between VLDL and
LDL and HDL. Subsequent hydrolysis of triglyceride
enriched LDL and HDL leads to the formation of small,
dense LDL particles and low HDL [3, 4]. Elevated TRL,
due to impaired clearance and increased production
from both the liver (apoB-100-containing VLDL) and
intestine (apoB-48-containing chylomicron), contribute to
hypertriglyceridemia [5]. In addition, it is now recognized
that postprandial lipemia, which is highly prevalent in obese
and insulin resistant individuals and T2D patients [6], is
associated with increased risks of atherosclerosis and CVD
[7,8]. Growing evidence suggests that increased intestinal
apoB-48-containing lipoprotein production is a feature of the
typical dyslipidemia of insulin resistance and T2D and may
contribute to the dyslipidemia that characterizes these
conditions [9].
*Address correspondence to this author at the Toronto General Hospital,
200 Elizabeth Street, EN12-218, Toronto, Ontario, M5G 2C4, Canada;
Tel: 1.416.340.4270; Fax: 1.416.340.3314; E-mail: gary.lewis@uhn.on.ca
1875-6182/12 $58.00+.00
Despite the availability of several commonly used antidiabetic drugs, achieving glycemic targets can be
challenging. It has been known for some time that oral
glucose ingestion results in increased insulin production
compared to when a similar blood glucose level is achieved
by intravenous glucose infusion, i.e. the incretin effect.
This effect is mediated mainly by two hormones: glucagonlike peptide-1 (GLP-1) and glucose-dependent insulinotropic
polypeptide (GIP) secreted by L cells and K cells of the
small intestine, respectively. Both hormones are rapidly
degraded by the enzyme dipeptidyl peptidase-4 (DPP-4).
Two new main types of incretin-based therapies have been
developed and therapeutic agents from these two main
classes are in increasing clinical use for treatment of T2D:
DPP-4 resistant GLP-1 receptor (GLP-1R) agonists (also
referred to as GLP-1 mimetics), and DPP-4 inhibitors or
incretin enhancers [10, 11]. Such incretin-based therapies
have shown promising efficacy in glycemic control [12].
GLP-1R agonists, such as exenatide, have multiple
glucoregulatory effects, including enhancing insulin
secretion and reducing glucagon secretion in a glucosedependent manner, reducing food intake (i.e. inducing
satiety), promoting weight loss, and inhibiting gastric
emptying [13, 14]. DPP-4 inhibitors also improve glycemic
control but differ from GLP-1R agonists in that they are
administered orally, do not reduce body weight and have
minimal effects on satiety [15]. Emerging evidence suggest
that incretin-based therapies offer benefits beyond glycemic
control, one of which is an improvement in the plasma lipid
profile [16]. This could potentially be of added therapeutic
value in the management of dyslipidemia and may translate
into reductions in CVD in patients with T2D. This review
aims to summarize clinical evidence, discuss possible
mechanisms and implications of incretin-based therapies in
lipid metabolism.
2012 Bentham Science Publishers
290 Cardiovascular & Hematological Agents in Medicinal Chemistry, 2012, Vol. 10, No. 4
ROLE OF INCRETIN-BASED
PLASMA LIPID PROFILE
THERAPIES
ON
Improvement in plasma lipid profile has been reported in
several clinical trials designed to examine efficacy of GLP1R agonists or DPP-4 inhibitors on glycemic control in T2D
patients. Since these studies were not specifically designed
to examine their effects on lipid metabolism, which are
likely to be far more pronounced in the postprandial than in
the fasting state, only minimal effects were noticed in most
of the studies that do report changes in lipid profile [16].
Nevertheless, incretin-based therapies have demonstrated
favorable changes in fasting lipid levels and several
cardiovascular risk factors.
Effect of Incretin-based Therapies on Fasting Plasma
Lipid Profile, Particle Size and other Cardiovascular
Risk Factors
Several studies have reported improved fasting plasma
lipid profile with incretin-based therapies. Treatment with
exenatide for 2 yrs and 3.5 yrs reduced fasting TG (-12%),
total cholesterol (-5%) and LDL-c (-6%), and increased
HDL-c (+24%) 1. Reduction in fasting TG (-15%) and
greater reduction of LDL-c (-0.13 mmol/L) from baseline
was also reported with the long-acting release form of
exenatide (once-weekly injection) during 30 week treatment
[17]. A direct comparison between once weekly and
twice daily exenatide for 52 wks demonstrated similar
improvement in fasting lipids (total cholesterol and TG)
[18]. In addition, adding exenatide to sulfonylurea therapy
led to small reductions in LDL-c and apoB concentrations
during 30 weeks of treatment [19]. In patients continuing
metformin and/or sulfonylureas, treatment with exenatide for
3.5 years decreased fasting TG (-12%), total cholesterol (5%) and LDL-c (-6%), and increased HDL-c (+24%) [20].
Exenatide once weekly or sitagliptin also slightly improved
HDL-c after 26 weeks treatment [21]. A retrospective
analysis of 110 patients treated with liraglutide indicates
reduced TG from 173 +/- 19 mg/dL at baseline to 151 +/- 15
mg/dL after 7.5 months of treatment [22]. The efficacy of
DPP-4 inhibitors on fasting lipids has been systematically
reviewed, showing either a small improvement in HDL,
LDL-c and TG, or no benefit [15]. Improved diabetes control
is generally accompanied by improvement in dyslipidemia.
The effects of incretin-based therapies on lipids have not
been directly compared with other commonly used antidiabetes medication, such as metformin or sulfonylurea.
However, in studies where exenatide improved fasting lipids
when added to metformin or sulfonylurea [19, 20] and where
exenatide attenuated postprandial TG and apoB-48 compared
with glargine [23], the observed improvement in lipid
parameters suggests additional benefits of incretin-based
therapies on dyslipidemia.
Besides fasting lipid levels, favorable changes in
lipoprotein particle size were reported with exenatide
treatment. For instance, one-year treatment with exenatide
increased particle size for LDL and HDL, increased
concentrations of large LDL and HDL, and reduced small
and very small LDL, which represents a less atherogenic
profile [24]. Increased small, dense LDL particles is an
important component of diabetic dyslipidemia, hence a
reduction in this entity would suggest reduced cardiovascular
risks. More detailed evaluation of the impact of incretinbased therapies on lipoprotein particle size requires further
study.
In addition, several studies have noted improvement in
blood pressure. For instance, exenatide treatment for 82
weeks improved cardiovascular risk factors, such as HDL-c,
TG and diastolic blood pressure [25]. Systolic blood pressure
was improved with once weekly administration of exenatide
or sitagliptin monotherapy [21]. Improved blood pressure
was also recorded with exenatide treatment over 3.5 yrs in
T2D patients continuing metformin and/or sulfonylureas
[20], as well as with liraglutide treatment of 7.5 months [22].
Improved blood pressure control reduces cardiovascular
complications in T2D patients [26]. Therefore, incretinbased therapies may provide benefits in reducing
cardiovascular risks in this regard, although the exact
mechanisms have not been clearly defined.
