Professional Documents
Culture Documents
0 ratings0% found this document useful (0 votes)
10 viewsCalculation of Relative Molecular Mass
Calculation of Relative Molecular Mass
Uploaded by
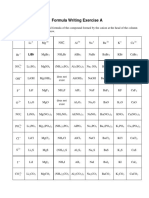
teoh6234This document lists common chemical formulas and calculates the relative molecular mass of hydrated copper(II) sulphate. It contains the formulas for zinc nitrate, aluminium nitrate, calcium carbonate, calcium hydroxide, hydrated copper(II) sulphate, hydrated sodium carbonate, sodium hydrogen sulphate, aluminium chloride, copper(II) sulphate, zinc carbonate, and potassium carbonate. It also shows the calculation of the relative molecular mass of hydrated copper(II) sulphate as being 250.
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
You might also like
- Chemical Formula List For Class 10Document12 pagesChemical Formula List For Class 10The Coding World100% (4)
- Chemical Formula Writing Worksheet SolutionsDocument3 pagesChemical Formula Writing Worksheet SolutionsReid Pineda Creencia100% (1)
- Chemical Formula Writing WorksheetDocument4 pagesChemical Formula Writing Worksheetprabhu4321100% (1)
- Symbol - Equations - Homework RMDocument2 pagesSymbol - Equations - Homework RMayaanrayhaanNo ratings yet
- Chemistry Notes (14 - 4 - 23)Document4 pagesChemistry Notes (14 - 4 - 23)Sanchay KumarNo ratings yet
- Booklet On Acid and Base and Redox MSDocument43 pagesBooklet On Acid and Base and Redox MShalahossam8899No ratings yet
- Predicting Reaction Products and NamingDocument2 pagesPredicting Reaction Products and Namingbistakenx2No ratings yet
- Ionic Compounds Worksheet-IiiDocument1 pageIonic Compounds Worksheet-IiitylerNo ratings yet
- Chemical Formulas List For Class 10Document5 pagesChemical Formulas List For Class 10Akshita Kamboj91% (65)
- ReactantsDocument2 pagesReactantsNick BesterNo ratings yet
- Chemical Formula - Oxidation NumberDocument47 pagesChemical Formula - Oxidation NumberZheng JoeyNo ratings yet
- Formulation and Balancing AnswersDocument3 pagesFormulation and Balancing AnswerskayraozlemNo ratings yet
- Howtowritechemicalformulacomplete 110302230114 Phpapp01Document86 pagesHowtowritechemicalformulacomplete 110302230114 Phpapp01VladimirNo ratings yet
- Baso Agno H S Cao H Co MG (Po) K Cro NaiDocument2 pagesBaso Agno H S Cao H Co MG (Po) K Cro NaiAmy BalicagNo ratings yet
- Chapter - 7 Correction Naming CompoundsDocument2 pagesChapter - 7 Correction Naming CompoundsMurad IsayevNo ratings yet
- 8A Salts - AnswerDocument14 pages8A Salts - AnswerWong Wai LunNo ratings yet
- List of Common CompoundsDocument9 pagesList of Common CompoundsEvann Myelle MontejoNo ratings yet
- CHEMICAL EQUATIONS I Balance Equation ExcDocument5 pagesCHEMICAL EQUATIONS I Balance Equation ExcMohd Hairil HaikalNo ratings yet
- Formula Kimia Dan Nama Bahan KimiaDocument2 pagesFormula Kimia Dan Nama Bahan Kimiahansm100% (5)
- Eleazar - Quiz#3Document2 pagesEleazar - Quiz#3ゆかりNo ratings yet
- Solubility ListDocument6 pagesSolubility ListpomegranatesoupNo ratings yet
- Chemical FormulaeDocument3 pagesChemical Formulaeparth chakre100% (2)
- Chemistry: Self Access Learning SheetDocument8 pagesChemistry: Self Access Learning SheetNooraini HusseinNo ratings yet
