Professional Documents
Culture Documents
Ajr 10 4865
Ajr 10 4865
Uploaded by
ChavdarOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Ajr 10 4865
Ajr 10 4865
Uploaded by
ChavdarCopyright:
Available Formats
Wo m e n s I m a g i n g O r i g i n a l R e s e a r c h
Sever et al.
Ultrasound Using Microbubbles in Breast Cancer
FOCUS ON:
Downloaded from www.ajronline.org by 78.83.20.38 on 03/10/13 from IP address 78.83.20.38. Copyright ARRS. For personal use only; all rights reserved
Womens Imaging
Original Research
W O M E N S
IMAGING
Ali R. Sever 1
Philippa Mills1
Susan E. Jones 2
Karina Cox 2
Jennifer Weeks1
David Fish 3
Peter A. Jones 2
Sever AR, Mills P, Jones SE, et al.
Preoperative Sentinel Node
Identification With Ultrasound
Using Microbubbles in Patients
With Breast Cancer
OBJECTIVE. Sentinel lymph node (SLN) biopsy is the standard procedure for axillary
staging in early breast cancer. Lymphatic imaging after peritumoral microbubble injection
has been described in animal models. The aim of this study was to identify and localize SLNs
preoperatively by contrast-enhanced sonography after intradermal injection of microbubbles
in patients with breast cancer.
SUBJECTS AND METHODS. Eighty consecutive consenting patients with primary breast cancer were recruited. Patients received a periareolar intradermal injection of microbubble contrast agent. Breast lymphatics were visualized by sonography and followed to
the axilla to identify SLNs. A guidewire was deployed to localize the SLN. The next day, patients underwent standard tumor excision and SLN biopsy.
RESULTS. In 71 (89%) of the 80 patients, SLNs were identified and guidewires were
inserted. In these patients, operative findings using conventional radioisotope and blue dye
techniques confirmed that the wired nodes were SLNs. Fourteen patients were found to have
metastases in SLNs. In these patients, the SLNs were identified correctly and were localized
with guidewires before surgery.
CONCLUSION. SLNs may be identified and localized before surgery using contrastenhanced sonography after injection of microbubbles.
Keywords: breast cancer, contrast-enhanced
ultrasound, microbubble contrast agents, preoperative
staging, sentinel lymph node
DOI:10.2214/AJR.10.4865
Received April 23, 2010; accepted after revision
July 6, 2010.
1
Department of Radiology, Maidstone Hospital,
Hermitage Ln., ME16 9QQ, Maidstone, United Kingdom.
Address correspondence to A. R. Sever (ali.sever@nhs.net).
2
Department of Surgery, Maidstone Hospital, Maidstone,
United Kingdom.
3
Department of Pathology, Maidstone Hospital,
Maidstone, United Kingdom.
AJR 2011; 196:251256
0361803X/11/1962251
American Roentgen Ray Society
AJR:196, February 2011
xillary nodal status remains the
most important prognostic factor
in patients with breast cancer
and determines adjuvant treatment [1, 2]. Axillary staging was traditionally performed by means of axillary lymph
node (ALN) dissection. With the widespread
use of high-quality screening by mammography and increased awareness, more patients with breast cancer now present with
smaller tumors than previously. The incidence of axillary nodal involvement is, therefore, also lower than before; almost two
thirds of patients present with an axilla that
is free from disease [3, 4]. Exposing all patients with breast cancer to the complications
associated with axillary dissection is no longer justified [5, 6].
Sentinel lymph node (SLN) biopsy is now
widely adopted as a less-invasive approach
in the staging of the axilla. The concept was
pioneered by Donald Morton and colleagues
[7] at the John Wayne Cancer Center in Santa Monica, California. Blue dye was used to
identify the SLNs in patients with malignant
melanoma. Giuliano et al. [8] used this tech-
nique in early-stage breast cancer with blue
dye only. Krag et al. [9] used a radiopharmaceutical (radiolabeled colloid) to identify the
SLN and introduced probe-guided surgery in
the operating room. Albertini et al. [10] were
the first group to use a combination of blue
dye and probe-guided surgery, showing that
these two techniques complement each other. When the combined tracing techniques
are used, the identification rate of SLN has
been shown to be over 95%, with false-negative rates between 5% and 10% after subsequent ALN dissection [11].
U.S. guidelines currently advise complete
ALN dissection for all cases in which SLN
biopsy reveals macro- and micrometastases
(> 0.2 mm) [11]. A meta-analysis by Kim et
al. [12] reported that more than half (53%) of
all patients with positive SLN biopsy results
were shown to have other ALN (non-SLN)
involvement after a complete ALN dissection. This represents an important drawback
of the SLN biopsy procedure because these
patients require a second procedure.
Clinical examination of the axilla is, unfortunately, unreliable for correct preoperative
251
Downloaded from www.ajronline.org by 78.83.20.38 on 03/10/13 from IP address 78.83.20.38. Copyright ARRS. For personal use only; all rights reserved
Sever et al.
staging of the lymph nodes [13]. The role of
ultrasound in the axilla has been investigated
in the preoperative assessment of lymph node
status. The sonographic assessment of axillary
nodes in combination with biopsy has been reported to increase the preoperative identification of axillary node metastasis by allowing
a reduction in the number of futile SLN biopsies, with considerable savings of time and
financial resources [14]. On the other hand, a
review study by Alvarez et al. [15] concluded
that axillary ultrasound should not be used in
isolation as a method for deciding whether to
perform ALN dissection.
The use of contrast-enhanced ultrasound
is well established in clinical practice; IV administration of contrast agent has provided enhanced imaging of the tissue microvasculature
in many organ systems. Ultrasound contrast
agents are composed of a dispersion of microbubbles (each of which is smaller than an
RBC) that act as reflectors of the ultrasound
beam [16]. In 2004, Goldberg et al. [17] published their results in a swine melanoma model, where microbubbles injected peritumorally were seen to enter lymphatic channels and
SLNs were identified accurately by sonography in 90% of cases. Recently published preliminary results of our group have shown that
this technique can be used in patients with
breast cancer to identify the SLN [18].
The aim of this article is to discuss from
a radiologic perspective the potential for intradermally injected microbubbles to enter
breast lymphatic channels and consequently permit the preoperative identification of
SLNs in patients with early breast cancer.
Subjects and Methods
This prospective study was approved by the local ethics committee and the Medicines and Healthcare Products Regulatory Agency. All patients gave
written informed consent. Between May 2008 and
November 2009, 80 consecutive consenting patients with breast cancer who were scheduled for
primary surgical treatment were recruited into the
study. Exclusion criteria included histologically or
cytologically proven ALN involvement diagnosed
clinically or using standard sonographic imaging,
receipt of neoadjuvant chemotherapy, and the presence of severe medical comorbidities.
