Professional Documents
Culture Documents
tmpFE04 TMP
tmpFE04 TMP
Uploaded by
FrontiersCopyright:
Available Formats
You might also like
- Sample Midterm 2 F303Document12 pagesSample Midterm 2 F303philmoreNo ratings yet
- AP CALCULUS BC - MC - Related RatesDocument4 pagesAP CALCULUS BC - MC - Related RatesShannon BurtonNo ratings yet
- Salicylic Acid Signalling in Plants: Molecular SciencesDocument6 pagesSalicylic Acid Signalling in Plants: Molecular SciencesKanishk KannaNo ratings yet
- Identification of Cell-Wall Stress As A Hexose-Dependent and Osmosensitive Regulator of Plant ResponsesDocument12 pagesIdentification of Cell-Wall Stress As A Hexose-Dependent and Osmosensitive Regulator of Plant ResponsesQuenguan FranklinNo ratings yet
- Salicylic Acid and Photosynthesis: Signalling and EffectsDocument31 pagesSalicylic Acid and Photosynthesis: Signalling and EffectsKanishk KannaNo ratings yet
- tmp1013 TMPDocument6 pagestmp1013 TMPFrontiersNo ratings yet
- Plant Hormone-Mediated Regulation of Stress Responses: Review Open AccessDocument10 pagesPlant Hormone-Mediated Regulation of Stress Responses: Review Open AccessEe EENo ratings yet
- Shetty Chamberry AntioxidantsDocument14 pagesShetty Chamberry AntioxidantsrosanelineliegeNo ratings yet
- 2017 Article 9685Document30 pages2017 Article 9685Mohamed HafezNo ratings yet
- Sanguinarine PDFDocument9 pagesSanguinarine PDFSara Yahia cherifNo ratings yet
- 2011 Dempsey SA-biosynthesisDocument24 pages2011 Dempsey SA-biosynthesisPriscila MezzomoNo ratings yet
- Regulation of Water, Salinity, and Cold Stress Responses by Salicylic AcidDocument12 pagesRegulation of Water, Salinity, and Cold Stress Responses by Salicylic AcidBastian SeguelNo ratings yet
- Evoluton Yellow FlopppyDocument30 pagesEvoluton Yellow Flopppydrdgrant6055No ratings yet
- Ecotoxicology and Environmental Safety: Xufei Li, Junyi Shi, Haoyu Sun, Zhifen LinDocument11 pagesEcotoxicology and Environmental Safety: Xufei Li, Junyi Shi, Haoyu Sun, Zhifen LinAbdul Wahab Assya RoniNo ratings yet
- Cross-Talk Between Gibberellins and Salicylic Acid in Early Stress Responses in Span Style "Font-StyleDocument3 pagesCross-Talk Between Gibberellins and Salicylic Acid in Early Stress Responses in Span Style "Font-StyleYohana SumiNo ratings yet
- Testosterona SericaDocument7 pagesTestosterona SericaUriel Avelar AparicioNo ratings yet
- Aps 20145Document18 pagesAps 20145Adelia RahmaNo ratings yet
- Jurnal PPT SimolDocument9 pagesJurnal PPT SimolDimana Mana Ada ManaNo ratings yet
- Rag Proteins Regulate Amino-Acid-InducedDocument2 pagesRag Proteins Regulate Amino-Acid-Inducedplastioid4079No ratings yet
- Plant Physiology and Biochemistry: Research ArticleDocument8 pagesPlant Physiology and Biochemistry: Research Articlefrancis3ndourNo ratings yet
- Systemic Acquired ResistanceDocument11 pagesSystemic Acquired Resistanceكركشندي أفندي Karkshndy afndyNo ratings yet
- Plants 12 01013 v2Document12 pagesPlants 12 01013 v2كركشندي أفندي Karkshndy afndyNo ratings yet
- Mechanisms of Tolerance To Salinity in Banana: Physiological, Biochemical, and Molecular AspectsDocument8 pagesMechanisms of Tolerance To Salinity in Banana: Physiological, Biochemical, and Molecular AspectsJavier Cobos MoraNo ratings yet
- tmp3E4E TMPDocument4 pagestmp3E4E TMPFrontiersNo ratings yet
- Sun 2014Document9 pagesSun 2014Jeff225No ratings yet
- 762 FullDocument16 pages762 Fullkorosuke1920No ratings yet
- Protective Effect of 5-Hydroxy-3, 4, 7-Trimethoxyflavone Against Inflammation Induced by Lipopolysaccharide in RAW 264.7 Macrophage: in Vitro Study and in Silico ValidationDocument14 pagesProtective Effect of 5-Hydroxy-3, 4, 7-Trimethoxyflavone Against Inflammation Induced by Lipopolysaccharide in RAW 264.7 Macrophage: in Vitro Study and in Silico ValidationsopmaNo ratings yet
- Proteomic Characterization of The Acid Tolerance Response in Lactococcus Lactis MG1363Document14 pagesProteomic Characterization of The Acid Tolerance Response in Lactococcus Lactis MG1363ThanasisNo ratings yet
- New Phytologist - 2010 - LeeDocument12 pagesNew Phytologist - 2010 - LeeVirgilio Gavicho UarrotaNo ratings yet
- 2022 Article 36Document15 pages2022 Article 36Lucas WillianNo ratings yet
- High Resistance To Oxidative Stress in The Fungal PathogenDocument12 pagesHigh Resistance To Oxidative Stress in The Fungal PathogenIvica PutrićNo ratings yet
- Alazem 2014 Roles of Plant Hormones in The Regulation of Host Virus InteractionsDocument12 pagesAlazem 2014 Roles of Plant Hormones in The Regulation of Host Virus InteractionsTriEka HeryaNo ratings yet
- Perception and Transduction of Abscisic Acid Signals: Keys To The Function of The Versatile Plant Hormone ABADocument9 pagesPerception and Transduction of Abscisic Acid Signals: Keys To The Function of The Versatile Plant Hormone ABAAnirban KunduNo ratings yet
- Pettko-Szandtner Et AlDocument13 pagesPettko-Szandtner Et AlAladár Pettkó-SzandtnerNo ratings yet
- Involvement of The Rab27 Binding Protein Slac2C/Myrip in Insulin ExocytosisDocument11 pagesInvolvement of The Rab27 Binding Protein Slac2C/Myrip in Insulin ExocytosisJoseGonzalezNo ratings yet
- Cárdenas-Manríquez Et Al. - 2016 - Environmental and Experimental BotanyDocument9 pagesCárdenas-Manríquez Et Al. - 2016 - Environmental and Experimental BotanyIsaac VegaNo ratings yet
- Abuaish 2021 The Role of Sex Differentiated Variations in Stress Hormones Antiox Neuroimmun Res in Rel To Soc Int Impair in Rodent Model of AutismDocument11 pagesAbuaish 2021 The Role of Sex Differentiated Variations in Stress Hormones Antiox Neuroimmun Res in Rel To Soc Int Impair in Rodent Model of AutismJortegloriaNo ratings yet
- 9 Sudemycins 2011Document8 pages9 Sudemycins 2011milenerato2240No ratings yet
- Gopika Suresh - Ims20121 Assignment 1Document3 pagesGopika Suresh - Ims20121 Assignment 1Gopika SureshNo ratings yet
- Chemical Composition, Antioxidant and Anticholinesterase Activity of MelissaDocument12 pagesChemical Composition, Antioxidant and Anticholinesterase Activity of MelissaOanaCristeaNo ratings yet
- Genomic Analysis of MAP Kinase Cascades In: Arabidopsis Defense ResponsesDocument13 pagesGenomic Analysis of MAP Kinase Cascades In: Arabidopsis Defense ResponsesMelvin PrasadNo ratings yet
- Dietary Regulation of Keap1 Nrf2 ARE Pathway NFKB.2014 GOODDocument25 pagesDietary Regulation of Keap1 Nrf2 ARE Pathway NFKB.2014 GOODBeirnes Fernando Sembiring M100% (1)
- Chirality, Drug Metabolism and ActionDocument7 pagesChirality, Drug Metabolism and ActionBrahma Hakim Yuanda HutabaratNo ratings yet
- 016 Scas Abstracts Listed Alphabetically by Abstractspdf 19 2016 Scas AbstractsDocument25 pages016 Scas Abstracts Listed Alphabetically by Abstractspdf 19 2016 Scas AbstractsdssagNo ratings yet
- Overexpression of Calmodulin-Like (Shcml44) Stress-Responsive Gene From Solanum Habrochaites Enhances Tolerance To Multiple Abiotic StressesDocument20 pagesOverexpression of Calmodulin-Like (Shcml44) Stress-Responsive Gene From Solanum Habrochaites Enhances Tolerance To Multiple Abiotic StressesFarahDeebaNo ratings yet
- Evaluation of Potential Antioxidant and Anti Inflammatory Effects - 2018 - PoultDocument16 pagesEvaluation of Potential Antioxidant and Anti Inflammatory Effects - 2018 - Poultlady mae rufinoNo ratings yet
- Tezisfuzet AngolDocument9 pagesTezisfuzet AngolYudistira PratamaNo ratings yet
