Professional Documents
Culture Documents
Jurnal
Jurnal
Uploaded by
Binta R. DikaraCopyright:
Available Formats
You might also like
- BCMB 415 Exam 3, Fall 2016 KeyDocument9 pagesBCMB 415 Exam 3, Fall 2016 KeyzzmasterNo ratings yet
- June 17 26 s001 PDFDocument9 pagesJune 17 26 s001 PDFBetul RojeabNo ratings yet
- The Entire Avengers Age of Ultron ScriptDocument51 pagesThe Entire Avengers Age of Ultron ScriptLebron JamesNo ratings yet
- Orac MethodDocument5 pagesOrac MethodPriscillaL.SilvaNo ratings yet
- Plant Tissue Culturelab Practices Made Easy (For Beginners) PDFDocument75 pagesPlant Tissue Culturelab Practices Made Easy (For Beginners) PDFSultan Ahmad Khan100% (1)
- Journal of Electroanalytical ChemistryDocument10 pagesJournal of Electroanalytical ChemistrySelin AktaşNo ratings yet
- Determination of Homogentisic Acid in Urine For Diagnosis of Alcaptonuria: Capillary Electrophoretic Method Optimization Using Experimental DesignDocument6 pagesDetermination of Homogentisic Acid in Urine For Diagnosis of Alcaptonuria: Capillary Electrophoretic Method Optimization Using Experimental DesigncristinaNo ratings yet
- Determination of PKa Values of Benzimidazole Derivatives From Mobility Obtained by Capillary ElectrophoresisDocument5 pagesDetermination of PKa Values of Benzimidazole Derivatives From Mobility Obtained by Capillary ElectrophoresisMario PosavecNo ratings yet
- Simultaneous Monitoring of Glucose and Uric Acid On A Single Test Strip WithDocument5 pagesSimultaneous Monitoring of Glucose and Uric Acid On A Single Test Strip WithMadalina FleancuNo ratings yet
- Selective Simultaneous Determination of Paracetamol and Uric Acid Using A Glassy Carbon Electrode Modified With..Document8 pagesSelective Simultaneous Determination of Paracetamol and Uric Acid Using A Glassy Carbon Electrode Modified With..ÁLVARO CARLOS AGUADO MALLQUINo ratings yet
- Determination of Pharmaceuticals ClassifiedDocument9 pagesDetermination of Pharmaceuticals ClassifiedsorinamotocNo ratings yet
- Using Cysteine-CdSe Nanoparticles For Quantification of Ovalbumin Thermal Denaturation by Cyclic and Differential Pulse VoltammetryDocument13 pagesUsing Cysteine-CdSe Nanoparticles For Quantification of Ovalbumin Thermal Denaturation by Cyclic and Differential Pulse VoltammetryInternational Journal of Academic Scientific ResearchNo ratings yet
- Aguilar 2002 LCRDocument5 pagesAguilar 2002 LCRiplabaNo ratings yet
- UntitledDocument7 pagesUntitledNancy HeshamNo ratings yet
- Baldelli 2004Document7 pagesBaldelli 2004davidlaglaNo ratings yet
- Protein Releasing Kinetics of Bakers' Yeast Cells by UltrasoundDocument6 pagesProtein Releasing Kinetics of Bakers' Yeast Cells by UltrasoundjainashahNo ratings yet
- Zhang 12Document11 pagesZhang 12Ayu Miftachul Jan'ahNo ratings yet
- Artikel Fito..Document10 pagesArtikel Fito..Nurul FuadyNo ratings yet
- Determination of Aniline and Its Derivatives in Environmental Water by Capillary Electrophoresis With On-Line ConcentrationDocument10 pagesDetermination of Aniline and Its Derivatives in Environmental Water by Capillary Electrophoresis With On-Line ConcentrationkareemNo ratings yet
- Uapcr DiagnosticDocument8 pagesUapcr DiagnosticYA MAAPNo ratings yet
- Sensors and Actuators B: Chemical: Jin Wang, Hiroaki Suzuki, Takaaki SatakeDocument5 pagesSensors and Actuators B: Chemical: Jin Wang, Hiroaki Suzuki, Takaaki SatakeMELISSA VERONICA TUDELANO OSPINANo ratings yet
- 2012-Biomedical Chromatography-AppelDocument7 pages2012-Biomedical Chromatography-AppelHaojie ZhuNo ratings yet
- Automated In-Tube Solid Phase Microextraction Coupled With HPLC-ES-MS For The Determination of Catechins and Caffeine in TeaDocument7 pagesAutomated In-Tube Solid Phase Microextraction Coupled With HPLC-ES-MS For The Determination of Catechins and Caffeine in TeaSeema YadavNo ratings yet
- Dialysis DoseDocument7 pagesDialysis DoseFotaf FolaNo ratings yet
- Turnell 1982Document5 pagesTurnell 1982Ammar MaryamabadiNo ratings yet
- Metabolomic Profiling of Amoebic and Pyogenic Liver Abscesses An in Vitro NMR StudyDocument16 pagesMetabolomic Profiling of Amoebic and Pyogenic Liver Abscesses An in Vitro NMR Studysantosh091283No ratings yet
- Analytica Chimica Acta: Taher Alizadeh, Mohammad Reza Ganjali, Faride Ra FieiDocument9 pagesAnalytica Chimica Acta: Taher Alizadeh, Mohammad Reza Ganjali, Faride Ra FieiwardaninurindahNo ratings yet
- Cold Atmospheric Plasma Discharged in Water and Its Potential Use in Cancer TherapyDocument18 pagesCold Atmospheric Plasma Discharged in Water and Its Potential Use in Cancer TherapySek PyroNo ratings yet
- Improving LC-MS Sensitivity Through Increases in Chromatographic Performance: Comparisons of UPLC-ES/MS/MS To Hplc-Es/Ms/MsDocument10 pagesImproving LC-MS Sensitivity Through Increases in Chromatographic Performance: Comparisons of UPLC-ES/MS/MS To Hplc-Es/Ms/MsgannysunNo ratings yet
- Tavana 2012Document6 pagesTavana 2012selia destianingrumNo ratings yet
- Jurnal RingerfundinDocument7 pagesJurnal RingerfundinRyan Arifin SuryantoNo ratings yet
- Piao 2008Document5 pagesPiao 2008Ellie satrianiNo ratings yet
- Scala Benuzzi2018Document10 pagesScala Benuzzi2018mohammad aidilNo ratings yet
- A Simple 96-Well Liquid-Liquid Extraction With A Mixture ofDocument10 pagesA Simple 96-Well Liquid-Liquid Extraction With A Mixture ofSowbhagya LaxmiNo ratings yet
- Nanocomposite Modified Electrochemical Sensor For Sensitive and Selective Determination of NoradrenalineDocument10 pagesNanocomposite Modified Electrochemical Sensor For Sensitive and Selective Determination of NoradrenalineLeau Sorina-AlexandraNo ratings yet
- 10 1016@j Colsurfa 2014 05 029Document7 pages10 1016@j Colsurfa 2014 05 029wora123potNo ratings yet
- Ultrasensitive Label Free Electrical Detection of Insulin in Neat BloodDocument6 pagesUltrasensitive Label Free Electrical Detection of Insulin in Neat BloodDusan SilniNo ratings yet
- Removal MikroorganismDocument9 pagesRemoval MikroorganismUkiNo ratings yet