- Garam Bab 8Document29 pagesGaram Bab 8ctohNo ratings yet
- Balancing Word Equations PracticeDocument3 pagesBalancing Word Equations PracticemmNo ratings yet
- Post 5.9. Ionic Compounds Practice - ANSWERSDocument3 pagesPost 5.9. Ionic Compounds Practice - ANSWERSAlan MartínNo ratings yet
- LMBNG Unsr, TTK DDH, LLHDocument3 pagesLMBNG Unsr, TTK DDH, LLHRoselinaNo ratings yet
- Molecular Ionic and Net IonicDocument7 pagesMolecular Ionic and Net IonicFarz21No ratings yet
- Molecular Ionic and Net IonicDocument7 pagesMolecular Ionic and Net IonicFarz21No ratings yet
- Write The Formulas For The Following Ionic Compounds:: Bonding and Naming WS 4Document2 pagesWrite The Formulas For The Following Ionic Compounds:: Bonding and Naming WS 4Bea Lha Zandra BesingaNo ratings yet
- Worksheet Balancing Word EquationsDocument2 pagesWorksheet Balancing Word EquationsSanti SamNo ratings yet
- Chemical Substance Formula: List of Chemical Substances With FormulaeDocument3 pagesChemical Substance Formula: List of Chemical Substances With FormulaeJaithri Mualakala100% (1)
- Nomenclature WS PennysmileDocument4 pagesNomenclature WS PennysmileMarilee HuntNo ratings yet
- Chemical Formula Writing WorksheetDocument5 pagesChemical Formula Writing WorksheetÂziz ShuvoNo ratings yet
- Simbol KimiaDocument8 pagesSimbol KimiaMOhd HisyamNo ratings yet
- Inorganic Nomenclature and StoichiometryDocument4 pagesInorganic Nomenclature and StoichiometryTjandrawati NugrahaNo ratings yet
- Chemical Formula Writing Worksheet2Document2 pagesChemical Formula Writing Worksheet2عابدهعلي100% (1)
- Salts PreparationDocument7 pagesSalts PreparationCynthia RoneyNo ratings yet
- QC1 - Naming Covalent Compounds and Ionic FormulaeDocument5 pagesQC1 - Naming Covalent Compounds and Ionic Formulaeykame4096No ratings yet
- Naming Inorganic Compound Practice SheetDocument4 pagesNaming Inorganic Compound Practice SheetWichel AnnNo ratings yet
- Naming Compounds Formulas For The Following Ionic CompoundsDocument1 pageNaming Compounds Formulas For The Following Ionic CompoundsJulie Trajano CortezNo ratings yet
- Inorganic Compounds: Chemical Name Chemical FormulaDocument6 pagesInorganic Compounds: Chemical Name Chemical FormulaFrendick LegaspiNo ratings yet
- IUPAC Name: Give The Chemical Formula For Each of The FollowingDocument1 pageIUPAC Name: Give The Chemical Formula For Each of The FollowingKiki ShofiaNo ratings yet
- Chemical Formula WorksheetDocument4 pagesChemical Formula WorksheetMaria adeelNo ratings yet
- Activity 8 NomenclatureDocument2 pagesActivity 8 NomenclatureCyruss MeranoNo ratings yet
- 2.4.3 Chemical Formula and Naming Practice QuestionsDocument7 pages2.4.3 Chemical Formula and Naming Practice Questionsphat.vuongNo ratings yet
- Write The Name of The Derived FormulaDocument1 pageWrite The Name of The Derived Formulaמתבונן בך אלוהיםNo ratings yet
- Chem16 Experiment # 1: Chemical Changes Post-Lab Discussion: 3 2 (Aq) (Aq) 2 (S) 3 (Aq) 2+ (Aq) - (Aq) 2 (S)Document1 pageChem16 Experiment # 1: Chemical Changes Post-Lab Discussion: 3 2 (Aq) (Aq) 2 (S) 3 (Aq) 2+ (Aq) - (Aq) 2 (S)DiyanikaNo ratings yet
- 2 WeekDocument2 pages2 WeekAna Carballo TrabazoNo ratings yet