- Centella Asiatica and Its Fractions Reduces Lipid Peroxidation Induced by Quinolinic Acid and Sodium Nitroprusside in Rat Brain RegionsDocument15 pagesCentella Asiatica and Its Fractions Reduces Lipid Peroxidation Induced by Quinolinic Acid and Sodium Nitroprusside in Rat Brain RegionsrhinaNo ratings yet
- Bioengineering For Salinity Tolerance in Plants State of The ArtDocument22 pagesBioengineering For Salinity Tolerance in Plants State of The ArtCesarRamNo ratings yet
- Sanchez Et Al 2010Document12 pagesSanchez Et Al 2010minervaNo ratings yet
- Campos Proposal 2012Document23 pagesCampos Proposal 2012Asim MughalNo ratings yet
- 2008 Smith-et-Al JA SA HerbivoryDocument7 pages2008 Smith-et-Al JA SA HerbivoryPriscila MezzomoNo ratings yet
- Lukaszuk2016 Article PhotosynthesisAndSucroseMetaboDocument12 pagesLukaszuk2016 Article PhotosynthesisAndSucroseMetaboghilbrania salsabilaNo ratings yet
- Food and Chemical Toxicology: Article InfoDocument7 pagesFood and Chemical Toxicology: Article InfoNathalia MojicaNo ratings yet
- To Blastand Leaf Blight Diseasesin RiceDocument10 pagesTo Blastand Leaf Blight Diseasesin RicevenkatscribdusernameNo ratings yet
- J. Biol. Chem.-2011-Pan-14575-87Document14 pagesJ. Biol. Chem.-2011-Pan-14575-87Henrique HolandaNo ratings yet
- Structure Activity Relationship: Related TermsDocument18 pagesStructure Activity Relationship: Related TermsSajjad Hossen ChowdhuryNo ratings yet
- Accepted Manuscript: Environmental and Experimental BotanyDocument29 pagesAccepted Manuscript: Environmental and Experimental BotanyRenganathan PrabhaharanNo ratings yet
- Erv 013Document13 pagesErv 013Aisha MurtazaNo ratings yet
- Changes in Membrane Fatty Acids Composition of MicrobialDocument5 pagesChanges in Membrane Fatty Acids Composition of MicrobialDaniel JimenezNo ratings yet
- Grant 1997Document10 pagesGrant 1997Django BoyeeNo ratings yet
- tmpF178 TMPDocument15 pagestmpF178 TMPFrontiersNo ratings yet
- Tmp1a96 TMPDocument80 pagesTmp1a96 TMPFrontiersNo ratings yet
- Tmpa077 TMPDocument15 pagesTmpa077 TMPFrontiersNo ratings yet
- tmpE3C0 TMPDocument17 pagestmpE3C0 TMPFrontiersNo ratings yet
- tmp27C1 TMPDocument5 pagestmp27C1 TMPFrontiersNo ratings yet
- tmp998 TMPDocument9 pagestmp998 TMPFrontiersNo ratings yet
- tmp3656 TMPDocument14 pagestmp3656 TMPFrontiersNo ratings yet
- tmp96F2 TMPDocument4 pagestmp96F2 TMPFrontiersNo ratings yet
- tmpA7D0 TMPDocument9 pagestmpA7D0 TMPFrontiersNo ratings yet
- tmp97C8 TMPDocument9 pagestmp97C8 TMPFrontiersNo ratings yet
- PROLYMERDocument2 pagesPROLYMERFlavio GonzalezNo ratings yet
- Outline Report PFR 2015 2018 CASESDocument5 pagesOutline Report PFR 2015 2018 CASESPaul ValerosNo ratings yet
- Navigating The Cooling, Heating and Reheat Process On The Psychrometric ChartDocument11 pagesNavigating The Cooling, Heating and Reheat Process On The Psychrometric ChartmohdkamalhaziqNo ratings yet
- Kebersihan Gigi Mulut Dan Kejadian Gingivitis Pada Anak Sekolah DasarDocument8 pagesKebersihan Gigi Mulut Dan Kejadian Gingivitis Pada Anak Sekolah DasarDeri Atau DariNo ratings yet
- Mwfly Business Magazine EN - CompressedDocument25 pagesMwfly Business Magazine EN - CompressedinfoNo ratings yet
- Physiology Past PapersDocument11 pagesPhysiology Past PapersIrfan Haider FaniNo ratings yet
- Acp CBC 1Document52 pagesAcp CBC 1Danny R. SalvadorNo ratings yet
- Delayed-Action AcceleratorDocument3 pagesDelayed-Action AcceleratorMOHD FIRDAUSNo ratings yet
- Nottingham Hip Fracture Score As A Predictor of One Year PDFDocument4 pagesNottingham Hip Fracture Score As A Predictor of One Year PDFjavieccrNo ratings yet
- 1 Introduction To Management Copy 2Document21 pages1 Introduction To Management Copy 2Angelica Camille B. AbaoNo ratings yet
- 10.A.Cools Kinetic Chain PDFDocument22 pages10.A.Cools Kinetic Chain PDFWalter PelaezNo ratings yet
- Conditioner Portatil Sams ClubDocument5 pagesConditioner Portatil Sams ClubJuan Carlos RodriguezNo ratings yet
- PDF SSCP Systems Security Certified Practitioner All in One Exam Guide 3Rd Edition Gibson Ebook Full ChapterDocument53 pagesPDF SSCP Systems Security Certified Practitioner All in One Exam Guide 3Rd Edition Gibson Ebook Full Chapterdiane.vant368100% (2)
- Ich Guidelines For Stability Testing of New Drug Substance and Drug ProductsDocument39 pagesIch Guidelines For Stability Testing of New Drug Substance and Drug ProductsRahul LakhaniNo ratings yet
- Discussion Q1Document7 pagesDiscussion Q1TASHANo ratings yet
- Motivation BisDocument20 pagesMotivation BishùhjgNo ratings yet
- Pipeline Defect Repair MethodsDocument20 pagesPipeline Defect Repair MethodsAyon RoyNo ratings yet
- RENOLD (Worm Gear - Models PH)Document60 pagesRENOLD (Worm Gear - Models PH)Rodrigo Leal GuzmanNo ratings yet
- Pilzzucht PDFDocument68 pagesPilzzucht PDFSu Per BingNo ratings yet
- Engelhardt BioethicsDocument9 pagesEngelhardt BioethicsAaron Lee WiegmannNo ratings yet
- Dup TemplateDocument5 pagesDup TemplateJoshua SouthNo ratings yet
- DCP-CZM (Detectores de Humo Convencionales)Document2 pagesDCP-CZM (Detectores de Humo Convencionales)alexropaNo ratings yet
- Personal FinanceDocument32 pagesPersonal FinancePrincess MogulNo ratings yet
- Lifting Operations AZDP Directive 008 - Edition 4.0: U:TobeusedasisDocument7 pagesLifting Operations AZDP Directive 008 - Edition 4.0: U:TobeusedasisSiddharth AsthanaNo ratings yet
- Sol-Gel Methods PDFDocument19 pagesSol-Gel Methods PDFDimitrios Tsiplakides100% (2)
- Mobile Crane SafetyDocument3 pagesMobile Crane SafetyAnonymous LFgO4WbID0% (1)
- 01 91 13 General Commissioning RequirementsDocument25 pages01 91 13 General Commissioning Requirementsvin ssNo ratings yet
- Chapter 6 Hexavalent ChromiumDocument56 pagesChapter 6 Hexavalent Chromiumgabriel100% (1)
tmpFE04 TMP
tmpFE04 TMP
Uploaded by
FrontiersOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
tmpFE04 TMP
tmpFE04 TMP
Uploaded by
FrontiersCopyright:
Available Formats
Plant Stress 2012 Global Science Books
Methyl Salicylate Esterases in Plant Immunity
Dhirendra Kumar1* Tazley Hotz2 Mir Hossain1 Pavan Chigurupati1
Amukta Mayakoti1 Nkongho Binda1 Bingqing Zhao1 Diwaker Tripathi3
1 Department of Biological Sciences, Box 70703, East Tennessee State University, Johnson City, TN 37614, USA
2 Quillen College of Medicine, East Tennessee State University, Johnson City, TN 37614, USA
3 Present Address: Department of Plant Pathology, Washington State University, Pullman, WA 99164, USA
Corresponding author: * kumard@etsu.edu
ABSTRACT
Salicylic acid (SA) is an important signal in various plant processes. It is well known and widely studied for its role in plant disease
resistance. Several proteins, which physically interact with SA has been identified and characterized for their possible role in disease
resistance signaling. These plant proteins bind to SA with varying affinity and they differ considerably in their structure and activity. The
protein, which binds to SA with highest affinity amongst all the characterized SA-binding proteins, is SABP2. It is a 29-kDa protein and
has esterase like enzymatic activity. It is able to use plant synthesized methyl salicylate as a substrate and convert it into SA, which
triggers disease resistance in plants. Silencing of SABP2 makes plants more susceptible to pathogens and their capacity to induce SAR is
severely compromised. The esterase activity of SABP2 is required to process the phloem mobile signal, MeSA in distal uninoculated
tissues to induce resistance. The binding of SA to SABP2 is important for activation of SAR in distal tissues.