There were 78 women and two men with a median age of 61 years (range, 3282 years). Of these,
51 patients had screen-detected cancers identified
by the Maidstone and Tunbridge Wells National
Health Service Trust Breast Screening Service and
29 were symptomatic referrals to the breast clinic. In the symptomatic group, three patients were
252
diagnosed with a nonpalpable lesion in the contra
lateral breast while having yearly follow-up mammograms because of their history of breast cancer,
and one woman was referred from a family history
clinic after an abnormal mammogram.
All sonographic examinations were performed
using four different ultrasound systems equipped
with small-parts high-frequency linear probes and
dedicated contrast software packages. The majority of the studies (n = 69) were performed with an
Acuson Sequoia 512 scanner (Siemens Healthcare).
This scanner provides conventional gray-scale imaging, pulse-inversion harmonic gray-scale, and
contrast-specific sonographic imaging with live
dual images of tissue only and contrast agent image (Cadence contrast pulse sequencing, Siemens
Healthcare). A high-frequency 14-MHz linear-array probe (15L8w) was used. To reduce microbubble destruction, low mechanical index values were
applied (0.20.4). The remaining 11 studies were
performed with three different ultrasound systemsfour with GE Logiq E9 using a 13-4-MHz
probe (ML615-D), three with a Philips Healthcare IU22 using an L125 probe, and four with an
Esaote MyLab70 XVG using a 13-4-MHz linear
probe (LA 523). All the machines produced very
satisfactory images with identical visualization.
The Acuson Sequoia scanner was on long-term
loan, and the other scanners were used in demonstration visits to determine personal preference.
The afternoon before surgery, patients attended
the nuclear medicine department for subdermal
injection of radioactive isotope (Nanocoll, GE
Healthcare; 40 mBq in the periareolar upper outer
quadrant region). Afterward, patients underwent
injection of ultrasound contrast agent (SonoVue,
Bracco Imaging) by an experienced breast radiologist. First, 1 mL of 1% lidocaine was injected
into the subcutaneous layer of the areola. The microbubble contrast agent was reconstituted using
2 mL of sterile saline, and the ampoule was shaken
vigorously to ensure a homogeneous suspension of
bubbles. The ultrasound contrast agent is composed of phospholipid stabilized microbubbles
containing sulfur hexafluoride gas with a mean
diameter of 2.5 m. Between 0.2 and 0.5 mL of
microbubble suspension was injected into and just
under the skin of the areola in the upper outer
quadrant using a 1-mL insulin syringe and a
26-gauge needle. Up to three consecutive injections
were given intradermally in a similar position to
the radioisotope.
Conventional gray-scale ultrasound examination of the axilla was performed before injection of
contrast agent, and the position of imaged lymph
nodes was noted. After injection of contrast agent,
lymphatic channels were visualized immediately
on contrast pulse sequencing and were followed
into the axilla. Areas of contrast agent accumulation were then imaged with gray-scale or live dual
images to confirm the presence of an architecturally defined lymph node. When no obvious lymphatic
filling was obtained, the areola area was massaged
for 1030 seconds and imaging was repeated. If no
lymph nodes could be identified after three consecutive injections, the study was abandoned and
accepted as a failure. In the patients whose nodes
were successfully identified, the transit time from
injection to arrival in the axillary node was 1545
seconds. Contrast agent remained in the putative
SLN for 13 minutes. Repeating the areola massage often produced a refill of the SLN without the
need for a second injection.
The technique was initially piloted in five
women with breast cancer to check the safety profile of intradermal microbubbles and to ensure
that the contrast agent successfully entered breast
lymphatics and accumulated in architecturally defined axillary nodes. Microbubbles have an excellent safety profile when used with conventional IV administration [1921]. On the basis of this
fact and the safety data from experimental models
when microbubbles were injected intradermally
[17], we received Medicines and Healthcare Products Regulatory Agency approval for this study.
We had concerns about possible local reactions at
injection sites, but these proved to be unfounded.
After this initial five-patient pilot study, in
all subsequent patients, contrast-enhanced ultrasound-identified SLNs were localized with a
19.5-gauge hookwire (X-Reidy, Cook). These are
the same guidewires we use for the localization
of nonpalpable screen-detected breast cancers.
The wires are 9 cm long and, after insertion, are
coiled and taped to the skin and covered by a sterile gauze and waterproof sterile dressing. There
were no dislodged wires or infections. Most procedures were completed in 15 minutes.
The next morning, patients underwent surgical
resection of the breast cancer. Immediately after
induction of anesthesia, patients received a 2-mL
subdermal injection of blue dye (Bleu Patente V
2.5%, Guerbet) in the periareolar upper outer
quadrant region. A standard axillary incision was
made, and a gamma probe (Navigator GPS, RMD
Instruments) was used to facilitate the identification of SLNs. All radioactive lymph nodes, all
blue dyecontaining lymph nodes, and all guidewire-containing lymph nodes within the axilla
were excised and sent for standard histologic analysis together with the breast cancer specimen.
Results
Clinical Data
There were 78 women and two men with a
median age of 61 years (range, 3282 years).
AJR:196, February 2011
Downloaded from www.ajronline.org by 78.83.20.38 on 03/10/13 from IP address 78.83.20.38. Copyright ARRS. For personal use only; all rights reserved
Ultrasound Using Microbubbles in Breast Cancer
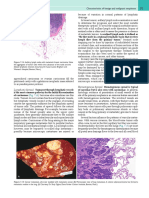
Fig. 169-year-old woman with grade II invasive
lobular carcinoma.
A, Contrast-enhanced ultrasound image obtained
by contrast-specific-mode Cadence contrast pulse
sequencing (Siemens Healthcare) reveals solitary
enhancing node in axilla (double arrow) with afferent
lymphatic vessel (single arrows).
B, Gray-scale sonogram of same axillary node (double
arrow), which was measured as 7 mm. Afferent
lymphatics cannot be recognized on gray-scale
images.
All but two patients had invasive disease on
the postoperative histologic examination.
The median diameter of the invasive breast
tumors was 14 mm (range, 470 mm); the
median diameter was 18 mm (range, 490
mm) when noninvasive tumor components
were also included. In 46% of cases, the primary breast lesion originated in the upper
outer quadrant of the breast, and in 26% of
cases, it originated in the upper inner quadrant. The final histologic analysis of invasive disease ranged from small tubular carcinomas to grade III ductal carcinomas. One
patient had ductal carcinoma in situ (DCIS)
with focal microinvasion and another had extensive DCIS. Lymphovascular invasion was
seen in 10 cases. Of the 80 patients, 65 (81%)
had estrogen receptor (ER)positive and
progesterone receptor (PR)positive tumors.
Five tumors (6%) were ER and PR negative.
One patient (1%) had ER-negative PR-positive tumors, and seven patients (9%) had ERpositive PR-negative tumors. ER and PR status was not tested in two DCIS cases (3%).
