Professional Documents
Culture Documents
Standard Enthalpy of Formation
Standard Enthalpy of Formation
Uploaded by
shiel175Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Standard Enthalpy of Formation
Standard Enthalpy of Formation
Uploaded by
shiel175Copyright:
Available Formats
8/23/2016
Standard Enthalpy of Formation - Chemistry LibreTexts
If you like us, please share us on social media or tell your professor!
Build or adopta LibreText for your course! Prof. Dianne Bennett from Sacramento City College shows you how, and
Prof. Delmar Larsen from UC Davis demonstrates.
Chemistry
Medicine
Biology
Geology
Mathematics
Statistics
Agriculture
Physics
Social Sciences
Engineering
PhotoSciences
| Periodic Table of the Elements | Reference Tables | Physical Constants | Units and Conversions | Organic
Chemistry
Glossary
|
Standard Enthalpy of Formation
Last updated: 09:06, 13 Jul 2016
Home
Core
Physical Chemistry
Expand/collapse
global
hierarchy
Thermodynamics
State Functions
Share
Table of contents
The standard enthalpy of formation is de ned as the change in enthalpy when one mole of a substance in the
standard state (1 atm of pressure and 298.15 K)is formed from its pure elements under the same conditions.
Introduction
The standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance
is created under standard conditions from its pure elements. The symbol of the standard enthalpy of formation is
Hf.
= A change in enthalpy
o = A degree signi
es that it's a standard enthalpy change.
f = The f indicates that the substance is formed from its elements
The equation for the standard enthalpy change of formation (originating from Enthalpy's being a State Function),
shown below, is commonly used:
H
reaction
= H
(products) H
(Reactants)
http://chem.libretexts.org/Core/Physical_Chemistry/Thermodynamics/State_Functions/Enthalpy/Standard_Enthalpy_Of_Formation
(1.1)
1/5
8/23/2016
Standard Enthalpy of Formation - Chemistry LibreTexts
This equationessentially states that the standard enthalpy change of formation is equal to the sum of the standard
enthalpies of formation of the products minus thesum of the standard enthalpies of formation of the reactants.
Example 1
Given a simple chemical equation with the variables A, B and C representing different compounds:
A + B C
andthe standard enthalpy of formation values:
Hfo[A] = 433 KJ/mol
Hfo[B] = -256 KJ/mol
Hfo[C] = 523 KJ/mol
the equation for the standard enthalpy change of formation is as follows:
Hreactiono = Hfo[C] - (Hfo[A] + Hfo[B])
Hreactiono = (1 mol)(523 kJ/mol) - ((1 mol)(433 kJ/mol) + (1 mol)(-256 kJ/mol)\)
Because there is one mole each of A, B and C, the standard enthalpy of formation of each reactant and product is
multiplied by 1 mole, which eliminates the mol denominator:
Hreactiono = 346 kJ
Howis346
can we
help you?
The result
kJ, which
is the standard enthalpy change of formation for the creation of variable "C".
Sign
in
Note
The standard enthalpy of formation of a pure element is in its reference form its standard enthalpy formation is
zero.
Carbon naturally exists as graphite and diamond. The enthalpy difference between graphite and diamond is too
large for both to have a standard enthalpy of formation of zero. To determine which form is zero, the more stable
form of carbon is chosen. This is also the form with the lowest enthalpy, so graphite has a standard enthalpy of
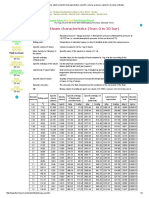
formation equal to zero. Table 1 provides sample values of standard enthalpies of formation of various compounds.
Table 1: Sample Table of Standard Enthalpy of Formation
Values. Table T1 is a more comprehensive table.
Compound
Hf o
O2(g)
0 kJ/mol
C(graphite)
0 kJ/mol
CO(g)
-110.5 kJ/mol
CO2(g)
-393.5 kJ/mol
H2(g)
0 kJ/mol
H2O(g)
-241.8 kJ/mol
http://chem.libretexts.org/Core/Physical_Chemistry/Thermodynamics/State_Functions/Enthalpy/Standard_Enthalpy_Of_Formation
2/5
8/23/2016
Standard Enthalpy of Formation - Chemistry LibreTexts
HF(g)
-271.1 kJ/mol
NO(g)
90.25 kJ/mol
NO2(g)
33.18 kJ/mol
N2O4(g)
9.16 kJ/mol
SO2(g)
-296.8 kJ/mol
SO3(g)
-395.7 kJ/mol
All values have units of kJ/mol and physical conditions of 298.15 K and 1 atm, referred to as the "standard state."
These are the conditions under which values of standard enthalpies of formation are typically given. Note that while
the majority of the values of standard enthalpies of formation are exothermic, or negative, there are a few
compounds such as NO(g) and N2O4(g) that actually require energy from its surroundings during its formation;
these endothermic compounds are generally unstable.