_____________________________________________________________________________________________________________
Keywords: disease resistance, systemic acquired resistance, SABP2, SA-methyl transferase, SA-binding protein
Abbreviations: MeSA, methyl salicylate; PR-1, pathogenesis-related protein-1; RNAi, ribonucleic acid interference; SA, salicylic acid;
SABP2, SA-binding protein 2; SAR, systemic acquired resistance; TMV, Tobacco mosaic virus
CONTENTS
INTRODUCTION ................................................................................................................................................................................ 1
SALICYLIC ACID-BINDING PROTEIN: CATALASE ........................................................................................................................ 1
SA-BINDING PROTEIN 3 (SABP3): A CHLOROPLASTIC CARBONIC ANYHYDRASE.................................................................. 2
SA-BINDING PROTEIN 2 (SABP2): A METHYL SALICYLATE ESTERASE..................................................................................... 2
Role of SABP2 in plant disease resistance......................................................................................................................................... 2
MeSA is a phloem mobile signal for SAR ......................................................................................................................................... 3
Effect of pathogen infection on SABP2 expression ............................................................................................................................ 3
Is SABP2 expression needed for basal defense?................................................................................................................................. 3
CONCLUSIONS .................................................................................................................................................................................. 4
ACKNOWLEDGEMENTS................................................................................................................................................................... 4
REFERENCES..................................................................................................................................................................................... 4
_____________________________________________________________________________________________________________
INTRODUCTION
SALICYLIC ACID-BINDING PROTEIN: CATALASE
Salicylic acid (SA) has been known to humans for a long
time as a plant compound, which could cure or relieve
symptoms associated with many diseases. It was encouraged by Hippocrates to be used as a child birth pain
reliever as early as fourth century B.C. (Vlot et al. 2009).
Extracts of willow bark, which contain Salicylate was
extensively used to relieve the pain. Possible role of
salicylic acid in plants was first demonstrated by the induction of resistance against tobacco mosaic virus by pretreating tobacco plants with aspirin (White 1979). Many
plants synthesize and accumulate high levels of SA upon
pathogen infection. How this newly synthesized SA activates downstream signaling pathway leading to activation
and establishment of robust resistance in plants has been
subject of interest for last several decades? Which components of signaling pathways are influenced by the increased levels of SA? Besides analysis of Arabidopsis
mutants defective in SA signaling pathways, another bold
approach was to identify and characterize cellular proteins,
which physically interact with SA. In this approach radioactive labeled SA was used as ligand to detect, identify and
purify proteins, which interacted with it.
Following this biochemical approach several plants proteins,
which physically interacted with SA were identified. 14Clabeled SA was used to detect and identify these proteins.
First such protein to be identified from extracts of tobacco
leaves was salicylic acid binding protein (SABP) (Chen and
Klessig 1991; Chen et al. 1993a). Further analysis and
characterization of SABP showed that it is a catalase like
protein, which could degrade H2O2 into H2O and O2 (Chen
1993; Conrath et al. 1995). Biochemical characterization of
tobacco catalase showed that binding of SA lead to the
inhibition of its H2O2 degrading activity. Other biologically
active (ability to induce resistance response in plants) analogs of SA also specifically inhibited H2O2 degrading activity of catalase. These observations lead Chen et al. to propose that inhibition of H2O2 degrading activity of catalase
resulted in accumulation of H2O2 triggering a resistance
response in plants known as SA mediated resistance response (Chen et al. 1993b). Durner and Klessig (1996)
showed that mammalian catalases are also inhibited by SA
and so are other scavenging enzymes e.g. ascorbate peroxidase in the cell (Durner and Klessig 1995, 1996). Inhibition of these enzymes by SA generates SA free radicals,
Received: 22 February, 2012. Accepted: 29 June, 2012.
Invited Mini-Review
Plant Stress 6 (Special Issue 1), x-y 2012 Global Science Books
which may induce lipid peroxidation leading to activation
of PR proteins and defense response (Anderson and Klessig
1998). This hypothesis was quickly challenged by the observations that concentrations of SA (10-3 M) necessary to
inhibit H2O2 degrading activity is never achieved in the cell
(Ruffer et al. 1995). Levels of SA in systemic plants tissue
are probably too low (0.5-9.0 M) to bind and cause inhibition of catalase and ascorbate peroxidase (Malamy et al.
1990; Enyedi et al. 1992; Vernooij et al. 1994). Other studies also did not support SA mediated inhibition of ascorbate peroxidase and catalase (Miyake 1996; Tenhaken and
Rubel 1997). Several other studies have also reported SA
mediated inhibition of catalase activity but its role in activation of disease resistance is unclear (Snchez-Casas and
Klessig 1994; Chen et al. 1997). More precise studies are
needed to understand the role of SA binding to catalase and
its effect on disease resistance or other plant processes.
SA-BINDING PROTEIN 3 (SABP3): A
CHLOROPLASTIC CARBONIC ANYHYDRASE
To detect binding proteins with much higher affinity for SA
compared to catalase (SABP), 3[H]-SA, which has higher
specific activity was used. Using 3[H]-SA as ligand, another
SA-binding protein (SABP3) was identified and characterized from tobacco leaves. SABP3 is a chloroplast localized
carbonic anyhydrase (Slaymaker et al. 2002). SABP3
showed higher binding affinity to SA (Kd of 3.7 M) compared to SABP (Kd of 14 M) (Chen and Klessig 1991; Du
and Klessig 1997; Slaymaker et al. 2002). Although biochemical analysis did not show any effect of SA binding on
the carbonic anhydrase activity of SABP3 but its silencing
resulted in suppression of pto:avrPto mediated hypersensitive response in disease resistance in Nicotiana benthamiana and enhanced susceptibility to Phytophthora infestans
(Slaymaker et al. 2002; Restrepo et al. 2005). Although binding of SA did not affect the enzymatic activity of SABP3
but its modification through S-nitrosylation in Arabidopsis
resulted in its reduced SA binding and enzymatic activity
(Wang et al. 2009). This may suggest that S-nitrosylation
and SA-binding may help modulate carbonic anhydrase
activity of SABP3.