- Nomenclature Assignment Part 1Document4 pagesNomenclature Assignment Part 1marNo ratings yet
- Jordan Paddock - Writing Polyatomic Names and Formulas WorksheetDocument1 pageJordan Paddock - Writing Polyatomic Names and Formulas Worksheetapi-675312232No ratings yet
- Laboratory Inventory 1 Chemistry Lab ChemicalsDocument4 pagesLaboratory Inventory 1 Chemistry Lab Chemicalspdqcmusa54No ratings yet
- Solution of Salt Analysis-13thDocument8 pagesSolution of Salt Analysis-13thRaju SinghNo ratings yet
- Selected Solubility Product Constants at 25°CDocument1 pageSelected Solubility Product Constants at 25°CJampier32No ratings yet
- Chemical Formulas Revision Activity Worksheet Yr10Document2 pagesChemical Formulas Revision Activity Worksheet Yr10Chloe WNo ratings yet
- Formula Writing - CambridgeDocument5 pagesFormula Writing - CambridgeQusai Saify100% (3)
- Valency Table: Cations (Positive Ions) Anions (Negative Ions)Document1 pageValency Table: Cations (Positive Ions) Anions (Negative Ions)ama100% (2)
- Chemical FormulaDocument1 pageChemical Formulaapi-516886039No ratings yet
- Compound Name Molecular Formula Compound Name Molecular FormulaDocument4 pagesCompound Name Molecular Formula Compound Name Molecular Formulamohamed ahmedNo ratings yet
- Main - Acids Bases and Salts Formulas and Names Cheat SheetDocument2 pagesMain - Acids Bases and Salts Formulas and Names Cheat SheetLorens NorNo ratings yet
- Intensive Revision SP 2019 NEWESTDocument1 pageIntensive Revision SP 2019 NEWESTteoh6234No ratings yet
- Solubility Equilibria: End of Chapter 16Document8 pagesSolubility Equilibria: End of Chapter 16teoh6234No ratings yet
- Ch39 Periodic Relationship Among The OxidesDocument69 pagesCh39 Periodic Relationship Among The Oxidesteoh6234No ratings yet
- STPM Trial 2012 Chemistry Qa KelantanDocument42 pagesSTPM Trial 2012 Chemistry Qa Kelantanteoh6234100% (2)
- A Sample of M Oxide Contains 2.7 G of Element X and 2.4 G of Oxygen. What Is The Empirical Formula of This Compound? (Relative Atomic Mass: X 27, O 16)Document1 pageA Sample of M Oxide Contains 2.7 G of Element X and 2.4 G of Oxygen. What Is The Empirical Formula of This Compound? (Relative Atomic Mass: X 27, O 16)teoh6234No ratings yet
- Chapter 2: Matter: Temperature, CDocument1 pageChapter 2: Matter: Temperature, Cteoh6234No ratings yet
- New AccountDocument1 pageNew Accountteoh6234No ratings yet
- Date:: Private Retirement Scheme Account Detai LSDocument1 pageDate:: Private Retirement Scheme Account Detai LSteoh6234No ratings yet
Calculation of Relative Molecular Mass
Calculation of Relative Molecular Mass
Uploaded by
teoh62340 ratings0% found this document useful (0 votes)
10 views1 pageThis document lists common chemical formulas and calculates the relative molecular mass of hydrated copper(II) sulphate. It contains the formulas for zinc nitrate, aluminium nitrate, calcium carbonate, calcium hydroxide, hydrated copper(II) sulphate, hydrated sodium carbonate, sodium hydrogen sulphate, aluminium chloride, copper(II) sulphate, zinc carbonate, and potassium carbonate. It also shows the calculation of the relative molecular mass of hydrated copper(II) sulphate as being 250.