Human epidermal growth factor receptor
2 status was negative in 66 patients (83%),
positive in five patients (6%), and was not
available in nine (11%) cases. Two patients
(3%) had triple negative tumors. The median
number of SLNs retrieved per patient was 2
(range, 15 SLNs).
With regard to adverse events, no allergic
reactions to ultrasound contrast agent were
identified. One patient reported a burning
sensation at the contrast agent injection site,
and two patients developed superficial bruising around the site of injection. Many patients
found insertion of the axillary guidewire uncomfortable, but at the time of surgery there
were no axillary hematomas and no other
signs of axillary trauma. A patient satisfaction survey of 30 consecutive cases yielded 22
responses and showed a high level of satisfaction, with 81% reporting no or only slight pain
associated with the procedure and subsequent
wire placement. Only 23% of patients needed
to take any analgesia after the procedure.
Technical Success
In 71 (89%) of the 80 patients, contrast-enhanced ultrasound images revealed visualization of lymphatic vessels, starting from the
site of injection and traced to the level of axilla, where contrast agent pooling was identified (Fig. 1). Guidewire insertion was performed in all 71 cases. The visibility of the
axillary nodes was quite variable both before
and after the administration of contrast agent.
In 43 patients, the gray-scale images revealed
a clearly visible lymph node corresponding to
the area of contrast agent pooling. This node
was also seen on gray-scale imaging before
injection of contrast agent. In 25 patients, a
guidewire was deployed into an SLN that was
recognized only after microbubble enhancement as an area of contrast agent accumulation and only then seen as a typical lymph
node on gray-scale imaging. In three cases,
the guidewire was inserted into the areas of
pooled contrast agent without any recognizable lymph node architecture even on the subsequent gray-scale imaging.
The procedure was not successful in nine
patients. In one, a technical error occurred
with the ultrasound machine and the procedure was abandoned. In three patients, the
breast lymphatics were not visualized by contrast-enhanced ultrasound. In three patients,
Fig. 256-year-old woman with grade II invasive ductal carcinoma.
A, Contrast-enhanced ultrasound image shows two synchronously enhancing nodes in axilla (arrows).
B, Gray-scale sonogram of same axillary nodes (double arrows) measuring 9 and 8 mm respectively. Single guidewire (single arrows) was used preoperatively to localize
these nodes.
C, Photograph of surgical specimen. Two sentinel nodes (double arrows) were excised using conventional techniques with blue dye and radioactive isotope. Both nodes
were reported as hot and blue at time of surgery. Preoperatively inserted guidewire (single arrow) correctly identified same two nodes.
AJR:196, February 2011
253
Downloaded from www.ajronline.org by 78.83.20.38 on 03/10/13 from IP address 78.83.20.38. Copyright ARRS. For personal use only; all rights reserved
Sever et al.
the breast lymphatics were visualized but a
draining lymph node was not identified. In
two patients, despite the fact that SLNs were
not identified by contrast agent accumulation, guidewires were deployed into prominent ALNs visualized only with gray-scale
imaging. At the time of surgery, these wired
lymph nodes were not SLNs. This emphasizes the importance of contrast enhancement
in assisting successful SLN identification.
Although only one SLN was visualized
on contrast-enhanced ultrasound in most patients, two patients had concordant lymphatic drainage to two SLNs. A single guidewire
was deployed into both enhancing lymph
nodes (Fig. 2).
The ability of preoperative contrast-enhanced ultrasound to identify axillary SLNs
correctly was validated at surgery by comparison with the standard SLN localization
techniques. At surgery, wired SLNs were radioactive and blue in 63 patients. In eight cases, the nodes were radioactive only and none
contained blue dye only. At histologic examination, 48% (38/80) of the excised sentinel
nodes were 10 mm or smaller, as measured on
their long axis (range, 530 mm). Insertion of
an axillary guidewire was deemed successful
if the tip was found in or on the surface of a
lymph node. The overall sensitivity of SLN
detection by microbubble contrast-enhanced
ultrasound was 89% (71/80 patients).
Histopathologic Results
Fourteen patients had metastatic deposits
in the excised SLNs. These results are summarized in Table 1. SLNs had been identified correctly and guidewires were inserted
into the node before surgery in all patients
with lymph node metastases. In three patients, only one SLN was retrieved that also
contained the guidewire. In one patient, two
axillary SLNs were identified and excised,
but the wired node contained isolated tumor
cells only. The second retrieved lymph node
contained a micrometastasis. In nine cases,
there were no further involved nodes found
in the axillary specimen on the completion
of ALN dissection, and, in six of these cases,
the wired node was the only abnormal node.
Of the nine patients in whom contrast-enhanced ultrasound did not identify the draining SLNs, two had small (< 20 mm) grade
II invasive lobular carcinomas on final histologic examination. One patient had a 34-mm
grade II invasive lobular carcinoma. The remaining six patients had small invasive ductal carcinomas (< 20 mm); three were grade
254
TABLE 1: Histopathologic Results of the Sentinel Lymph NodePositive Cases
Sentinel Lymph Node Axillary Lymph Node
No. of Status, No. of Positive
Dissection, No. of
Size of Wired
Cases
Nodes/Total
Nodes Dissected/Total Node (mm)
Comments
1/1
0/21
10
Micrometastasis in wired node
1/1
0/18
12
Macrometastasis in wired node
1/1
6/25
Macrometastasis in wired node
1/2
0/23
Macrometastasis only in wired
node
1/2
1/25
10
Micrometastasis only in wired
node
1/3
0/13
15
Micrometastasis only in wired
node
1/3
0/30
10
Macrometastasis only in wired
node
1/4
0/14
Macrometastasis only in wired
node
2/2
0/17
11
Micrometastasis in both sentinel
lymph nodes
10
2/2
1/22
14
Isolated tumor cells in wired
node and micrometastasis in
other node
11
2/2
3/23
20
Macrometastasis in wired node
12
2/2
13/19
10
Macrometastasis in both sentinel
lymph nodes
13
2/4
0/26
10
Macrometastasis in wired node
14
3/5
0/15
15
Macrometastasis in all sentinel
lymph nodes
I, one was grade II, and two were grade III. A
total of 26 SLNs were removed in these nine
patients and all were negative on final histologic examination; hence, no ALN dissections were performed in this group.
Discussion
SLN biopsy has become widely accepted as the standard procedure for lymphatic
staging in patients with early breast cancer.
Patients whose SLN biopsy results are negative do not require further axillary treatment. However, if metastases are detected in
the SLN, then a complete ALN dissection is
advised. Preoperative confirmation of lymph
node metastases has the potential to prevent
such patients undergoing a second surgery
because ALN dissection can be performed
at the same time as the initial breast surgery.
