Professional Documents
Culture Documents
Ampiginosa 1
Ampiginosa 1
Uploaded by
Juan BianchiCopyright:
Available Formats
You might also like
- PCCN PrepDocument133 pagesPCCN Prepdaniel100% (2)
- Clinical Atlas of Muscle and Musculocutaneous FlapsDocument741 pagesClinical Atlas of Muscle and Musculocutaneous FlapsVlad TaralaNo ratings yet
- 2014 Article 33Document5 pages2014 Article 33SusPa NarahaNo ratings yet
- Diagnosis and Treatment of Bacterial Meningitis: Arch. Dis. ChildDocument7 pagesDiagnosis and Treatment of Bacterial Meningitis: Arch. Dis. Childapi-214563632No ratings yet
- Clinical Profile of Tubercular Uveitis in A Tertiary Care Ophthalmic CentreDocument3 pagesClinical Profile of Tubercular Uveitis in A Tertiary Care Ophthalmic CentreArif BudimanNo ratings yet
- Syphilitic Uveitis - EyeWikiDocument5 pagesSyphilitic Uveitis - EyeWikitgrrwccj98No ratings yet
- Zoster Sine HerpeteDocument7 pagesZoster Sine HerpeteLalo RCNo ratings yet
- 2019 Glomerulonefritis Pos-Inf Pedi Clin NADocument14 pages2019 Glomerulonefritis Pos-Inf Pedi Clin NA0511821012 KATHERIN YULEIMA CAMACHO MARIN ESTUDIANTE ACTIVONo ratings yet
- Post Streptococus GNDocument14 pagesPost Streptococus GNRahma Cita HalidaNo ratings yet
- Investigations Related To Uveitis: D - M. U SDocument48 pagesInvestigations Related To Uveitis: D - M. U SUsman Sadiq100% (1)
- A 45-Year-Old Man With Shortness of Breath, Cough, and Fever BackgroundDocument7 pagesA 45-Year-Old Man With Shortness of Breath, Cough, and Fever BackgroundFluffyyy BabyyyNo ratings yet
- Contoh Lapsus DR Aries PDFDocument6 pagesContoh Lapsus DR Aries PDFshe laNo ratings yet
- Postinfectious Glomerulonephritis With Crescents in An Elderly Diabetic Patient After Acute Gastroenteritis: Case ReportDocument8 pagesPostinfectious Glomerulonephritis With Crescents in An Elderly Diabetic Patient After Acute Gastroenteritis: Case ReportNabil Sangga BuanaNo ratings yet
- Postinfectious Glomerulonephritis With Crescents in An Elderly Diabetic Patient After Acute Gastroenteritis: Case ReportDocument8 pagesPostinfectious Glomerulonephritis With Crescents in An Elderly Diabetic Patient After Acute Gastroenteritis: Case ReportNabil Sangga BuanaNo ratings yet
- Gnaps EmedicineDocument13 pagesGnaps Emedicineharyanti lupitaNo ratings yet
- Case Study 1: Group 1Document27 pagesCase Study 1: Group 1Jennylen TorresNo ratings yet
- Correlation of Clinical and Laboratory Parameters of Acute Glomerulonephritis in ChildrenDocument8 pagesCorrelation of Clinical and Laboratory Parameters of Acute Glomerulonephritis in ChildrenResi OfzanNo ratings yet
- Conjunctivitis As A Manifestation of Wegener 'S GranulomatosisDocument5 pagesConjunctivitis As A Manifestation of Wegener 'S GranulomatosisAsd DsaNo ratings yet
- 2016 Article 310Document6 pages2016 Article 310Gracie ShemaNo ratings yet
- Journal of Clinical and Diagnostic ResearchDocument3 pagesJournal of Clinical and Diagnostic Researchmutya yulindaNo ratings yet
- AdultOI CryptococcosisDocument18 pagesAdultOI CryptococcosisricardoatejassNo ratings yet
- Diagnostic and Prognostic Role of Procalcitonin in CAPDocument5 pagesDiagnostic and Prognostic Role of Procalcitonin in CAPKarito FloresNo ratings yet
- Jaad O: NlineDocument2 pagesJaad O: NlinepunishNo ratings yet
- 18-Acute Dacryoadenitis (Partab Rai) 71-76Document6 pages18-Acute Dacryoadenitis (Partab Rai) 71-76Yuni PurwatiNo ratings yet
- Imp bn1Document5 pagesImp bn1Karito FloresNo ratings yet
- Ecthyma Gangrenosum - StatPearls - NCBI BookshelfDocument5 pagesEcthyma Gangrenosum - StatPearls - NCBI BookshelfSyafira Laila NurulitaNo ratings yet
- Diagnostic Approach To Ocular ToxoplasmosisDocument7 pagesDiagnostic Approach To Ocular ToxoplasmosisQoshrina AdenitaNo ratings yet
- Our Experience With Bicytopenia in Patients Treated at The Ankara Hospital Pediatric ClinicDocument5 pagesOur Experience With Bicytopenia in Patients Treated at The Ankara Hospital Pediatric ClinicRonny DoankNo ratings yet
- Severe Cytomegalovirus Infection in An Immunocompetent Host Potentially Related To Flood Water ExposureDocument6 pagesSevere Cytomegalovirus Infection in An Immunocompetent Host Potentially Related To Flood Water ExposureEtnoNo ratings yet
- Ophthal Notes PGDocument6 pagesOphthal Notes PGSwati TyagiNo ratings yet