- Effect of He Self-Organized Pattern Plasma-Activated Media With Different Conductivity On Cancer CellsDocument23 pagesEffect of He Self-Organized Pattern Plasma-Activated Media With Different Conductivity On Cancer CellsSek PyroNo ratings yet
- Molecularly Imprinted Electrochemical Sensing of Urinary Melatonin in A Microfluidic SystemDocument11 pagesMolecularly Imprinted Electrochemical Sensing of Urinary Melatonin in A Microfluidic SystemgpaivNo ratings yet
- Square Wave Adsorptive Cathodic Stripping Voltammetry Automated by Sequential Injection Analysis Potentialities and Limitations Exemplified by The Determination of Methyl Parathion in Water SamplesDocument8 pagesSquare Wave Adsorptive Cathodic Stripping Voltammetry Automated by Sequential Injection Analysis Potentialities and Limitations Exemplified by The Determination of Methyl Parathion in Water SamplesSHERLY KIMBERLY RAMOS JESUSNo ratings yet
- 9-Aryl Substituted Hydroxylated Xanthen-3-Ones: Synthesis, Structure and Antioxidant Potency EvaluationDocument7 pages9-Aryl Substituted Hydroxylated Xanthen-3-Ones: Synthesis, Structure and Antioxidant Potency EvaluationUmar IjazNo ratings yet
- Online Extraction LC-MSMS Method For The Simultaneous Quantitative...Document11 pagesOnline Extraction LC-MSMS Method For The Simultaneous Quantitative...qketzalNo ratings yet
- Biosensors: Single-Drop Analysis of Epinephrine and Uric Acid On A Screen-Printed Carbon ElectrodeDocument17 pagesBiosensors: Single-Drop Analysis of Epinephrine and Uric Acid On A Screen-Printed Carbon Electrodewidia waatiNo ratings yet
- Pnas-A Fully Integrated Microfluidic Genetic Analysis SystemDocument6 pagesPnas-A Fully Integrated Microfluidic Genetic Analysis Systemapi-3723328No ratings yet
- Single Determination of A-Ketoglutaric Acid and Pyruvic Acid in Beer by HPLC With UV DetectionDocument6 pagesSingle Determination of A-Ketoglutaric Acid and Pyruvic Acid in Beer by HPLC With UV DetectionPatrícia MontenegroNo ratings yet
- Analisis de PQ by LC-MSDocument5 pagesAnalisis de PQ by LC-MSalina esther diaz arrietaNo ratings yet
- Litopenaeus Vannamei (Boone, 1931) Closed-Hatchery System: Application of An Electrolytic Water Treatment Technique in ADocument10 pagesLitopenaeus Vannamei (Boone, 1931) Closed-Hatchery System: Application of An Electrolytic Water Treatment Technique in AHiral PatelNo ratings yet
- Effect of Zeolite Types LTX and Lta On Physicochemical Parameters of Drinking Water Samples in Ghana, Assisted by Light Transmission ExperiDocument7 pagesEffect of Zeolite Types LTX and Lta On Physicochemical Parameters of Drinking Water Samples in Ghana, Assisted by Light Transmission ExperiesatjournalsNo ratings yet
- 2010 - Recent Advances of Capillary Electrophoresis in Pharmaceutical AnalysisDocument24 pages2010 - Recent Advances of Capillary Electrophoresis in Pharmaceutical AnalysisStefana SzántóNo ratings yet
- Sobre Ação Do EDC NHSDocument6 pagesSobre Ação Do EDC NHSAlberto Galdino SparrowNo ratings yet
- Method 1611 Enterococci in Water by TaqMan Quantitative Polymerase Chain Reaction (QPCR) AssayDocument56 pagesMethod 1611 Enterococci in Water by TaqMan Quantitative Polymerase Chain Reaction (QPCR) AssayJBNo ratings yet
- In Vitro and in Vivo Trials of Wearable Artificial KidneyDocument5 pagesIn Vitro and in Vivo Trials of Wearable Artificial KidneyMIGUEL ANGEL PALOMINO CAMPONo ratings yet
- Determination of Amoxicillin in Plasma Samples by Capillary ElectrophoresisDocument7 pagesDetermination of Amoxicillin in Plasma Samples by Capillary ElectrophoresisJose Manuel Rito GonzalezNo ratings yet
- Caarvedilol Electrochimic - Grupa 3 PDFDocument4 pagesCaarvedilol Electrochimic - Grupa 3 PDFdanielafarmacie_1617No ratings yet
- Darroudi 2022 J. Electrochem. Soc. 169 026501Document7 pagesDarroudi 2022 J. Electrochem. Soc. 169 026501abdellahbe41No ratings yet
- Estudo Hepasfera Versus BeadDocument6 pagesEstudo Hepasfera Versus BeadAlexandre Campos Moraes AmatoNo ratings yet
- Molecules 19 18923Document13 pagesMolecules 19 18923dikadika_tansNo ratings yet
- Electron Spin Resonance SpectrosDocument6 pagesElectron Spin Resonance SpectrossakuraleeshaoranNo ratings yet
- Use of A Blood Gas Analyzer and A Laboratory Autoanalyzer in Routine Practice To Measure Electrolytes in Intensive Care Unit PatientsDocument7 pagesUse of A Blood Gas Analyzer and A Laboratory Autoanalyzer in Routine Practice To Measure Electrolytes in Intensive Care Unit PatientsZheng GuNo ratings yet
- Electrochemical Determination of Uric Acid in Human Urine Using Nickel Hexa-Cyano Ferrate Modified Carbon Paste ElectrodeDocument18 pagesElectrochemical Determination of Uric Acid in Human Urine Using Nickel Hexa-Cyano Ferrate Modified Carbon Paste ElectrodeMohammad MiyanNo ratings yet
- Development and Evaluation of A Bio Ion Measureme - 2013 - Journal of AcupuncturDocument9 pagesDevelopment and Evaluation of A Bio Ion Measureme - 2013 - Journal of AcupuncturMatias DamiánNo ratings yet
- Application of Real-Time Quantitative PCR For The Detection of SelectedDocument9 pagesApplication of Real-Time Quantitative PCR For The Detection of SelectedMiriam LeiNo ratings yet
- Urinary Stones: Medical and Surgical ManagementFrom EverandUrinary Stones: Medical and Surgical ManagementMichael GrassoNo ratings yet
- 10th Science Maharashtra Board Animal Classification IntroDocument12 pages10th Science Maharashtra Board Animal Classification IntroAditya BorateNo ratings yet
- Human EyeDocument7 pagesHuman Eyeofmir154No ratings yet
- AO No. 156 S. 1971 AO No. 291 S. 1976 Section 7 Section 7Document5 pagesAO No. 156 S. 1971 AO No. 291 S. 1976 Section 7 Section 7Clark RamosNo ratings yet
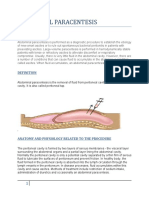
- Abdominal ParacentesisDocument14 pagesAbdominal ParacentesisManisha Thakur100% (1)
- A Cost Optimization-Based Design of Precast Concrete Floors Using Genetic AlgorithmsDocument9 pagesA Cost Optimization-Based Design of Precast Concrete Floors Using Genetic AlgorithmsRajesh RanjanNo ratings yet