SA-BINDING PROTEIN 2 (SABP2): A METHYL
SALICYLATE ESTERASE
Of all the SABPs studied to date, SABP2 showed the highest affinity (Kd of 90 nM) for SA (Du and Klessig 1997).
Affinity of SABP2 to SA is 150 times higher compared to
SABP (catalase). SABP2 is present in excruciatingly low
abundance (0.01 pmol/mg) in tobacco leaves compared to
SABP (5 pmol/mg soluble protein) and SABP3 (0.4
pmol/mg). Low abundance of SABP2 made its purification
much harder compared other SA-binding proteins. Indeed,
it took several years to standardize a working purification
scheme to purify small amounts of SABP2 enough to determine its amino acid sequence (Kumar and Klessig 2003).
Based on size exclusion chromatography, SABP2 appeared
to be a monomeric protein of 25-30 kDa as reported by Du
et al. (Du and Klessig 1997). A combination of degenerate
primers were synthesized based on the peptide sequences
obtained and RT-PCR was used to amplify a partial sequence
of putative SABP2. Full- length cDNA was obtained following 5-RACE.
For biochemical characterization of putative SABP2, its
full length coding sequence was cloned into E.coli expression vector and protein was expressed and purified. Recombinant purified SABP2 bound specifically to SA and its
active analogs and not to its inactive analogs. DNA analysis
showed that SABP2 belongs to / hydrolase superfamily
of proteins. This family of proteins exhibit a range of enzymatic activities, which include carboxylic acid ester hydrolases e.g. cholineterases; lipid hydrolases e.g. lipases; thioester, hydrolases e.g. thioesterases; peptide hydrolases e.g.
serine carboxipeptidases; hydroxylnitrile lyases etc. Recom-
binant SABP2 showed low level activity with several artificial substrates, which included paranitrophenol acetate,
paranitrophenol butyrate, paranitrophenol myristate, paranitrophenol palmitate and 4-methylumbelliferone butyrate
(Kumar and Klessig 2003). Several of the compounds,
which are naturally produced by plants, were also tested as
potential substrates of SABP2. Of the three potential substrates e.g. methyl salicylate (MeSA), methyl jasmonate
(MeJA) and methyl indole acetic acid (MeIAA) tested;
SABP2 showed highest activity with MeSA as a substrate
(Forouhar et al. 2005). MeSA, commonly known as oil of
wintergreen is naturally synthesized by plants and its level
increases upon pathogen infection (Shulaev et al. 1997).
To understand the effect of SA binding to SABP2, its 3D structure alone and in complex form with SA was determined at 2.1 resolution (Forouhar et al. 2005). As expected based on its similarity to other known proteins, SABP2
belonged to / hydrolase superfamily of proteins. The
structure of SABP2 consisted of six-stranded parallel sheet flanked on both sides by six -helices. It has catalytic
triad consisting of active site Ser-81, His-238 and Asp-210.
Another surprising observation was existence of SABP2 as
a dimer. Size exclusion chromatography of purified recombinant SABP2 suggested it to be a dimer while native
SABP2 purified from tobacco leaves was reported to be a
monomer (Du and Klessig 1997; Forouhar et al. 2005).
Most surprising observation was the binding of SA in the
active site of SABP2. SABP2 was completely shielded from
the solvent. SABP2 bound SA was located in the hydrophobic environment surrounded by side chains of Asn-123,
Trp-131, Phe-136, Met-149 and Leu-181. This observation
explained why SABP2 bound to SA with such a high
affinity (Kd of 90 nM). Binding of SA to SABP2 did not
result in a major structural change. This observation was
also supported by NMR analysis, which showed only minor
chemical shifts for few residues in presence of SA (Forouhar et al. 2005). MeSA could also readily fit into the active
site of SABP2 while MeJA and MeIAA were not easy to fit.
Tight binding of SA in the active site of SABP2 suggested
that it could inhibit enzymatic activity of SABP2. In vitro
studies using recombinant SABP2 clearly suggested that its
enzymatic activity is inhibited by SA (Forouhar et al. 2005).
Synthetic SA analog, acibenzolar-S-methyl (ASM) binds to
SABP2 with higher affinity compared to SA. SABP2 catalyzes the conversion of ASM to its acid form acibenzolar to
induce resistance in plants (Tripathi et al. 2010).
Role of SABP2 in plant disease resistance
To determine if SABP2 has any role in disease resistance
signaling, tobacco plants silenced in SABP2 expression
were used. RNAi was used to stably silence the expression
of SABP2 in tobacco plants (Smith et al. 2000). Transgenic
SABP2 silenced tobacco plants upon challenge with
Tobacco mosaic virus (TMV) resulted in compromise in
their ability to mount robust systemic acquired resistance
compared to the control plants containing only empty silencing vector (Kumar and Klessig 2003). Expression of pathogenesis-related protein-1 (PR-1) was also compromised in
SABP2-silenced plants. These silenced plants allowed enhanced replication/movement of TMV but did not allow
systemic movement to distal tissues. Results from silencing
experiments made it clear that SA synthesized by SABP2 in
tobacco plants is essential for a robust SAR response. A
synthetic version of SABP2 was used to trans-complement
the loss of SAR inducing activity of SABP2 silenced
(Kumar et al. 2006). This synthetic version of SABP2 escaped silencing in SABP2 silenced plants due to sufficiently
different DNA sequence compared to wild type tobacco
SABP2 sequence while coding for the same amino acid
sequence (Kumar et al. 2006). Arabidopsis contains a gene
family of 18 members sharing 32-57% identity with tobacco
SABP2 (Vlot et al. 2008). Five of these show preference for
MeSA as a substrate and their activity is inhibited by SA
binding. Three of these family members (At2g23620,
MES in plant defenses. Kumar et al.
At2g23560 and At4g37150) when expressed in SABP2
silenced tobacco plants could complement for its SAR deficiency. These results suggest that several Arabidopsis genes
(At2g23620, At2g23560 and At4g37150) are orthologs of
tobacco SABP2 and are redundant for hydrolysis of MeSA
and SAR. This, in part explains why SABP2 like proteins
were never identified in several exhaustive genetic screens
for Arabidopsis mutants with enhanced disease susceptibility phenotype (Glazebrook et al. 1996; Volko et al. 1998;
Ton et al. 2002).
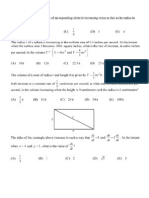
Pathogen
Rproteins
ICS1
Plastid
SAMT
Systemic
tissue
SA
MeSA
MeSA is a phloem mobile signal for SAR
Since SABP2 catalyzes conversion of MeSA to SA and
MeSA is a volatile compound, question arises if MeSA instead of SA is the signal for SAR in distal uninfected tissue.
The earlier observation that SA accumulates in phloem
tissues in plants exhibiting SAR led to the believe that SA is
the mobile signal for SAR (Yalpani et al. 1991). However,
later studies using grafting experiments with NahG transgenic plants did not support this hypothesis because NahG
rootstock containing chimera plants lacking SA were still
able to generate signal for SAR (Vernooij et al. 1994).
MeSA has been shown to be an airborne signal involved in
inducing disease resistance in neighboring plants (Shulaev
et al. 1997). MeSA does not induce SAR by itself and
works by being converted to salicylic acid. It was observed
that levels of MeSA increases dramatically in TMV infected
tissue and also in uninfected distal tissues (Seskar et al.
1998). This increase in MeSA levels paralleled the increase
in SA levels in plants infected with TMV. Chimera plants
were made by grafting SABP2-silenced plants, and control
tobacco plants expressing wild type levels of SABP2.
Grafted plants were assayed for SAR response and plants
lacking SABP2 in distal tissues (scion) failed to convert
MeSA to SA and was defective in inducing resistance to
pathogen and expression of PR-1 was also compromised
(Park et al. 2007). Chimera plants lacking SABP2 in rootstock and expressing wild type levels of SABP2 in scion
developed normal SAR. These observations suggested that
while SABP2 is not required for generating systemic signal
for SAR but it is required for processing the SAR signal in
distal tissues (Park et al. 2007; Kumar and Klessig 2008).