- Case Report Epididymal Tuberculosis Abscess in Patient Immunocompetent (2021)Document2 pagesCase Report Epididymal Tuberculosis Abscess in Patient Immunocompetent (2021)Nghi VõNo ratings yet
- Ophthalmology 2008 115:292 - 297 © 2008 by The American Academy of OphthalmologyDocument3 pagesOphthalmology 2008 115:292 - 297 © 2008 by The American Academy of OphthalmologySamuel Cruz ValdrramaNo ratings yet
- Clinico-Epidemiological Profile of Childhood Cutaneous TuberculosisDocument7 pagesClinico-Epidemiological Profile of Childhood Cutaneous TuberculosisFajar SatriaNo ratings yet
- USMLE Step 3 Answers To ARCHER Q Bank 1-25Document15 pagesUSMLE Step 3 Answers To ARCHER Q Bank 1-25rashmiajNo ratings yet
- Systemic Disorders Associated With Peripheral Corneal UlcerationDocument4 pagesSystemic Disorders Associated With Peripheral Corneal UlcerationYoga KharismaNo ratings yet
- Moro Lago2017Document6 pagesMoro Lago2017Yamel MartínezNo ratings yet
- Jurding Mata EndophthalamitisDocument24 pagesJurding Mata EndophthalamitisBenk Setsuna F. SeieiNo ratings yet
- TBR PCPDocument28 pagesTBR PCPAyahnya Rafli100% (1)
- 6 - Infectious DiseasesDocument35 pages6 - Infectious DiseasesTalaat OmranNo ratings yet
- 10 1016@j Jaad 2012 12 513Document1 page10 1016@j Jaad 2012 12 513Kuro ChanNo ratings yet
- Blau Int ReviewDocument9 pagesBlau Int ReviewAslıhan Yılmaz ÇebiNo ratings yet
- 1 s2.0 S2468124519301184 MainDocument1 page1 s2.0 S2468124519301184 Maindewi sumbangNo ratings yet
- Duttamajumder 2017Document10 pagesDuttamajumder 2017YanJuniorNo ratings yet
- Case ReportDocument5 pagesCase ReportFerdina NidyasariNo ratings yet
- Cde 0006 0119Document5 pagesCde 0006 0119Mati AjaNo ratings yet
- Mode of Presentation TBDocument7 pagesMode of Presentation TBSohaib Abbas MalikNo ratings yet
- Aspiracion EspinalDocument6 pagesAspiracion EspinalYuriko Castro SanchezNo ratings yet
- Herpes Zoster Ophthalmicus: Limits Advanced Journal List Help Journal List West J Emerg Med v.9 (3) 2008 AugDocument7 pagesHerpes Zoster Ophthalmicus: Limits Advanced Journal List Help Journal List West J Emerg Med v.9 (3) 2008 AugGio Vano NaihonamNo ratings yet
- Elizabeth King I ADocument3 pagesElizabeth King I AAlba UrbinaNo ratings yet
- Typhoid Blood Test ReportDocument3 pagesTyphoid Blood Test ReportPranay BhosaleNo ratings yet
- Clinmed International Library CMRCR 2 045Document2 pagesClinmed International Library CMRCR 2 045Ivan RisnaNo ratings yet
- Serum Procalcitonin Levels in Bacterial and Abacterial MeningitisDocument5 pagesSerum Procalcitonin Levels in Bacterial and Abacterial MeningitisSaul Gonzalez HernandezNo ratings yet
- Dr. Rina - CMV RetinitisDocument32 pagesDr. Rina - CMV RetinitisRonni HandokoNo ratings yet
- Jceh 12 30 021Document3 pagesJceh 12 30 021Tatik HandayaniNo ratings yet
- Clinical OncologyDocument8 pagesClinical OncologyHector VillaseñorNo ratings yet
- Photo QuizDocument21 pagesPhoto Quizabas_maytham1021100% (4)
- Reflective Journal ReadingDocument13 pagesReflective Journal Readingعبداللة العمراني الحويطيNo ratings yet
- Herpes Ophthalmicus: Review: ZosterDocument4 pagesHerpes Ophthalmicus: Review: ZosterAndika Wahyu UtomoNo ratings yet
- Journal Raf SurgicalDocument4 pagesJournal Raf SurgicalRAFNo ratings yet
- Direct Immmuno Flourescence Study in Auto Immune Bullous DisordersDocument3 pagesDirect Immmuno Flourescence Study in Auto Immune Bullous DisordersInternational Organization of Scientific Research (IOSR)No ratings yet
- 1quarterly Journal of Medicine, 1993Document8 pages1quarterly Journal of Medicine, 1993Maha OmarNo ratings yet
- Biophysical Properties in Glaucoma: Diagnostic TechnologiesFrom EverandBiophysical Properties in Glaucoma: Diagnostic TechnologiesIngrida JanulevicieneNo ratings yet
- Pediatric X RaysDocument88 pagesPediatric X RaysprinceejNo ratings yet
- Orthopedics BoneDisordersDocument20 pagesOrthopedics BoneDisordersannapanna1No ratings yet
- Stroke - OT EB Stroke InterventionsDocument14 pagesStroke - OT EB Stroke InterventionsVanessa CarmodyNo ratings yet
- Creation or Evolution?: Comparison and ContrastDocument12 pagesCreation or Evolution?: Comparison and ContrastJeanne Marie ValleNo ratings yet
- ClaireHadfield VictimImpactStatementDocument3 pagesClaireHadfield VictimImpactStatementClaire Hadfield100% (1)
- Velopharyngeal DefectsDocument66 pagesVelopharyngeal DefectsVivek ShankarNo ratings yet