- Autogenics Therapy For Dynamic Relaxation and Stress ReliefDocument17 pagesAutogenics Therapy For Dynamic Relaxation and Stress ReliefNickRhodos100% (2)
- Priming and Pretreatment of Seeds and SeedlingsDocument606 pagesPriming and Pretreatment of Seeds and SeedlingsFadhillah ArsaniNo ratings yet
- Brain Plasticity - How Experience Changes The BrainDocument5 pagesBrain Plasticity - How Experience Changes The BrainWilson OTONo ratings yet
- Microsoft Word - Biology - Genetics UnitDocument20 pagesMicrosoft Word - Biology - Genetics Unitapi-233894872No ratings yet
- ENDO) Pancreas. For The ClassDocument58 pagesENDO) Pancreas. For The ClassromaloiNo ratings yet
- iBT TOEFL 托福词汇 词以类记Document15 pagesiBT TOEFL 托福词汇 词以类记beat2013No ratings yet
- Bioinformatic Tools For Next Generation DNA Sequencing - PHD ThesisDocument237 pagesBioinformatic Tools For Next Generation DNA Sequencing - PHD ThesisDan PintilescuNo ratings yet
- Minerva Anestesiol 2017 GruenewaldDocument15 pagesMinerva Anestesiol 2017 GruenewaldMarjorie Lisseth Calderón LozanoNo ratings yet
- MCB 7200: Molecular BiologyDocument27 pagesMCB 7200: Molecular BiologytasniyanNo ratings yet
- Longing To BelongDocument6 pagesLonging To BelongKhalid KhalilNo ratings yet
- Structure of Chromosomes, Cell Cycle, Cell DivisionDocument35 pagesStructure of Chromosomes, Cell Cycle, Cell DivisionXyzNo ratings yet
- Laboratory Diagnosis of Leptospirosis: A Challenge: Didier Musso, Bernard La ScolaDocument8 pagesLaboratory Diagnosis of Leptospirosis: A Challenge: Didier Musso, Bernard La ScolaAnna Diaz YuberoNo ratings yet
- FDS Component 3 PDFDocument307 pagesFDS Component 3 PDFmarielNo ratings yet
- Psychotria ElataDocument2 pagesPsychotria ElataJevon ChengNo ratings yet
- Land Suitability AnalysisDocument79 pagesLand Suitability AnalysisVasilică-Dănuţ HorodnicNo ratings yet
- Poem Book 2012 PDFDocument89 pagesPoem Book 2012 PDFAnshNo ratings yet
- Childhood and Growing UpDocument27 pagesChildhood and Growing UpjikooNo ratings yet
- What Is A Pirate'S Favorite Amino Acid? Arginine. ArgDocument1 pageWhat Is A Pirate'S Favorite Amino Acid? Arginine. ArgSimona PetranNo ratings yet
- Gits Systems Anaphy DisordersDocument23 pagesGits Systems Anaphy DisordersIlawNo ratings yet
- Alprazolam TMaxDocument14 pagesAlprazolam TMaxErwin FernándezNo ratings yet
- Media Preparation SterilizationDocument13 pagesMedia Preparation SterilizationJessica GutierrezNo ratings yet
Jurnal
Jurnal
Uploaded by
Binta R. DikaraOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Jurnal
Jurnal
Uploaded by
Binta R. DikaraCopyright:
Available Formats
Journal of Chromatography B, 821 (2005) 229234
Short communication
Determination of uric acid and p-aminohippuric acid in human saliva and
urine using capillary electrophoresis with electrochemical detection
Potential application in fast diagnosis of renal disease
Yueqing Guan, Ting Wu, Jiannong Ye
Department of Chemistry, East China Normal University, Shanghai 200062, PR China
Received 30 November 2004; accepted 3 March 2005
Available online 23 May 2005
Abstract
The monitoring of uric acid (UA) and p-aminohippuric acid (PAH) levels in biological samples is routinely carried out in clinical laboratories
as an indication of renal disease. With the aim of investigation of the correlation between the trace amounts of UA and PAH in human saliva
or urine and renal diseases, we carried out the determination of UA and PAH in human saliva and urine by using capillary electrophoresis
with electrochemical detection (CE-ED) in this work. Under the optimum conditions, UA, PAH and three coexisting analytes could be
well separated within 21 min at the separation voltage of 14 kV in 80 mmol/L borax running buffer (pH 9.2). Good linear relationship was
established between peak current and concentration of analytes over two orders of magnitude with detection limits (S/N = 3) ranged from
5.01 107 to 2.00 106 mol/L for all analytes. The result shows that this proposed method could be successfully applied for the study on
the correlation between the levels of UA and PAH in human saliva and urine and renal diseases, and provide an alternative and convenient
method for the fast diagnosis of renal disease.
2005 Elsevier B.V. All rights reserved.
Keywords: Capillary electrophoresis; Electrochemical detection; Renal disease; Saliva; Urine; Uric acid; p-Aminohippuric acid
1. Introduction
The kidneys play a vital role in maintaining the bodys
stable internal chemical environment. They are responsible
for filtering impurities from the body, draining waste products of the body and maintaining the necessary balance of
chemicals in the liquid form [1]. Once kidneys are disorder,
people will suffer from loss of role within the family and
workplace, loss of renal function and mobility, loss of physical skills and cognitive abilities, loss of sexual function [2,3]
and medical disorders of diabetes and hypertension [4,5]. Recent years, renal disease has been the focus of many diseases.
In the USA, a survey has shown that more than 372 000 people suffer from renal failure at the end of 2000 (about 0.13%
of the population) and the number of patients is growing at a
Corresponding author. Tel.: +86 21 6223 2254; fax: +86 21 62576217.
E-mail address: jnye@ecnu.edu.cn (J. Ye).
1570-0232/$ see front matter 2005 Elsevier B.V. All rights reserved.
doi:10.1016/j.jchromb.2005.03.049
rate of 6% per year. In 2010, the number of patients will get
to 650 000 [6,7]. In Europe and Asia, there are less detailed
analyses for trends, but the data available indicate a similar
pattern to that in the USA [8].
Several compounds in biological fluids can serve as
marker for diagnose of renal function such as UA and PHA
[9]. The presence of elevated UA levels [10] and an imbalance in PAH levels [1113] have been acknowledged as a
sign of renal diseases. Thus, monitoring of UA and PAH in
human physiological fluids is indispensable for the diagnosis
of renal disease.
