Professional Documents
Culture Documents
Những vấn đề lí thuyết thường gặp trong Hóa Vô Cơ PDF
Những vấn đề lí thuyết thường gặp trong Hóa Vô Cơ PDF
Uploaded by
Lê Quang VũCopyright:
Available Formats
You might also like
- kĩ thuật khăn trải bànDocument6 pageskĩ thuật khăn trải bànLê Quang VũNo ratings yet
- Chuyen Lam Son Thanh Hoa Lan 2Document7 pagesChuyen Lam Son Thanh Hoa Lan 2Lê Quang VũNo ratings yet
- Kĩ Thuật Kwl Và KwlhDocument2 pagesKĩ Thuật Kwl Và KwlhLê Quang VũNo ratings yet
- De-Thi-Giua-Ki-2-Hoa-9-De-4 (1) 12421643Document6 pagesDe-Thi-Giua-Ki-2-Hoa-9-De-4 (1) 12421643Lê Quang VũNo ratings yet
- 62471.Phục lục 1 hoá học 8 cv 5512Document7 pages62471.Phục lục 1 hoá học 8 cv 5512Lê Quang VũNo ratings yet
- Trac Nghiem Ly Thuyet Su Dien Li Acid Va Base Hoa Lop 11 HayDocument4 pagesTrac Nghiem Ly Thuyet Su Dien Li Acid Va Base Hoa Lop 11 HayLê Quang VũNo ratings yet
- De Thi HK1 Hoa 11 So GD Bac Ninh 23 24Document3 pagesDe Thi HK1 Hoa 11 So GD Bac Ninh 23 24Lê Quang VũNo ratings yet
- De Cuong On Tap Giua HK2 Hoa 11 KNTT HayDocument5 pagesDe Cuong On Tap Giua HK2 Hoa 11 KNTT HayLê Quang VũNo ratings yet
- De Cuong On Thi TN (Co Giai)Document185 pagesDe Cuong On Thi TN (Co Giai)phatdangquang60% (5)
- 100 de Tu On Luyen Hoa Hoc THCSDocument172 pages100 de Tu On Luyen Hoa Hoc THCSLê Quang VũNo ratings yet
Những vấn đề lí thuyết thường gặp trong Hóa Vô Cơ PDF
Những vấn đề lí thuyết thường gặp trong Hóa Vô Cơ PDF
Uploaded by
Lê Quang VũOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Những vấn đề lí thuyết thường gặp trong Hóa Vô Cơ PDF
Những vấn đề lí thuyết thường gặp trong Hóa Vô Cơ PDF
Uploaded by
Lê Quang VũCopyright:
Available Formats
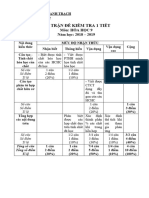
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
Li ni u
Cc em hc sinh thn mn!
Theo xu th thi nh nm 2013, ta thy cc bi ton ha hc d dn, ng ngha vi vic khong cch
gia hc sinh kh v gii cng gim dn trong vic gii bi ton. V vy th sinh chnh lch im nhau
ch yu phn l thuyt. Hin nay, theo xu hng d nhiu th sinh c im sn c nhiu c
hi i hc, cng l iu cc em cn ch nh hng hc tp cho hiu qu.
Hin nay kin thc ca cc em vn cha c tnh tng hp v ph hp vi vic thi, v vy khc
phc im yu ca cc em, thy bin son ti liu cu hi l thuyt thng gp trong ha v c ny
c ti liu tt trong vic n thi. Ti liu c chia ra thnh cc vn thng gp trong thi c, v c
ly v d minh ha trong cc thi i hc t nm 2007 2013 c p n cc em tham kho.
y l mt trong nhng cun ti liu m u thy vit dnh cho cc em, do yu t thi gian nn
cun ti liu cha c hon chnh, mt s vn cha hon thin, tuy vy y cng l ti liu hu ch
c th gip cc em trong vic hc tp mn ha hc.
Nhiu em t cu hi lm th no hc tt mn ha? Thy ch khuyn cc em 2 sau
- hc t tng qut ri hc n chi tit, cui cng l tng hp.
- Khi hc cht lun phi tr li c cht c tnh cht g? V sao? Cht tc dng c vi
nhng cht no? Phn ng to sn phm g? iu kin ca phn ng. V ly c v d minh
ha.