- Daflon Addition in Treatment Coventional MedicalDocument8 pagesDaflon Addition in Treatment Coventional MedicalTha Vila Le ColloNo ratings yet
- Chapter 019Document13 pagesChapter 019api-281340024100% (1)
- List of Herbal Industries in Karnataka: #49/12 1st Floor, 31st Cross, IV T-BlockDocument25 pagesList of Herbal Industries in Karnataka: #49/12 1st Floor, 31st Cross, IV T-BlockDr. ChannabasavaNo ratings yet
- WordRoot MedicalAbbreviationsDocument9 pagesWordRoot MedicalAbbreviationsJouriel Kristoffer GriarteNo ratings yet
- CHAPTER 8 Student With Physical DisabilitiesDocument17 pagesCHAPTER 8 Student With Physical DisabilitiesEarl Joseph TrinidadNo ratings yet
- The Role of Nurse Practitioners in Care Coordination and TransitionDocument10 pagesThe Role of Nurse Practitioners in Care Coordination and Transitionapi-312364579No ratings yet
- Meralgia ParestheticaDocument22 pagesMeralgia ParestheticaWahyu Tri KusprasetyoNo ratings yet
- Case Report Rehabilitation Program in A Patient With Bilateral Arthroplasty For Hip OsteoarthritisDocument26 pagesCase Report Rehabilitation Program in A Patient With Bilateral Arthroplasty For Hip OsteoarthritisNatalia LoredanaNo ratings yet
- Graduate Syllabus For Genetic EngineeringDocument3 pagesGraduate Syllabus For Genetic EngineeringAnuj SharmaNo ratings yet
- Introduction To Social Skills TrainingDocument9 pagesIntroduction To Social Skills TrainingNabiha100% (1)
- Joy To Live EbookDocument417 pagesJoy To Live Ebookmarkuswolf11No ratings yet
- ChemotherapyDocument102 pagesChemotherapyJoseph John K Pothanikat100% (1)
- Ebook Psychiatric Drugs Explained 7Th PDF Full Chapter PDFDocument67 pagesEbook Psychiatric Drugs Explained 7Th PDF Full Chapter PDFandrew.weidert647100% (37)
- Practice Bulletin Summary: Multifetal Gestations: Twin, Triplet, and Higher-Order Multifetal PregnanciesDocument3 pagesPractice Bulletin Summary: Multifetal Gestations: Twin, Triplet, and Higher-Order Multifetal PregnancieselmerNo ratings yet
- Kundu - Pearls in Medicine For Students@helpmedico PDFDocument303 pagesKundu - Pearls in Medicine For Students@helpmedico PDFSwarnadeep Mani100% (1)
- What Is Hypertensive NephropathyDocument11 pagesWhat Is Hypertensive NephropathyIvy Pamanian DeldaNo ratings yet
- PleuritisDocument32 pagesPleuritisBinot's TogamaNo ratings yet
- List of Janseva Aushadh MedicinesDocument16 pagesList of Janseva Aushadh MedicinesShrinivasan ParthasarathyNo ratings yet
- 6 Weeks Observership ProgrammeDocument20 pages6 Weeks Observership ProgrammeAbdelrahman GalalNo ratings yet
- Kako Napisati I Publikovati Naucni RadDocument14 pagesKako Napisati I Publikovati Naucni RadJovan Z. PopovićNo ratings yet
- BOD Sample Dilution Memo 2015Document2 pagesBOD Sample Dilution Memo 2015Mohamed SalemNo ratings yet
- Applications of HPLC in Clinical DiagnosticsDocument2 pagesApplications of HPLC in Clinical Diagnosticsmanan259050% (2)
Ampiginosa 1
Ampiginosa 1
Uploaded by
Juan BianchiOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Ampiginosa 1
Ampiginosa 1
Uploaded by
Juan BianchiCopyright:
Available Formats
Case Rep Ophthalmol 2015;6:453457
DOI: 10.1159/000442742
Published online: December 12, 2015
2015 The Author(s)
Published by S. Karger AG, Basel
16632699/15/00630453$39.50/0
www.karger.com/cop
This article is licensed under the Creative Commons Attribution-NonCommercial 4.0
International License (CC BY-NC) (http://www.karger.com/Services/OpenAccessLicense).
Usage and distribution for commercial purposes requires written permission.
A Case of Ampiginous Choroiditis
Pieter Lambrecht
Marnix Claeys
Ilse De Schryver
Department of Ophthalmology, Ghent University Hospital, Ghent, Belgium
Key Words
Ampiginous choroiditis Relentless placoid chorioretinitis Primary inflammatory
choriocapillaropathy
Abstract
Purpose: Presentation of a case report of a unilateral ampiginous choroiditis. Methods: This
is an observational case report. Results: A 70-year-old woman was referred to us with unilateral scattered chorioretinal lesions. The multifocal pattern of the lesions and the angiographic features led to the diagnosis of ampiginous choroiditis. Conclusion: Ampiginous choroiditis is a primary inflammatory choriocapillaropathy with characteristics of both acute posterior
multifocal placoid pigment epitheliopathy and serpiginous choroiditis.