Mutant plants with uncontrolled esterase activity in primary
tissue were compromised in SAR response. These plants
did not have increased levels of MeSA in primary inoculated tissue, phloem exudates and upper distal tissues. Transgenic tobacco plants silenced in SA-methyl transferase
expression failed to develop SAR (Park et al. 2007). SAmethyl transferase is required to convert SA into MeSA. All
these results indicated that MeSA is phloem mobile signal
for SAR and SABP2 is required to convert this signal into
SA leading to resistance response (Park et al. 2007). A
synthetic analog of SA, 2,2,2,2-tetrafluoroacetophenone
(tetraFA), binds SABP2 with high affinity as SA and
inhibits its esterase activity but it could not induce resistance response as SA. Treatment with tetraFA effectively
blocked SAR in tobacco and Arabidopsis plants showing
the importance of MeSA and SABP2 in SAR (Park et al.
2009).
Several studies using Arabidopsis mutants have suggested a lipid-derived signal for SAR. SAR was compromised in Arabidopsis mutant dir1-1 defective in an apoplastic lipid transfer protein (Maldonado et al. 2002). Recent
studies have implicated plant volatile compound JA in SAR
(Truman et al. 2007). Several recent studies however rule
out the possibility of JA as an SAR signal (Cui et al. 2005;
Mishina and Zeier 2007; Chaturvedi et al. 2008; Attaran et
al. 2009). Another study completely ruled out MeSA and JA
as a phloem mobile signal in Arabidopsis (Attaran et al.
2009). A new study suggests role of length exposure to light
as a deciding factor if plant uses MeJA or MeSA mediated
pathway for SAR (Liu et al. 2011).
PAL
SABP2
SABP2
SA
SA
SAR
Redox
Cytoplasm
NPR1 NPR1
NPR1
NPR1
NPR1
Nucleus
TGA1
TGACG
PR-1
SAresponsivegenes
Resistance
Fig. 1 A working model for SA-dependent resistance pathway. Interaction between pathogen derived avr (avirulence) molecule and plant
derived R (resistance) protein results in synthesis of SA via ICS1/PAL
pathways. SA is then converted into MeSA by an SAMT (SA-methyl
transferase). SABP2 catalyzes conversion of MeSA to SA, which triggers
downstream signaling leading to expression of SA responsive genes.
MeSA being lipid mobile, travels to uninfected (systemic) parts of the
plant where it is converted to SA by SABP2, which triggers expression of
resistance proteins leading to SAR. ICS1: isochorismate synthase 1; PAL:
phenyl ammonia lyase; NPR1: non expressor of pathogenesis related
protein 1.
Effect of pathogen infection on SABP2 expression
Tobacco SABP2 expression was induced upon infection by
TMV (Kumar and Klessig, 2003). This expression was
dependent on SA (Kumar and Klessig unpublished results).
Three of the Arabidopsis orthologs of SABP2, AtMES1
(At2g23620), AtMES7 (At2g23560) and AtMES9
(At4g37150) were also induced upon infection by avirulent
Pseudomonas syringae. Treatments with SA did not induce
the expression of tobacco SABP2 but MeJA treatment
induced its expression (Kumar and Klessig, unpublished
results). Induction of SABP2 expression by MeJA suggests
a possible crosstalk between SA and JA pathway. Further
studies may be required to explore this role of SABP2.
Recently, SABP2 homlogs of poplar plants, PtSABP2-1 and
PtSABP2-2 were shown to be differentially expressed.
PtSABP2-1 was highly expressed in leaves while
PtSABP2-2 was in roots (Zhao et al. 2009). PtSABP2-2
was significantly upregulated by wounding and MeJA treatments. In a global gene expression analysis of tomato genes,
a SABP2 homolog was highly expressed in NaCl treated
plants indicating a possible role in abiotic stress (Sun et al.
2010).
Is SABP2 expression needed for basal defense?
Role of SA is well established in basal defense in plants.
SA-mediated basal defense is specifically targeted by plants
pathogens to gain entry into the cell. Analysis of SABP2
silenced plants for expression of defense genes revealed
that these transgenic plants were compromised in expres-
Plant Stress 6 (Special Issue 1), x-y 2012 Global Science Books
sion of PR-1 (D Kumar unpublished results). This may indicate role of SABP2 in synthesis of basal levels of SA in cell.
This SABP2 synthesized SA may help in maintaining basal
defense levels in tobacco plants.
CONCLUSIONS
Discovery of tobacco SABP2 has helped to identify the
long sought phloem mobile signal in plant immunity. Plants
appear to communicate by synthesizing MeSA as a signal
and require SABP2 to decipher this plant immunity signal.
High affinity binding of SA to SABP2 is critical in enhancing the levels of MeSA in local and distal tissues. In distal
tissues MeSA is converted back to SA, which results in the
downstream signaling leading to activation of resistance
(Fig. 1). SABP2 proteins are present in many of the plant
species. Its role in tobacco, Arabidopsis and potato is now
well studied (Kumar and Klessig 2003; Vlot et al. 2008;
Manosalva et al. 2010). SA synthesized due to activity of
SABP2 appears to be critical for activation of defense responses because plants silenced in SABP2 expression are
compromised in local and systemic resistance (Kumar and
Klessig 2003). Further studies may be needed to determine
if SABP2 is also required for other SA mediated plant processes.
ACKNOWLEDGEMENTS
This work was supported by grant from National Science Foundation (MCB1022077), small RDC and major RDC grants (E82023,
10-018M) from East Tennessee State University, Johnson City, TN.
REFERENCES
Anderson M, Chen Z, Klessig DF (1998) Possible involvement of lipid
peroxidation in salicylic acid-mediated induction of PR-1 gene expression.
Phytochemistry 47, 555-566
Attaran E, Zeier TE, Griebel T, Zeier J (2009) Methyl salicylate production
and jasmonate signaling are not essential for systemic acquired resistance in
Arabidopsis. Plant Cell 21, 954-971
Chaturvedi R, Krothapalli K, Makandar R, Nandi A, Sparks AA, Roth MR,
Welti R, Shah J (2008) Plastid omega3-fatty acid desaturase-dependent
accumulation of a systemic acquired resistance inducing activity in petiole
exudates of Arabidopsis thaliana is independent of jasmonic acid. Plant
Journal 54, 106-117
Chen Z, Klessig DF (1991) Identification of a soluble salicylic acid-binding
protein that may function in signal transduction in the plant disease-resistance response. Proceedings of the National Academy of Sciences USA 88,
8179-8183
Chen Z, Ricigliano JW, Klessig DF (1993a) Purification and characterization
of a soluble salicylic acid-binding protein from tobacco. Proceedings of the
National Academy of Sciences USA 90, 9533-9537
Chen Z, Silva H, Klessig DF (1993b) Active oxygen species in the induction of
plant systemic acquired resistance by salicylic acid. Science 262, 1883-1886
Chen Z, Iyer S, Caplan A, Klessig DF, Fan B (1997) Differential accumulation of salicylic acid and salicylic acid-sensitive catalase in different rice tissues. Plant Physiology 114, 193-201
Chen Z, Silva H, Klessig DF (1993) Active oxygen species in the induction of
plant systemic acquired resistance by salicylic acid. Science 262, 1883-1886
Conrath U, Chen Z, Ricigliano JR, Klessig DF (1995) Two inducers of plant
defense responses, 2,6-dichloroisonicotinec acid and salicylic acid, inhibit
catalase activity in tobacco. Proceedings of the National Academy of Sciences
USA 92, 7143-7147
Cui J, Bahrami AK, Pringle EG, Hernandez-Guzman G, Bender CL, Pierce
NE, Ausubel FM (2005) Pseudomonas syringae manipulates systemic plant
defenses against pathogens and herbivores. Proceedings of the National
Academy of Sciences USA 102, 1791-1796
Du H, Klessig DF (1997) Identification of a soluble, high-affinity salicylic
acid-binding protein in tobacco. Plant Physiology 113, 1319-1327
Durner J, Klessig DF (1995) Inhibition of ascorbate peroxidase by salicylic
acid and 2,6-dichloroisonicotinic acid, two inducers of plant defense responses. Proceedings of the National Academy of Sciences USA 92, 11312-11316
Durner J, Klessig DF (1996) Salicylic acid is a modulator of tobacco and
mammalian catalases. Journal of Biological Chemistry, 271 28492-28501
Enyedi AJ, Yalpani N, Silverman P, Raskin I (1992) Signal molecules in systemic plant resistance to pathogens and pests. Cell 70, 879-886
Forouhar F, Yang Y, Kumar D, Chen Y, Fridman, E, Park SW, Chiang Y,
Acton TB, Montelione GT, Pichersky E, Klessig DF, Tong L (2005) Structural and biochemical studies identify tobacco SABP2 as a methyl salicylate
esterase and implicate it in plant innate immunity. Proceedings of the National Academy of Sciences USA 102, 1773-1778
Glazebrook J, Rogers EE, Ausubel FM (1996) Isolation of Arabidopsis mutants with enhanced disease susceptibility by direct screening. Genetics 143,
973-982
Kumar D, Klessig DF (2003) High-affinity salicylic acid-binding protein 2 is
required for plant innate immunity and has salicylic acid-stimulated lipase
activity. Proceedings of the National Academy of Sciences USA 100, 1610116106
Kumar D, Gustafsson C, Klessig DF (2006) Validation of RNAi silencing specificity using synthetic genes: Salicylic acid-binding protein 2 is required for
innate immunity in plants. Plant Journal 45, 863-868
Kumar D, Klessig DF (2008) The search for the salicylic acid receptor led to
discovery of the SAR signal receptor. Plant Signaling Behavior 3, 691-692
Liu PP, von Dahl CC, Klessig DF (2011) The extent to which methyl salicylate
is required for signaling systemic acquired resistance is dependent on exposure to light after infection. Plant Physiology 157, 2216-2226
Malamy J, Carr JP, Klessig DF, Raskin I (1990) Salicylic Acid: A likely
endogenous signal in the resistance response of tobacco to viral infection.