Example 2
Between Br2(l) and Br2(g) at 298.15 K, which substance has a nonzero standard enthalpy of formation?
SOLUTION
Br2(l) is the more stable form, which means it has the lower enthalpy; thus, Br2(l) has Hf = 0. Consequently,Br2(g)
has a nonzero standard enthalpy of formation.
Note: that the element phosphorus is a unique case. The reference form in phosphorus is not the most stable form,
red phosphorus, but the less stable form, white phosphorus.
Recall thatstandard enthalpies of formation can be either positive or negative.
Example 3
The enthalpy of formation of carbon dioxide at 298.15K is Hf = -393.5 kJ/mol CO2(g). Write the chemical equation
for the formation of CO2.
SOLUTION
This equation must be writtenfor one mole of CO2(g). In this case, the reference forms of the constituent elements
are O2(g) and graphite for carbon.
O2 (g) + C(graphite) C O2 (g)
(1.2)
The general equation for the standard enthalpy change of formation is given below:
H
reaction
= H
(products) H
(Reactants)
(1.3)
Plugging in theequation for the formation of CO2gives the following:
o
o
o
o
http://chem.libretexts.org/Core/Physical_Chemistry/Thermodynamics/State_Functions/Enthalpy/Standard_Enthalpy_Of_Formation
3/5
8/23/2016
Standard Enthalpy of Formation - Chemistry LibreTexts
Hreactiono= Hfo[CO2(g)] - (Hfo[O2(g)] + Hfo[C(graphite)]
Because O2(g) and C(graphite) are in their most elementally stable forms, they each have a standard enthalpy of
formation equal to 0:
Hreactiono= -393.5 kJ = Hfo[CO2(g)] - ((1 mol)(0 kJ/mol) + (1 mol)(0 kJ/mol))
Hfo[CO2(g)]= -393.5 kJ
Example 4
Using the values in the above table of standard enthalpies of formation, calculate the Hreactiono for the formation of
NO2(g).
SOLUTION
N O2(g)
is formed from the combination of N O
and O
(g)
2(g)
in the following reaction:
2N O(g) + O2 (g) 2N O2 (g)
To nd the Hreactiono,use the formula for the standard enthalpy change of formation:
H
reaction
= H
(products) H
(Reactants)
(1.4)
The relevantstandard enthalpy of formation valuesfrom Table 1 are:
O2(g): 0 kJ/mol
NO(g): 90.25 kJ/mol
NO2(g): 33.18 kJ/mol
Plugging these values into the formula above gives the following:
H
reaction
= (2 mol )(33.18 kJ / mol ) [(2 mol )(90.25kJ / mol ) + (1 mol )(0 kJ / mol )]
reaction
= 114.1 kJ
(1.5)
(1.6)
References
1. Pisanty, Alejandro. "The electronic structure of graphite: A chemist's introduction to band theory." J. Chem. Educ.
1991, 68, 804.
2. Petrucci, et al. General Chemistry: Principles & Modern Applications. 9th ed. Upper Saddle River, New Jersey
2007.
Contributors
Jonathan Nguyen (UCD), Garrett Larimer (UCD)
Back to top
http://chem.libretexts.org/Core/Physical_Chemistry/Thermodynamics/State_Functions/Enthalpy/Standard_Enthalpy_Of_Formation
4/5
8/23/2016
Standard Enthalpy of Formation - Chemistry LibreTexts
Simple Measurement o...
Entropy
Recommended articles
Heat of Vaporization
Heat of Fusion
Heat of Reaction
Heat of Sublimation
Entropy
Article type:
topic
Tags:
Fundamental, Vet1
Copyright 2016 Chemistry LibreTexts
Powered by MindTouch
Unless otherwise noted, the LibreTexts library is licensed under a Creative Commons
Attribution-Noncommercial-Share Alike 3.0 United States License. Permissions beyond the scope of this license
may be available at copyright@ucdavis.edu.