2015 The Author(s)
Published by S. Karger AG, Basel
Case Report
This case report is about a 70-year-old Caucasian woman who is in good general health
although she has medically controlled arterial hypertension and is overweight. The patient
has a 3-dpt hyperopic refraction and there are no important past general or ophthalmologic
antecedents in the patients history. The patient developed progressive painful visual blurring in the right eye during an episode of severe sinusitis. Consultation with her local ophthalmologist showed anterior chamber inflammation, macular subretinal fluid with cystoid
macular oedema (fig. 1) and scattered whitish lesions in the fundus of the right eye. Ophthalmologic examination of the left eye was unremarkable. Anterior chamber inflammation
disappeared after treatment with topical steroids, after which the patient was referred to us
because of manifestation of new lesions and evolution of the scattered chorioretinal lesions
towards atrophy.
Pieter Lambrecht
Department of Ophthalmology, Ghent University Hospital
De Pintelaan 185
BE9000 Ghent (Belgium)
E-Mail pieter.lambrecht@uzgent.be
Case Rep Ophthalmol 2015;6:453457
DOI: 10.1159/000442742
2015 The Author(s). Published by S. Karger AG, Basel
www.karger.com/cop
Lambrecht et al.: A Case of Ampiginous Choroiditis
Consultation at our hospital showed a best-corrected visual acuity of 20/40 for the right
and 20/20 for the left eye. Slit lamp biomicroscopy showed bilateral nuclear sclerosis but no
endothelial precipitates or posterior synechiae. Eye pressure was 12 and 14 mm Hg in the
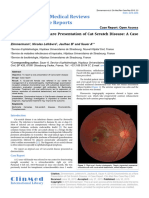
right and left eye, respectively. Fundus examination of the right eye showed a peripapillary
lesion with pseudopodial extensions and multiple yellow-greyish irregularly bordered lesions (fig. 2a). Fluorescein angiography demonstrated these lesions to be early hypofluorescent with late hyperfluorescent staining of the borders (fig. 2b). Indocyanine green (ICG)
angiography demonstrated these lesions to be early and late hypofluorescent (fig. 2c). Blue
light autofluorescence showed these lesions to be hypoautofluorescent with some being
mildly granular hyperautofluorescent (fig. 2d). An OCT scan of the macula showed residual
macular oedema. An OCT scan through these lesions displayed outer retinal atrophy. Ophthalmologic examination of the left eye was unremarkable.
Workup at her local ophthalmologist and with us showed that antinuclear factor, rheumatoid factor, angiotensin-converting enzyme, anti-proteinase 3 and anti-myeloperoxidase,
calcemia and syphilis serology were all normal. There was no history of previous exposure
to tuberculosis. A tuberculin skin test and chest radiography were both normal. There were
no systemic signs or symptoms that were suggestive for a diagnosis of sarcoidosis. The multifocal pattern and the angiographic features led us to the diagnosis of ampiginous choroiditis. An intravitreal injection with triamcinolone acetonide was given as a treatment for the
macular oedema with resolution of the macular oedema as a consequence (fig. 3). We started
immunomodulatory treatment with azathioprine 2 50 mg/day to prevent recurrence of
inflammation. After 4 months, we decided to stop therapy with azathioprine since it was not
well tolerated by the patient. At the time of writing this case report, the patient has been
followed for more than 1 year. During this period, visual acuity has remained stable and no
appearance of new lesions or enlargement of the known lesions has occurred.
Discussion
We present a case report of unilateral uveitis in a healthy 70-year-old woman. Visual
symptoms and eye pain developed during an episode of sinusitis. Consultation with her local
ophthalmologist showed scattered retinal lesions reminiscent of acute posterior multifocal
placoid pigment epitheliopathy (APMPPE). These lesions, however, evolved to outer retinal
atrophy with angiographic characteristics reminiscent of serpiginous choroiditis. The combination of characteristics of both APMPPE and serpiginous choroiditis led to the diagnosis
of ampiginous choroiditis [1]. A similar disease entity has been called relentless placoid chorioretinitis [2]. Diagnostic criteria used by Jyotirmay et al. [3] help to delineate this disease.
The clinical profile of 16 patients with ampiginous choroiditis showed a male preponderance
(7:3), a median age of 34 years, 81% bilateral involvement and 35% suffered from recurrences [3].