Cc em nn tham kho tip cc cun ti liu khc ca thy na
Cc ti liu vit
1. Cu hi l thuyt thng gp trong ha hu c
2. Cu hi l thuyt thng gp trong ha v c
Cc ti liu ang vit
3. Tuyn tp 460 cu hi l thuyt hu c theo dng cu hi thng gp
4. Tuyn tp 580 cu hi l thuyt v c theo dng cu hi thng gp
5. Cc dng bi tp thng gp v cng thc gii.
Chc cc em hc tt!
ti thm ti liu, cc em vui lng truy cp http://tailieulovebook.com
GV: Trn c Tun ( 01695 178 188) Trang: 1.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
VN 1: CHT LNG TNH
L THUYT
1. Cht/Ion lng tnh
- Cht/Ion lng tnh l nhng cht/ion va c kh nng nhng va c kh nng nhn proton ( H+)
- Cht/ ion lng tnh va tc dng c vi dung dch axit ( nh HCl, H2SO4 long), va tc dng c
vi dung dch baz ( nh NaOH, KOH, Ba(OH)2)
Lu : Cht va tc dng c vi dung dch axit, va tc dng c vi dung dch baz nhng cha
chc phi cht lng tnh nh: Al, Zn, Sn, Pb, Be
2. Cc cht lng tnh thng gp.
- Oxit nh: Al2O3, ZnO, BeO, SnO, PbO, Cr2O3.
- Hidroxit nh: Al(OH)3, Zn(OH)2, Be(OH)2, Pb(OH)2, Cr(OH)3
- Mui cha ion lng tnh nh: Mui HCO3-, HSO3-, HS-, H2PO4-
- Mui amoni ca axit yu nh: (NH4)2CO3, (NH4)2SO3, (NH4)2S, CH3COONH4
3. Cc phn ng ca cc cht lng vi dd HCl, NaOH
- Gi s: X ( l Al, Cr), Y l ( Zn, Be, Sn, Pb)
a. Oxit:
* Tc dng vi HCl
X2O3 + 6HCl 2MCl3 + 3H2O
YO + 2HCl YCl2 + H2O
* Tc dng vi NaOH
X2O3 + NaOH NaXO2 + 2H2O
YO + 2NaOH Na2YO2 + H2O
b. Hidroxit lng tnh
* Tc dng vi HCl
X(OH)3 + 3HCl XCl3 + 3H2O
Y(OH)2 + 2HCl YCl2 + 2H2O
* Tc dng vi NaOH
X(OH)3 + NaOH NaXO2 + 2H2O
Y(OH)2 + 2NaOH Na2YO2 + 2H2O
c. Mui cha ion lng tnh
* Tc dng vi HCl
HCO3- + H+ H2O + CO2
HSO3- + H+ H2O + SO2
HS- + H+ H2S
* Tc dng vi NaOH
HCO3- + OH- CO32- + H2O
HSO3- + OH- SO32- + H2O
HS- + OH- S2- + H2O
d. Mui ca NH4+ vi axit yu
* Tc dng vi HCl
(NH4)2RO3 + 2HCl 2NH4Cl + H2O + RO2 ( vi R l C, S)
(NH4 )2S + 2HCl 2NH4Cl + H2S
* Tc dng vi NaOH
NH4 + + OH- NH3 + H2O
Lu : Kim loi Al, Zn, Be, Sn, Pb khng phi cht lng tnh nhng cng tc ng c vi c axit v
dung dch baz
n
M + nHCl MCln + H2 ( M l kim loi Al, Zn, Be, Sn, Pb; n l ha tr ca M)
2
n
M + (4 - n)NaOH + (n 2) H2O Na4-nMO2 + H2
2
GV: Trn c Tun ( 01695 178 188) Trang: 2.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
CU HI
Cu 1.Cu 4-A7-748: Cho dy cc cht: Ca(HCO3)2, NH4Cl, (NH4)2CO3, ZnSO4, Al(OH)3, Zn(OH)2. S
cht trong dy c tnh cht lng tnh l
A. 3. B. 4. C. 2. D. 5.
Cu 2.Cu 56-CD7-439: Cc hp cht trong dy cht no di y u c tnh lng tnh?