In 2004, Goldberg et al. [17] published
their experimental results in Radiology indicating that, in a swine melanoma model, microbubbles have the potential to identify the
sentinel nodes. Despite the relatively small
number of patients, our clinical feasibility
study has shown that microbubble contrast
agent readily enters breast lymphatic chan-
nels, passing into the draining SLN, and can
be clearly visualized by contrast-enhanced
ultrasound. SLNs were identified successfully before surgery in 89% of patients.
In the majority of solid tumors, demonstration of regional or distant metastases provides prognostic information that supersedes
any primary tumor characteristics. Current
methods of lymph node staging in breast
cancer are heavily dependent on operative
excision. Before surgery, gray-scale axillary
sonography and fine-needle aspiration cytology (FNAC) of enlarged or morphologically
abnormal lymph nodes have some value in
diagnosing nodal metastases but may have a
sensitivity of only 3163% [15]. The results
of this study have shown that, in 28 (39%) of
71 successful cases, SLNs were recognized
as a lymph node only after microbubble enhancement. This finding is also supported
by the results of Mustonen et al. [22], who
found that, when any sonographically visible
lymph nodes are used as criteria for positivity on gray-scale imaging, the sensitivity of
ultrasound does not exceed 67%. This finding suggests that, even if a protocol to sample
every imaged axillary LN were to be adopt-
AJR:196, February 2011
Downloaded from www.ajronline.org by 78.83.20.38 on 03/10/13 from IP address 78.83.20.38. Copyright ARRS. For personal use only; all rights reserved
Ultrasound Using Microbubbles in Breast Cancer
ed, without microbubble contrastenhanced
ultrasound, a large proportion of SLNs would
remain invisible to gray-scale ultrasound imaging. Similar findings were also reported by
Britton et al. [23], who found that gray-scale
ultrasound-guided biopsy would miss the
sentinel node in 36% of cases. They concluded that there was a need for a better identification and sampling method for the SLN.
The current combined method of using
radioactive isotope and blue dye to identify
SLNs at the time of surgical excision has several limitations. Although lymphoscintigraphy can give an indication of the position of
the SLN, by the time a surgical incision is
made, the small size of the radioactive colloid means that the isotope may have passed
through the SLN and entered other regional lymph nodes [24]. No consensus exists
about the number of SLNs that should be removed on the basis of radioactivity counts,
because several reports suggest that the most
radioactive node is not always the metastatic
one [2527]. In addition, there are logistical
problems related to obtaining, handling, and
disposing of radioactive material.
Injection of blue dye may lead to staining of adjacent lymph nodes as well as the
SLN. In addition, there is a growing recognition that blue dye injection can lead to breast
tattooing, which can persist for more than 1
year [28]. There is also a small, but serious,
risk of anaphylaxis associated with blue dye
injection [29].
Ultrasound examination is an economically viable, safe, and well-accepted technique
for both patients and health care providers.
We have shown that contrast-enhanced ultrasound represents a practical method for
mapping the lymphatic drainage of a breast
tumor in real time. At present, as many as
35% of patients who undergo SLN excision
biopsy will require additional surgery because of nodal metastasis [30]. Unlike conventional ultrasound, the use of microbubble
contrastenhanced ultrasound could enable
targeted preoperative ultrasound-guided biopsy of SLNs in patients with breast cancer.
Individuals who are then found to have SLN
metastasis would avoid the prospect of a futile SLN excision biopsy and proceed directly to ALN dissection.
We have also shown that this technique is
feasible with different commercially available ultrasound systems. Most of the examinations (n = 69) were performed using
an Acuson Sequoia 512 scanner (Siemens
Healthcare). In the remaining 11 cases, we
AJR:196, February 2011
used other ultrasound scanners, including
GE Logiq E9, Philips Healthcare IU22, and
Esaote MyLab70 XVG. Similarly successful
results were obtained with these systems.
In the current study, the aim was simply to
identify the SLN with the help of microbubble
enhancement. We did not assess the enhancement pattern or comment on unenhanced areas (filling defects) within the node. In the
swine melanoma model, however, Goldberg
et al. [17] have successfully correlated the intranodal filling defects with tumor deposits.
This may be a valuable addition to this technique, particularly when a targeted biopsy is
planned. However, it needs to be considered
that ALNs are deep in location in human tissue and therefore may be more difficult to interpret than shown in the animal model.
The use of microbubbles may also be of value in patients with DCIS, for whom operative
SLN biopsy remains controversial. Some published articles do not justify SLN biopsy for patients with DCIS, whereas other contradicting
studies emphasize the importance of SLN biopsy for this group of patients because, in almost
20% of cases, an invasive component is found
at surgery [31, 32]. On the other hand, these patients may benefit from contrast-enhanced ultrasound before surgery where a small marker
can be deployed into the microbubble-enhanced
SLN, enabling postoperative SLN biopsy to be
performed if an invasive component is found in
the final histologic examination. There are currently such breast tissue markers in use after a
breast biopsy to help locate the site for future
reference, which potentially may also be used
for axillary nodes (e.g., UltraClip, Bard Biopsy
Systems; and UltraCor, SenoRx). Similar arguments could also be made for patients undergoing risk-reducing mastectomies, where preoperative identification and marking of SLNs
may be of value [33].
It has been reported that neoadjuvant
treatment lowers the SLN identification rate,
possibly because of fibrosis within the axilla,
and increases the false-negative rate because
of down-staging [34, 35]. For the same reasons, we have excluded this group of patients
from our study. On the other hand, among
patients for whom neoadjuvant chemotherapy is planned, pretreatment staging of the
axilla by identification and FNAC of SLNs
before the start of chemotherapy is another
potential application of contrast-enhanced
ultrasound. If the FNAC result is positive,
then the patient will be scheduled for ALN
dissection after completion of chemotherapy. However, a patient with a negative FNAC
result will be scheduled for a routine SLN
biopsy. Insertion of a marker into the microbubble-enhancing SLN before commencing neoadjuvant chemotherapy may help in
identifying the node afterward if the conventional techniques fail.
Dynamic visualization of lymphatics and
SLNs by contrast-enhanced ultrasound is
theoretically possible for other superficial
tumors, such as melanoma, vulval and penile
carcinoma, and head and neck malignancies.
FNAC of these nodes may give crucial information in preoperative nodal staging as well
as surgical planning.
There are a few shortcomings in our study
that deserve comment. Although this is a pioneering study and the results are interesting,
they reflect a single institutional experience
performed by two dedicated breast radiologists. Larger patient data and multicenter trials are required to optimize the technique
should this become an established practice.
We presume that a more extensive use of this
procedure will improve our current 11% failure rate. Another issue that merits further assessment is a comparison study using other
commercially available microbubble agents
to optimize the technique.