Postprandial Lipemia
Significant benefits of GLP-1R agonists on lipid
metabolism have been demonstrated in several studies in
humans. Although fasting lipid profile was not affected,
exenatide treatment for 1 year significantly decreased
postprandial incremental area under the curve of TG, free
fatty acids, apoB-48 and VLDL-c, with a smaller decline in
postprandial HDL-c compared to patients treated with
glargine (an ultra-long acting insulin), despite similar
glycemic control [23]. Meier et al., reported that continuous
GLP-1 infusion in healthy subjects for 390 min completely
abolished postprandial TG excursion (concentrations and
area under the curve) in response to a test meal [27]. In
individuals with impaired glucose tolerance or T2D, a single
s.c. dose of exenatide attenuated postprandial elevation of
serum TG, apoB-48, apoC-III, and cholesterol and TG in
remnant-like particles [28]. Such acute effects on
postprandial lipemia have not been directly examined with
DPP-4 inhibitors. However, 4-wk treatment of T2D patients
with vildagliptin decreased plasma TG, chylomicron TG and
apoB-48, and chylomicron cholesterol response to a fat-rich
meal, despite unchanged fasting lipids, total apoB, and TRL
apoB-48 and B-100 levels [29]. These studies thus point to a
more significant role of incretin-based therapies on
postprandial lipemia as compared to fasting lipids.
Potential
Mechanisms
Whereby
Therapies Improve Lipid Metabolism
Kendall, D.; Blonde, L.; Mac, S.; Guan, X.; Holcombe, J. H.; Okerson, T.;
Kim, D.; Bhole, D. Improvements in cardiovascular risk factors
accompanied improved glycemic control and weight reduction in patients
with type 2 diabetes treated with exenatide for 3.5 y. Diabetes 2007, 56
(Suppl.1), A149.
Xiao et al.
Incretin-based
Modulation of Pancreatic Function
GLP-1R agonists acutely modulate the secretion of
pancreatic hormones. GLP-1 is a potent modulator of islet
function, stimulating glucose-dependent insulin secretion
Incretin Effects on Lipids
Cardiovascular & Hematological Agents in Medicinal Chemistry, 2012, Vol. 10, No. 4
and suppressing glucagon secretion [11]. Improved
pancreatic beta-cell function was also reported in several
studies with chronic administration of liraglutide [30, 31],
exenatide [25] and vildagliptin [32-34]. In addition,
vildagliptin enhances alpha-cell responsiveness to both the
suppressive effects of hyperglycemia and the stimulatory
effects of hypoglycemia [35]. TRL production in liver and
intestine is acutely inhibited by insulin [36-41] and glucagon
may mediate lipid homeostasis under certain conditions [42,
43]. The mechanisms whereby insulin suppresses TRL
production have been studied extensively in vitro and in vivo
in animals and humans [5]. Collectively, insulin may
indirectly suppress TRL production by lowering circulating
free fatty acids, which stimulate TRL production, and
directly mediate lipoprotein synthesis through insulin
signaling pathway. The latter includes decreased microsomal
triglyceride transfer protein gene expression and activity
and increased posttranslational apoB degradation. Acute
stimulation of insulin secretion and suppression of glucagon
secretion or chronic improvement in beta-cell function or
with GLP-1R agonists or DPP-4 inhibitors thus may account
for at least part of their effects on lipid homeostasis.
Improved Metabolic Control
Improved metabolic status, especially glycemia control,
may contribute to improved lipid homeostasis with incretinbased therapies. Overproduction of hepatic lipoprotein
particles in T2D is exacerbated by hyperglycemia [44].
Glucose lowering with GLP-1R agonists or DPP-4 inhibitors
is thus expected to attenuate TRL production, at least in the
liver. Improved glycemic control by incretin-based therapies
may be attributed to enhanced pancreatic function (see
above) and attenuated insulin resistance. Both GLP-1R
agonists and DPP-4 inhibitors may improve insulin
sensitivity, possibly secondary to reduction in glucose
toxicity but an acute, insulin sensitizing effect of these
agents, independent of improvement in glycemic control, has
also been described [45-48]. Both hepatic and intestinal TRL
production are increased in insulin resistance and T2D [5, 9].
In humans, TRL production correlates with degree of insulin
resistance [37]. Improving insulin sensitivity with
rosiglitazone attenuated TRL overproduction in animals
[38]. In this regard, amelioration of insulin resistance with
incretin-based therapies is expected to attenuate hepatic and
intestinal lipoprotein production. In addition, improvement
in lipid profile with exenatide treatment for 1-yr was
accompanied by lower postprandial oxidative stress markers
malondialdehyde and oxidized LDL [23]. Since postprandial
hyperglycemia and dyslipidemia increase oxidative stress
which has been associated with CVD risks in T2D [49],
reduction in oxidative stress along with improvement in
hyperglycemia and dyslipidemia with incretin mimetics or
enhancers may contribute to improved cardiovascular
outcomes. Therefore, incretin-based therapies may improve
lipid homeostasis through mechanisms beyond glycemic
control, although the relative contribution of these effects
needs more detailed studies.
Chronically, incretin-based therapies may also indirectly
modulate lipid profile through their well-established effects
on weight loss (in the case of GLP-1R agonists but not DPP4 inhibitors), increased satiety and reduced food intake [16].
291
Improved fasting lipid profile with long-term use of such
agents generally parallels weight loss [20]. The greatest
improvement in cardiovascular risk factors was seen in those
patients who had the greatest reductions in weight [25]. On
the other hand, improved lipid profiles were also observed in
patients with minimal or no reduction in body weight [20,
25], suggesting mechanisms beyond weight loss. Compared
with GLP-1R agonists, DPP-4 inhibitors are relatively
weight neutral. Therefore, DPP-4 inhibitors and GLP-1R
agonists may not have fully overlapping mechanisms.
Evidence in Support of a Direct Effect of Incretin-based
Therapies on Lipoprotein Production
Aside from the above-discussed mechanisms, a direct
role of GLP-1R on enterocytes and hepatocytes in
modulating lipid metabolism is emerging. We have recently
examined the mechanism of GLP-1R on TRL kinetics in
healthy humans [50]. The study was designed to assess the
direct effects of exenatide on TRL metabolism in the
absence of weight loss, improvements in glycemia, changes
in pancreatic hormone secretion and gastric emptying.
A single s.c. dose (10 ug) of exenatide was given to
non-diabetic, healthy individuals and TRL kinetics were
assessed under conditions of a pancreatic clamp (in which
somatostatin was infused to suppress insulin and glucagon
secretion and these hormones were replaced at a basal rate).
To circumvent the well-described inhibition of gastric
emptying by exenatide, we infused a lipid/carbohydrate
liquid formula directly into the duodenum through a
nasoduodenal tube. Preliminary results indicate that acute
exenatide directly suppressed apoB-48 production rate, an
effect independent of gastric emptying and not fully
accounted for by changes in pancreatic hormone secretion.