A. Cr(OH)3, Zn(OH)2, Pb(OH)2. B. Cr(OH)3, Pb(OH)2, Mg(OH)2.
C. Cr(OH)3, Zn(OH)2, Mg(OH)2. D. Cr(OH)3, Fe(OH)2, Mg(OH)2.
Cu 3.Cu 53-CD8-216: Cho dy cc cht: Cr(OH)3, Al2(SO4)3, Mg(OH)2, Zn(OH)2, MgO, CrO3. S
cht trong dy c tnh cht lng tnh l
A. 5. B. 2. C. 3. D. 4.
Cu 4.Cu 35-CD9-956: Dy gm cc cht va tan trong dung dch HCl, va tan trong dung dch NaOH
l:
A. NaHCO3, MgO, Ca(HCO3)2. B. NaHCO3, Ca(HCO3)2, Al2O3.
C. NaHCO3, ZnO, Mg(OH)2. D. Mg(OH)2, Al2O3, Ca(HCO3)2.
Cu 5.Cu 14-A11-318: Cho dy cc cht: NaOH, Sn(OH)2, Pb(OH)2, Al(OH)3, Cr(OH)3. S cht trong
dy c tnh cht lng tnh l
A. 1. B. 2. C. 4. D. 3.
Cu 6.Cu 45-B11-846: Cho dy cc cht sau: Al, NaHCO3, (NH4)2CO3, NH4Cl, Al2O3, Zn, K2CO3,
K2SO4. C bao nhiu cht trong dy va tc dng c vi dung dch HCl, va tc dng c vi dung
dch NaOH?
A. 5. B. 2. C. 3. D. 4.
Cu 7.Cu 33-A12-296: Cho dy cc cht: Al, Al(OH)3, Zn(OH)2, NaHCO3, Na2SO4. S cht trong
dy va phn ng c vi dung dch HCl, va phn ng c vi dung dch NaOH l
A. 5. B. 4. C. 3. D. 2
VN 2: MI TRNG CA DUNG DCH MUI
L THUYT
1. Mui trung ha
- Mui trung ha to bi cation ca baz mnh v anion gc axit mnh khng b thy phn. Dung dch
thu c c mi trng trung tnh ( pH = 7)
VD: NaNO3, KCl, Na2SO4,
- Mui trung ha to bi cation ca baz mnh v anion gc axit yu b thy phn. Dung dch thu c
c mi trng baz ( pH > 7)
VD: Na2CO3, K2S
- Mui trung ha to bi cation ca baz yu v anion gc axit mnh b thy phn. Dung dch thu c
c mi trng axit ( pH < 7)
VD: NH4Cl, CuSO4, AlCl3
- Mui trung ha to bi cation ca baz yu v anion gc axit yu b thy phn ( c hai b thy phn).
Ty thuc vo thy phn ca hai ion m dung dch c pH = 7 hoc pH > 7 hoc pH < 7
VD: (NH4)2CO3, (NH4)2S
2. Mui axit
- Mui HSO4- c mi trng axit ( pH < 7) VD: NaHSO4
- Mui HCO3-, HSO3-, HS- vi cation baz mnh c mi trng baz VD: NaHCO3,
CU HI
Cu 1.Cu 32-CD7-439: Trong s cc dung dch: Na2CO3, KCl, CH3COONa, NH4Cl, NaHSO4,
C6H5ONa, nhng dung dch c pH > 7 l
A. KCl, C6H5ONa, CH3COONa. B. NH4Cl, CH3COONa, NaHSO4.
C. Na2CO3, NH4Cl, KCl. D. Na2CO3, C6H5ONa, CH3COONa
GV: Trn c Tun ( 01695 178 188) Trang: 3.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
Cu 2.Cu 27-CD8-216: Cho cc dung dch c cng nng : Na2CO3 (1), H2SO4 (2), HCl (3), KNO3
(4). Gi tr pH ca cc dung dch c sp xp theo chiu tng t tri sang phi l:
A. 3, 2, 4, 1. B. 4, 1, 2, 3. C. 1, 2, 3, 4. D. 2, 3, 4, 1.
Cu 3.Cu 54-CD10-824: Dung dch no sau y c pH > 7?
A. Dung dch NaCl. B. Dung dch Al2(SO4)3.
C. Dung dch NH4Cl. D. Dung dch CH3 COONa.
Cu 4.Cu 49-B13-279: Trong s cc dung dch c cng nng 0,1M di y, dung dch cht no
c gi tr pH nh nht?
A. NaOH. B. HCl. C. H2SO4. D. Ba(OH)2.
Cu 5.Cu 57-CD13-415: Dung dch cht no di y c mi trng kim?