APMPPE and serpiginous choroiditis are both classified as a primary inflammatory choriocapillaropathy [4]. Inflammation at the level of the choriocapillaris leads to areas of choriocapillaris non- or hypoperfusion with ischaemic consequences both at the level of the
choroid and at the level of the outer retina. Rarely, an intermediary form between these two
diseases, i.e. ampiginous choroiditis, can be seen, but the reason why some patients develop
a blend of both diseases is unknown. It has been proposed that the behaviour of each type of
primary inflammatory choriocapillaropathy has to do with the level and severity of vascular
damage to the choriocapillaris [4]. An argument used in favour of this proposition is the fact
that in a large series of serpiginous choroiditis, some lesions had initial features of APMPPE
454
Case Rep Ophthalmol 2015;6:453457
DOI: 10.1159/000442742
2015 The Author(s). Published by S. Karger AG, Basel
www.karger.com/cop
Lambrecht et al.: A Case of Ampiginous Choroiditis
but showed progression towards serpiginous choroiditis during follow-up [5]. The aetiology
of serpiginous choroiditis is unclear, with degenerative, autoimmune and/or infectious processes considered [6]. The aetiology of ampiginous choroiditis, which is considered by some
to be a subclass of serpiginous choroiditis, is likewise unclear, with some speculating it to be
an ocular manifestation of a larger systemic autoimmune condition [3]. A case of ampiginous
choroiditis following a human papillomavirus vaccine was reported, although a causal relationship is not sure [7]. An ampiginous-like case report showed positive serology in blood
and polymerase chain reaction (PCR) in aqueous humour for Toxoplasma gondii infection.
That patient also had a tuberculin skin test with 15 mm of induration, but negative aqueous
humour PCR for Mycobacterium tuberculosis [8]. Our patients workup could not identify an
underlying cause for this disease, making the origin of this disease unclear. Since our patient
developed symptoms during an episode of sinusitis, it is tempting to suspect an infectious
trigger as a cause.
An important differential diagnostic of ampiginous choroiditis is multifocal serpiginoid
choroiditis caused by Mycobacterium tuberculosis, Treponema pallidum or a herpesvirus [6].
Syphilis was excluded as a cause since syphilis serology was negative. Mycobacterium tuberculosis infection is unlikely since there was no history of previous exposure to tuberculosis
and since a tuberculin skin test and chest radiography were both negative [9]. Multifocal
lesions, unilaterality and considerable vitreous or anterior chamber inflammation, as in our
patient, point towards a diagnosis of tuberculosis multifocal serpiginoid choroiditis instead
of a serpiginous choroiditis [6]. Peripapillary involvement from the early phases of the disease, as in our patient, is, however, not typical for a tuberculosis multifocal serpiginoid choroiditis [6]. We did not perform an anterior chamber tap to do a PCR for Mycobacterium tuberculosis or herpesviruses since the patient did not consult us during active inflammation,
making a positive PCR result more unlikely. Two other important differential diagnoses are
sarcoidosis and granulomatosis with polyangiitis, formerly known as Wegeners granulomatosis. The patient had no systemic manifestations that were suggestive for sarcoidosis.
Workup with serum angiotensin-converting enzyme, serum calcium and a chest radiograph
also showed no arguments for sarcoidosis. Granulomatosis with polyangiitis is unlikely since
the patient did not have systemic manifestations suggestive of this disease and since antiproteinase 3 and anti-myeloperoxidase testing were normal.
We chose to treat the macular oedema with an intravitreal injection with triamcinolone
acetonide. We started immunomodulatory therapy with azathioprine to prevent recurrences, but after 4 months, this medication was stopped since the patient did not tolerate it well.
After consulting the patient, we decided not to start another immunomodulatory agent because there are indications that only one third of patients suffer relapses [3]. After having
followed up this patient for more than 1 year, we have not seen an evolution in number or
size of retinal lesions. The absence of diffuse choroidal hyperfluorescence with ICG angiography at the edges of the lesions indicates no progression of the lesions [4]. Since some of
these lesions still have a hyperautofluorescent aspect, we do suspect some smouldering residual activity in these lesions. We plan to follow up our patient closely and in the event of
any development of activity of an ampiginous lesion towards the macula, we will start systemic therapy with corticosteroids and immunomodulatory therapy.
455
Case Rep Ophthalmol 2015;6:453457
DOI: 10.1159/000442742
2015 The Author(s). Published by S. Karger AG, Basel
www.karger.com/cop
Lambrecht et al.: A Case of Ampiginous Choroiditis
Statement of Ethics
The patient has given voluntary written informed consent. During treatment, the health
of the patient was our first consideration and we have acted in the patients best interest
when providing medical care.
Disclosure Statement
The authors declare no conflicts of interest. There are no financial or nonfinancial interests/relationships that may be interpreted to have influenced the manuscript.
References
1
2
3
4
5
6
7
8
9
Nussenblatt RB: Serpiginous choroidopathy; in Nussenblatt RB, Whitcup SM (eds): Uveitis, ed 4. Edinburgh,
Mosby, 2010, pp 373382.
Jones BE, Jampol LM, Yannuzzi LA, Tittl M, Johnson MW, Han DP, Davis JL, Williams DF: Relentless placoid
chorioretinitis: a new entity or an unusual variant of serpiginous chorioretinitis? Arch Ophthalmol
2000;118:931938.
Jyotirmay B, Jafferji SS, Sudharshan S, Kalpana B: Clinical profile, treatment, and visual outcome of
ampiginous choroiditis. Ocul Immunol Inflamm 2010;18:4651.
Cimino L, Mantovani A, Herbort C: Primary inflammatory choriocapillaropathies; in Krieglstein G, Weinreb
R, Pleyer U, Mondino B (eds): Uveitis and Immunological Disorders. Berlin, Springer, 2005, pp 209231.
Gupta V, Agarwal A, Gupta A, Bambery P, Narang S: Clinical characteristics of serpiginous choroidopathy in
North India. Am J Ophthalmol 2002;134:4756.