No effect on apoB-100 was observed in our study. These
results are in line with the report that exenatide treatment for
1-yr in T2D patients decreased postprandial serum apoB-48
but not VLDL apoB-100 concentration [23]. In animal
models, a GLP-1R agonist decreased VLDL production [51],
an observation yet to be established in humans. Suppression
of VLDL production is expected to decrease circulating
levels of intestinal lipoprotein via increased clearance, since
TRL of hepatic and intestinal origins share a common,
saturable removal pathway [52]. Since TRL TG levels were
not affected in our study, the results suggested secretion of
fewer numbers but greater lipid enrichment of lipoprotein
particles with exenatide treatment. It seems that exenatide
suppressed intestinal TRL production at least partly by
suppressing not only apoB-48 protein synthesis but also
chylomicron packaging with neutral lipids, without
suppressing microsomal triglyceride transfer protein.
Whether exenatide suppressed apoB mRNA translation,
apoB intracellular trafficking and stability in the enterocyte
remains unknown and further studies are required to
elucidate the underlining molecular mechanisms.
The effect of exenatide on intestinal lipoprotein
production is supported by several other studies in animal
models in vivo and ex vivo. GLP-1 infusion in rats decreased
intestinal lipid absorption, lymph flow and lymphatic apoB
secretion [53]. In this study, absorption of lipids infused into
the duodenum was reduced; therefore it cannot be attributed
to slowed gastric emptying. GLP-1R signaling has been
292 Cardiovascular & Hematological Agents in Medicinal Chemistry, 2012, Vol. 10, No. 4
Xiao et al.
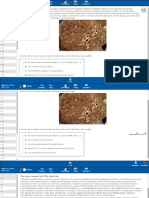
Mechanisms of Incretin-based Therapies on TG-rich Lipoprotein Metabolism
GLP-1 mimetics
Satiety
Weight loss
Slowed gastric
emptying and fat
absorption
GLP-1 activity
Inhibited gut and /
or liver TRL
particle production
DPP-4 inhibitors
Improved
glycemic
control
Stimulated
insulin
secretion
Inhibited
glucagon
secretion
Improved
lipid profile
Fig. (1). Potential mechanisms of incretin effects on lipids. Incretin-based therapies, i.e. GLP-1R agonists (incretin mimetics) and DPP-4
inhibitors (incretin enhancers) elicit improvements in lipid profile via several potential mechanisms. Activation of GLP-1R may slow gastric
emptying and intestinal motility, induce satiety, result in weight loss, improve glycemic control, and modulate pancreatic hormone secretion.
DPP-4 inhibitors are thought to exert their effects by increasing circulating levels of incretins. Emerging evidence points to possible direct
effects of incretin-based therapeutic agents on intestinal lipoprotein production.
shown to be important for postprandial TRL lipoprotein
synthesis in animal models [54], thus TRL TG and apoB-48
and plasma TG were decreased by the GLP-1R agonist
exendin-4 and the DPP-4 inhibitor sitagliptin, but were
increased by the GLP-1R antagonist exendin (9-39) [55]. In
addition, Glp-1r knockout mice had increased TRL TG and
apoB and exendin-4 inhibited apoB-48 synthesis in isolated
primary hamster enterocytes [55], further indicating a direct
role of GLP-1R in intestinal lipoprotein metabolism.
DPP-4 inhibitors may affect lipid metabolism by
increasing circulating levels of intact, biologically active
incretins [29, 33], subsequent modulation of pancreatic
hormone secretion, improved metabolic status, attenuated
insulin resistance, slowed gastric emptying, and possibly
direct effects of incretins on lipid metabolism. DPP-4
inhibitors not only enhance GLP-1, but also GIP [33]
and other substances, including neuropeptides, other
gastrointestinal hormones, cytokines or chemokines [56].
Contribution of these other peptides to lipid metabolism is
difficult to determine but cannot be ruled out. In dogs,
infusion of GIP attenuated postprandial TG excursion by
stimulating chylomicron clearance [57]. The relative
contribution of intestinal lipoprotein production and removal
require further elucidation, particularly for DPP-4 inhibitors.
In addition, it remains to be determined whether DPP-4
inhibitors affect lipid metabolism independently of their
effects on GLP-1 and GIP.
CONCLUDING REMARKS
Incretin-based therapies have been used primarily as
glucose-lowering agents in T2D, but additional therapeutic
benefits beyond improvements in glycemic control are
increasingly being recognized. One of the recently
appreciated benefits is improvement of the fasting plasma
lipid profile (in the case of GLP-1R agonists) and reduction
of postprandial lipemia (with both GLP-1R agonists and
DPP-4 inhibitors). The exact mechanisms whereby incretin
mimetics or enhancers modulate lipid homeostasis are still
under active investigation. Although their effects on glucose,
body weight, pancreatic hormone secretion and intestinal
motility may account for some of their lipid benefits, a direct
role of these agents on intestinal (and less so hepatic) lipid
metabolism is increasingly being recognized (Fig. 1).
Incretin-based therapies are recommended as a second line
treatment option in patients with T2D [58]. Considering the
multiple benefits of these agents, including improved
pancreatic beta-cell function, insulin sensitivity, lipid profile
and cardiovascular risk factors, use of incretin mimetics or
enhancers at earlier stage has been proposed [59]. Studies are
currently underway to determine whether these agents are
associated with reduction in cardiovascular events.
CONFLICT OF INTEREST
The author(s) confirm that this article content has no
conflicts of interest.
ACKNOWLEDGEMENTS
Research in our lab has been supported by operating
grants from the Canadian Institutes of Health Research.
G.F.L. holds the Drucker Family Chair in Diabetes Research.
S.D. is the recipient of a postdoctoral fellowship award
of the Banting and Best Diabetes Centre, University of
Toronto.
Incretin Effects on Lipids
Cardiovascular & Hematological Agents in Medicinal Chemistry, 2012, Vol. 10, No. 4
[17]
ABBREVIATIONS
Apo
apolipoprotein
CVD
cardiovascular disease
DPP-4
dipeptidyl peptidase-4
GIP
glucose-dependent
peptide
GLP-1
glucagon-like peptide-1
GLP-1R
GLP-1 receptor
HDL
high-density lipoprotein
LDL
low-density lipoprotein
T2D
type 2 diabetes
TG
triglycerides
TRL
triglyceride-rich lipoprotein
VLDL
very low-density lipoprotein
insulinotropic
[18]
poly[19]
[20]
[21]
[22]
REFERENCES
[1]
[2]
[3]
[4]
[5]
[6]
[7]
[8]
[9]
[10]
[11]
[12]
[13]
[14]
[15]
[16]
Klein, R. Hyperglycemia and microvascular and macrovascular
disease in diabetes. Diabetes Care, 1995, 18(2), 258-268.
Ginsberg, H.N.; Zhang, Y.L.; Hernandez-Ono, A. Regulation of
plasma triglycerides in insulin resistance and diabetes. Arch. Med.
Res., 2005, 36(3), 232-240.