A. Al(NO3)3. B. NH4Cl. C. HCl. D. CH3COONa.
VN 3: CC CHT PHN NG VI NC NHIT THNG
L THUYT
1. Cc cht phn ng vi H2O nhit thng.
- Kim loi Kim + Ca, Sr, Ba tc dng vi H2O nhit thng to baz + H2
VD: Na + H2O NaOH + H2
Ba + 2H2O Ba(OH)2 + H2
n
TQ: M + n H2O M(OH)n + H2
2
- Oxit ca KLK v CaO, SrO, BaO tc dng vi H2O nhit thng to baz
VD: Na2O + H2O 2NaOH
BaO + H2O Ba(OH)2
- Cc oxit: CO2, SO2, SO3, P2O5, N2O5, NO2 tc dng vi H2O nhit thng to axit
VD: CO2 + H2O H2CO3
SO3 + H2O H2SO4
P2O5 + 3H2O 2H3PO4
N2O5 + H2O 2HNO3
3NO2 + H2O 2HNO3 + NO
4NO2 + 2H2O + O2 4HNO3
- Cc kh HCl, HBr, HI, H2S khng c tnh axit, khi ha tan vo nc s to dung dch axit tng ng.
- Kh NH3 tc dng vi H2O rt yu: NH3 + H2O NH4 + + OH-.
- Mt s mui ca cation Al3+, Zn2+, Fe3+ vi anion gc axit yu nh CO32-, HCO3-, SO32-, HSO3-, S2-, HS-
b thy phn to baz + axit tng ng.
VD: Al2S3 + 6H2O 2Al(OH)3 + 3H2S
Fe2(CO3)3 + 3H2O 2Fe(OH)3 + 3CO2
2. Tc dng vi H2O nhit cao.
- nhit cao, kh nng phn ng ca cc cht vi H2O cao hn, nhng cc em ch mt s phn ng
dunnong
sau: Mg + 2H2O Mg(OH)2 + H2
o
570 C
3Fe + 4H2O Fe3O4 + 4H2
570o C
Fe + H2O FeO + H2
nungdothan
C + H2O CO + H2
nungdothan
C + 2H2O CO2 + 2H2
CU HI
Cu 1.Cu 25-B07-285: Hn hp X cha Na2O, NH4Cl, NaHCO3 v BaCl2 c s mol mi cht u
bng nhau. Cho hn hp X vo H2O (d), un nng, dung dch thu c cha
A. NaCl, NaOH, BaCl2. B. NaCl, NaOH.
C. NaCl, NaHCO3, NH4Cl, BaCl2. D. NaCl.
GV: Trn c Tun ( 01695 178 188) Trang: 4.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
Cu 2.Cu 2-B11-846: Cho dy cc oxit sau: SO2, NO2, NO, SO3, CrO3, P2O5, CO, N2O5, N2O. S oxit
trong dy tc dng c vi H2O iu kin thng l
A. 5. B. 6. C. 8. D. 7.
Cu 3.Cu 35-B13-279: Khi ha tan hon ton m gam mi kim loi vo nc d, t kim loi no sau
y thu c th tch kh H2 (cng iu kin nhit v p sut) l nh nht?
A. K. B. Na. C. Li. D. Ca.
VN 4: NC CNG
L THUYT
1. Khi nim
- Nc cng l nc cha nhiu cation Ca2+ v Mg2+
- Nc mm l nc cha t hoc khng cha cation Ca2+ v Mg2+
2. Phn loi
- Da vo c anion trong nc cng ta chia 3 loi:
a. Nc cng tm thi l nc cng cha ion HCO3- ( dng mui Ca(HCO3)2 v Mg(HCO3)2 )
- nc cng tm thi un nng s lm mt tnh cng ca nc
b. Nc cng vnh cu l nc cng cha ion Cl-, SO42- ( dng mui CaCl2, MgCl2, CaSO4, v MgSO4)
- nc cng vnh cu un nng s khng lm mt tnh cng ca nc
c. Nc cng ton phn l nc cng cha c anion HCO3- ln Cl-, SO42-.
- nc cng ton phn un nng s lm gim tnh cng ca nc
3. Tc hi
- Lm hng cc thit b ni hi, ng dn nc
- Lm gim mi v thc n
- Lm mt tc dng ca x phng
4. Phng php lm mm
a. Phng php kt ta.