Nazari Khanamiri H, Rao NA: Serpiginous choroiditis and infectious multifocal serpiginoid choroiditis. Surv
Ophthalmol 2013;58:203232.
Khalifa YM, Monahan PM, Acharya NR: Ampiginous choroiditis following quadrivalent human papilloma
virus vaccine. Br J Ophthalmol 2010;94:137139.
Mahendradas P, Kamath G, Mahalakshmi B, Shetty KB: Serpiginous choroiditis-like picture due to ocular
toxoplasmosis. Ocul Immunol Inflamm 2007;15:127130.
Vasconcelos-Santos DV, Zierhut M, Rao NA: Strengths and weaknesses of diagnostic tools for tuberculous
uveitis. Ocul Immunol Inflamm 2009;17:351355.
Fig. 1. OCT scan of the macula of the right eye at the time of presentation.
456
Case Rep Ophthalmol 2015;6:453457
DOI: 10.1159/000442742
2015 The Author(s). Published by S. Karger AG, Basel
www.karger.com/cop
Lambrecht et al.: A Case of Ampiginous Choroiditis
Fig. 2. Fundus pictures of the right eye at the time of referral. a Colour picture. b Fluorescein angiography.
c ICG angiography. d Blue light autofluorescence.
Fig. 3. OCT scan of the macula of the right eye after treatment with intravitreal triamcinolone acetonide.
457
You might also like
- PCCN PrepDocument133 pagesPCCN Prepdaniel100% (2)
- Clinical Atlas of Muscle and Musculocutaneous FlapsDocument741 pagesClinical Atlas of Muscle and Musculocutaneous FlapsVlad TaralaNo ratings yet
- 2014 Article 33Document5 pages2014 Article 33SusPa NarahaNo ratings yet
- Diagnosis and Treatment of Bacterial Meningitis: Arch. Dis. ChildDocument7 pagesDiagnosis and Treatment of Bacterial Meningitis: Arch. Dis. Childapi-214563632No ratings yet
- Clinical Profile of Tubercular Uveitis in A Tertiary Care Ophthalmic CentreDocument3 pagesClinical Profile of Tubercular Uveitis in A Tertiary Care Ophthalmic CentreArif BudimanNo ratings yet
- Syphilitic Uveitis - EyeWikiDocument5 pagesSyphilitic Uveitis - EyeWikitgrrwccj98No ratings yet
- Zoster Sine HerpeteDocument7 pagesZoster Sine HerpeteLalo RCNo ratings yet
- 2019 Glomerulonefritis Pos-Inf Pedi Clin NADocument14 pages2019 Glomerulonefritis Pos-Inf Pedi Clin NA0511821012 KATHERIN YULEIMA CAMACHO MARIN ESTUDIANTE ACTIVONo ratings yet
- Post Streptococus GNDocument14 pagesPost Streptococus GNRahma Cita HalidaNo ratings yet
- Investigations Related To Uveitis: D - M. U SDocument48 pagesInvestigations Related To Uveitis: D - M. U SUsman Sadiq100% (1)
- A 45-Year-Old Man With Shortness of Breath, Cough, and Fever BackgroundDocument7 pagesA 45-Year-Old Man With Shortness of Breath, Cough, and Fever BackgroundFluffyyy BabyyyNo ratings yet
- Contoh Lapsus DR Aries PDFDocument6 pagesContoh Lapsus DR Aries PDFshe laNo ratings yet
- Postinfectious Glomerulonephritis With Crescents in An Elderly Diabetic Patient After Acute Gastroenteritis: Case ReportDocument8 pagesPostinfectious Glomerulonephritis With Crescents in An Elderly Diabetic Patient After Acute Gastroenteritis: Case ReportNabil Sangga BuanaNo ratings yet
- Postinfectious Glomerulonephritis With Crescents in An Elderly Diabetic Patient After Acute Gastroenteritis: Case ReportDocument8 pagesPostinfectious Glomerulonephritis With Crescents in An Elderly Diabetic Patient After Acute Gastroenteritis: Case ReportNabil Sangga BuanaNo ratings yet
- Gnaps EmedicineDocument13 pagesGnaps Emedicineharyanti lupitaNo ratings yet
- Case Study 1: Group 1Document27 pagesCase Study 1: Group 1Jennylen TorresNo ratings yet
- Correlation of Clinical and Laboratory Parameters of Acute Glomerulonephritis in ChildrenDocument8 pagesCorrelation of Clinical and Laboratory Parameters of Acute Glomerulonephritis in ChildrenResi OfzanNo ratings yet
- Conjunctivitis As A Manifestation of Wegener 'S GranulomatosisDocument5 pagesConjunctivitis As A Manifestation of Wegener 'S GranulomatosisAsd DsaNo ratings yet
- 2016 Article 310Document6 pages2016 Article 310Gracie ShemaNo ratings yet
- Journal of Clinical and Diagnostic ResearchDocument3 pagesJournal of Clinical and Diagnostic Researchmutya yulindaNo ratings yet
- AdultOI CryptococcosisDocument18 pagesAdultOI CryptococcosisricardoatejassNo ratings yet
- Diagnostic and Prognostic Role of Procalcitonin in CAPDocument5 pagesDiagnostic and Prognostic Role of Procalcitonin in CAPKarito FloresNo ratings yet