So far, a limited number of methods including colorimetry
[13,14], HPLC [1519] and CE with UV [2022] have been
only developed for the quantitative analysis of these renal
markers in human urine or serum, most of them suffer from
long analysis time, low resolution, short column lifetime and
etc. Capillary electrophoresis is becoming increasingly recognized as an important analytical separation technique due
230
Y. Guan et al. / J. Chromatogr. B 821 (2005) 229234
to its speed, efficiency, reproducibility, ultra-small sample
volume, and ease of clearing up the contaminants. In combination with electrochemical detection, CE-ED offers high
sensitivity and good selectivity for electroactive species. The
advantages of capillary electrophoresis include fast analysis
time, high resolution, and ultra-small sample volume. Besides, diluted sample solutions can often be directly injected
into the capillary without complicated sample pretreatment.
Garcia et al. has described the separation of creatine, creatinine, and UA in urine by microchip CE with pulsed ED [23].
Fanguy et al. have separated UA by microchip CE-ED [24]. In
addition, CE with polarity reversal and ED have been applied
for the determination of UA in human serum [25]. However,
none of them have examined the saliva samples as well as
uncovered the relativity between the levels of UA and PAH
in human saliva and urine samples.
In this work, we reported a sensitive and reliable method
for the simultaneous determination of UA, PAH and their exogenous and endogenous co-existing potential interferences
namely xanthine (Xan), hypoxanthine (HX) and ascorbic acid
(Vc) in human saliva and urine by CE-ED, and furthermore,
confirmed the close relativity between the levels of UA and
PAH in human saliva and urine and the renal disease, which
suggests a potential application in fast primary diagnosis of
renal disease. The molecular structures of above ingredients
were shown in Fig. 1.
2. Experimental
2.1. Reagent and solutions
HX, PAH, Vc, UA and Xan were all purchased from Sigma
(St. Louis, MO, USA), and were all used as received. All
chemicals were of analytical grade.
Stock solution of Vc and PAH (1.0 102 mol/L) was
prepared in doubly distilled water, and those of HX, UA and
Xan were dissolved in 0.05 mol/L NaOH aqueous solution
to reach the final concentration of 0.01 mol/L. All standard
solutions were stored in a 4 C refrigerator and were stable for
4 days. All solutions were diluted to the desired concentration
with the running buffer (80 mmol/L H3 BO3 Na2 B4 O7 buffer
with pH value from 8.2 to 9.2). Before use, all solutions were
filtered through 0.22 m nylon filters. All experiments were
performed in a air-conditioned room at 25 C.
2.2. Apparatus
A CE-ED system has been described previously [26,27].
A 30 kV high-voltage power supply (Shanghai Institute of
Nuclear Research, China) provided a separation voltage between the ends of the capillary. The inlet end of the capillary was held at a positive potential and the outlet end of
capillary was maintained at ground. The separations were
proceeded in a 75 cm length of 25 m i.d. and 360 m o.d.
fused-silica capillary (Polymicro Technologies, Phoenix, AZ,
Fig. 1. Molecular structures of HX, PAH, Vc, UA and Xan.
USA). Samples were all injected electrokinetically, applying
14 kV for 6 s.
A three-electrode electrochemical cell consisting of a
laboratory-made 300 m diameter carbon disc working
electrode, a platinum auxiliary electrode and a saturated
calomel electrode (SCE) as the reference electrode, was used
in combination with a BAS LC-4C amperometric detector
(Bioanalytical Systems Inc., West Lafayette, IN, USA).
The carbon disc electrode was made of a piece of 300 m
diameter graphite rod from polishing technique as descried
in a previous report [28]. Before use, the surface of the
carbon-disk electrode was successively polished with emery
paper and alumina power, sonicated in doubly distilled water,
and finally was positioned carefully opposite the outlet of the
capillary with the aid of a micromanipulator (CORRECT,
Tokyo, Japan) and arranged in a wall-jet configuration.
The electropherograms were recorded using a chart record
(Shanghai Dahua Instrumental Factory, China). A YS
38-1000 220 V alternate constant-voltage power supply
(Shanghai Instrumental Transformer Factory, Shanghai,
Y. Guan et al. / J. Chromatogr. B 821 (2005) 229234
Fig. 2. Hydrodynamic voltammograms (HDVs) of HX (1), PAH (2), Vc
(3), UA (4) and Xan (5) in CE-ED. Fused-silica capillary: 25 m i.d.
75 cm; working electrode: 300 m diameter carbon disk electrode; running buffer: 80 mmol/L(BB. pH 9.2); separation voltage: 14 kV; injection
time: 14 kV/6 s; concentrations of five analytes: HX (5.0 105 mol/L),
PAH (1.0 104 mol/L), Vc (1.0 104 mol/L), UA (1.0 104 mol/L)
and Xan (1.0 104 mol/L).
China) was employed to suppress the voltage fluctuation
of the power line. The whole system was assembled in
a air-conditioned room at 25 C in order to minimize the
variation of running buffer viscosity, which is important to
the reproducibility of the experiment results.
2.3. Sample preparation
Urine and saliva samples from apparently healthy volunteers and patients with renal disease were collected from
Shanghai Changzheng Hospital. None of them suffered from
systemic or salivary gland disease that could affect the saliva.
They were requested to fast 30 min before saliva withdrawal.
Urine and saliva samples were diluted with running buffer,
then filtered through 0.22 m nylon filters, and the filtrate
were injected directly to the CE-ED system for analysis. Before use, all solutions were stored in a 4 C refrigerator.
3. Results and discussion
3.1. Effect of the potentials applied to the working
electrode
In amperometric detection, the potential applied to the
working electrode directly affects the sensitivity, detection
limit and stability of this method. Therefore, hydrodynamic
voltammetry experiment was investigated to obtain optimum
detection. As shown in Fig. 2, the peak current of Vc does not
have obvious changes, while, the peak current of other three
analytes increases rapidly when the applied potential exceed
+100 mV for UA, +500 mV for Xan, +600 mV for PAH, and
+700 mV for HX, respectively. However, when applied potential is greater than +950 mV (versus SCE), although the
231
Fig. 3. Effect of the running buffer pH on the migration time of the analytes.
Working potential was +950 mV (vs. SCE); concentrations of five analytes:
HX (1.0 104 mol/L), PAH (1.0 104 mol/L), Vc (2.0 104 mol/L),
UA (1.0 104 mol/L) and Xan (1.0 104 mol/L); other experimental
conditions and labels are the same as in Fig. 2.
peak current of the analytes still have certain increase, both
the baseline noise and the background current increase substantially, which is a big disadvantage for sensitive and stable detection. Therefore, the potential applied to the working
electrode is maintained at +950 mV (versus SCE), where the
background current is not too high and the signal-to-noise
(S/N = 3) ratio is the highest.
3.2. Effects of the pH and concentration of the running
buffer
The acidity of the running buffer affects the zeta potential
(), the electro osmotic flow (EOF) as well as the migration
time and the separation of the analytes. The effect of the running buffer pH on the migration time of the analytes was
investigated in the pH range of 8.29.2. As shown in Fig. 3,
the migration time of all analytes increase with the increasing
pH value. When pH is below 8.4, Xan and Vc cannot be separated. When pH value is 8.79.0, Xan cannot be separated
from UA. At pH 9.2, the five analytes can be well separated,
however, higher pH value results in longer analysis times, and
the analytes are more susceptible to oxidation. Therefore, pH
9.2 was selected as the optimum pH value for this work.