The LibreTexts library isPowered by MindTouch
http://chem.libretexts.org/Core/Physical_Chemistry/Thermodynamics/State_Functions/Enthalpy/Standard_Enthalpy_Of_Formation
5/5
You might also like
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeFrom EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeRating: 4 out of 5 stars4/5 (5820)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreFrom EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreRating: 4 out of 5 stars4/5 (1093)
- Never Split the Difference: Negotiating As If Your Life Depended On ItFrom EverandNever Split the Difference: Negotiating As If Your Life Depended On ItRating: 4.5 out of 5 stars4.5/5 (845)
- Grit: The Power of Passion and PerseveranceFrom EverandGrit: The Power of Passion and PerseveranceRating: 4 out of 5 stars4/5 (590)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceFrom EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceRating: 4 out of 5 stars4/5 (898)
- Shoe Dog: A Memoir by the Creator of NikeFrom EverandShoe Dog: A Memoir by the Creator of NikeRating: 4.5 out of 5 stars4.5/5 (540)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersFrom EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersRating: 4.5 out of 5 stars4.5/5 (349)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureFrom EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureRating: 4.5 out of 5 stars4.5/5 (474)
- Her Body and Other Parties: StoriesFrom EverandHer Body and Other Parties: StoriesRating: 4 out of 5 stars4/5 (822)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)From EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Rating: 4.5 out of 5 stars4.5/5 (122)
- The Emperor of All Maladies: A Biography of CancerFrom EverandThe Emperor of All Maladies: A Biography of CancerRating: 4.5 out of 5 stars4.5/5 (271)
- The Little Book of Hygge: Danish Secrets to Happy LivingFrom EverandThe Little Book of Hygge: Danish Secrets to Happy LivingRating: 3.5 out of 5 stars3.5/5 (401)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyFrom EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyRating: 3.5 out of 5 stars3.5/5 (2259)
- The Yellow House: A Memoir (2019 National Book Award Winner)From EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Rating: 4 out of 5 stars4/5 (98)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaFrom EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaRating: 4.5 out of 5 stars4.5/5 (266)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryFrom EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryRating: 3.5 out of 5 stars3.5/5 (231)
- Team of Rivals: The Political Genius of Abraham LincolnFrom EverandTeam of Rivals: The Political Genius of Abraham LincolnRating: 4.5 out of 5 stars4.5/5 (234)
- On Fire: The (Burning) Case for a Green New DealFrom EverandOn Fire: The (Burning) Case for a Green New DealRating: 4 out of 5 stars4/5 (74)
- The Unwinding: An Inner History of the New AmericaFrom EverandThe Unwinding: An Inner History of the New AmericaRating: 4 out of 5 stars4/5 (45)
- 25 Unit Test Acid and Base Answer KeyDocument2 pages25 Unit Test Acid and Base Answer KeyMyrna AppalNo ratings yet
- Industrial Cleaning Source BookDocument58 pagesIndustrial Cleaning Source Bookshiel175No ratings yet
- StyreneDocument8 pagesStyreneshiel175No ratings yet
- Steam Heating Processes - Load CalculatingDocument5 pagesSteam Heating Processes - Load Calculatingshiel175No ratings yet
- Glycerol (Data Page) : From Wikipedia, The Free EncyclopediaDocument6 pagesGlycerol (Data Page) : From Wikipedia, The Free Encyclopediashiel175No ratings yet
- Overall Heat Transfer Coefficient - TLV - A Steam Specialist Company (International)Document4 pagesOverall Heat Transfer Coefficient - TLV - A Steam Specialist Company (International)shiel175No ratings yet
- Properties of Steam and Water PDFDocument6 pagesProperties of Steam and Water PDFshiel175No ratings yet
- Calculate Volume of A Hollow Cylinder and Its Surface AreaDocument4 pagesCalculate Volume of A Hollow Cylinder and Its Surface Areashiel175No ratings yet
- Glycerol (Data Page) : From Wikipedia, The Free EncyclopediaDocument6 pagesGlycerol (Data Page) : From Wikipedia, The Free Encyclopediashiel175No ratings yet
- Steam Heating Processes - Load CalculatingDocument5 pagesSteam Heating Processes - Load Calculatingshiel175No ratings yet
- Reactor TypesDocument81 pagesReactor Typesshiel175No ratings yet
- Transesterification - Chemistry LibreTextsDocument5 pagesTransesterification - Chemistry LibreTextsshiel175No ratings yet
- Coconut Methyl Ester As An Alternative FuelDocument8 pagesCoconut Methyl Ester As An Alternative Fuelshiel175No ratings yet