Although the identification of SLNs under
ultrasound in the axilla yields the possibility for performing an imaging-guided biopsy, we do acknowledge that FNAC of nodes
has a recognized false-negative rate resulting from sampling error [23]. The next stage
in this study is to remove the entire contrastidentified node intact to permit detailed histologic analysis to adequately diagnose cancer cells; only then may the conventional
SLN biopsy be totally obviated.
In conclusion, our results have shown that
SLNs can be correctly identified and localized using preoperative contrast-enhanced
ultrasound. Additional research is being
undertaken to determine whether this new
technique may be used to perform a targeted
needle biopsy of these nodes to stage the axilla more accurately preoperatively.
References
1. Fisher B, Bauer M, Wickerham D, Redmond CK,
Fisher ER. Relation of number of positive axillary
nodes to the prognosis of patients with primary
breast cancer: an NSABP update. Cancer 1983;
52:15511557
2. Carter CL, Allen C, Henderson DE. Relation of tumor size, lymph node status and survival in 24,740
breast cancer cases. Cancer 1989; 63:181187
3. Cady B, Stone MD, Schuler JG, Thakur R, Wan-
255
Downloaded from www.ajronline.org by 78.83.20.38 on 03/10/13 from IP address 78.83.20.38. Copyright ARRS. For personal use only; all rights reserved
Sever et al.
ner MA, Lavin PT. The new era in breast cancer:
invasion, size, and nodal involvement dramatically decreasing as a result of mammographic
screening. Arch Surg 1996; 131:301308
4. Silverstein MJ, Skinner KA, Lomis TJ. Predicting
axillary nodal positivity in 2282 patients with
breast carcinoma. World J Surg 2001; 25:767772
5. Schrenk P, Rieger R, Shamiyeh A, Wayand W.
Morbidity following sentinel lymph node biopsy
versus axillary lymph node dissection for patients
with breast carcinoma. Cancer 2000; 88:608614
6. Schijven MP, Vingerhoets AJ, Rutten HJ, et al.
Comparison of morbidity between axillary lymph
node dissection and sentinel node biopsy. Eur J
Surg Oncol 2003; 29:341350
7. Morton DL, Wen DR, Wong JH, et al. Technical details of intraoperative lymphatic mapping for early
stage melanoma. Arch Surg 1992; 127:392399
8. Giuliano AE, Kirgan DM, Guenther JM, et al. Lymphatic mapping and sentinel lymphadenectomy for
breast cancer. Ann Surg 1994; 220:391398
9. Krag DN, Weaver DL, Alex JC, Fairbank JT. Surgical resection and radiolocalization of the sentinel lymph node in breast cancer using a gamma
probe. Surg Oncol 1993; 2:335340
10. Albertini JJ, Lyman GH, Cox C, et al. Lymphatic
mapping and sentinel node biopsy in the patient
with breast cancer. JAMA 1996; 276:18181822
11. Lyman GH, Giuliano AE, Somerfield MR, et al.
American Society of Clinical Oncology guideline
recommendations for sentinel lymph node biopsy
in early-stage breast cancer. J Clin Oncol 2005;
23:77037720
12. Kim T, Giuliano AE, Lyman GH. Lymphatic
mapping and sentinel lymph node sampling in
early stage breast cancer: a meta-analysis. Cancer
2006; 106:416
13. De Freitas R, Costa MV, Schneider SV, et al. Accuracy of ultrasound and clinical examination in the
diagnosis of axillary lymph node metastases in
breast cancer. Eur J Surg Oncol 1991; 17:240244
14. Nori J, Vanzi E, Bazzocchi M, et al. Role of axillary ultrasound examination in the selection of
breast cancer patients for sentinel node biopsy. Am
J Surg 2007; 193:1620
15. Alvarez S, Aorbe E, Alcorta P, Lpez F, Alonso
I, Corts J. Role of sonography in the diagnosis of
axillary lymph node metastases in breast cancer:
a systematic review. AJR 2006; 186:13421348
16. Cosgrove D. Ultrasound contrast agents: an overview. Eur J Radiol 2006; 60:324330
17. Goldberg BB, Merton DA, Liu JB, et al. Sentinel
lymph nodes in a swine model with melanoma:
contrast-enhanced lymphatic US. Radiology
2004; 230:727734
18. Sever A, Jones S, Cox K, Weeks J, Mills P, Jones
P. Preoperative localization of sentinel lymph
nodes using intradermal microbubbles and contrast-enhanced ultrasonography in patients with
breast cancer. Br J Surg 2009; 96:12951299
19. Wilson SR, Greenbaum LD, Goldberg BB. Contrast-enhanced ultrasound: what is the evidence
and what are the obstacles? AJR 2009; 193:5560
20. Piscaglia F, Bolondi L. The safety of SonoVue in
abdominal applications: retrospective analysis of
23188 investigations. Ultrasound Med Biol 2006;
32:13691375
21. Correas JM, Bridal L, Lesavre A, et al. Ultrasound contrast agents: properties, principles of
action, tolerance, and artifacts. Eur Radiol 2001;
11:13161328
22. Mustonen P, Farin P, Kosunen O. Ultrasonographic detection of metastatic axillary lymph nodes in
breast cancer. Ann Chir Gynaecol 1990; 79:1518
23. Britton PD, Provenzano E, Barter S, et al. Ultrasound guided percutaneous axillary lymph node
core biopsy: how often is the sentinel lymph node
being biopsied? Breast 2009; 18:1316
24. Goldfarb LR, Alazraki NP, Eshima D, Eshima
LA, Herda SC, Halkar RK. Lymphoscintigraphic
identification of sentinel lymph nodes: clinical
evaluation of 0.22-micron filtration of Tc-99m
sulphur colloid. Radiology 1998; 208:505509
25. Camp ER, Cendan JC, Feezor R, Lind DS,
Wilkinson E, Copeland EM. The hottest sentinel
lymph node is not always the positive node. Am
Surg 2004; 70:475478
26. Liu LC, Lang JE, Jenkins T, et al. Is it necessary to
harvest additional lymph nodes after resection of
the most radioactive sentinel lymph node in breast
cancer? J Am Coll Surg 2008; 207:853858
27. Bourgeois P, Nogaret JM, Veys I, et al. How hot
is the pathologically positive sentinel lymph node
in breast cancer patients? Nucl Med Commun
2003; 24:513518
28. Govaert GAM, Oostenbroek RJ, Plaisier PW. Prolonged skin staining after intradermal use of patent blue in sentinel lymph node biopsy for breast