Original Description:
Relativie Molecular Mass
Original Title
Relativie Molecular Mass
Copyright
© © All Rights Reserved
Available Formats
DOCX, PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentThis document lists common chemical formulas and calculates the relative molecular mass of hydrated copper(II) sulphate. It contains the formulas for zinc nitrate, aluminium nitrate, calcium carbonate, calcium hydroxide, hydrated copper(II) sulphate, hydrated sodium carbonate, sodium hydrogen sulphate, aluminium chloride, copper(II) sulphate, zinc carbonate, and potassium carbonate. It also shows the calculation of the relative molecular mass of hydrated copper(II) sulphate as being 250.
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
Download as docx, pdf, or txt
0 ratings0% found this document useful (0 votes)
10 views1 pageCalculation of Relative Molecular Mass
Calculation of Relative Molecular Mass
Uploaded by
teoh6234This document lists common chemical formulas and calculates the relative molecular mass of hydrated copper(II) sulphate. It contains the formulas for zinc nitrate, aluminium nitrate, calcium carbonate, calcium hydroxide, hydrated copper(II) sulphate, hydrated sodium carbonate, sodium hydrogen sulphate, aluminium chloride, copper(II) sulphate, zinc carbonate, and potassium carbonate. It also shows the calculation of the relative molecular mass of hydrated copper(II) sulphate as being 250.
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
Download as docx, pdf, or txt
You are on page 1of 1
Zinc nitrate Zn(NO3)2
Aluminium nitrate Al(NO3)3
Calcium carbonate CaCO3
Calcium hydroxide Ca(OH)2
Hydrated copper(II) CuSO4.5H2O 64 + 32 + 4(16) + 5[2(1) + 16]=250
sulphate
Hydrated sodium Na2CO3.10H2O
carbonate
Sodium hydrogen NaHSO4
sulphate
Aluminium chloride AlCl3
Copper(II) sulphate CuSO4
Zinc carbonate ZnCO3
Potassium K2CO3
carbonate
CALCULATION OF RELATIVE MOLECULAR MASS
You might also like
- Chemical Formula List For Class 10Document12 pagesChemical Formula List For Class 10The Coding World100% (4)
- Chemical Formula Writing Worksheet SolutionsDocument3 pagesChemical Formula Writing Worksheet SolutionsReid Pineda Creencia100% (1)
- Chemical Formula Writing WorksheetDocument4 pagesChemical Formula Writing Worksheetprabhu4321100% (1)
- Symbol - Equations - Homework RMDocument2 pagesSymbol - Equations - Homework RMayaanrayhaanNo ratings yet
- Chemistry Notes (14 - 4 - 23)Document4 pagesChemistry Notes (14 - 4 - 23)Sanchay KumarNo ratings yet
- Booklet On Acid and Base and Redox MSDocument43 pagesBooklet On Acid and Base and Redox MShalahossam8899No ratings yet
- Predicting Reaction Products and NamingDocument2 pagesPredicting Reaction Products and Namingbistakenx2No ratings yet
- Ionic Compounds Worksheet-IiiDocument1 pageIonic Compounds Worksheet-IiitylerNo ratings yet
- Chemical Formulas List For Class 10Document5 pagesChemical Formulas List For Class 10Akshita Kamboj91% (65)
- ReactantsDocument2 pagesReactantsNick BesterNo ratings yet
- Chemical Formula - Oxidation NumberDocument47 pagesChemical Formula - Oxidation NumberZheng JoeyNo ratings yet
- Formulation and Balancing AnswersDocument3 pagesFormulation and Balancing AnswerskayraozlemNo ratings yet
- Howtowritechemicalformulacomplete 110302230114 Phpapp01Document86 pagesHowtowritechemicalformulacomplete 110302230114 Phpapp01VladimirNo ratings yet
