Professional Documents
Culture Documents
Hidrolisis & Penyangga
Hidrolisis & Penyangga
Uploaded by
Ardhi Omen0 ratings0% found this document useful (0 votes)
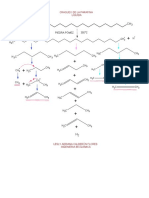
5 views1 pageAcid + base → salt + water. Specifically, acetic acid (CH3COOH) plus sodium hydroxide (NaOH) yields sodium acetate (CH3COONa) plus water (H2O) through a process called hydrolysis. This chemical reaction demonstrates that a weak acid plus a strong base produces a salt and water.
Original Description:
soal
Original Title
HIDROLISIS & PENYANGGA
Copyright
© © All Rights Reserved
Available Formats
DOC, PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentAcid + base → salt + water. Specifically, acetic acid (CH3COOH) plus sodium hydroxide (NaOH) yields sodium acetate (CH3COONa) plus water (H2O) through a process called hydrolysis. This chemical reaction demonstrates that a weak acid plus a strong base produces a salt and water.
Copyright:
© All Rights Reserved
Available Formats
Download as DOC, PDF, TXT or read online from Scribd
Download as doc, pdf, or txt
0 ratings0% found this document useful (0 votes)
5 views1 pageHidrolisis & Penyangga
Hidrolisis & Penyangga
Uploaded by
Ardhi OmenAcid + base → salt + water. Specifically, acetic acid (CH3COOH) plus sodium hydroxide (NaOH) yields sodium acetate (CH3COONa) plus water (H2O) through a process called hydrolysis. This chemical reaction demonstrates that a weak acid plus a strong base produces a salt and water.
Copyright:
© All Rights Reserved
Available Formats
Download as DOC, PDF, TXT or read online from Scribd
Download as doc, pdf, or txt
You are on page 1of 1
HIDROLISIS
ASAM LEMAH + BASA KUAT GARAM + H2O
CH3COOH + NaOH CH3COONa + H2O
You might also like
- Chemsheets KS3 063 Reactions of AcidsDocument1 pageChemsheets KS3 063 Reactions of AcidsNour BersyNo ratings yet
- Quiz BalanceoDocument7 pagesQuiz BalanceoCecilia Delgado GaleanoNo ratings yet
- Sodium HydroxideDocument3 pagesSodium HydroxideISLAM I. Fekry100% (2)
- Cramming NotesDocument7 pagesCramming NotesMuhammad ali WasimNo ratings yet
- PDocument4 pagesPIca zaharaNo ratings yet
- Balance Equation4Document1 pageBalance Equation4Amal JavedNo ratings yet
- STPM Chemistry Topic 16 Haloalkanes Short Notes PDFDocument2 pagesSTPM Chemistry Topic 16 Haloalkanes Short Notes PDFbendanNo ratings yet
- STPM Chemistry Topic 16 Haloalkanes (Short Notes)Document2 pagesSTPM Chemistry Topic 16 Haloalkanes (Short Notes)Chris LauNo ratings yet
- Rumus Asam Nama Asam Rumus Ionisasi KeteranganDocument4 pagesRumus Asam Nama Asam Rumus Ionisasi Keteranganelsa noviyantiNo ratings yet
- NOTES Inorganic ReactionsDocument43 pagesNOTES Inorganic Reactionsvaishnavi singhNo ratings yet
- As Equations: Lithium Monoxide (Contains O) Sodium Peroxide (Contains O) Potassium Superoxide (Contains O)Document4 pagesAs Equations: Lithium Monoxide (Contains O) Sodium Peroxide (Contains O) Potassium Superoxide (Contains O)arcmikeNo ratings yet
- Inter-Link Chapter 11-DwiDocument1 pageInter-Link Chapter 11-Dwirudi_zNo ratings yet
- Flow Chart of Organic Reactions: Substitution (+NHDocument1 pageFlow Chart of Organic Reactions: Substitution (+NHAhhhhhhhhhhhNo ratings yet
- Grap Nhom Halogen Hay So 1Document4 pagesGrap Nhom Halogen Hay So 1Anonymous hsV1btNo ratings yet
- CH 10Document11 pagesCH 10Vishal KushwahNo ratings yet
- Red Ox BalancingDocument2 pagesRed Ox BalancingLuis Panduro AlvaNo ratings yet
- Aspirin Cheat SheetDocument2 pagesAspirin Cheat SheetReacher ElliotNo ratings yet
- Environmental Engineering Industrial WaterDocument28 pagesEnvironmental Engineering Industrial WaterMd Afif AbrarNo ratings yet
- Science 1.5 Answers Worksheet Five Acids: Acid + Base Salt + WaterDocument2 pagesScience 1.5 Answers Worksheet Five Acids: Acid + Base Salt + Watermintesinot esayasNo ratings yet
- Redox ReactionDocument3 pagesRedox ReactionPranjal GuptaNo ratings yet
- SPM Chemistry Formula List Form5 PDFDocument15 pagesSPM Chemistry Formula List Form5 PDFshuyiNo ratings yet