cancer. Eur J Surg Oncol 2005; 31:373375
29. Scherer K, Studer W, Figueiredo V, Bircher AJ.
Anaphylaxis to isosulphan blue and cross-reactivity to patent blue V: case report and review of the
nomenclature of vital blue dyes. Ann Allergy
Asthma Immunol 2006; 96:497500
30. Veronesi U, Paganelli G, Viale G, et al. A randomised comparison of sentinel-node biopsy with
routine axillary dissection in breast cancer. N
Engl J Med 2003; 349:546553
31. Intra M, Rotmensz N, Veronesi P, et al. Sentinel
node biopsy is not a standard procedure in ductal
carcinoma in situ of the breast: the experience of
the European institute of oncology on 854 patients
in 10 years. Ann Surg 2008; 247:315319
32. Van la Parra RF, Ernst MF, Barneveld PC, Broekman JM, Rutten MJ, Bosscha K. The value of sentinel lymph node biopsy in ductal carcinoma in
situ (DCIS) and DCIS with microinvasion of the
breast. Eur J Surg Oncol 2008; 34:631635
33. Boughey JC, Khakpour N, Meric-Bernstam F, et
al. Selective use of sentinel lymph node surgery
during prophylactic mastectomy. Cancer 2006;
107:14401447
34. Papa MZ, Zippel D, Kaufman B, et al. Timing of
sentinel lymph node biopsy in patients receiving
neoadjuvant chemotherapy for breast cancer. J
Surg Oncol 2008; 98:403406
35. Schrenk P, Tausch C, Wlfl S, Bogner S, Fridrik
M, Wayand W. Sentinel node mapping performed
before preoperative chemotherapy may avoid axillary dissection in breast cancer patients with
negative or micrometastatic sentinel nodes. Am J
Surg 2008; 196:176183
F O R YO U R I N F O R M AT I O N
The readers attention is directed to the commentary on
this article, which appears on the preceding pages.
256
AJR:196, February 2011
You might also like
- Herrmann - Radioguided Surgery - Current Applications and Innovative Directions in Clinical PracticeDocument494 pagesHerrmann - Radioguided Surgery - Current Applications and Innovative Directions in Clinical Practicesimona mariana dutuNo ratings yet
- 380 (Olive Peart) Lange QA Mammography Examination PDFDocument186 pages380 (Olive Peart) Lange QA Mammography Examination PDFGabriel Callupe100% (2)
- Lymph Nodes Esophageal Cancer Staging: Improved Accuracy by Endoscopic Ultrasound of CeliacDocument5 pagesLymph Nodes Esophageal Cancer Staging: Improved Accuracy by Endoscopic Ultrasound of CeliacPoonam JoshiNo ratings yet
- Sentinel Lymph Node Methods in Breast CancerDocument10 pagesSentinel Lymph Node Methods in Breast CancerZuriNo ratings yet
- The Diagnostic Accuracy of Abdominal Ultrasound Imaging For Detection of Ovarian MassesDocument5 pagesThe Diagnostic Accuracy of Abdominal Ultrasound Imaging For Detection of Ovarian MassesHanny TanzilNo ratings yet
- Linfonodo CentinelaDocument8 pagesLinfonodo CentinelaKaren Palma CarrascoNo ratings yet
- Abstracts 991: ST STDocument1 pageAbstracts 991: ST STAli Sibra MulluziNo ratings yet
- Lessismore:Minimally Invasiveandquality Surgicalmanagementof GynecologiccancerDocument12 pagesLessismore:Minimally Invasiveandquality Surgicalmanagementof GynecologiccancerNita AgarwalNo ratings yet
- Simple Ultrasound Rules To Distinguish Between Benign and Malignant Adnexal Masses Before Surgery: Prospective Validation by IOTA GroupDocument16 pagesSimple Ultrasound Rules To Distinguish Between Benign and Malignant Adnexal Masses Before Surgery: Prospective Validation by IOTA GroupherryNo ratings yet
- Breast Tomosynthesis in Clinical PracticeDocument9 pagesBreast Tomosynthesis in Clinical PracticedeadcorpsesNo ratings yet
- Image Features of True Positive and False Negative Cancers in Screening MammogramsDocument9 pagesImage Features of True Positive and False Negative Cancers in Screening MammogramsTher RayNo ratings yet
- Safety and Efficacy of Ultrasound-Guided Fiducial Marker Implantation For Cyberknife Radiation TherapyDocument7 pagesSafety and Efficacy of Ultrasound-Guided Fiducial Marker Implantation For Cyberknife Radiation TherapyAngelito SalinasNo ratings yet
- External Validation of Diagnostic Models To Estimate The Risk of Malignancy in Adnexal MassesDocument12 pagesExternal Validation of Diagnostic Models To Estimate The Risk of Malignancy in Adnexal Massesalvin bramantyoNo ratings yet
- Four Risk of Malignancy Indices in Evaluation of Pelvic MassesDocument8 pagesFour Risk of Malignancy Indices in Evaluation of Pelvic MassesherryNo ratings yet
- Comparison of Ultrasound and Mammography For Early DiagnosisDocument8 pagesComparison of Ultrasound and Mammography For Early Diagnosishdh1224No ratings yet
- Laparoscopic-Assisted Radical Vaginal Hysterectomy (LARVH) : Prospective Evaluation of 200 Patients With Cervical CancerDocument7 pagesLaparoscopic-Assisted Radical Vaginal Hysterectomy (LARVH) : Prospective Evaluation of 200 Patients With Cervical CancerHari NugrohoNo ratings yet
- Detection and Characterization of Breast Masses With Ultrasound Tomography - Clinical Results - Duric Et Al - 2009Document8 pagesDetection and Characterization of Breast Masses With Ultrasound Tomography - Clinical Results - Duric Et Al - 2009samouille6666No ratings yet
- Bladder 8Document9 pagesBladder 8Duddy Ari HardiantoNo ratings yet
- Breast CA ScreeningDocument8 pagesBreast CA Screeningchristelm_1No ratings yet
- PDF DIR 378Document6 pagesPDF DIR 378Sidgi BadjriNo ratings yet
- Breast Cancer Missed at Screening Hindsight or MiDocument6 pagesBreast Cancer Missed at Screening Hindsight or MideaNo ratings yet
- BGC Impronta 2021en t1 OriginalDocument10 pagesBGC Impronta 2021en t1 OriginalGlenda Marina Falcon PachecoNo ratings yet
- Cancer de MamaDocument11 pagesCancer de MamaIng SánchezNo ratings yet
- Exact ArticleDocument6 pagesExact ArticleMuhammad WaqasNo ratings yet
- FNAC Axillary 2012Document9 pagesFNAC Axillary 2012Dima PathNo ratings yet