Science 250, 1002-1004
Maldonado AM, Doerner P, Dixon, RA, Lamb CJ, Cameron RK (2002) A
putative lipid transfer protein involved in systemic resistance signalling in
Arabidopsis. Nature 419, 399-403
Manosalva PM, Park SW, Forouhar F, Tong L, Fry WE, Klessig DF (2010)
Methyl esterase 1 (StMES1) is required for systemic acquired resistance in
potato. Molecular Plant-Microbe Interactions 23, 1151-1163
Mishina TE, Zeier J (2007) Pathogen-associated molecular pattern recognition
rather than development of tissue necrosis contributes to bacterial induction
of systemic acquired resistance in Arabidopsis. Plant Journal 50, 500-513
Miyake C, Sano S, Asada K (1996) A new assay of ascorbate peroxidase using
the coupled system with monodehydroascorbate radical reductase. Plant
Peroxidases: Biochemistry and Physiology, University of Geneva, Vienna
Park SW, Kaimoyo E, Kumar D, Mosher S, Klessig DF (2007) Methyl Salicylate is a critical mobile signal for plant systemic acquired resistance. Science 318, 113-116
Park SW, Liu PP, Forouhar F, Vlot AC, Tong L, Tietjen K, Klessig DF
(2009) Use of a synthetic salicylic acid analog to investigate the roles of
methyl salicylate and its esterases in plant disease resistance. Journal of Biological Chemistry 284, 7307-7317
Restrepo S, Myers KL, del Pozo O, Martin GB, Hart AL, Buell CR, Fry
WE, Smart CD (2005) Gene profiling of a compatible interaction between
Phytophthora infestans and Solanum tuberosum suggests a role for carbonic
anhydrase. Molecular Plant-Microbe Interactions 18, 913-922.
Ruffer M, Steipe B, Zenk MH (1995) Evidence against specific binding of
salicylic acid to plant catalase. FEBS Letters 377, 175-180
Snchez-Casas P, Klessig DF (1994) A salicylic acid-binding activity and a
salicylic acid-inhibitable catalase activity are present in a variety of plant species. Plant Physiology 106 1675-1679
Seskar M, Shulaev V, Raskin I (1998) Endogenous methyl salicylate in pathogen-inoculated tobacco plants. Plant Physiology 116, 387-392
Shulaev V, Silverman P, Raskin I (1997) Airborne signalling by methyl salicylate in plant pathogen resistance. Nature 385, 718-721
Slaymaker DH, Navarre DA, Clark D, del Pozo O, Martin GB Klessig DF
(2002) The tobacco salicylic acid-binding protein 3 (SABP3) is the chloroplast carbonic anhydrase, which exhibits antioxidant activity and plays a role
in the hypersensitive defense response. Proceedings of the National Academy
of Sciences USA 99, 11640-11645
Smith NA, Singh SP, Wang MB, Stoutjesdijk PA, Green AG, Waterhouse
PM (2000) Total silencing by intron-spliced hairpin RNAs. Nature 407, 319320
Sun W, Xu X, Zhu H, Liu A. Liu L, Li J, Hua X (2010) Comparative transcriptomic profiling of a salt-tolerant wild tomato species and a salt-sensitive
tomato cultivar. Plant Cell Physiology 51, 997-1006
Tenhaken R, Rubel C (1997) Salicylic acid is needed in hypersensitive cell
death in soybean but does not act as a catalase inhibitor. Plant Physiology
115, 291-298
Ton J, De Vos M, Robben C, Buchala A, Metraux JP, Van Loon LC, Pieterse CM (2002) Characterization of Arabidopsis enhanced disease susceptibility mutants that are affected in systemically induced resistance. Plant
Journal 29, 11-21
Tripathi D, Jiang YL, Kumar D (2010) SABP2, a methyl salicylate esterase is
required for the systemic acquired resistance induced by acibenzolar-Smethyl in plants. FEBS Letters 584, 3458-3463
Truman W, Bennett MH, Kubigsteltig I, Turnbull C, Grant M (2007) Arabidopsis systemic immunity uses conserved defense signaling pathways and is
mediated by jasmonates. Proceedings of the National Academy of Sciences
USA 104, 1075-1080
Vernooij B, Friedrich L, Morse A, Reist R, Kolditz-Jawhar R, Ward E,
Uknes S, Kessmann H, Ryals J (1994) Salicylic acid is not the translocated
signal responsible for inducing systemic acquired resistance but is required in
signal transduction. Plant Cell 6, 959-965
Vlot AC, Liu PP, Cameron RK, Park SW, Yang Y, Kumar D, Zhou F,
Padukkavidana T, Gustafsson C, Pichersky E, Klessig DF (2008) Identi-
MES in plant defenses. Kumar et al.
fication of likely orthologs of tobacco salicylic acid-binding protein 2 and
their role in systemic acquired resistance in Arabidopsis thaliana. Plant Journal 56, 445-456
Vlot AC, Dempsey DA, Klessig DF (2009) Salicylic acid, a multifaceted hormone to combat disease. Annual Review of Phytopathology 47, 177-206
Volko SM, Boller T, Ausubel FM (1998) Isolation of new Arabidopsis mutants
with enhanced disease susceptibility to Pseudomonas syringae by direct
screening. Genetics 149, 537-548
Wang YQ, Feechan A, Yun BW, Shafiei R, Hofmann A, Taylor P, Xue P,
Yang FQ, Xie ZS, Pallas JA, Chu CC, Loake GJ (2009) S-nitrosylation of
AtSABP3 antagonizes the expression of plant immunity. Journal of Biological Chemistry, 284 2131-2137
White RF (1979) Acetylsalicylic acid (aspirin) induces resistance to tobacco
mosaic virus in tobacco. Virology 99, 410-412
Yalpani N, Silverman P, Wilson TM, Kleier DA, Raskin I (1991) Salicylic
acid is a systemic signal and an inducer of pathogenesis-related proteins in
virus-infected tobacco. Plant Cell 3, 809-818
Zhao N, Guan J, Forouhar F, Tschaplinski TJ, Cheng ZM, Tong L, Chen F
(2009) Two poplar methyl salicylate esterases display comparable biochemical properties but divergent expression patterns. Phytochemistry 70, 32-39
The author has requested enhancement of the downloaded file. All in-text references underlined in blue are linked to publications on ResearchGate.