- Reactii RedoxDocument1 pageReactii RedoxMaria MihaelaNo ratings yet
- Activity No. 3 Worksheet Types of Redox ReactionDocument3 pagesActivity No. 3 Worksheet Types of Redox ReactionMaynard CortezNo ratings yet
- Arrangement of Coefficients in The Equations of Redox ReactionsDocument11 pagesArrangement of Coefficients in The Equations of Redox Reactionslorenlori2004No ratings yet
- Uace Chem Guide To Mechanism and SynthesisDocument60 pagesUace Chem Guide To Mechanism and SynthesisNelima Stella mercyNo ratings yet
- Uace Chem Guide To Mechanism and SynthesisDocument60 pagesUace Chem Guide To Mechanism and SynthesisNelima Stella mercy100% (1)
- EqnPS 1Document1 pageEqnPS 1phiridanford621No ratings yet
- Mind Map Organic ChemistryDocument1 pageMind Map Organic ChemistryWaqar AhmedNo ratings yet
- Chemistry 10 Simplifed NotesDocument15 pagesChemistry 10 Simplifed NotesFaina VeraNo ratings yet
- Given Below Are Word EquationsDocument2 pagesGiven Below Are Word Equationstrevin79saiNo ratings yet
- Red Ox BalancingDocument2 pagesRed Ox BalancingAhmed KazazNo ratings yet
- QUALITATIVE ANALYSIS OF IONS (USP ID Tests)Document12 pagesQUALITATIVE ANALYSIS OF IONS (USP ID Tests)Kirsten Joyce VillajuanNo ratings yet
- Test Ahhg1Document1 pageTest Ahhg1Ahmed OmarNo ratings yet
- Balancing EquationsDocument6 pagesBalancing EquationspkrajenpillaiNo ratings yet
- Balancing Questions PracticeDocument5 pagesBalancing Questions PracticeZunaira SafdarNo ratings yet
- Balancing Questions PracticeDocument5 pagesBalancing Questions PracticeZunaira SafdarNo ratings yet
- Craqueo de La ParafinaDocument1 pageCraqueo de La ParafinaLesly Adriana Calderon FloresNo ratings yet
- Druganalysis 201031101345Document24 pagesDruganalysis 201031101345Ata Ur RahmanNo ratings yet
- Equations Worksheet 1Document2 pagesEquations Worksheet 1jaikovskyNo ratings yet
- A2 Organic ReactionsDocument3 pagesA2 Organic ReactionsAli AlanniNo ratings yet
- Exer Ajust Reac TGLDocument2 pagesExer Ajust Reac TGLPaula CortesNo ratings yet
- P-Block Elements: Boron Family (Group 13 Elements)Document19 pagesP-Block Elements: Boron Family (Group 13 Elements)Indonesia HarshNo ratings yet
- Actividad 3 T4Document6 pagesActividad 3 T4Jafeth Rodriguez LeonNo ratings yet
- Untitled Spreadsheet - Google SheetsDocument3 pagesUntitled Spreadsheet - Google SheetsVIV GamingNo ratings yet
- Reactions You Need To Know - Inorganic ChemDocument7 pagesReactions You Need To Know - Inorganic ChemSaad AatirNo ratings yet
- Tabel Data PercobaanDocument13 pagesTabel Data PercobaanikeuNo ratings yet
- Tabel Data PercobaanDocument13 pagesTabel Data PercobaanikeuNo ratings yet
- Chemistry 1302: CHAPTER 3.3 - Acid Base ChemistryDocument15 pagesChemistry 1302: CHAPTER 3.3 - Acid Base Chemistryzak mahmoudNo ratings yet
- Syntezy OrganiczneDocument8 pagesSyntezy OrganiczneWojtek LinertowiczNo ratings yet
- Balance ArDocument2 pagesBalance ArIvan Dario Pineda PatiñoNo ratings yet
- Basic Chemistry - Final NotesDocument1 pageBasic Chemistry - Final NotesJerry G0% (1)
- WTP Chemical EquationDocument10 pagesWTP Chemical EquationSivakumar NagarathinamNo ratings yet
- SLOP - Writing Word and Symbol Equations For Reactions of MetalsDocument2 pagesSLOP - Writing Word and Symbol Equations For Reactions of Metalsmarcos.vaqueNo ratings yet
- Physics NoteDocument3 pagesPhysics NoteJitul konchNo ratings yet
- MINDMAP Alkene, Benzene, HaloalkaneDocument3 pagesMINDMAP Alkene, Benzene, HaloalkaneLeow JiashengNo ratings yet
- Regulation of Hydrogen Ion ConcentrationDocument15 pagesRegulation of Hydrogen Ion Concentrationmostafadiab074No ratings yet
- Carbon Carbon Dioxide Carbon Carbon Dioxide: MG MGDocument3 pagesCarbon Carbon Dioxide Carbon Carbon Dioxide: MG MGDSE No WorriesNo ratings yet
- All Named ReactionsDocument3 pagesAll Named ReactionsSamrathsingh Hayer100% (1)
- All As Chem ReactionsDocument11 pagesAll As Chem Reactionsklo9d ninjaNo ratings yet
- Chemistry EquationsDocument4 pagesChemistry Equationssakchham agrawalNo ratings yet