In addition to the pH value, the concentration of the running buffer is another important factor affecting the resolution, migration time and the peak current of the analytes. So
the effect of the running buffer concentration on migration
time was also studied, and the optimum running buffer concentration is 80 mmol/L (pH 9.2).
3.3. Effect of separation voltage and injection time
For a given capillary length, the separation voltage determines the electric field strength, which affects both the
velocity of electroosmotic flow (EOF) and the migration velocity of the analytes, which in turn determines the migration
232
Y. Guan et al. / J. Chromatogr. B 821 (2005) 229234
Fig. 4. Effect of injection time on the peak current of the analytes. Experimental conditions and labels are the same as in Fig. 3.
time of the analytes. As expected, higher separation voltage
gives shorter migration time for all analytes. However, when
the separation voltage exceeds 16 kV, Xan, Vc, and UA cannot be well separated, and baseline noise becomes larger.
Therefore the optimum separation voltage selected is 14 kV,
at which good separation can be obtained for all analytes
within 21 min.
The injection time determining the amount of sampling
affects both peak current and peak shape. The effect of injection time on peak current is studied by varying injection time
from 2 to 10 s at 14 kV. As shown in Fig. 4, the peak current
increases with increasing sampling time. When the injection
time is longer than 8 s, peak current nearly levels off and
peak broadening becomes more severe. In this experiment,
6 s (14 kV) is selected as the optimum injection time.
Through the experiments above, the optimum conditions
for HX, PAH, Vc, UA and Xan have been decided. The typical
electropherogram for a standard mixture solution of the five
analytes is shown in Fig. 5A, from which we can see good
separation can be achieved within 21 min.
3.4. Reproducibility, linearity, detection limits and
recovery
The reproducibility of the peak current is estimated by
making repetitive injections of a standard mixture solution
(2.0 104 mol/L for Vc, 1.0 104 mol/L for HX, PAH,
UA and Xan, respectively) under the selected optimum conditions (n = 7). The relative standard deviations (R.S.D.s) for
the peak current are 0.39, 0.66, 2.39, 0.63 and 0.55% for HX,
Vc, PAH, UA and Xan, respectively. The high reproducibility
indicates that this method is accurate and sensitive.
A series of the standard mixture solutions of HX, PAH,
Vc, UA and Xan with concentration ranging from 2.0 106
to 5.0 104 mol/L are tested to determine the linearity for
all analytes at the carbon disc electrode in this method. The
results of regression analysis on calibration curves and detec-
Fig. 5. Electropherograms of the standard mixture solution (A), and samples
of patient urine sample (B), and patient saliva (C). Experimental conditions
and labels are the same as in Fig. 3. Peak identification: (1) HX; (2) PAH;
(3) Vc; (4) UA; (5) Xan.
tion limits are presented in Table 1. Determination limits are
evaluated on the basis of a signal-to-noise ratio of 3. The calibration curves exhibit satisfactory linear behavior over two
orders of magnitude with the detection limit ranging from
Y. Guan et al. / J. Chromatogr. B 821 (2005) 229234
233
Table 1
The results of regression analysis on calibration curves and the detection limitsa
Compound
Regression equation Y = aX + bb
Correlation coefficient
Linear range (104 mol/L)
Detection limit (106 mol/L)
HY
PAH
Vc
UA
Xan
Y = 1.04 105
Y = 3.13 104
Y = 1.26 104
Y = 3.86 104
Y = 2.72 104
0.9996
0.9999
0.9999
0.9997
0.9998
0.012
0.012
0.022
0.012
0.012
0.50
0.80
2.00
0.66
0.92
a
b
X + 0.0962
X 0.0177
X 0.0010
X + 0.0655
X 0.0267
CE-ED conditions are the same as in Fig. 3.
In the regression equation, the X value is the concentration of analytes (mol/L) and the Y value is the peak current (nA).
Table 2
Assay results of UA in urine and saliva of healthy volunteers and patients with renal diseasea
Healthy volunteer (mol/L)
Urine
Sample 1
Sample 2
Sample 3
a
b
1.99 103
Patient (mol/L)
Saliva
(96.9%)b
1.93 103 (97.5%)
0.76 103 (98.3%)
Urine
9.60 105
103
(94.2%)
9.25 105 (90.2%)
8.80 105 (95.3%)
6.39
(97.2%)
5.10 103 (97.6%)
4.59 103 (99.1%)
Saliva
3.55 104 (96.3%)
2.42 104 (95.2%)
2.19 104 (93.2%)
CE-ED conditions are the same as in Fig. 3.
The data in parentheses refer to the recovery.
5.01 107 to 2.00 106 mol/L for all five analytes, and
the correlation coefficients are in the range of 0.99960.9999.
To further evaluate the precision and accuracy of the
method, the recovery experiments under the optimum conditions are also conducted with the urine and saliva samples
(n = 3). Accurate amounts of standard were added to the actual samples, and the recovery values can be obtained by
comparing the increase of the peak height before and after
the addition of standards. The average recoveries are listed
in Table 2.
3.5. Application and discussion
HX and Xan are the metabolites of purine bases in nucleic acids through a series of biochemical pathway, and Vc
is also a common compound in human fluids. Furthermore,
HX, Xan and Vc are electroactive compounds which might
interfere the determination of PHA and UA, therefore PAH
and UA were determined at the presence of these three coexisting potential interferences. Under the optimum conditions, CE-ED is employed for the determination of UA and
PAH in human saliva and urine, where the exogenous and
endogenous co-existing compounds produce no interference
for the quantitation. Typical electropherograms for real-life
samples are shown in Fig. 5B and C. By comparing the migration times of analytes with those of the standard mixture
solution (Fig. 5A), UA content in human saliva and urine can
be determined, and PAH content can be determined in human urine, this lack of PAH in saliva may be the result of
levels of PAH below the limit of detection after dilution. The
experimental results of healthy volunteers and patients with
renal disease are presented in Tables 2 and 3, from which we
can readily find the close relativity of UA and PAH levels in
human saliva and urine for both healthy volunteers and renal
Table 3
Assay results of PAH in urine of healthy volunteers and patients with renal
diseasea
Sample 1
Sample 2
Sample 3
a
b
Healthy volunteer (mol/L)
Patient (mol/L)
0.44 104 (97.9%)b
0.37 104 (97.9%)
0.22 104 (98.8%)
2.82 104 (97.3%)
1.02 104 (98.6%)
0.97 104 (98.1%)
CE-ED conditions are the same as in Fig. 3.
The data in parentheses refer to the recovery.
disease patients. The UA and PAH levels in urine samples
collected from renal disease patients is about several times
higher than that collected from healthy volunteers, similarly
the UA levels in saliva samples collected from renal disease
patients is also about several times higher than that collected
from healthy volunteers.
4. Conclusions
We developed a sensitive and reliable method for the simultaneous determination of UA, PAH and their exogenous
and endogenous co-existing ingredients namely HX, Vc and
Xan in human saliva and urine by CE-ED, and especially we
confirmed the close relativity between the levels of UA and
PAH and the renal disease, and developed a fast method for
primary diagnosis of renal disease in clinic.