- i vi mi loi nc cng ta dng Na2CO3 hoc Na3PO4 lm mm nc
M2+ + CO32- MCO3
2M2+ + 2PO43- M3(PO4)2
- i vi nc cng tm thi, ngoi phng php dng Na2CO3, Na3PO4 ta c th dng thm NaOH
hoc Ca(OH)2 va , hoc l un nng.
+ Dng NaOH va .
Ca(HCO3)2 + 2NaOH CaCO3 + Na2CO3 + 2H2O
Mg(HCO3)2 + 2NaOH MgCO3 + Na2CO3 + 2H2O
+ Dng Ca(OH)2 va
Ca(HCO3)2 + Ca(OH)2 2CaCO3 + 2H2O
Mg(HCO3)2 + Ca(OH)2 MgCO3 + CaCO3 + 2H2O
+ un si nc, phn hy Ca(HCO3)2 v Mg(HCO3)2 to thnh mui cacbonat khng tan.
lng gn b k ta c nc mm.
to
Ca(HCO3)2 CaCO3 + CO2 + H2O
o
t
Mg(HCO3)2 MgCO3 + CO2 + H2O
CU HI
Cu 1.Cu 3-B8-371: Mt mu nc cng cha cc ion: Ca2+ , Mg2+, HCO3-, Cl-, SO42-. Cht c
dng lm mm mu nc cng trn l
A. NaHCO3. B. Na2CO3. C. HCl. D. H2SO4.
Cu 2.Cu 3-CD8-216: Hai cht c dng lm mm nc cng vnh cu l
A. Na2CO3 v HCl. B. Na2CO3 v Na3PO4.
C. Na2CO3 v Ca(OH)2. D. NaCl v Ca(OH)2.
GV: Trn c Tun ( 01695 178 188) Trang: 5.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
Cu 3.Cu 23-CD11-259: Mt cc nc c cha cc ion: Na+ 0,02 mol), Mg2+0,02 mol), Ca2+ 0,04 mol),
Cl 0,02 mol), HCO 0,10 mol) v SO 2 0,01 mol). un si cc nc trn cho n khi cc phn ng
3 4
xy ra hon
ton th nc cn li trong cc
A. l nc mm. B. c tnh cng vnh cu.
C. c tnh cng ton phn. D. c tnh cng tm thi.
Cu 4.Cu 6-A11-318: Dy gm cc cht u c th lm mt tnh cng tm thi ca nc l:
A. HCl, NaOH, Na2CO3. B. KCl, Ca(OH)2, Na2CO3.
C. NaOH, Na3PO4, Na2CO3. D. HCl, Ca(OH)2, Na2CO3.
Cu 5.Cu 26-B13-279: Mt loi nc cng khi un si th mt tnh cng. Trong loi nc cng ny c ha
tan nhng hp cht no sau y?
A. Ca(HCO3)2, Mg(HCO3)2. B. Ca(HCO3)2, MgCl2.
C. CaSO4, MgCl2. D. Mg(HCO3)2, CaCl2.
VN 5: N MN KIM LOI
L THUYT
1. n mn kim loi: l s ph hy kim loi do tc dng ca cc cht trong mi trng
- n mn kim loi c 2 dng chnh: n mn ha hc v n mn in ha.
2. n mn ha hc: l qu trnh oxi ha kh, trong cc electron ca kim loi c chuyn trc tip
n cc cht trong mi trng.
- n mn ha hc thng xy ra nhng b phn ca thit b l t hoc nhng thit b thng xuyn
phi tip xc v hi nc v kh oxi
Kinh nghim: nhn bit n mn ha hc, ta thy n mn kim loi m khng thy xut hin cp kim loi
hay cp KL-C th l n mn kim loi.
3. n mn in ha: l qu trnh oxi ha kh, trong kim loi b n mn do tc dng ca dung dch
cht in li v to nn ong electron chuyn di t cc m n cc dng.
- iu kin xy ra n mn in ha: phi tha mn ng thi 3 iu sau
+ Cc in cc phi khc nhau v bn cht
+ Cc nh cc phi tip xc trc tip hoc gin tip vi nhau qua dy dn
+ Cc in cc cng tip xc vi dung dch cht in li
- n mn in ha thng xy ra khi cp kim loi ( hoc hp kim) ngoi khng kh m, hoc nhng
trong dung dch axit, dung dch mui, trong nc khng nguyn cht
4. Cc bin php chng n mn kim loi.
a. Phng php bo v b mt
- Ph ln b mt kim loi mt lp sn, du m, cht do
- Lau chi, ni kh do thong
b. Phng php in ha
- dng mt kim loi l vt hi sinh bo v vt liu kim loi.