- 2015 Article 269Document8 pages2015 Article 269Hiba MimineNo ratings yet
- (Review) Comparison of The Clinical Application Value of Mo-Targeted X-Ray, Color Doppler Ultrasound and MRI in Preoperative Comprehensive Evaluation of Breast CancerDocument5 pages(Review) Comparison of The Clinical Application Value of Mo-Targeted X-Ray, Color Doppler Ultrasound and MRI in Preoperative Comprehensive Evaluation of Breast Cancer최윤No ratings yet
- Ajr 10 4615Document7 pagesAjr 10 4615rizkiNo ratings yet
- Trends in Delayed Breast Cancer Diagnosis After ReDocument9 pagesTrends in Delayed Breast Cancer Diagnosis After ReeugeniaNo ratings yet
- FNAC Axillary 2012bDocument7 pagesFNAC Axillary 2012bDima PathNo ratings yet
- Role of Computed Tomography (CT) Scan in Staging of Cervical CarcinomaDocument6 pagesRole of Computed Tomography (CT) Scan in Staging of Cervical CarcinomaTriponiaNo ratings yet
- AxilaDocument3 pagesAxilaEmilio SanchezNo ratings yet
- Axillary ClearenceDocument9 pagesAxillary ClearencelakanthNo ratings yet
- Outcomes Following Surgery For Colorectal Live 2016 International Journal ofDocument1 pageOutcomes Following Surgery For Colorectal Live 2016 International Journal ofoomculunNo ratings yet
- Baek 2017Document10 pagesBaek 2017witaNo ratings yet
- Predicting The Risk of Malignancy in Adnexal Masses Based On IOTA AJOG 2016Document14 pagesPredicting The Risk of Malignancy in Adnexal Masses Based On IOTA AJOG 2016Fernando Suarez ChumaceroNo ratings yet
- 1339-Article Text-24955-2-10-20220330Document6 pages1339-Article Text-24955-2-10-20220330Kevean Kimi LimNo ratings yet
- Evaluation of Axillary Lymphadenopathy With Carcinoma BreastDocument42 pagesEvaluation of Axillary Lymphadenopathy With Carcinoma BreastMashrufNo ratings yet
- Residual Breast Tissue After Mastectomy, How Often and Where It Is LocatedDocument11 pagesResidual Breast Tissue After Mastectomy, How Often and Where It Is LocatedBunga Tri AmandaNo ratings yet
- Jurnal CNFDocument36 pagesJurnal CNFAndrianorMuhammadNo ratings yet
- MRI Criteria For The Diagnosis of Pleomorphic Adenoma: A Validation StudyDocument6 pagesMRI Criteria For The Diagnosis of Pleomorphic Adenoma: A Validation Studysarah iriamanaNo ratings yet
- JurnalradiologieDocument7 pagesJurnalradiologieBella AmaliaNo ratings yet
- Esophageal Cancer Recurrence Patterns and Implicati 2013 Journal of ThoracicDocument5 pagesEsophageal Cancer Recurrence Patterns and Implicati 2013 Journal of ThoracicFlorin AchimNo ratings yet
- Research ArticleDocument8 pagesResearch ArticleGrace Juniaty GozaliNo ratings yet
- Jurnal Abdel GawadDocument9 pagesJurnal Abdel GawadSeptiana meta handayaniNo ratings yet
- Diagnostics 10 00103Document13 pagesDiagnostics 10 00103Ahmed NomanNo ratings yet
- Diagnostic Parameters To Differentiate Benign From Malignant Ovarian Masses With Contrast-Enhanced Transvaginal SonographyDocument8 pagesDiagnostic Parameters To Differentiate Benign From Malignant Ovarian Masses With Contrast-Enhanced Transvaginal SonographyRush32No ratings yet
- Nej Mo A 1414882Document9 pagesNej Mo A 1414882Corina Hagiu-RaduNo ratings yet
- HN 03-2011 Submadibular Salivary Gland Transfer PDFDocument7 pagesHN 03-2011 Submadibular Salivary Gland Transfer PDFVikas VatsNo ratings yet
- Evaluation of Breast Masses Using Mammography and Sonography As First Line InvestigationsDocument3 pagesEvaluation of Breast Masses Using Mammography and Sonography As First Line InvestigationsNata SanjayaNo ratings yet
- Human Colonic TissuesDocument9 pagesHuman Colonic TissuesAnomar MRgNo ratings yet
- Radiology: World Journal ofDocument7 pagesRadiology: World Journal ofWenny EudensiaNo ratings yet
- Magnetic Resonance Imaging For The Detection of Nasopharyngeal CarcinomaDocument4 pagesMagnetic Resonance Imaging For The Detection of Nasopharyngeal CarcinomaFebri W FinisiaNo ratings yet
- The Diagnostic Accuracy of PDFDocument5 pagesThe Diagnostic Accuracy of PDFfrnsskNo ratings yet
- Su H. Et Al.,2023Document9 pagesSu H. Et Al.,2023Mai M. AlshalNo ratings yet
- Journal of Surgical Oncology - 2013 - Fortunato - When Mastectomy Is Needed Is The Nipple Sparing Procedure A New StandardDocument6 pagesJournal of Surgical Oncology - 2013 - Fortunato - When Mastectomy Is Needed Is The Nipple Sparing Procedure A New StandardJethro ConcepcionNo ratings yet
- Saunders 2010Document5 pagesSaunders 2010Evan QuirozNo ratings yet
- Giuliano 1997Document6 pagesGiuliano 1997angelica cuevasNo ratings yet
- The Benefit of Tru-Cut Biopsy in Breast Masses: Poster No.: Congress: Type: Authors: KeywordsDocument8 pagesThe Benefit of Tru-Cut Biopsy in Breast Masses: Poster No.: Congress: Type: Authors: Keywordsم.محمدولدعليNo ratings yet
- Analysis of Ovarian Tumor Pathology by Fourier Transform Infrared SpectrosDocument6 pagesAnalysis of Ovarian Tumor Pathology by Fourier Transform Infrared SpectrosDAVID ROSAS VARANo ratings yet
- A Randomized Trial of Laparoscopic Versus Open Surgery For Rectal CancerDocument9 pagesA Randomized Trial of Laparoscopic Versus Open Surgery For Rectal CancerOtoyGethuNo ratings yet
- Maximum CT ScoreDocument11 pagesMaximum CT ScoreChavdarNo ratings yet
- CT Chest ScoringDocument10 pagesCT Chest ScoringChavdarNo ratings yet
- Clinical Significance of Azygos VeinDocument6 pagesClinical Significance of Azygos VeinChavdarNo ratings yet
- Tu of The OrbitDocument12 pagesTu of The OrbitChavdarNo ratings yet
- Orbital Peripheral Nerve SheathDocument15 pagesOrbital Peripheral Nerve SheathChavdarNo ratings yet
- Pulmonary Cavitation An Under-RecognizedDocument8 pagesPulmonary Cavitation An Under-RecognizedChavdarNo ratings yet
- CT Evaluation of Congenital and AcquiredDocument14 pagesCT Evaluation of Congenital and AcquiredChavdarNo ratings yet