Miller, M.; Stone, N.J.; Ballantyne, C.; Bittner, V.; Criqui, M.H.;
Ginsberg, H.N.; Goldberg, A.C.; Howard, W.J.; Jacobson, M.S.;
Kris-Etherton, P.M.; Lennie, T.A.; Levi, M.; Mazzone, T.;
Pennathur, S. Triglycerides and cardiovascular disease: a scientific
statement from the American Heart Association. Circulation, 2011,
123(20), 2292-2333.
Lewis, G.F.; Rader, D.J. New insights into the regulation of HDL
metabolism and reverse cholesterol transport. Circ. Res., 2005,
96(12), 1221-1232.
Xiao, C.; Hsieh, J.; Adeli, K.; Lewis, G.F. Gut-liver interaction in
triglyceride-rich lipoprotein metabolism. Am. J. Physiol. Endocrinol.
Metab., 2011, 301(3), E429-E446.
Taskinen, M.R. Diabetic dyslipidaemia: from basic research to
clinical practice. Diabetologia, 2003, 46(6), 733-749.
Bansal, S.; Buring, J.E.; Rifai, N.; Mora, S.; Sacks, F.M.; Ridker,
P.M. Fasting compared with nonfasting triglycerides and risk of
cardiovascular events in women. JAMA, 2007, 298(3), 309-316.
Mero, N.; Malmstrom, R.; Steiner, G.; Taskinen, M. R.; Syvanne,
M. Postprandial metabolism of apolipoprotein B-48- and B-100containing particles in type 2 diabetes mellitus: relations to
angiographically verified severity of coronary artery disease.
Atherosclerosis, 2000, 150(1), 167-177.
Adeli, K.; Lewis, G.F. Intestinal lipoprotein overproduction in
insulin-resistant states. Curr. Opin. Lipidol., 2008, 19(3), 221-228.
Drucker, D.J. The biology of incretin hormones. Cell Metab., 2006,
3(3), 153-165.
Drucker, D.J.; Nauck, M.A. The incretin system: glucagon-like
peptide-1 receptor agonists and dipeptidyl peptidase-4 inhibitors in
type 2 diabetes. Lancet, 2006, 368(9548), 1696-1705.
Lovshin, J.A.; Drucker, D.J. Incretin-based therapies for type 2
diabetes mellitus. Nat. Rev. Endocrinol., 2009, 5(5), 262-269.
Nielsen, L.L.; Young, A.A.; Parkes, D.G. Pharmacology of
exenatide (synthetic exendin-4): a potential therapeutic for
improved glycemic control of type 2 diabetes. Regul. Pept., 2004,
117(2), 77-88.
Gentilella, R.; Bianchi, C.; Rossi, A.; Rotella, C.M. Exenatide: a
review from pharmacology to clinical practice. Diabetes Obes.
Metab., 2009, 11(6), 544-556.
Amori, R.E.; Lau, J.; Pittas, A.G. Efficacy and safety of incretin
therapy in type 2 diabetes: systematic review and meta-analysis.
JAMA, 2007, 298(2), 194-206.
Mudaliar, S.; Henry, R. R. Incretin therapies: effects beyond
glycemic control. Eur. J. Intern. Med., 2009, 20(Suppl. 2), S319S328.
[23]
[24]
[25]
[26]
[27]
[28]
[29]
[30]
[31]
[32]
293
Drucker, D.J.; Buse, J.B.; Taylor, K.; Kendall, D.M.; Trautmann,
M.; Zhuang, D.; Porter, L. Exenatide once weekly versus twice
daily for the treatment of type 2 diabetes: a randomised, open-label,
non-inferiority study. Lancet, 2008, 372(9645), 1240-1250.
Buse, J.B.; Drucker, D.J.; Taylor, K.L.; Kim, T.; Walsh, B.; Hu, H.;
Wilhelm, K.; Trautmann, M.; Shen, L.Z.; Porter, L.E.
DURATION-1: exenatide once weekly produces sustained
glycemic control and weight loss over 52 weeks. Diabetes Care
2010, 33(6), 1255-1261.
Buse, J.B.; Henry, R.R.; Han, J.; Kim, D.D.; Fineman, M.S.;
Baron, A.D. Effects of exenatide (exendin-4) on glycemic control
over 30 weeks in sulfonylurea-treated patients with type 2 diabetes.
Diabetes Care, 2004, 27(11), 2628-2635.
Klonoff, D.C.; Buse, J.B.; Nielsen, L.L.; Guan, X.; Bowlus, C.L.;
Holcombe, J.H.; Wintle, M.E.; Maggs, D.G. Exenatide effects on
diabetes, obesity, cardiovascular risk factors and hepatic
biomarkers in patients with type 2 diabetes treated for at least 3
years. Curr. Med. Res. Opin., 2008, 24(1), 275-286.
Bergenstal, R.M.; Wysham, C.; MacConell, L.; Malloy, J.; Walsh,
B.; Yan, P.; Wilhelm, K.; Malone, J.; Porter, L.E. Efficacy and
safety of exenatide once weekly versus sitagliptin or pioglitazone
as an adjunct to metformin for treatment of type 2 diabetes
(DURATION-2): a randomised trial. Lancet, 2010, 376(9739),
431-439.
Varanasi, A.; Patel, P.; Makdissi, A.; Dhindsa, S.; Chaudhuri, A.;
Dandona, P. Clinical use of liraglutide in type 2 diabetes, and its
effects on cardiovascular risk factors. Endocr. Pract., 2012, 18(2),
104-145.
Bunck, M.C.; Corner, A.; Eliasson, B.; Heine, R.J.; Shaginian,
R.M.; Wu, Y.; Yan, P.; Smith, U.; Yki-Jarvinen, H.; Diamant, M.;
Taskinen, M.R. One-year treatment with exenatide vs. insulin
glargine: effects on postprandial glycemia, lipid profiles, and
oxidative stress. Atherosclerosis, 2010, 212(1), 223-229.
Kapitza, C.; Nauck, M.A.; Kim, D.; Trautmann, M.; Johns, D.;
Festa, A. Long-term treatment with exenatide improved
postprandial glyceric control and was associated with a shift from
small to large HDL and LDL particles. Diabetologia, 2006,
49(Suppl.1), 140-141.
Blonde, L.; Klein, E.J.; Han, J.; Zhang, B.; Mac, S.M.; Poon, T.H.;
Taylor, K.L.; Trautmann, M.E.; Kim, D.D.; Kendall, D.M. Interim
analysis of the effects of exenatide treatment on A1C, weight and
cardiovascular risk factors over 82 weeks in 314 overweight
patients with type 2 diabetes. Diabetes Obes. Metab., 2006, 8(4),
436-447.
Tight blood pressure control and risk of macrovascular and
microvascular complications in type 2 diabetes: UKPDS 38.
UK Prospective Diabetes Study Group. BMJ, 1998, 317(7160),
703-713.
Meier, J.J.; Gethmann, A.; Gotze, O.; Gallwitz, B.; Holst, J.J.;
Schmidt, W.E.; Nauck, M.A. Glucagon-like peptide 1 abolishes the
postprandial rise in triglyceride concentrations and lowers levels of
non-esterified fatty acids in humans. Diabetologia 2006, 49(3),
452-458.