- Hess's Law and Enthalpy Change CalculationsDocument8 pagesHess's Law and Enthalpy Change Calculationsshiel175No ratings yet
- Steam, Vapor, Density, Latent, Sensible Heat Vaporization, Specific, Volume, Pressure, Dynamic Viscosity, EnthalpyDocument2 pagesSteam, Vapor, Density, Latent, Sensible Heat Vaporization, Specific, Volume, Pressure, Dynamic Viscosity, Enthalpyshiel175No ratings yet
- Boiling Water, Density, Specific Enthalpy, Heat, Dynamic Viscosity, Vaporization, Volume Heat 2Document3 pagesBoiling Water, Density, Specific Enthalpy, Heat, Dynamic Viscosity, Vaporization, Volume Heat 2shiel175No ratings yet
- Enthalpy - Chemistry LibreTextsDocument5 pagesEnthalpy - Chemistry LibreTextsshiel175No ratings yet
- Heat (Enthalpy) of ReactionDocument4 pagesHeat (Enthalpy) of Reactionshiel175No ratings yet
- Shell and Tube Exchangers - General Design ConsiderationsDocument2 pagesShell and Tube Exchangers - General Design Considerationsshiel175100% (1)
- Chemical Kinetics TutorialDocument2 pagesChemical Kinetics TutorialFormer TorrentNo ratings yet
- Market Research For Linear Alkylbenzene Sulfonic AcidDocument9 pagesMarket Research For Linear Alkylbenzene Sulfonic Acidoba.ajjNo ratings yet
- Corrosion of Materials and Its Prevention: Dr. Abdul ShakoorDocument40 pagesCorrosion of Materials and Its Prevention: Dr. Abdul ShakoorAbdulNo ratings yet
- Haselrieder 2015Document8 pagesHaselrieder 2015Daiana Medone AcostaNo ratings yet
- Crescent PPG Lining Pvt. LTD: We Define Trust...... ... RelaxDocument29 pagesCrescent PPG Lining Pvt. LTD: We Define Trust...... ... RelaxKamatchi NathanNo ratings yet
- Msds-Calcium - Hypochlorite - 65% (Enochlor)Document10 pagesMsds-Calcium - Hypochlorite - 65% (Enochlor)tito rahman100% (1)
- 13 Carbohydrates Part I 2023Document33 pages13 Carbohydrates Part I 2023Kurstein FerriolNo ratings yet
- Biocontamination Control Techniques For Purified Water SystemDocument4 pagesBiocontamination Control Techniques For Purified Water SystemAli KureishiNo ratings yet
- CLS Aipmt-18-19 XI Che Study-Package-1 SET-2 Chapter-1Document30 pagesCLS Aipmt-18-19 XI Che Study-Package-1 SET-2 Chapter-1parvez khanNo ratings yet
- NLC Intern Dec-JanDocument39 pagesNLC Intern Dec-JanSurya SuryaNo ratings yet
- Battery Recycling Technologies Recycling Waste LitDocument16 pagesBattery Recycling Technologies Recycling Waste LitYong JiangNo ratings yet
- Chapter 5Document41 pagesChapter 5Eshita SharmaNo ratings yet
- Formate Brine TableDocument12 pagesFormate Brine Tables v poyil100% (1)
- Annexure IV Report On Hydrogen ProductionDocument224 pagesAnnexure IV Report On Hydrogen ProductionPtl BhaiyuNo ratings yet
- FireDocument2 pagesFireYi Long MuskNo ratings yet
- Student Exploration: Physical and Chemical Changes of MatterDocument5 pagesStudent Exploration: Physical and Chemical Changes of MatterTopherNo ratings yet
- A Review On Nanomaterial Based Humidity SensingDocument16 pagesA Review On Nanomaterial Based Humidity SensingParth GargNo ratings yet
- Class - X Science MCQ at 2021-22 (Answer Key)Document4 pagesClass - X Science MCQ at 2021-22 (Answer Key)Navyaa ChandraNo ratings yet
- Polymer Grade 12Document107 pagesPolymer Grade 12Hetal ShahNo ratings yet
- Cork Properties Capabilities and ApplicationsDocument22 pagesCork Properties Capabilities and ApplicationsVijay AnandNo ratings yet
- Zener DiodeDocument20 pagesZener DiodeMaizatul Hanisah RoziNo ratings yet
- Chemistry Sulphuric Acid SolutionsDocument7 pagesChemistry Sulphuric Acid SolutionsGurjapsingh SandhuNo ratings yet
- Fischer 2013Document7 pagesFischer 2013sukanyagggNo ratings yet
- Essay On Hydrogen TechnologyDocument22 pagesEssay On Hydrogen TechnologyBruno EggertNo ratings yet
- Acid, Base, and SaltDocument12 pagesAcid, Base, and SaltTamoghna DeyNo ratings yet
- Co2 Capture PHD ThesisDocument5 pagesCo2 Capture PHD Thesisnibaditapalmerpaterson100% (2)
- IGCSE Chemistry Lecture 16 - Group 1 ElementsDocument12 pagesIGCSE Chemistry Lecture 16 - Group 1 Elementsnazzlor08No ratings yet
- 0009 d9 SB Zinc Silicate Pds March 08Document2 pages0009 d9 SB Zinc Silicate Pds March 08Prashant MalveNo ratings yet
- Annona muricata Linn. leaf as a source of antioxidant compounds with in vitro antidiabetic and inhibitory potential against α-amylase, α-glucosidase, lipase, non-enzymatic glycation and lipid peroxidationDocument10 pagesAnnona muricata Linn. leaf as a source of antioxidant compounds with in vitro antidiabetic and inhibitory potential against α-amylase, α-glucosidase, lipase, non-enzymatic glycation and lipid peroxidationDanny. JayNo ratings yet