- COVID-19-Associated Carotid Atherothrombosis and StrokeDocument3 pagesCOVID-19-Associated Carotid Atherothrombosis and StrokeChavdarNo ratings yet
- Ann Clin Transl Neurol - 2019 - Campbell - Hearing Impairment After Subarachnoid HemorrhageDocument11 pagesAnn Clin Transl Neurol - 2019 - Campbell - Hearing Impairment After Subarachnoid HemorrhageChavdarNo ratings yet
- Annals of Clinical Medicine and Research: Pulmonary Cavitation Post-COVID-19 Pneumonia: A CaseDocument3 pagesAnnals of Clinical Medicine and Research: Pulmonary Cavitation Post-COVID-19 Pneumonia: A CaseChavdarNo ratings yet
- Endovasc Jug BulbDocument7 pagesEndovasc Jug BulbChavdarNo ratings yet
- Clavicle and Sternoclavicular JointDocument14 pagesClavicle and Sternoclavicular JointChavdarNo ratings yet
- Imaging of Palatal LumpsDocument11 pagesImaging of Palatal LumpsChavdarNo ratings yet
- Intraluminal Carotid Artery Thrombus in COVID-19 AnotherDocument6 pagesIntraluminal Carotid Artery Thrombus in COVID-19 AnotherChavdarNo ratings yet
- Geniculate Artery Embolization For Recurrent Postarthroplasty Hemarthrosis of The KneeDocument4 pagesGeniculate Artery Embolization For Recurrent Postarthroplasty Hemarthrosis of The KneeChavdarNo ratings yet
- Contemporary Surgical Treatment of Benign Prostatic Hyperplasia VAJNODocument6 pagesContemporary Surgical Treatment of Benign Prostatic Hyperplasia VAJNOChavdarNo ratings yet
- Bone Metastases From Thyroid Carcinoma of Follicular Origin A Single Institutional ExperienceDocument6 pagesBone Metastases From Thyroid Carcinoma of Follicular Origin A Single Institutional ExperienceChavdarNo ratings yet
- Common Carotid Artery Occlusion in A Young Patient Can Large-Vessel Stroke Be The Initial Clinical Manifestation of Coronavirus Disease 2019Document4 pagesCommon Carotid Artery Occlusion in A Young Patient Can Large-Vessel Stroke Be The Initial Clinical Manifestation of Coronavirus Disease 2019ChavdarNo ratings yet
- Angiographic Analysis of The Anatomical Variants IDocument5 pagesAngiographic Analysis of The Anatomical Variants IChavdarNo ratings yet
- AJCC 8thed TNMDocument34 pagesAJCC 8thed TNMPrarthna PatelNo ratings yet
- Lymph FullDocument4 pagesLymph Fullmayada elmakkiNo ratings yet
- Axillary ClearenceDocument9 pagesAxillary ClearencelakanthNo ratings yet
- OSNA For Lymph Node Analysis in Breast Cancer: One Step - One DecisionDocument8 pagesOSNA For Lymph Node Analysis in Breast Cancer: One Step - One DecisionStacey Lee100% (1)
- MCQ Breast Surgery Bordeaux 2010Document9 pagesMCQ Breast Surgery Bordeaux 2010Ismail Evren100% (1)
- Volum Rezumate ZMN 2023 1Document56 pagesVolum Rezumate ZMN 2023 1LuthienFreyNo ratings yet
- Responsive Document - CREW: FDA: Regarding Communications Between The FDA and Martin Shkreli: 11/30/2012 - 11005576 - Enc - EJLDocument12 pagesResponsive Document - CREW: FDA: Regarding Communications Between The FDA and Martin Shkreli: 11/30/2012 - 11005576 - Enc - EJLCREWNo ratings yet
- Anatomy of The AxillarDocument43 pagesAnatomy of The AxillarJustine NyangaresiNo ratings yet
- Sentinel Lymph Node BiopsyDocument6 pagesSentinel Lymph Node Biopsycoolash1010No ratings yet
- Society of Surgical Oncology 67th Annual Cancer SymposiumDocument198 pagesSociety of Surgical Oncology 67th Annual Cancer Symposiummc_nsNo ratings yet
- Axillary Lymph NodesDocument2 pagesAxillary Lymph NodesNiqabi NextdoorNo ratings yet
- SentinaDocument10 pagesSentinaakayaNo ratings yet
- Pathology Reporting of Breast Cancer A GuideDocument52 pagesPathology Reporting of Breast Cancer A Guidewht36No ratings yet
- Hematogenous Spread. Lymphatic Spread.: 275 Characteristics of Benign and Malignant NeoplasmsDocument4 pagesHematogenous Spread. Lymphatic Spread.: 275 Characteristics of Benign and Malignant NeoplasmsGin TokiNo ratings yet
- Breast Cancer Staging PDFDocument12 pagesBreast Cancer Staging PDFDanu BagoesNo ratings yet
- The BreastDocument4,358 pagesThe BreastluficorNo ratings yet
- Breast Cancer PresentationDocument52 pagesBreast Cancer Presentationapi-341607639100% (1)
- Evaluation of Axillary Lymphadenopathy With Carcinoma BreastDocument42 pagesEvaluation of Axillary Lymphadenopathy With Carcinoma BreastMashrufNo ratings yet
- Important Surgical Concepts and Techniques in Inguinal Lymph Node DissectionDocument8 pagesImportant Surgical Concepts and Techniques in Inguinal Lymph Node DissectionVlad GrigoreNo ratings yet
- Frozen Section DiagnosisDocument290 pagesFrozen Section DiagnosisDima Path100% (2)
- Breast Operative Standards OSCS ManualDocument5 pagesBreast Operative Standards OSCS ManualVlad GrigoreNo ratings yet
- Cody, Hiram S - Sentinel Lymph Node Biopsy (2003, Distributed in The USA by Taylor & Francis, Martin Dunitz) PDFDocument384 pagesCody, Hiram S - Sentinel Lymph Node Biopsy (2003, Distributed in The USA by Taylor & Francis, Martin Dunitz) PDFJaime AlonsoNo ratings yet
- Presentaciones Congreso EANM 2018Document844 pagesPresentaciones Congreso EANM 2018Puig Cozar100% (1)
- Role of Sentinel Lymph Node Biopsy in Head and Neck CancerDocument78 pagesRole of Sentinel Lymph Node Biopsy in Head and Neck CancernasimNo ratings yet
- 5 Axillary and Epitrochlear Lymph Node Dissection For MelanomaDocument11 pages5 Axillary and Epitrochlear Lymph Node Dissection For MelanomajuanrangoneNo ratings yet
- Spect/Ct Imaging: Benefits To Clinical Nuclear MedicineDocument32 pagesSpect/Ct Imaging: Benefits To Clinical Nuclear MedicineVladut BibanNo ratings yet
- Chapter 6 NeoplasiaDocument24 pagesChapter 6 Neoplasiahenna patelNo ratings yet
- ASiT Conference Hull 2010 - Abstract BookDocument114 pagesASiT Conference Hull 2010 - Abstract BookAssociation of Surgeons in TrainingNo ratings yet