Schwartz, E.A.; Koska, J.; Mullin, M.P.; Syoufi, I.; Schwenke,
D.C.; Reaven, P.D. Exenatide suppresses postprandial elevations in
lipids and lipoproteins in individuals with impaired glucose
tolerance and recent onset type 2 diabetes mellitus. Atherosclerosis,
2010, 212(1), 217-222.
Matikainen, N.; Manttari, S.; Schweizer, A.; Ulvestad, A.;
Mills, D.; Dunning, B.E.; Foley, J.E.; Taskinen, M.R. Vildagliptin
therapy reduces postprandial intestinal triglyceride-rich lipoprotein
particles in patients with type 2 diabetes. Diabetologia, 2006,
49(9), 2049-2057.
Mari, A.; Nielsen, L.L.; Nanayakkara, N.; DeFronzo, R.A.;
Ferrannini, E.; Halseth, A. Mathematical modeling shows
exenatide improved beta-cell function in patients with type 2
diabetes treated with metformin or metformin and a sulfonylurea.
Horm. Metab Res., 2006, 38(12), 838-844.
Mari, A.; Degn, K.; Brock, B.; Rungby, J.; Ferrannini, E.; Schmitz,
O. Effects of the long-acting human glucagon-like peptide-1 analog
liraglutide on beta-cell function in normal living conditions.
Diabetes Care, 2007, 30(8), 2032-2033.
Foley, J.E.; Bunck, M.C.; Moller-Goede, D.L.; Poelma, M.;
Nijpels, G.; Eekhoff, E.M.; Schweizer, A.; Heine, R.J.; Diamant,
M. Beta cell function following 1 year vildagliptin or placebo
treatment and after 12 week washout in drug-naive patients with
294 Cardiovascular & Hematological Agents in Medicinal Chemistry, 2012, Vol. 10, No. 4
[33]
[34]
[35]
[36]
[37]
[38]
[39]
[40]
[41]
[42]
[43]
[44]
[45]
type 2 diabetes and mild hyperglycaemia: a randomised controlled
trial. Diabetologia 2011, 54(8), 1985-1991.
Mari, A.; Sallas, W.M.; He, Y.L.; Watson, C.; Ligueros-Saylan,
M.; Dunning, B.E.; Deacon, C.F.; Holst, J.J.; Foley, J.E.
Vildagliptin, a dipeptidyl peptidase-IV inhibitor, improves modelassessed beta-cell function in patients with type 2 diabetes. J. Clin.
Endocrinol. Metab., 2005, 90(8), 4888-4894.
Ahren, B.; Schweizer, A.; Dejager, S.; Villhauer, E.B.; Dunning,
B.E.; Foley, J.E. Mechanisms of action of the dipeptidyl peptidase4 inhibitor vildagliptin in humans. Diabetes Obes. Metab., 2011,
13(9), 775-783.
Ahren, B.; Schweizer, A.; Dejager, S.; Dunning, B.E.; Nilsson,
P.M.; Persson, M.; Foley, J.E. Vildagliptin enhances islet
responsiveness to both hyper- and hypoglycemia in patients with
type 2 diabetes. J. Clin. Endocrinol. Metab., 2009, 94(4), 12361243.
Adiels, M.; Westerbacka, J.; Soro-Paavonen, A.; Hakkinen, A.M.;
Vehkavaara, S.; Caslake, M.J.; Packard, C.; Olofsson, S.O.; YkiJarvinen, H.; Taskinen, M.R.; Boren, J. Acute suppression of
VLDL1 secretion rate by insulin is associated with hepatic fat
content and insulin resistance. Diabetologia, 2007, 50(11), 23562365.
Duez, H.; Lamarche, B.; Uffelman, K.D.; Valero, R.; Cohn, J.S.;
Lewis, G.F. Hyperinsulinemia is associated with increased
production rate of intestinal apolipoprotein B-48-containing
lipoproteins in humans. Arterioscler. Thromb. Vasc. Biol., 2006,
26(6), 1357-1363.
Lewis, G.F.; Uffelman, K.; Naples, M.; Szeto, L.; Haidari, M.;
Adeli, K. Intestinal lipoprotein overproduction, a newly recognized
component of insulin resistance, is ameliorated by the insulin
sensitizer rosiglitazone: studies in the fructose-fed Syrian golden
hamster. Endocrinology, 2005, 146(1), 247-255.
Malmstrom, R.; Packard, C.J.; Watson, T.D.; Rannikko, S.;
Caslake, M.; Bedford, D.; Stewart, P.; Yki-Jarvinen, H.; Shepherd,
J.; Taskinen, M.R. Metabolic basis of hypotriglyceridemic effects
of insulin in normal men. Arterioscler. Thromb. Vasc. Biol., 1997,
17(7), 1454-1464.
Malmstrom, R.; Packard, C.J.; Caslake, M.; Bedford, D.; Stewart,
P.; Yki-Jarvinen, H.; Shepherd, J.; Taskinen, M.R. Effects of
insulin and acipimox on VLDL1 and VLDL2 apolipoprotein B
production in normal subjects. Diabetes, 1998, 47(5), 779-787.
Pavlic, M.; Xiao, C.; Szeto, L.; Patterson, B.W.; Lewis, G.F.
Insulin acutely inhibits intestinal lipoprotein secretion in humans in
part by suppressing plasma free fatty acids. Diabetes, 2010, 59(3),
580-587.
Longuet, C.; Sinclair, E.M.; Maida, A.; Baggio, L.L.; Maziarz, M.;
Charron, M.J.; Drucker, D. J. The glucagon receptor is required for
the adaptive metabolic response to fasting. Cell Metab., 2008, 8(5),
359-371.
Xiao, C.; Pavlic, M.; Szeto, L.; Patterson, B.W.; Lewis, G.F.
Effects of acute hyperglucagonemia on hepatic and intestinal
lipoprotein production and clearance in healthy humans. Diabetes,
2011, 60(2), 383-390.
Adiels, M.; Boren, J.; Caslake, M.J.; Stewart, P.; Soro, A.;
Westerbacka, J.; Wennberg, B.; Olofsson, S.O.; Packard, C.;
Taskinen, M.R. Overproduction of VLDL1 driven by
hyperglycemia is a dominant feature of diabetic dyslipidemia.
Arterioscler. Thromb. Vasc. Biol., 2005, 25(8), 1697-1703.
Prigeon, R.L.; Quddusi, S.; Paty, B.; D'Alessio, D.A. Suppression
of glucose production by GLP-1 independent of islet hormones: a
novel extrapancreatic effect. Am. J. Physiol. Endocrinol. Metab.,
2003, 285(4), E701-E707.
Received: January 23, 2012
[46]
[47]
[48]
[49]
[50]
[51]
[52]
[53]
[54]
[55]
[56]
[57]
[58]
[59]
Revised: June 01, 2012
Xiao et al.