- Baso Agno H S Cao H Co MG (Po) K Cro NaiDocument2 pagesBaso Agno H S Cao H Co MG (Po) K Cro NaiAmy BalicagNo ratings yet
- Chapter - 7 Correction Naming CompoundsDocument2 pagesChapter - 7 Correction Naming CompoundsMurad IsayevNo ratings yet
- 8A Salts - AnswerDocument14 pages8A Salts - AnswerWong Wai LunNo ratings yet
- List of Common CompoundsDocument9 pagesList of Common CompoundsEvann Myelle MontejoNo ratings yet
- CHEMICAL EQUATIONS I Balance Equation ExcDocument5 pagesCHEMICAL EQUATIONS I Balance Equation ExcMohd Hairil HaikalNo ratings yet
- Formula Kimia Dan Nama Bahan KimiaDocument2 pagesFormula Kimia Dan Nama Bahan Kimiahansm100% (5)
- Eleazar - Quiz#3Document2 pagesEleazar - Quiz#3ゆかりNo ratings yet
- Solubility ListDocument6 pagesSolubility ListpomegranatesoupNo ratings yet
- Chemical FormulaeDocument3 pagesChemical Formulaeparth chakre100% (2)
- Chemistry: Self Access Learning SheetDocument8 pagesChemistry: Self Access Learning SheetNooraini HusseinNo ratings yet
- Garam Bab 8Document29 pagesGaram Bab 8ctohNo ratings yet
- Balancing Word Equations PracticeDocument3 pagesBalancing Word Equations PracticemmNo ratings yet
- Post 5.9. Ionic Compounds Practice - ANSWERSDocument3 pagesPost 5.9. Ionic Compounds Practice - ANSWERSAlan MartínNo ratings yet
- LMBNG Unsr, TTK DDH, LLHDocument3 pagesLMBNG Unsr, TTK DDH, LLHRoselinaNo ratings yet
- Molecular Ionic and Net IonicDocument7 pagesMolecular Ionic and Net IonicFarz21No ratings yet
- Molecular Ionic and Net IonicDocument7 pagesMolecular Ionic and Net IonicFarz21No ratings yet
- Write The Formulas For The Following Ionic Compounds:: Bonding and Naming WS 4Document2 pagesWrite The Formulas For The Following Ionic Compounds:: Bonding and Naming WS 4Bea Lha Zandra BesingaNo ratings yet
- Worksheet Balancing Word EquationsDocument2 pagesWorksheet Balancing Word EquationsSanti SamNo ratings yet
- Chemical Substance Formula: List of Chemical Substances With FormulaeDocument3 pagesChemical Substance Formula: List of Chemical Substances With FormulaeJaithri Mualakala100% (1)
- Nomenclature WS PennysmileDocument4 pagesNomenclature WS PennysmileMarilee HuntNo ratings yet
- Chemical Formula Writing WorksheetDocument5 pagesChemical Formula Writing WorksheetÂziz ShuvoNo ratings yet
- Simbol KimiaDocument8 pagesSimbol KimiaMOhd HisyamNo ratings yet
- Inorganic Nomenclature and StoichiometryDocument4 pagesInorganic Nomenclature and StoichiometryTjandrawati NugrahaNo ratings yet
- Chemical Formula Writing Worksheet2Document2 pagesChemical Formula Writing Worksheet2عابدهعلي100% (1)
- Salts PreparationDocument7 pagesSalts PreparationCynthia RoneyNo ratings yet
- QC1 - Naming Covalent Compounds and Ionic FormulaeDocument5 pagesQC1 - Naming Covalent Compounds and Ionic Formulaeykame4096No ratings yet
- Naming Inorganic Compound Practice SheetDocument4 pagesNaming Inorganic Compound Practice SheetWichel AnnNo ratings yet
- Naming Compounds Formulas For The Following Ionic CompoundsDocument1 pageNaming Compounds Formulas For The Following Ionic CompoundsJulie Trajano CortezNo ratings yet
- Inorganic Compounds: Chemical Name Chemical FormulaDocument6 pagesInorganic Compounds: Chemical Name Chemical FormulaFrendick LegaspiNo ratings yet
- IUPAC Name: Give The Chemical Formula For Each of The FollowingDocument1 pageIUPAC Name: Give The Chemical Formula For Each of The FollowingKiki ShofiaNo ratings yet