- Jaad O: NlineDocument2 pagesJaad O: NlinepunishNo ratings yet
- 18-Acute Dacryoadenitis (Partab Rai) 71-76Document6 pages18-Acute Dacryoadenitis (Partab Rai) 71-76Yuni PurwatiNo ratings yet
- Imp bn1Document5 pagesImp bn1Karito FloresNo ratings yet
- Ecthyma Gangrenosum - StatPearls - NCBI BookshelfDocument5 pagesEcthyma Gangrenosum - StatPearls - NCBI BookshelfSyafira Laila NurulitaNo ratings yet
- Diagnostic Approach To Ocular ToxoplasmosisDocument7 pagesDiagnostic Approach To Ocular ToxoplasmosisQoshrina AdenitaNo ratings yet
- Our Experience With Bicytopenia in Patients Treated at The Ankara Hospital Pediatric ClinicDocument5 pagesOur Experience With Bicytopenia in Patients Treated at The Ankara Hospital Pediatric ClinicRonny DoankNo ratings yet
- Severe Cytomegalovirus Infection in An Immunocompetent Host Potentially Related To Flood Water ExposureDocument6 pagesSevere Cytomegalovirus Infection in An Immunocompetent Host Potentially Related To Flood Water ExposureEtnoNo ratings yet
- Ophthal Notes PGDocument6 pagesOphthal Notes PGSwati TyagiNo ratings yet
- Case Report Epididymal Tuberculosis Abscess in Patient Immunocompetent (2021)Document2 pagesCase Report Epididymal Tuberculosis Abscess in Patient Immunocompetent (2021)Nghi VõNo ratings yet
- Ophthalmology 2008 115:292 - 297 © 2008 by The American Academy of OphthalmologyDocument3 pagesOphthalmology 2008 115:292 - 297 © 2008 by The American Academy of OphthalmologySamuel Cruz ValdrramaNo ratings yet
- Clinico-Epidemiological Profile of Childhood Cutaneous TuberculosisDocument7 pagesClinico-Epidemiological Profile of Childhood Cutaneous TuberculosisFajar SatriaNo ratings yet
- USMLE Step 3 Answers To ARCHER Q Bank 1-25Document15 pagesUSMLE Step 3 Answers To ARCHER Q Bank 1-25rashmiajNo ratings yet
- Systemic Disorders Associated With Peripheral Corneal UlcerationDocument4 pagesSystemic Disorders Associated With Peripheral Corneal UlcerationYoga KharismaNo ratings yet
- Moro Lago2017Document6 pagesMoro Lago2017Yamel MartínezNo ratings yet
- Jurding Mata EndophthalamitisDocument24 pagesJurding Mata EndophthalamitisBenk Setsuna F. SeieiNo ratings yet
- TBR PCPDocument28 pagesTBR PCPAyahnya Rafli100% (1)
- 6 - Infectious DiseasesDocument35 pages6 - Infectious DiseasesTalaat OmranNo ratings yet
- 10 1016@j Jaad 2012 12 513Document1 page10 1016@j Jaad 2012 12 513Kuro ChanNo ratings yet
- Blau Int ReviewDocument9 pagesBlau Int ReviewAslıhan Yılmaz ÇebiNo ratings yet
- 1 s2.0 S2468124519301184 MainDocument1 page1 s2.0 S2468124519301184 Maindewi sumbangNo ratings yet
- Duttamajumder 2017Document10 pagesDuttamajumder 2017YanJuniorNo ratings yet
- Case ReportDocument5 pagesCase ReportFerdina NidyasariNo ratings yet
- Cde 0006 0119Document5 pagesCde 0006 0119Mati AjaNo ratings yet
- Mode of Presentation TBDocument7 pagesMode of Presentation TBSohaib Abbas MalikNo ratings yet
- Aspiracion EspinalDocument6 pagesAspiracion EspinalYuriko Castro SanchezNo ratings yet
- Herpes Zoster Ophthalmicus: Limits Advanced Journal List Help Journal List West J Emerg Med v.9 (3) 2008 AugDocument7 pagesHerpes Zoster Ophthalmicus: Limits Advanced Journal List Help Journal List West J Emerg Med v.9 (3) 2008 AugGio Vano NaihonamNo ratings yet
- Elizabeth King I ADocument3 pagesElizabeth King I AAlba UrbinaNo ratings yet
- Typhoid Blood Test ReportDocument3 pagesTyphoid Blood Test ReportPranay BhosaleNo ratings yet
- Clinmed International Library CMRCR 2 045Document2 pagesClinmed International Library CMRCR 2 045Ivan RisnaNo ratings yet
- Serum Procalcitonin Levels in Bacterial and Abacterial MeningitisDocument5 pagesSerum Procalcitonin Levels in Bacterial and Abacterial MeningitisSaul Gonzalez HernandezNo ratings yet
- Dr. Rina - CMV RetinitisDocument32 pagesDr. Rina - CMV RetinitisRonni HandokoNo ratings yet
- Jceh 12 30 021Document3 pagesJceh 12 30 021Tatik HandayaniNo ratings yet
- Clinical OncologyDocument8 pagesClinical OncologyHector VillaseñorNo ratings yet
- Photo QuizDocument21 pagesPhoto Quizabas_maytham1021100% (4)
- Reflective Journal ReadingDocument13 pagesReflective Journal Readingعبداللة العمراني الحويطيNo ratings yet