Balas, B.; Baig, M.R.; Watson, C.; Dunning, B.E.; LiguerosSaylan, M.; Wang, Y.; He, Y. L.; Darland, C.; Holst, J.J.; Deacon,
C.F.; Cusi, K.; Mari, A.; Foley, J.E.; DeFronzo, R. A. The
dipeptidyl peptidase IV inhibitor vildagliptin suppresses
endogenous glucose production and enhances islet function after
single-dose administration in type 2 diabetic patients. J. Clin.
Endocrinol. Metab., 2007, 92(4), 1249-1255.
Azuma, K.; Radikova, Z.; Mancino, J.; Toledo, F.G.; Thomas, E.;
Kangani, C.; Dalla, M.C.; Cobelli, C.; Holst, J.J.; Deacon, C.F.; He,
Y.; Ligueros-Saylan, M.; Serra, D.; Foley, J.E.; Kelley, D.E.
Measurements of islet function and glucose metabolism with the
dipeptidyl peptidase 4 inhibitor vildagliptin in patients with type 2
diabetes. J. Clin. Endocrinol. Metab., 2008, 93(2), 459-464.
Duez, H.; Smith, A.C.; Xiao, C.; Giacca, A.; Szeto, L.; Drucker,
D.J.; Lewis, G.F. Acute dipeptidyl peptidase-4 inhibition rapidly
enhances insulin-mediated suppression of endogenous glucose
production in mice. Endocrinology, 2009, 150(1), 56-62.
Tushuizen, M.E.; Diamant, M.; Heine, R.J. Postprandial
dysmetabolism and cardiovascular disease in type 2 diabetes.
Postgrad. Med. J., 2005, 81(951), 1-6.
Xiao, C.; Bandsma, R.H.; Dash, S.; Szeto, L.; Lewis, G.F.
Exenatide, a Glucagon-like Peptide-1 Receptor Agonist, Acutely
Inhibits Intestinal Lipoprotein Production in Healthy Humans.
Arterioscler. Thromb. Vasc. Biol., 2012, 32(6), 1513-1519.
Parlevliet, E.T.; Schroder-van der Elst, J.P.; Corssmit, E.P.; Picha,
K.; O'Neil, K.; Stojanovic-Susulic, V.; Ort, T.; Havekes, L.M.;
Romijn, J.A.; Pijl, H. CNTO736, a novel glucagon-like peptide-1
receptor agonist, ameliorates insulin resistance and inhibits very
low-density lipoprotein production in high-fat-fed mice. J.
Pharmacol. Exp. Ther., 2009, 328(1), 240-248.
Bjorkegren, J.; Packard, C.J.; Hamsten, A.; Bedford, D.; Caslake,
M.; Foster, L.; Shepherd, J.; Stewart, P.; Karpe, F. Accumulation of
large very low density lipoprotein in plasma during intravenous
infusion of a chylomicron-like triglyceride emulsion reflects
competition for a common lipolytic pathway. J. Lipid. Res., 1996,
37(1), 76-86.
Qin, X.; Shen, H.; Liu, M.; Yang, Q.; Zheng, S.; Sabo, M.;
D'Alessio, D.A.; Tso, P. GLP-1 reduces intestinal lymph flow,
triglyceride absorption, and apolipoprotein production in rats. Am.
J. Physiol. Gastrointest. Liver Physiol., 2005, 288(5), G943-G949.
Farr, S.; Adeli, K. Incretin-based therapies for treatment of
postprandial dyslipidemia in insulin-resistant states. Curr. Opin.
Lipidol., 2012, 23(1), 56-61.
Hsieh, J.; Longuet, C.; Baker, C.L.; Qin, B.; Federico, L.M.;
Drucker, D.J.; Adeli, K. The glucagon-like peptide 1 receptor is
essential for postprandial lipoprotein synthesis and secretion in
hamsters and mice. Diabetologia, 2010, 53(3), 552-561.
Fadini, G.P.; Avogaro, A. Cardiovascular effects of DPP-4
inhibition: beyond GLP-1. Vascul. Pharmacol., 2011, 55(1-3), 1016.
Wasada, T.; McCorkle, K.; Harris, V.; Kawai, K.; Howard, B.;
Unger, R.H. Effect of gastric inhibitory polypeptide on plasma
levels of chylomicron triglycerides in dogs. J. Clin. Invest., 1981,
68(4), 1106-1107.
Nathan, D.M.; Buse, J.B.; Davidson, M.B.; Ferrannini, E.; Holman,
R.R.; Sherwin, R.; Zinman, B. Medical management of
hyperglycemia in type 2 diabetes: a consensus algorithm for the
initiation and adjustment of therapy: a consensus statement of the
American Diabetes Association and the European Association for
the Study of Diabetes. Diabetes Care, 2009, 32(1), 193-203.
Cernea, S.; Raz, I. Therapy in the early stage: incretins. Diabetes
Care, 2011, 34(Suppl. 2), S264-S271.
Accepted: June 01, 2012
You might also like
- David Dent - Insect Pest ManagementDocument425 pagesDavid Dent - Insect Pest Managementsaphito83% (6)
- Effect of Sunlight Against Green Beans GrowthDocument4 pagesEffect of Sunlight Against Green Beans GrowthDanny Ramadhan0% (1)
- History of Psychiatry: Hypnosis, False Memory and Multiple Personality: A Trinity of AffinityDocument10 pagesHistory of Psychiatry: Hypnosis, False Memory and Multiple Personality: A Trinity of AffinityJulianna BartalusNo ratings yet
- Amess Management of Hyper Triglyceride Mia in The Diabetic PatientDocument5 pagesAmess Management of Hyper Triglyceride Mia in The Diabetic Patientjimrod1950No ratings yet
- Effect of Diuretics On The Plasma Lipid Profile: KEY WORDS: Hypertension, Diuretics, Dyslipidaemia, Glucose IntoleranceDocument7 pagesEffect of Diuretics On The Plasma Lipid Profile: KEY WORDS: Hypertension, Diuretics, Dyslipidaemia, Glucose IntoleranceVims BatchNo ratings yet
- NursingresearchreportDocument12 pagesNursingresearchreportapi-300699057No ratings yet
- Markel 2011 - The Resurgence of Niacin, From Nicotinic Acid To NiaspanlaropiprantDocument7 pagesMarkel 2011 - The Resurgence of Niacin, From Nicotinic Acid To NiaspanlaropiprantAlbert CalvetNo ratings yet
- Diretrizes de Prática Clínica para o Tratamento de HipertrigliceriemiaDocument24 pagesDiretrizes de Prática Clínica para o Tratamento de Hipertrigliceriemiaqmatheusq wsantoswNo ratings yet
- Cardiovascular Outcomes StudiesDocument5 pagesCardiovascular Outcomes StudiesAnonymous LOeICSOqNo ratings yet
- Optimizing The Care of Patients With Type 2 Diabetes Using Incretin-Based Therapy: Focus On GLP-1 Receptor AgonistsDocument12 pagesOptimizing The Care of Patients With Type 2 Diabetes Using Incretin-Based Therapy: Focus On GLP-1 Receptor AgonistsRidha Surya NugrahaNo ratings yet