You might also like
- Sample Midterm 2 F303Document12 pagesSample Midterm 2 F303philmoreNo ratings yet
- AP CALCULUS BC - MC - Related RatesDocument4 pagesAP CALCULUS BC - MC - Related RatesShannon BurtonNo ratings yet
- Salicylic Acid Signalling in Plants: Molecular SciencesDocument6 pagesSalicylic Acid Signalling in Plants: Molecular SciencesKanishk KannaNo ratings yet
- Identification of Cell-Wall Stress As A Hexose-Dependent and Osmosensitive Regulator of Plant ResponsesDocument12 pagesIdentification of Cell-Wall Stress As A Hexose-Dependent and Osmosensitive Regulator of Plant ResponsesQuenguan FranklinNo ratings yet
- Salicylic Acid and Photosynthesis: Signalling and EffectsDocument31 pagesSalicylic Acid and Photosynthesis: Signalling and EffectsKanishk KannaNo ratings yet
- tmp1013 TMPDocument6 pagestmp1013 TMPFrontiersNo ratings yet
- Plant Hormone-Mediated Regulation of Stress Responses: Review Open AccessDocument10 pagesPlant Hormone-Mediated Regulation of Stress Responses: Review Open AccessEe EENo ratings yet
- Shetty Chamberry AntioxidantsDocument14 pagesShetty Chamberry AntioxidantsrosanelineliegeNo ratings yet
- 2017 Article 9685Document30 pages2017 Article 9685Mohamed HafezNo ratings yet
- Sanguinarine PDFDocument9 pagesSanguinarine PDFSara Yahia cherifNo ratings yet
- 2011 Dempsey SA-biosynthesisDocument24 pages2011 Dempsey SA-biosynthesisPriscila MezzomoNo ratings yet
- Regulation of Water, Salinity, and Cold Stress Responses by Salicylic AcidDocument12 pagesRegulation of Water, Salinity, and Cold Stress Responses by Salicylic AcidBastian SeguelNo ratings yet
- Evoluton Yellow FlopppyDocument30 pagesEvoluton Yellow Flopppydrdgrant6055No ratings yet
- Ecotoxicology and Environmental Safety: Xufei Li, Junyi Shi, Haoyu Sun, Zhifen LinDocument11 pagesEcotoxicology and Environmental Safety: Xufei Li, Junyi Shi, Haoyu Sun, Zhifen LinAbdul Wahab Assya RoniNo ratings yet
- Cross-Talk Between Gibberellins and Salicylic Acid in Early Stress Responses in Span Style "Font-StyleDocument3 pagesCross-Talk Between Gibberellins and Salicylic Acid in Early Stress Responses in Span Style "Font-StyleYohana SumiNo ratings yet
- Testosterona SericaDocument7 pagesTestosterona SericaUriel Avelar AparicioNo ratings yet
- Aps 20145Document18 pagesAps 20145Adelia RahmaNo ratings yet
- Jurnal PPT SimolDocument9 pagesJurnal PPT SimolDimana Mana Ada ManaNo ratings yet
- Rag Proteins Regulate Amino-Acid-InducedDocument2 pagesRag Proteins Regulate Amino-Acid-Inducedplastioid4079No ratings yet
- Plant Physiology and Biochemistry: Research ArticleDocument8 pagesPlant Physiology and Biochemistry: Research Articlefrancis3ndourNo ratings yet
- Systemic Acquired ResistanceDocument11 pagesSystemic Acquired Resistanceكركشندي أفندي Karkshndy afndyNo ratings yet
- Plants 12 01013 v2Document12 pagesPlants 12 01013 v2كركشندي أفندي Karkshndy afndyNo ratings yet
- Mechanisms of Tolerance To Salinity in Banana: Physiological, Biochemical, and Molecular AspectsDocument8 pagesMechanisms of Tolerance To Salinity in Banana: Physiological, Biochemical, and Molecular AspectsJavier Cobos MoraNo ratings yet
- tmp3E4E TMPDocument4 pagestmp3E4E TMPFrontiersNo ratings yet
- Sun 2014Document9 pagesSun 2014Jeff225No ratings yet
- 762 FullDocument16 pages762 Fullkorosuke1920No ratings yet
- Protective Effect of 5-Hydroxy-3, 4, 7-Trimethoxyflavone Against Inflammation Induced by Lipopolysaccharide in RAW 264.7 Macrophage: in Vitro Study and in Silico ValidationDocument14 pagesProtective Effect of 5-Hydroxy-3, 4, 7-Trimethoxyflavone Against Inflammation Induced by Lipopolysaccharide in RAW 264.7 Macrophage: in Vitro Study and in Silico ValidationsopmaNo ratings yet
- Proteomic Characterization of The Acid Tolerance Response in Lactococcus Lactis MG1363Document14 pagesProteomic Characterization of The Acid Tolerance Response in Lactococcus Lactis MG1363ThanasisNo ratings yet
- New Phytologist - 2010 - LeeDocument12 pagesNew Phytologist - 2010 - LeeVirgilio Gavicho UarrotaNo ratings yet
- 2022 Article 36Document15 pages2022 Article 36Lucas WillianNo ratings yet
- High Resistance To Oxidative Stress in The Fungal PathogenDocument12 pagesHigh Resistance To Oxidative Stress in The Fungal PathogenIvica PutrićNo ratings yet
- Alazem 2014 Roles of Plant Hormones in The Regulation of Host Virus InteractionsDocument12 pagesAlazem 2014 Roles of Plant Hormones in The Regulation of Host Virus InteractionsTriEka HeryaNo ratings yet
- Perception and Transduction of Abscisic Acid Signals: Keys To The Function of The Versatile Plant Hormone ABADocument9 pagesPerception and Transduction of Abscisic Acid Signals: Keys To The Function of The Versatile Plant Hormone ABAAnirban KunduNo ratings yet
- Pettko-Szandtner Et AlDocument13 pagesPettko-Szandtner Et AlAladár Pettkó-SzandtnerNo ratings yet
- Involvement of The Rab27 Binding Protein Slac2C/Myrip in Insulin ExocytosisDocument11 pagesInvolvement of The Rab27 Binding Protein Slac2C/Myrip in Insulin ExocytosisJoseGonzalezNo ratings yet
- Cárdenas-Manríquez Et Al. - 2016 - Environmental and Experimental BotanyDocument9 pagesCárdenas-Manríquez Et Al. - 2016 - Environmental and Experimental BotanyIsaac VegaNo ratings yet
- Abuaish 2021 The Role of Sex Differentiated Variations in Stress Hormones Antiox Neuroimmun Res in Rel To Soc Int Impair in Rodent Model of AutismDocument11 pagesAbuaish 2021 The Role of Sex Differentiated Variations in Stress Hormones Antiox Neuroimmun Res in Rel To Soc Int Impair in Rodent Model of AutismJortegloriaNo ratings yet
- 9 Sudemycins 2011Document8 pages9 Sudemycins 2011milenerato2240No ratings yet
- Gopika Suresh - Ims20121 Assignment 1Document3 pagesGopika Suresh - Ims20121 Assignment 1Gopika SureshNo ratings yet
- Chemical Composition, Antioxidant and Anticholinesterase Activity of MelissaDocument12 pagesChemical Composition, Antioxidant and Anticholinesterase Activity of MelissaOanaCristeaNo ratings yet
- Genomic Analysis of MAP Kinase Cascades In: Arabidopsis Defense ResponsesDocument13 pagesGenomic Analysis of MAP Kinase Cascades In: Arabidopsis Defense ResponsesMelvin PrasadNo ratings yet
- Dietary Regulation of Keap1 Nrf2 ARE Pathway NFKB.2014 GOODDocument25 pagesDietary Regulation of Keap1 Nrf2 ARE Pathway NFKB.2014 GOODBeirnes Fernando Sembiring M100% (1)
- Chirality, Drug Metabolism and ActionDocument7 pagesChirality, Drug Metabolism and ActionBrahma Hakim Yuanda HutabaratNo ratings yet