- Herpes Ophthalmicus: Review: ZosterDocument4 pagesHerpes Ophthalmicus: Review: ZosterAndika Wahyu UtomoNo ratings yet
- Journal Raf SurgicalDocument4 pagesJournal Raf SurgicalRAFNo ratings yet
- Direct Immmuno Flourescence Study in Auto Immune Bullous DisordersDocument3 pagesDirect Immmuno Flourescence Study in Auto Immune Bullous DisordersInternational Organization of Scientific Research (IOSR)No ratings yet
- 1quarterly Journal of Medicine, 1993Document8 pages1quarterly Journal of Medicine, 1993Maha OmarNo ratings yet
- Biophysical Properties in Glaucoma: Diagnostic TechnologiesFrom EverandBiophysical Properties in Glaucoma: Diagnostic TechnologiesIngrida JanulevicieneNo ratings yet
- Pediatric X RaysDocument88 pagesPediatric X RaysprinceejNo ratings yet
- Orthopedics BoneDisordersDocument20 pagesOrthopedics BoneDisordersannapanna1No ratings yet
- Stroke - OT EB Stroke InterventionsDocument14 pagesStroke - OT EB Stroke InterventionsVanessa CarmodyNo ratings yet
- Creation or Evolution?: Comparison and ContrastDocument12 pagesCreation or Evolution?: Comparison and ContrastJeanne Marie ValleNo ratings yet
- ClaireHadfield VictimImpactStatementDocument3 pagesClaireHadfield VictimImpactStatementClaire Hadfield100% (1)
- Velopharyngeal DefectsDocument66 pagesVelopharyngeal DefectsVivek ShankarNo ratings yet
- Daflon Addition in Treatment Coventional MedicalDocument8 pagesDaflon Addition in Treatment Coventional MedicalTha Vila Le ColloNo ratings yet
- Chapter 019Document13 pagesChapter 019api-281340024100% (1)
- List of Herbal Industries in Karnataka: #49/12 1st Floor, 31st Cross, IV T-BlockDocument25 pagesList of Herbal Industries in Karnataka: #49/12 1st Floor, 31st Cross, IV T-BlockDr. ChannabasavaNo ratings yet
- WordRoot MedicalAbbreviationsDocument9 pagesWordRoot MedicalAbbreviationsJouriel Kristoffer GriarteNo ratings yet
- CHAPTER 8 Student With Physical DisabilitiesDocument17 pagesCHAPTER 8 Student With Physical DisabilitiesEarl Joseph TrinidadNo ratings yet
- The Role of Nurse Practitioners in Care Coordination and TransitionDocument10 pagesThe Role of Nurse Practitioners in Care Coordination and Transitionapi-312364579No ratings yet
- Meralgia ParestheticaDocument22 pagesMeralgia ParestheticaWahyu Tri KusprasetyoNo ratings yet
- Case Report Rehabilitation Program in A Patient With Bilateral Arthroplasty For Hip OsteoarthritisDocument26 pagesCase Report Rehabilitation Program in A Patient With Bilateral Arthroplasty For Hip OsteoarthritisNatalia LoredanaNo ratings yet
- Graduate Syllabus For Genetic EngineeringDocument3 pagesGraduate Syllabus For Genetic EngineeringAnuj SharmaNo ratings yet
- Introduction To Social Skills TrainingDocument9 pagesIntroduction To Social Skills TrainingNabiha100% (1)
- Joy To Live EbookDocument417 pagesJoy To Live Ebookmarkuswolf11No ratings yet
- ChemotherapyDocument102 pagesChemotherapyJoseph John K Pothanikat100% (1)
- Ebook Psychiatric Drugs Explained 7Th PDF Full Chapter PDFDocument67 pagesEbook Psychiatric Drugs Explained 7Th PDF Full Chapter PDFandrew.weidert647100% (37)
- Practice Bulletin Summary: Multifetal Gestations: Twin, Triplet, and Higher-Order Multifetal PregnanciesDocument3 pagesPractice Bulletin Summary: Multifetal Gestations: Twin, Triplet, and Higher-Order Multifetal PregnancieselmerNo ratings yet
- Kundu - Pearls in Medicine For Students@helpmedico PDFDocument303 pagesKundu - Pearls in Medicine For Students@helpmedico PDFSwarnadeep Mani100% (1)
- What Is Hypertensive NephropathyDocument11 pagesWhat Is Hypertensive NephropathyIvy Pamanian DeldaNo ratings yet
- PleuritisDocument32 pagesPleuritisBinot's TogamaNo ratings yet
- List of Janseva Aushadh MedicinesDocument16 pagesList of Janseva Aushadh MedicinesShrinivasan ParthasarathyNo ratings yet
- 6 Weeks Observership ProgrammeDocument20 pages6 Weeks Observership ProgrammeAbdelrahman GalalNo ratings yet
- Kako Napisati I Publikovati Naucni RadDocument14 pagesKako Napisati I Publikovati Naucni RadJovan Z. PopovićNo ratings yet
- BOD Sample Dilution Memo 2015Document2 pagesBOD Sample Dilution Memo 2015Mohamed SalemNo ratings yet
- Applications of HPLC in Clinical DiagnosticsDocument2 pagesApplications of HPLC in Clinical Diagnosticsmanan259050% (2)