VD: bo v v tu bin bng thp, ngi ta gn cc l Zn vo pha ngoi v tu phn chm trong
nc bin ( nc bin l dung dch cht in li). Km b n mn, v tu c bo v.
CU HI
Cu 1.Cu 31-B07-285: C 4 dung dch ring bit: a) HCl, b) CuCl2, c) FeCl3, d) HCl c ln CuCl2.
Nhng vo mi dung dch mt thanh Fe nguyn cht. S trng hp xut hin n mn in ho l
A. 0. B. 1. C. 2. D. 3.
Cu 2.Cu 9-CD7-439: Cho cc cp kim loi nguyn cht tip xc trc tip vi nhau: Fe v Pb; Fe v
Zn; Fe v Sn; Fe v Ni. Khi nhng cc cp kim loi trn vo dung dch axit, s cp kim loi trong Fe
b ph hu trc l
A. 2. B. 1. C. 4. D. 3.
GV: Trn c Tun ( 01695 178 188) Trang: 6.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
Cu 3.Cu 48-A8-329: Bit rng ion Pb2+ trong dung dch oxi ha c Sn. Khi nhng hai thanh kim
loi Pb v Sn c ni vi nhau bng dy dn in vo mt dung dch cht in li th
A. ch c Pb b n mn in ho. B. ch c Sn b n mn in ho.
C. c Pb v Sn u khng b n mn in ho. D. c Pb v Sn u b n mn in ho.
Cu 4.Cu 55-A8-329: Mt pin in ho c in cc Zn nhng trong dung dch ZnSO4 v in cc Cu
nhng trong dung dch CuSO4. Sau mt thi gian pin phng in th khi lng
A. in cc Zn gim cn khi lng in cc Cu tng.
B. c hai in cc Zn v Cu u tng.
C. in cc Zn tng cn khi lng in cc Cu gim.
D. c hai in cc Zn v Cu u gim.
Cu 5.Cu 46-B8-371: Tin hnh bn th nghim sau:
- Th nghim 1: Nhng thanh Fe vo dung dch FeCl3;
- Th nghim 2: Nhng thanh Fe vo dung dch CuSO4;
- Th nghim 3: Nhng thanh Cu vo dung dch FeCl3;
- Th nghim 4: Cho thanh Fe tip xc vi thanh Cu ri nhng vo dung dch HCl. S trng hp xut
hin n mn in ho l
A. 1. B. 2. C. 4. D. 3.
Cu 6.Cu 8-A9-438: Cho cc hp kim sau: Cu-Fe (I); Zn-Fe (II); Fe-C (III); Sn-Fe (IV). Khi tip
xc vi dung dch cht in li th cc hp kim m trong Fe u b n mn trc l:
A. I, II v IV. B. I, III v IV. C. I, II v III. D. II, III v IV.
Cu 7.Cu 12-B9-148: C cc th nghim sau:
(I) Nhng thanh st vo dung dch H2SO4 long, ngui.
(II) Sc kh SO2 vo nc brom.
(III) Sc kh CO2 vo nc Gia-ven.
(IV) Nhng l nhm vo dung dch H2SO4 c, ngui.
S th nghim xy ra phn ng ho hc l
A. 2. B. 1. C. 3. D. 4.
Cu 8.Cu 30-B10-937: C 4 dung dch ring bit: CuSO4, ZnCl2, FeCl3, AgNO3. Nhng vo mi dung
dch mt thanh Ni. S trng hp xut hin n mn in ho l
A. 2. B. 4. C. 3. D. 1.
Cu 9.Cu 30-CD11-259: Nu vt lm bng hp kim Fe-Zn b n mn in ho th trong qu trnh n
mn
A. km ng vai tr catot v b oxi ha. B. st ng vai tr anot v b oxi ho.
C. st ng vai tr catot v ion H+ b oxi ha. D. km ng vai tr anot v b oxi ho.
Cu 10.Cu 53-B11-846: Trong qu trnh hot ng ca pin in ho Zn Cu th
A. khi lng ca in cc Zn tng. B. nng ca ion Zn2+ trong dung dch tng.
C. khi lng ca in cc Cu gim. D. nng ca ion Cu 2+ trong dung dch tng.
Cu 11.Cu 26-B12-359: Trng hp no sau y xy ra n mn in ho?