- Chemical Formula WorksheetDocument4 pagesChemical Formula WorksheetMaria adeelNo ratings yet
- Activity 8 NomenclatureDocument2 pagesActivity 8 NomenclatureCyruss MeranoNo ratings yet
- 2.4.3 Chemical Formula and Naming Practice QuestionsDocument7 pages2.4.3 Chemical Formula and Naming Practice Questionsphat.vuongNo ratings yet
- Write The Name of The Derived FormulaDocument1 pageWrite The Name of The Derived Formulaמתבונן בך אלוהיםNo ratings yet
- Chem16 Experiment # 1: Chemical Changes Post-Lab Discussion: 3 2 (Aq) (Aq) 2 (S) 3 (Aq) 2+ (Aq) - (Aq) 2 (S)Document1 pageChem16 Experiment # 1: Chemical Changes Post-Lab Discussion: 3 2 (Aq) (Aq) 2 (S) 3 (Aq) 2+ (Aq) - (Aq) 2 (S)DiyanikaNo ratings yet
- 2 WeekDocument2 pages2 WeekAna Carballo TrabazoNo ratings yet
- Nomenclature Assignment Part 1Document4 pagesNomenclature Assignment Part 1marNo ratings yet
- Jordan Paddock - Writing Polyatomic Names and Formulas WorksheetDocument1 pageJordan Paddock - Writing Polyatomic Names and Formulas Worksheetapi-675312232No ratings yet
- Laboratory Inventory 1 Chemistry Lab ChemicalsDocument4 pagesLaboratory Inventory 1 Chemistry Lab Chemicalspdqcmusa54No ratings yet
- Solution of Salt Analysis-13thDocument8 pagesSolution of Salt Analysis-13thRaju SinghNo ratings yet
- Selected Solubility Product Constants at 25°CDocument1 pageSelected Solubility Product Constants at 25°CJampier32No ratings yet
- Chemical Formulas Revision Activity Worksheet Yr10Document2 pagesChemical Formulas Revision Activity Worksheet Yr10Chloe WNo ratings yet
- Formula Writing - CambridgeDocument5 pagesFormula Writing - CambridgeQusai Saify100% (3)
- Valency Table: Cations (Positive Ions) Anions (Negative Ions)Document1 pageValency Table: Cations (Positive Ions) Anions (Negative Ions)ama100% (2)
- Chemical FormulaDocument1 pageChemical Formulaapi-516886039No ratings yet
- Compound Name Molecular Formula Compound Name Molecular FormulaDocument4 pagesCompound Name Molecular Formula Compound Name Molecular Formulamohamed ahmedNo ratings yet
- Main - Acids Bases and Salts Formulas and Names Cheat SheetDocument2 pagesMain - Acids Bases and Salts Formulas and Names Cheat SheetLorens NorNo ratings yet
- Intensive Revision SP 2019 NEWESTDocument1 pageIntensive Revision SP 2019 NEWESTteoh6234No ratings yet
- Solubility Equilibria: End of Chapter 16Document8 pagesSolubility Equilibria: End of Chapter 16teoh6234No ratings yet
- Ch39 Periodic Relationship Among The OxidesDocument69 pagesCh39 Periodic Relationship Among The Oxidesteoh6234No ratings yet
- STPM Trial 2012 Chemistry Qa KelantanDocument42 pagesSTPM Trial 2012 Chemistry Qa Kelantanteoh6234100% (2)
- A Sample of M Oxide Contains 2.7 G of Element X and 2.4 G of Oxygen. What Is The Empirical Formula of This Compound? (Relative Atomic Mass: X 27, O 16)Document1 pageA Sample of M Oxide Contains 2.7 G of Element X and 2.4 G of Oxygen. What Is The Empirical Formula of This Compound? (Relative Atomic Mass: X 27, O 16)teoh6234No ratings yet
- Chapter 2: Matter: Temperature, CDocument1 pageChapter 2: Matter: Temperature, Cteoh6234No ratings yet
- New AccountDocument1 pageNew Accountteoh6234No ratings yet
- Date:: Private Retirement Scheme Account Detai LSDocument1 pageDate:: Private Retirement Scheme Account Detai LSteoh6234No ratings yet