- Lipid Effects of Antihypertensive Medications: Nonstatin Drugs (W Borden, Section Editor)Document8 pagesLipid Effects of Antihypertensive Medications: Nonstatin Drugs (W Borden, Section Editor)Septi Fadhilah SPNo ratings yet
- Secondary Causes of DyslipidemiaDocument3 pagesSecondary Causes of DyslipidemiaAnghelo Aldair Velásquez CarrilloNo ratings yet
- Heart Views-Cardio GlicemicDocument24 pagesHeart Views-Cardio Glicemicsupit1No ratings yet
- Metabolic Syndrome in SaudisDocument4 pagesMetabolic Syndrome in SaudisOnon EssayedNo ratings yet
- Srikant H 2016Document10 pagesSrikant H 2016aditya sekarNo ratings yet
- Lipids Module 4Document16 pagesLipids Module 4angelmedurNo ratings yet
- ECBCM - Oral Meds T2 Diab - Clinician06282011Document8 pagesECBCM - Oral Meds T2 Diab - Clinician06282011Ridha Surya NugrahaNo ratings yet
- ProjectDocument32 pagesProjectNithya sudevNo ratings yet
- Precision MedicineDocument23 pagesPrecision MedicineLavina Jain100% (1)
- Linagliptin - Farmacology, Efficacy and SafetyDocument7 pagesLinagliptin - Farmacology, Efficacy and SafetyMisaelDominguezNo ratings yet
- Reduction of Plasma Triglycerides by Diet in Subjects With Chronic Renal FailureDocument15 pagesReduction of Plasma Triglycerides by Diet in Subjects With Chronic Renal FailureDina sayunaNo ratings yet
- Type 2 Diabetes Mellitus (Article) Author United States Department of Veterans AffairsDocument9 pagesType 2 Diabetes Mellitus (Article) Author United States Department of Veterans Affairsnaljnaby9No ratings yet
- Hyperlipidemia in Diabetes-Etiology, Consequences and Treatment Edward Shahady MD Athero-Thrombosis in DiabetesDocument6 pagesHyperlipidemia in Diabetes-Etiology, Consequences and Treatment Edward Shahady MD Athero-Thrombosis in DiabetesindahkurNo ratings yet
- Endocrinology and Metabolism Clinics-Lípidos 2009Document234 pagesEndocrinology and Metabolism Clinics-Lípidos 2009Tony Miguel Saba SabaNo ratings yet
- Meds DiabetesDocument5 pagesMeds DiabetesAnjangsari 'aRie' WijayantiNo ratings yet
- New Practice in Semaglutide On Type-2 Diabetes and Obesity: Clinical Evidence and ExpectationDocument8 pagesNew Practice in Semaglutide On Type-2 Diabetes and Obesity: Clinical Evidence and ExpectationBoris ThielmannNo ratings yet
- Green Cardamom... Improves Serum Irisin, Glucose Indices, and Lipid Profiles In... Non-Alcoholic Fatty Liver Disease Patients ...Document11 pagesGreen Cardamom... Improves Serum Irisin, Glucose Indices, and Lipid Profiles In... Non-Alcoholic Fatty Liver Disease Patients ...Daniel GilbertoNo ratings yet
- F 2433 CMED Saxagliptin A Selective DPP 4 Inhibitor For The Treatment of Type 2 D.PDF 3311Document12 pagesF 2433 CMED Saxagliptin A Selective DPP 4 Inhibitor For The Treatment of Type 2 D.PDF 3311Ridha Surya NugrahaNo ratings yet
- Beyond Blood Lipids: Phytosterols, Statins and Omega-3 Polyunsaturated Fatty Acid Therapy For HyperlipidemiaDocument13 pagesBeyond Blood Lipids: Phytosterols, Statins and Omega-3 Polyunsaturated Fatty Acid Therapy For HyperlipidemiaChristian Ivan SantosoNo ratings yet
- Management of Persistent Hyperglycemia in Type 2 Diabetes MellitusDocument47 pagesManagement of Persistent Hyperglycemia in Type 2 Diabetes MellitusEslam khedrNo ratings yet
- Bays 2013Document26 pagesBays 2013Lukman Al-HafizNo ratings yet
- Incretin Mimetics and Dipeptidyl Peptidase-4 Inhibitors: Innovative Treatment Therapies For Type 2 DiabetesDocument11 pagesIncretin Mimetics and Dipeptidyl Peptidase-4 Inhibitors: Innovative Treatment Therapies For Type 2 DiabetesRidha Surya NugrahaNo ratings yet
- Practical Laboratory Medicine: Subashini C. Thambiah, Leslie Charles LaiDocument21 pagesPractical Laboratory Medicine: Subashini C. Thambiah, Leslie Charles LaiAdina BrahaNo ratings yet
- Pathology of Type 2 DiabetesDocument4 pagesPathology of Type 2 DiabetesJenny SembranoNo ratings yet
- Profil Lipid Dan Risiko Penyakit Jantung Koroner Andi Surya AmalDocument5 pagesProfil Lipid Dan Risiko Penyakit Jantung Koroner Andi Surya AmalAyu Dewi SuwantiNo ratings yet
- DM2 and Thiazide 2018Document13 pagesDM2 and Thiazide 2018Osvaldo VillarNo ratings yet
- Cardio Metabolic Risk in DiabetesDocument29 pagesCardio Metabolic Risk in DiabetesoctoindradjajaNo ratings yet
- 2017 Article 551Document11 pages2017 Article 551Albert EdoNo ratings yet
- AntioksidanDocument5 pagesAntioksidanRakasiwi GalihNo ratings yet
- 2 - Metabolic Syndrome-Role of Dietary Fat TypeDocument7 pages2 - Metabolic Syndrome-Role of Dietary Fat TypeJuliana ZapataNo ratings yet
- Does Metformin Raise Cholesterol Levels - DIQDocument2 pagesDoes Metformin Raise Cholesterol Levels - DIQFathima Sheik KatherNo ratings yet
- Clinical Efficacy Evaluation and Longterm Prognosis of Glucagonlike Peptide1 Combined With Sodium Glucose CotransportersDocument9 pagesClinical Efficacy Evaluation and Longterm Prognosis of Glucagonlike Peptide1 Combined With Sodium Glucose CotransportersvinayNo ratings yet
- Original Article: Journal of Clinical Lipidology (2017)Document12 pagesOriginal Article: Journal of Clinical Lipidology (2017)IrhamNo ratings yet
- Glucagon-Like Peptide 1 수용체작용제 최신지견Document8 pagesGlucagon-Like Peptide 1 수용체작용제 최신지견이호원No ratings yet
- Retrospective Case Series of Patients With Diabetes or Prediabetes Who Were Switched From Omega-3-Acid Ethyl Esters To Icosapent EthylDocument11 pagesRetrospective Case Series of Patients With Diabetes or Prediabetes Who Were Switched From Omega-3-Acid Ethyl Esters To Icosapent EthylAgus SetiawanNo ratings yet