A. Si dy bc nhng trong dung dch HNO3. B. t l st trong kh Cl2.
C. Thanh nhm nhng trong dung dch H2SO4 long. D. Thanh km nhng trong dung dch CuSO4.
Cu 12.Cu 26-CD12-169: Tin hnh cc th nghim sau:
(a) Cho l Fe vo dung dch gm CuSO4 v H2SO4 long; (b) t dy Fe trong bnh ng kh O2;
(c) Cho l Cu vo dung dch gm Fe(NO3)3 v HNO3; (d) Cho l Zn vo dung dch HCl.
S th nghim c xy ra n mn in ha l
A. 1. B. 4. C. 2. D. 3.
Cu 13.Cu 60-A13-193: Trng hp no sau y, kim loi b n mn in ha hc?
A. Kim loi st trong dung dch HNO3 long. B. Thp cacbon trong khng kh m.
C. t dy st trong kh oxi kh. D. Kim loi km trong dung dch HCl.
GV: Trn c Tun ( 01695 178 188) Trang: 7.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
VN 6: PHN NG NHIT PHN
L THUYT
1. Nhit phn mui nitrat
- Tt c cc mui nitrat u b nhit phn to sn phm X + O2
a. Nhit phn mui nitrat ca kim loi K, Ba,Ca, Na( kim loi tan) th sn phm X l mui nitrit (
NO2-)
to
VD: 2NaNO3 2NaNO2 + O2
o
t
2KNO3 2KNO2 + O2
b. Nhit phn mui nitrat ca kim loi Mg Cu th sn phm X l oxit + NO2
to
VD: 2Cu(NO3)2 2CuO + 4NO2 + O2
to 3
2Fe(NO3)3 Fe2O3 + 6NO2 + O2
2
Lu : nhit phn mui Fe(NO3)2 thu c Fe2O3 ( khng to ra FeO )
to
2Fe(NO3)2 Fe2O3 + 4NO2 + O2
c. Nhit phn mui nitrat ca kim loi sau Cu th sn phm X l KL + NO2
to
VD: 2AgNO3 2Ag + 2NO2 + O2
2. Nhit phn mui cacbonat ( CO32- )
- Mui cacbonat ca kim loi kim khng b phn hy nh Na2CO3, K2CO3
- Mui cacbonat ca kim loi khc trc Cu b nhit phn thnh oxit + CO2
to
VD: CaCO3 CaO + CO2
to
MgCO3 MgO + CO2
- Mui cacbonat ca kim loi sau Cu b nhit phn thnh KL + O2 + CO2
to
VD: Ag2CO3 2Ag + O2 + CO2
o
t
- Mui (NH4)2CO3 2NH3 + CO2 + H2O
3. Nhit phn mui hidrocacbonat ( HCO3-)
- Tt c cc mui hidrocacbonat u b nhit phn.
- Khi un nng dung dch mui hidrocacbonat:
to
Hidrocacbonat Cacbonat trung ha + CO2 + H2O
to
VD: 2NaHCO3 Na2CO3 + CO2 + H2O
o
t
Ca(HCO3)2 CaCO3 + CO2 + H2O
- Nu nhit phn hon ton mui hidrocacbonat
to
+ Mui hidrocacbonat ca kim loi kim Cacbonat trung ha + CO2 + H2O
to
VD: 2NaHCO3 Na2CO3 + CO2 + H2O
o
t
+ Mui hidrocacbonat ca kim loi khc Oxit kim loi + CO2 + H2O
o
t , hontoan
VD: Ca(HCO3)2 CaO + 2CO2 + H2O
3. Nhit phn mui amoni
to
- Mui amoni ca gc axit khng c tnh oxi ha Axit + NH3
o
t
VD: NH4Cl NH3 + HCl
to
(NH4)2CO3 2NH3 + H2O + CO2
o
t
- Mui amoni ca gc axit c tnh oxi ha N2 hoc N2O + H2O
o
t
VD: NH4NO3 N2O + 2H2O
o
t
NH4NO2 N2 + 2H2O
to
(NH4)2Cr2O7 Cr2O3 + N2 + 2H2O
4. Nhit phn baz
- Baz tan nh NaOH, KOH, Ba(OH)2, Ca(OH)2 khng b nhit phn hy.