- 016 Scas Abstracts Listed Alphabetically by Abstractspdf 19 2016 Scas AbstractsDocument25 pages016 Scas Abstracts Listed Alphabetically by Abstractspdf 19 2016 Scas AbstractsdssagNo ratings yet
- Overexpression of Calmodulin-Like (Shcml44) Stress-Responsive Gene From Solanum Habrochaites Enhances Tolerance To Multiple Abiotic StressesDocument20 pagesOverexpression of Calmodulin-Like (Shcml44) Stress-Responsive Gene From Solanum Habrochaites Enhances Tolerance To Multiple Abiotic StressesFarahDeebaNo ratings yet
- Evaluation of Potential Antioxidant and Anti Inflammatory Effects - 2018 - PoultDocument16 pagesEvaluation of Potential Antioxidant and Anti Inflammatory Effects - 2018 - Poultlady mae rufinoNo ratings yet
- Tezisfuzet AngolDocument9 pagesTezisfuzet AngolYudistira PratamaNo ratings yet
- Centella Asiatica and Its Fractions Reduces Lipid Peroxidation Induced by Quinolinic Acid and Sodium Nitroprusside in Rat Brain RegionsDocument15 pagesCentella Asiatica and Its Fractions Reduces Lipid Peroxidation Induced by Quinolinic Acid and Sodium Nitroprusside in Rat Brain RegionsrhinaNo ratings yet
- Bioengineering For Salinity Tolerance in Plants State of The ArtDocument22 pagesBioengineering For Salinity Tolerance in Plants State of The ArtCesarRamNo ratings yet
- Sanchez Et Al 2010Document12 pagesSanchez Et Al 2010minervaNo ratings yet
- Campos Proposal 2012Document23 pagesCampos Proposal 2012Asim MughalNo ratings yet
- 2008 Smith-et-Al JA SA HerbivoryDocument7 pages2008 Smith-et-Al JA SA HerbivoryPriscila MezzomoNo ratings yet
- Lukaszuk2016 Article PhotosynthesisAndSucroseMetaboDocument12 pagesLukaszuk2016 Article PhotosynthesisAndSucroseMetaboghilbrania salsabilaNo ratings yet
- Food and Chemical Toxicology: Article InfoDocument7 pagesFood and Chemical Toxicology: Article InfoNathalia MojicaNo ratings yet
- To Blastand Leaf Blight Diseasesin RiceDocument10 pagesTo Blastand Leaf Blight Diseasesin RicevenkatscribdusernameNo ratings yet
- J. Biol. Chem.-2011-Pan-14575-87Document14 pagesJ. Biol. Chem.-2011-Pan-14575-87Henrique HolandaNo ratings yet
- Structure Activity Relationship: Related TermsDocument18 pagesStructure Activity Relationship: Related TermsSajjad Hossen ChowdhuryNo ratings yet
- Accepted Manuscript: Environmental and Experimental BotanyDocument29 pagesAccepted Manuscript: Environmental and Experimental BotanyRenganathan PrabhaharanNo ratings yet
- Erv 013Document13 pagesErv 013Aisha MurtazaNo ratings yet
- Changes in Membrane Fatty Acids Composition of MicrobialDocument5 pagesChanges in Membrane Fatty Acids Composition of MicrobialDaniel JimenezNo ratings yet
- Grant 1997Document10 pagesGrant 1997Django BoyeeNo ratings yet
- tmpF178 TMPDocument15 pagestmpF178 TMPFrontiersNo ratings yet
- Tmp1a96 TMPDocument80 pagesTmp1a96 TMPFrontiersNo ratings yet
- Tmpa077 TMPDocument15 pagesTmpa077 TMPFrontiersNo ratings yet
- tmpE3C0 TMPDocument17 pagestmpE3C0 TMPFrontiersNo ratings yet
- tmp27C1 TMPDocument5 pagestmp27C1 TMPFrontiersNo ratings yet
- tmp998 TMPDocument9 pagestmp998 TMPFrontiersNo ratings yet
- tmp3656 TMPDocument14 pagestmp3656 TMPFrontiersNo ratings yet
- tmp96F2 TMPDocument4 pagestmp96F2 TMPFrontiersNo ratings yet
- tmpA7D0 TMPDocument9 pagestmpA7D0 TMPFrontiersNo ratings yet
- tmp97C8 TMPDocument9 pagestmp97C8 TMPFrontiersNo ratings yet
- PROLYMERDocument2 pagesPROLYMERFlavio GonzalezNo ratings yet
- Outline Report PFR 2015 2018 CASESDocument5 pagesOutline Report PFR 2015 2018 CASESPaul ValerosNo ratings yet
- Navigating The Cooling, Heating and Reheat Process On The Psychrometric ChartDocument11 pagesNavigating The Cooling, Heating and Reheat Process On The Psychrometric ChartmohdkamalhaziqNo ratings yet
- Kebersihan Gigi Mulut Dan Kejadian Gingivitis Pada Anak Sekolah DasarDocument8 pagesKebersihan Gigi Mulut Dan Kejadian Gingivitis Pada Anak Sekolah DasarDeri Atau DariNo ratings yet
- Mwfly Business Magazine EN - CompressedDocument25 pagesMwfly Business Magazine EN - CompressedinfoNo ratings yet
- Physiology Past PapersDocument11 pagesPhysiology Past PapersIrfan Haider FaniNo ratings yet
- Acp CBC 1Document52 pagesAcp CBC 1Danny R. SalvadorNo ratings yet
- Delayed-Action AcceleratorDocument3 pagesDelayed-Action AcceleratorMOHD FIRDAUSNo ratings yet
- Nottingham Hip Fracture Score As A Predictor of One Year PDFDocument4 pagesNottingham Hip Fracture Score As A Predictor of One Year PDFjavieccrNo ratings yet
- 1 Introduction To Management Copy 2Document21 pages1 Introduction To Management Copy 2Angelica Camille B. AbaoNo ratings yet
- 10.A.Cools Kinetic Chain PDFDocument22 pages10.A.Cools Kinetic Chain PDFWalter PelaezNo ratings yet
- Conditioner Portatil Sams ClubDocument5 pagesConditioner Portatil Sams ClubJuan Carlos RodriguezNo ratings yet
- PDF SSCP Systems Security Certified Practitioner All in One Exam Guide 3Rd Edition Gibson Ebook Full ChapterDocument53 pagesPDF SSCP Systems Security Certified Practitioner All in One Exam Guide 3Rd Edition Gibson Ebook Full Chapterdiane.vant368100% (2)
- Ich Guidelines For Stability Testing of New Drug Substance and Drug ProductsDocument39 pagesIch Guidelines For Stability Testing of New Drug Substance and Drug ProductsRahul LakhaniNo ratings yet
- Discussion Q1Document7 pagesDiscussion Q1TASHANo ratings yet
- Motivation BisDocument20 pagesMotivation BishùhjgNo ratings yet
- Pipeline Defect Repair MethodsDocument20 pagesPipeline Defect Repair MethodsAyon RoyNo ratings yet
- RENOLD (Worm Gear - Models PH)Document60 pagesRENOLD (Worm Gear - Models PH)Rodrigo Leal GuzmanNo ratings yet
- Pilzzucht PDFDocument68 pagesPilzzucht PDFSu Per BingNo ratings yet
- Engelhardt BioethicsDocument9 pagesEngelhardt BioethicsAaron Lee WiegmannNo ratings yet
- Dup TemplateDocument5 pagesDup TemplateJoshua SouthNo ratings yet
- DCP-CZM (Detectores de Humo Convencionales)Document2 pagesDCP-CZM (Detectores de Humo Convencionales)alexropaNo ratings yet
- Personal FinanceDocument32 pagesPersonal FinancePrincess MogulNo ratings yet
- Lifting Operations AZDP Directive 008 - Edition 4.0: U:TobeusedasisDocument7 pagesLifting Operations AZDP Directive 008 - Edition 4.0: U:TobeusedasisSiddharth AsthanaNo ratings yet
- Sol-Gel Methods PDFDocument19 pagesSol-Gel Methods PDFDimitrios Tsiplakides100% (2)
- Mobile Crane SafetyDocument3 pagesMobile Crane SafetyAnonymous LFgO4WbID0% (1)
- 01 91 13 General Commissioning RequirementsDocument25 pages01 91 13 General Commissioning Requirementsvin ssNo ratings yet
- Chapter 6 Hexavalent ChromiumDocument56 pagesChapter 6 Hexavalent Chromiumgabriel100% (1)