Acknowledgement
The authors are grateful for the financial supports provided
by the National Nature Science Foundation of China (grant
No. 20375013).
234
Y. Guan et al. / J. Chromatogr. B 821 (2005) 229234
References
[1] J. Clark, The Kidneys, Balancing the Fluids, Torstar Books, New
York, 1985.
[2] P.L. Kimmel, Kidney Int. 59 (2001) 1599.
[3] P.L. Kimmel, K.L. Weihs, R.A. Peterson, J. Am. Soc. Nephrol. 4
(1993) 12.
[4] United States Renal Data System, Annual Data Report, National
Institutes of Health, National Institutes of Diabetes and Digestive
and Kidney Diseases, Bethesda, MD, 1999.
[5] H.W. Smith, The Kidney, Structure and Function in Health and Disease, Oxford University Press, New York, 1951.
[6] Anon, Am. J. Kidney Dis. 32 (Suppl.) (1998) S20.
[7] A. Collins, J.L. Xue, J.Z. Ma, T. Louis, J. Am. Soc. Nephrol. 11
(2000) 133A.
[8] F. Valderrabano, F.C. Berthoux, E.H. Jones, O. Mehls, Nephrol. Dial.
Transplant 11 (Suppl.) (1996) 2.
[9] G. Dryhurst, Electrochemistry of Biological Molecules, Academic
Press, New York, 1977.
[10] J.M. Zen, J.J. Jou, G. Ilangovan, Analyst 123 (1998) 1345.
[11] A. Whelton, A.J. Watson, R.C. Rock, in: C.A. Burtis, E.R. Ashwood (Eds.), Tietz Fundamentals of Clinical Chemistry, Saunders,
Philadelphia, 1996, p. 569.
[12] R.F. Pitts, Physiology of the Kidney and Body Fluids, Year Bool
Medical Publishers, Chicago, 1965.
[13] N. Baccard, G. Hoizey, C. Frances, D. Lamiable, T. Trenque, H.
Millart, Analyst 124 (1999) 833.
[14] G.F. Domagk, H.H. Schlicke, Anal. Biochem. 22 (1968) 219.
[15] X. Liu, W.M. Lin, X.H. Yan, X.H. Chen, J.R. Hoidal, P. Xu, J.
Chromatogr. B 785 (2003) 101.
[16] M. Czauderna, J. Kowalczyk, J. Chromatogr. B 704 (1997) 89.
[17] M. Czauderna, J. Kowalczyk, J. Chromatogr. B 744 (2000) 129.
[18] A. Pastore, S. Bernardini, D.L. Strologo, G. Rizzoni, C. Cortese, G.
Federici, J. Chromatogr. B 751 (2001) 187.
[19] T.C. Dowling, R.F. Frye, M.A. Zemaitis, J. Chromatogr. B 716
(1998) 305.
[20] T. Fujii, S. Kawabe, T. Horike, T. Taguchi, M. Ogata, J. Chromatogr.
B 730 (1999) 41.
[21] M. Pizzichini, L. Arezzini, C. Billarelli, F. Carlucci, L. Terzuoli,
Clin. Biochem. 30 (1997) 274.
[22] E.A. Clark, J.C. Fanguy, C.S. Henry, J. Pharm. Biomed. Anal. 25
(2001) 795.
[23] C.D. Garcia, C.S. Henry, Analyst 129 (2004) 579.
[24] J.C. Fanguy, C.S. Henry, Electrophoresis 23 (2002) 767.
[25] J.L. Boughton, B.W. Robinson, T.G. Strein, Electrophoresis 23
(2002) 3705.
[26] G. Chen, X.H. Ding, Z.G. Chao, J.N. Ye, Anal. Chim. Acta 408
(2000) 249.
[27] G. Chen, J.X. Zhang, J.N. Ye, J. Chromatogr. A 923 (2001) 255.
[28] G. Chen, X.H. Ding, J.N. Ye, Chem. J. Chin. Univ. 21 (2000) 1364.
You might also like
- BCMB 415 Exam 3, Fall 2016 KeyDocument9 pagesBCMB 415 Exam 3, Fall 2016 KeyzzmasterNo ratings yet
- June 17 26 s001 PDFDocument9 pagesJune 17 26 s001 PDFBetul RojeabNo ratings yet
- The Entire Avengers Age of Ultron ScriptDocument51 pagesThe Entire Avengers Age of Ultron ScriptLebron JamesNo ratings yet
- Orac MethodDocument5 pagesOrac MethodPriscillaL.SilvaNo ratings yet
- Plant Tissue Culturelab Practices Made Easy (For Beginners) PDFDocument75 pagesPlant Tissue Culturelab Practices Made Easy (For Beginners) PDFSultan Ahmad Khan100% (1)
- Journal of Electroanalytical ChemistryDocument10 pagesJournal of Electroanalytical ChemistrySelin AktaşNo ratings yet
- Determination of Homogentisic Acid in Urine For Diagnosis of Alcaptonuria: Capillary Electrophoretic Method Optimization Using Experimental DesignDocument6 pagesDetermination of Homogentisic Acid in Urine For Diagnosis of Alcaptonuria: Capillary Electrophoretic Method Optimization Using Experimental DesigncristinaNo ratings yet
- Determination of PKa Values of Benzimidazole Derivatives From Mobility Obtained by Capillary ElectrophoresisDocument5 pagesDetermination of PKa Values of Benzimidazole Derivatives From Mobility Obtained by Capillary ElectrophoresisMario PosavecNo ratings yet
- Simultaneous Monitoring of Glucose and Uric Acid On A Single Test Strip WithDocument5 pagesSimultaneous Monitoring of Glucose and Uric Acid On A Single Test Strip WithMadalina FleancuNo ratings yet
- Selective Simultaneous Determination of Paracetamol and Uric Acid Using A Glassy Carbon Electrode Modified With..Document8 pagesSelective Simultaneous Determination of Paracetamol and Uric Acid Using A Glassy Carbon Electrode Modified With..ÁLVARO CARLOS AGUADO MALLQUINo ratings yet
- Determination of Pharmaceuticals ClassifiedDocument9 pagesDetermination of Pharmaceuticals ClassifiedsorinamotocNo ratings yet
- Using Cysteine-CdSe Nanoparticles For Quantification of Ovalbumin Thermal Denaturation by Cyclic and Differential Pulse VoltammetryDocument13 pagesUsing Cysteine-CdSe Nanoparticles For Quantification of Ovalbumin Thermal Denaturation by Cyclic and Differential Pulse VoltammetryInternational Journal of Academic Scientific ResearchNo ratings yet
- Aguilar 2002 LCRDocument5 pagesAguilar 2002 LCRiplabaNo ratings yet
- UntitledDocument7 pagesUntitledNancy HeshamNo ratings yet
- Baldelli 2004Document7 pagesBaldelli 2004davidlaglaNo ratings yet
- Protein Releasing Kinetics of Bakers' Yeast Cells by UltrasoundDocument6 pagesProtein Releasing Kinetics of Bakers' Yeast Cells by UltrasoundjainashahNo ratings yet
- Zhang 12Document11 pagesZhang 12Ayu Miftachul Jan'ahNo ratings yet
- Artikel Fito..Document10 pagesArtikel Fito..Nurul FuadyNo ratings yet
- Determination of Aniline and Its Derivatives in Environmental Water by Capillary Electrophoresis With On-Line ConcentrationDocument10 pagesDetermination of Aniline and Its Derivatives in Environmental Water by Capillary Electrophoresis With On-Line ConcentrationkareemNo ratings yet