- j3 2010Document12 pagesj3 2010Taha FransNo ratings yet
- Diabetes and Hyperlipidemia: A Direct Quantitative AnalysisDocument6 pagesDiabetes and Hyperlipidemia: A Direct Quantitative AnalysisAnonymous Kc2tM8No ratings yet
- Clinical Trial 2016Document11 pagesClinical Trial 2016guugle gogleNo ratings yet
- "Dyslipidemia" : Dyslipidemia Caused by Excessive Hepatic Secretion of VLDLDocument5 pages"Dyslipidemia" : Dyslipidemia Caused by Excessive Hepatic Secretion of VLDLDANEKKA TANNo ratings yet
- Lipid Lowering DrugsDocument38 pagesLipid Lowering Drugsmannona200357No ratings yet
- Bipin 2023Document10 pagesBipin 2023Rajendra KulyeNo ratings yet
- Terapia Farmacologica Dell'obesità, 2008Document11 pagesTerapia Farmacologica Dell'obesità, 2008todino32No ratings yet
- Review Inhibition of Dipeptidyl Peptidase-4 With Vildagliptin A Potential New Treatment For Type 2 DiabetesDocument8 pagesReview Inhibition of Dipeptidyl Peptidase-4 With Vildagliptin A Potential New Treatment For Type 2 DiabetesAndhi FahrurrojiNo ratings yet
- Type 2 Diabetes TugaskuDocument4 pagesType 2 Diabetes Tugaskuzuhaela iqbalNo ratings yet
- Alessandro MantovaniDocument16 pagesAlessandro Mantovaniasma.almhsnNo ratings yet
- Hba1C: Predictor of Dyslipidemia and Atherogenicity in Diabetes MellitusDocument3 pagesHba1C: Predictor of Dyslipidemia and Atherogenicity in Diabetes MellitusPramod Reddy EvuriNo ratings yet
- SGLT2 Inhibitors in The Management of Type 2 Diabetes With CardiovascularRenal ComorbiditiDocument5 pagesSGLT2 Inhibitors in The Management of Type 2 Diabetes With CardiovascularRenal ComorbiditiShekharNo ratings yet
- Biomedicines 10 01090 v2Document18 pagesBiomedicines 10 01090 v2irvinpereda1997No ratings yet
- Effect of Insulin Analog Initiation Therapy On LDL/ HDL Subfraction Profile and HDL Associated Enzymes in Type 2 Diabetic PatientsDocument11 pagesEffect of Insulin Analog Initiation Therapy On LDL/ HDL Subfraction Profile and HDL Associated Enzymes in Type 2 Diabetic PatientsAan AlawiyahNo ratings yet
- Dyson LineDocument10 pagesDyson LineSharly DwijayantiNo ratings yet
- Objective:: BackgroundDocument23 pagesObjective:: BackgroundJanine DimaangayNo ratings yet
- Diabetic 2 Update Review !!Document82 pagesDiabetic 2 Update Review !!Patrick CommettantNo ratings yet
- Semaglutide Unveiled: A Comprehensive Guide to Diabetes Treatment and Weight ManagementFrom EverandSemaglutide Unveiled: A Comprehensive Guide to Diabetes Treatment and Weight ManagementNo ratings yet
- Vulovic - Brskovo Final PDFDocument27 pagesVulovic - Brskovo Final PDFDraganaNo ratings yet
- Kanker Ovarium 2 PDFDocument6 pagesKanker Ovarium 2 PDFhestiNo ratings yet
- Poster Session v3Document11 pagesPoster Session v3Fiola FinandakasihNo ratings yet
- WJ Ms Paper Sir 2013Document13 pagesWJ Ms Paper Sir 2013Rohan sharmaNo ratings yet
- High Risk NeonateDocument102 pagesHigh Risk NeonateJaya Prabha100% (2)
- Virology SummaryDocument3 pagesVirology SummaryvictorNo ratings yet
- (Download PDF) Essentials of Anatomy Physiology Kenneth Saladin Online Ebook All Chapter PDFDocument42 pages(Download PDF) Essentials of Anatomy Physiology Kenneth Saladin Online Ebook All Chapter PDFnigel.allen933100% (12)
- A Whole-Of-Nation Approach To COVID-19 Taiwan's National Epidemic Prevention TeamDocument16 pagesA Whole-Of-Nation Approach To COVID-19 Taiwan's National Epidemic Prevention Teamgrahma96No ratings yet
- Pneumonia: Nur Yahya, T 05/1087561/KU/11522Document19 pagesPneumonia: Nur Yahya, T 05/1087561/KU/11522Taufik Nur YahyaNo ratings yet
- Jurnal 5Document14 pagesJurnal 5mellvinNo ratings yet
- Selected Instructional Course LecturesDocument27 pagesSelected Instructional Course LecturesAhmad ShakirNo ratings yet
- Holoxan Pi PDFDocument18 pagesHoloxan Pi PDFKarol IonasNo ratings yet
- Human Biology 13Th Edition Mader Test Bank Full Chapter PDFDocument56 pagesHuman Biology 13Th Edition Mader Test Bank Full Chapter PDFninhdermotc1u100% (11)
- Icd 11 Introduction ClinicalDocument50 pagesIcd 11 Introduction ClinicalMuhammad Farooq Ul HassanNo ratings yet
- DocxDocument53 pagesDocxkuro hanabusaNo ratings yet
- COVID - 19 Treatment and VaccinesDocument2 pagesCOVID - 19 Treatment and Vaccinesridwan_olawale_1No ratings yet
- Prepared By: M.Bilal BSN, Rn. MSN UhsDocument52 pagesPrepared By: M.Bilal BSN, Rn. MSN UhsXweet AdilNo ratings yet
- Uterine Anomaly, Fibroid Uterus, Ovarian Tumor, Uterine ProlapseDocument30 pagesUterine Anomaly, Fibroid Uterus, Ovarian Tumor, Uterine ProlapseVijith.V.kumar80% (5)
- Assignment and Portfolio Consent FormDocument16 pagesAssignment and Portfolio Consent Formapi-549168092No ratings yet
- What Is A Root CanalDocument5 pagesWhat Is A Root CanaljjahaddinNo ratings yet
- Discharge PlaningDocument28 pagesDischarge PlaningRuhiyat Spd100% (1)
- Definitions and Reporting Framework For Tuberculosis - 2013 RevisionDocument47 pagesDefinitions and Reporting Framework For Tuberculosis - 2013 RevisionDavien UtoyoNo ratings yet
- LRM 1 Iv Bone Loss and Patterns of Bone DestructionDocument36 pagesLRM 1 Iv Bone Loss and Patterns of Bone DestructionSamridhi SrivastavaNo ratings yet
- Richa Shrivastava Assistant Professor Department of Pharmacy BITS PilaniDocument18 pagesRicha Shrivastava Assistant Professor Department of Pharmacy BITS PilaniCHOUGULE KISHOR SURYAKANTNo ratings yet
- Pulmonary Patho PDFDocument623 pagesPulmonary Patho PDFNabeel Shahzad100% (1)
- CHN - 1Document23 pagesCHN - 1Raagul SaravanaKumarNo ratings yet