- Uapcr DiagnosticDocument8 pagesUapcr DiagnosticYA MAAPNo ratings yet
- Sensors and Actuators B: Chemical: Jin Wang, Hiroaki Suzuki, Takaaki SatakeDocument5 pagesSensors and Actuators B: Chemical: Jin Wang, Hiroaki Suzuki, Takaaki SatakeMELISSA VERONICA TUDELANO OSPINANo ratings yet
- 2012-Biomedical Chromatography-AppelDocument7 pages2012-Biomedical Chromatography-AppelHaojie ZhuNo ratings yet
- Automated In-Tube Solid Phase Microextraction Coupled With HPLC-ES-MS For The Determination of Catechins and Caffeine in TeaDocument7 pagesAutomated In-Tube Solid Phase Microextraction Coupled With HPLC-ES-MS For The Determination of Catechins and Caffeine in TeaSeema YadavNo ratings yet
- Dialysis DoseDocument7 pagesDialysis DoseFotaf FolaNo ratings yet
- Turnell 1982Document5 pagesTurnell 1982Ammar MaryamabadiNo ratings yet
- Metabolomic Profiling of Amoebic and Pyogenic Liver Abscesses An in Vitro NMR StudyDocument16 pagesMetabolomic Profiling of Amoebic and Pyogenic Liver Abscesses An in Vitro NMR Studysantosh091283No ratings yet
- Analytica Chimica Acta: Taher Alizadeh, Mohammad Reza Ganjali, Faride Ra FieiDocument9 pagesAnalytica Chimica Acta: Taher Alizadeh, Mohammad Reza Ganjali, Faride Ra FieiwardaninurindahNo ratings yet
- Cold Atmospheric Plasma Discharged in Water and Its Potential Use in Cancer TherapyDocument18 pagesCold Atmospheric Plasma Discharged in Water and Its Potential Use in Cancer TherapySek PyroNo ratings yet
- Improving LC-MS Sensitivity Through Increases in Chromatographic Performance: Comparisons of UPLC-ES/MS/MS To Hplc-Es/Ms/MsDocument10 pagesImproving LC-MS Sensitivity Through Increases in Chromatographic Performance: Comparisons of UPLC-ES/MS/MS To Hplc-Es/Ms/MsgannysunNo ratings yet
- Tavana 2012Document6 pagesTavana 2012selia destianingrumNo ratings yet
- Jurnal RingerfundinDocument7 pagesJurnal RingerfundinRyan Arifin SuryantoNo ratings yet
- Piao 2008Document5 pagesPiao 2008Ellie satrianiNo ratings yet
- Scala Benuzzi2018Document10 pagesScala Benuzzi2018mohammad aidilNo ratings yet
- A Simple 96-Well Liquid-Liquid Extraction With A Mixture ofDocument10 pagesA Simple 96-Well Liquid-Liquid Extraction With A Mixture ofSowbhagya LaxmiNo ratings yet
- Nanocomposite Modified Electrochemical Sensor For Sensitive and Selective Determination of NoradrenalineDocument10 pagesNanocomposite Modified Electrochemical Sensor For Sensitive and Selective Determination of NoradrenalineLeau Sorina-AlexandraNo ratings yet
- 10 1016@j Colsurfa 2014 05 029Document7 pages10 1016@j Colsurfa 2014 05 029wora123potNo ratings yet
- Ultrasensitive Label Free Electrical Detection of Insulin in Neat BloodDocument6 pagesUltrasensitive Label Free Electrical Detection of Insulin in Neat BloodDusan SilniNo ratings yet
- Removal MikroorganismDocument9 pagesRemoval MikroorganismUkiNo ratings yet
- Effect of He Self-Organized Pattern Plasma-Activated Media With Different Conductivity On Cancer CellsDocument23 pagesEffect of He Self-Organized Pattern Plasma-Activated Media With Different Conductivity On Cancer CellsSek PyroNo ratings yet
- Molecularly Imprinted Electrochemical Sensing of Urinary Melatonin in A Microfluidic SystemDocument11 pagesMolecularly Imprinted Electrochemical Sensing of Urinary Melatonin in A Microfluidic SystemgpaivNo ratings yet
- Square Wave Adsorptive Cathodic Stripping Voltammetry Automated by Sequential Injection Analysis Potentialities and Limitations Exemplified by The Determination of Methyl Parathion in Water SamplesDocument8 pagesSquare Wave Adsorptive Cathodic Stripping Voltammetry Automated by Sequential Injection Analysis Potentialities and Limitations Exemplified by The Determination of Methyl Parathion in Water SamplesSHERLY KIMBERLY RAMOS JESUSNo ratings yet
- 9-Aryl Substituted Hydroxylated Xanthen-3-Ones: Synthesis, Structure and Antioxidant Potency EvaluationDocument7 pages9-Aryl Substituted Hydroxylated Xanthen-3-Ones: Synthesis, Structure and Antioxidant Potency EvaluationUmar IjazNo ratings yet
- Online Extraction LC-MSMS Method For The Simultaneous Quantitative...Document11 pagesOnline Extraction LC-MSMS Method For The Simultaneous Quantitative...qketzalNo ratings yet
- Biosensors: Single-Drop Analysis of Epinephrine and Uric Acid On A Screen-Printed Carbon ElectrodeDocument17 pagesBiosensors: Single-Drop Analysis of Epinephrine and Uric Acid On A Screen-Printed Carbon Electrodewidia waatiNo ratings yet
- Pnas-A Fully Integrated Microfluidic Genetic Analysis SystemDocument6 pagesPnas-A Fully Integrated Microfluidic Genetic Analysis Systemapi-3723328No ratings yet
- Single Determination of A-Ketoglutaric Acid and Pyruvic Acid in Beer by HPLC With UV DetectionDocument6 pagesSingle Determination of A-Ketoglutaric Acid and Pyruvic Acid in Beer by HPLC With UV DetectionPatrícia MontenegroNo ratings yet
- Analisis de PQ by LC-MSDocument5 pagesAnalisis de PQ by LC-MSalina esther diaz arrietaNo ratings yet
- Litopenaeus Vannamei (Boone, 1931) Closed-Hatchery System: Application of An Electrolytic Water Treatment Technique in ADocument10 pagesLitopenaeus Vannamei (Boone, 1931) Closed-Hatchery System: Application of An Electrolytic Water Treatment Technique in AHiral PatelNo ratings yet
- Effect of Zeolite Types LTX and Lta On Physicochemical Parameters of Drinking Water Samples in Ghana, Assisted by Light Transmission ExperiDocument7 pagesEffect of Zeolite Types LTX and Lta On Physicochemical Parameters of Drinking Water Samples in Ghana, Assisted by Light Transmission ExperiesatjournalsNo ratings yet
- 2010 - Recent Advances of Capillary Electrophoresis in Pharmaceutical AnalysisDocument24 pages2010 - Recent Advances of Capillary Electrophoresis in Pharmaceutical AnalysisStefana SzántóNo ratings yet
- Sobre Ação Do EDC NHSDocument6 pagesSobre Ação Do EDC NHSAlberto Galdino SparrowNo ratings yet
- Method 1611 Enterococci in Water by TaqMan Quantitative Polymerase Chain Reaction (QPCR) AssayDocument56 pagesMethod 1611 Enterococci in Water by TaqMan Quantitative Polymerase Chain Reaction (QPCR) AssayJBNo ratings yet
- In Vitro and in Vivo Trials of Wearable Artificial KidneyDocument5 pagesIn Vitro and in Vivo Trials of Wearable Artificial KidneyMIGUEL ANGEL PALOMINO CAMPONo ratings yet
- Determination of Amoxicillin in Plasma Samples by Capillary ElectrophoresisDocument7 pagesDetermination of Amoxicillin in Plasma Samples by Capillary ElectrophoresisJose Manuel Rito GonzalezNo ratings yet
- Caarvedilol Electrochimic - Grupa 3 PDFDocument4 pagesCaarvedilol Electrochimic - Grupa 3 PDFdanielafarmacie_1617No ratings yet
- Darroudi 2022 J. Electrochem. Soc. 169 026501Document7 pagesDarroudi 2022 J. Electrochem. Soc. 169 026501abdellahbe41No ratings yet
- Estudo Hepasfera Versus BeadDocument6 pagesEstudo Hepasfera Versus BeadAlexandre Campos Moraes AmatoNo ratings yet
- Molecules 19 18923Document13 pagesMolecules 19 18923dikadika_tansNo ratings yet
- Electron Spin Resonance SpectrosDocument6 pagesElectron Spin Resonance SpectrossakuraleeshaoranNo ratings yet
- Use of A Blood Gas Analyzer and A Laboratory Autoanalyzer in Routine Practice To Measure Electrolytes in Intensive Care Unit PatientsDocument7 pagesUse of A Blood Gas Analyzer and A Laboratory Autoanalyzer in Routine Practice To Measure Electrolytes in Intensive Care Unit PatientsZheng GuNo ratings yet
- Electrochemical Determination of Uric Acid in Human Urine Using Nickel Hexa-Cyano Ferrate Modified Carbon Paste ElectrodeDocument18 pagesElectrochemical Determination of Uric Acid in Human Urine Using Nickel Hexa-Cyano Ferrate Modified Carbon Paste ElectrodeMohammad MiyanNo ratings yet
- Development and Evaluation of A Bio Ion Measureme - 2013 - Journal of AcupuncturDocument9 pagesDevelopment and Evaluation of A Bio Ion Measureme - 2013 - Journal of AcupuncturMatias DamiánNo ratings yet
- Application of Real-Time Quantitative PCR For The Detection of SelectedDocument9 pagesApplication of Real-Time Quantitative PCR For The Detection of SelectedMiriam LeiNo ratings yet
- Urinary Stones: Medical and Surgical ManagementFrom EverandUrinary Stones: Medical and Surgical ManagementMichael GrassoNo ratings yet
- 10th Science Maharashtra Board Animal Classification IntroDocument12 pages10th Science Maharashtra Board Animal Classification IntroAditya BorateNo ratings yet
- Human EyeDocument7 pagesHuman Eyeofmir154No ratings yet
- AO No. 156 S. 1971 AO No. 291 S. 1976 Section 7 Section 7Document5 pagesAO No. 156 S. 1971 AO No. 291 S. 1976 Section 7 Section 7Clark RamosNo ratings yet
- Abdominal ParacentesisDocument14 pagesAbdominal ParacentesisManisha Thakur100% (1)
- A Cost Optimization-Based Design of Precast Concrete Floors Using Genetic AlgorithmsDocument9 pagesA Cost Optimization-Based Design of Precast Concrete Floors Using Genetic AlgorithmsRajesh RanjanNo ratings yet
- Autogenics Therapy For Dynamic Relaxation and Stress ReliefDocument17 pagesAutogenics Therapy For Dynamic Relaxation and Stress ReliefNickRhodos100% (2)
- Priming and Pretreatment of Seeds and SeedlingsDocument606 pagesPriming and Pretreatment of Seeds and SeedlingsFadhillah ArsaniNo ratings yet
- Brain Plasticity - How Experience Changes The BrainDocument5 pagesBrain Plasticity - How Experience Changes The BrainWilson OTONo ratings yet
- Microsoft Word - Biology - Genetics UnitDocument20 pagesMicrosoft Word - Biology - Genetics Unitapi-233894872No ratings yet
- ENDO) Pancreas. For The ClassDocument58 pagesENDO) Pancreas. For The ClassromaloiNo ratings yet
- iBT TOEFL 托福词汇 词以类记Document15 pagesiBT TOEFL 托福词汇 词以类记beat2013No ratings yet
- Bioinformatic Tools For Next Generation DNA Sequencing - PHD ThesisDocument237 pagesBioinformatic Tools For Next Generation DNA Sequencing - PHD ThesisDan PintilescuNo ratings yet
- Minerva Anestesiol 2017 GruenewaldDocument15 pagesMinerva Anestesiol 2017 GruenewaldMarjorie Lisseth Calderón LozanoNo ratings yet
- MCB 7200: Molecular BiologyDocument27 pagesMCB 7200: Molecular BiologytasniyanNo ratings yet
- Longing To BelongDocument6 pagesLonging To BelongKhalid KhalilNo ratings yet
- Structure of Chromosomes, Cell Cycle, Cell DivisionDocument35 pagesStructure of Chromosomes, Cell Cycle, Cell DivisionXyzNo ratings yet
- Laboratory Diagnosis of Leptospirosis: A Challenge: Didier Musso, Bernard La ScolaDocument8 pagesLaboratory Diagnosis of Leptospirosis: A Challenge: Didier Musso, Bernard La ScolaAnna Diaz YuberoNo ratings yet
- FDS Component 3 PDFDocument307 pagesFDS Component 3 PDFmarielNo ratings yet
- Psychotria ElataDocument2 pagesPsychotria ElataJevon ChengNo ratings yet
- Land Suitability AnalysisDocument79 pagesLand Suitability AnalysisVasilică-Dănuţ HorodnicNo ratings yet
- Poem Book 2012 PDFDocument89 pagesPoem Book 2012 PDFAnshNo ratings yet
- Childhood and Growing UpDocument27 pagesChildhood and Growing UpjikooNo ratings yet
- What Is A Pirate'S Favorite Amino Acid? Arginine. ArgDocument1 pageWhat Is A Pirate'S Favorite Amino Acid? Arginine. ArgSimona PetranNo ratings yet
- Gits Systems Anaphy DisordersDocument23 pagesGits Systems Anaphy DisordersIlawNo ratings yet
- Alprazolam TMaxDocument14 pagesAlprazolam TMaxErwin FernándezNo ratings yet
- Media Preparation SterilizationDocument13 pagesMedia Preparation SterilizationJessica GutierrezNo ratings yet