GV: Trn c Tun ( 01695 178 188) Trang: 8.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
- Baz khng tan nhit phn to oxit + H2O
to
VD: 2Al(OH)3 Al2O3 + 3H2O
o
t
Cu(OH)2 CuO + H2O
o
t , khngcokhongkhi
Lu : Fe(OH)2 FeO + H2O
o
t
2Fe(OH)2 + O2 Fe2O3 + 2H2O
CU HI
Cu 1.Cu 16-A7-748: Khi nung hn hp cc cht Fe(NO3)2, Fe(OH)3 v FeCO3 trong khng kh n
khi lng khng i, thu c mt cht rn l
A. Fe3O4. B. FeO. C. Fe2O3. D. Fe.
Cu 2.Cu 33-B8-371: Phn ng nhit phn khng ng l
to to
A. NH4NO2 N2 + 2H2O B. NaHCO3 NaOH + CO2
o o
t t
B. 2KNO3 2KNO2 + O2 C. NH4Cl NH3 + HCl
Cu 3. Cu 11-B9-148: Khi nhit phn hon ton tng mui X, Y th u to ra s mol kh nh hn s
mol mui tng ng. t mt lng nh tinh th Y trn n kh khng mu, thy ngn la c mu vng.
Hai mui X, Y ln lt l:
A. KMnO4, NaNO3. B. Cu(NO3)2, NaNO3. C. CaCO3, NaNO3. D. NaNO3, KNO3.
Cu 4.Cu 47-CD10-824: Sn phm ca phn ng nhit phn hon ton AgNO3 l:
A. Ag, NO2, O2. B. Ag2O, NO, O2. C. Ag, NO, O2. D. Ag2O, NO2, O2.
VN 7: PHN NG IN PHN
L THUYT
I. in phn nng chy
- Thng in phn mui clorua ca kim loi mnh, baz ca kim loi kim, hoc oxit nhm
dpnc n
+ Mui halogen: RCln R + Cl2 ( R l kim loi kim, kim th)
2
dpnc
+ Baz: 2MOH 2M + O2 + H2O
dpnc
+ Oxit nhm: 2Al2O3 4Al + 3O2
II. in phn dung dch.
1. Mui ca kim loi tan
- in phn dung dch mui halogenua ( gc Cl, -Br ) c mng ngn, to baz + halogen + H2
dpdd
VD: 2NaCl + H2O comangngan
2NaOH + Cl2 + H2
- in phn dung dch mui halogen nu khng c mng ngn, Cl2 sinh ra phn ng vi dung dch kim
to nc giaven.
dpdd
VD: 2NaCl + H2O khongmangngan
NaCl + NaClO + H2
2. Mui ca kim loi trung bnh yu: khi in phn dung dch sinh kim loi
a. Nu mui cha gc halogenua ( gc Cl, - Br ): Sn phm l KL + phi kim
dpdd
VD: CuCl2 Cu + Cl2
b. Nu mui cha gc c oxi: Sn phm l KL + Axit + O2
dpdd
VD: 2Cu(NO3)2 + 2H2O 2Cu + 4HNO3 + O2
dpdd
2CuSO4 + 2H2O 2Cu + 2H2SO4 + O2
3. Mui ca kim loi tan vi gc axit c oxi, axit c oxi, baz tan nh NaNO3, NaOH, H2SO4
dpdd
- Coi nc b in phn: 2H2O 2H2 + O2
CU HI
GV: Trn c Tun ( 01695 178 188) Trang: 9.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
Cu 1.Cu 32-B07-285: in phn dung dch cha a mol CuSO4 v b mol NaCl (vi in cc tr, c
mng ngn xp). dung dch sau in phn lm phenolphtalein chuyn sang mu hng th iu kin
ca a v b l (bit ion SO42 khng b in phn trong dung dch)
A. b > 2a. B. b = 2a. C. b < 2a. D. 2b = a.
Cu 2.Cu 43-A8-329: Khi in phn NaCl nng chy (in cc tr), ti catt xy ra
A. s oxi ho ion Cl-. B. s oxi ho ion Na+. C. s kh ion Cl-. D. s kh ion Na+.
Cu 3.Cu 33-A10-684: Phn ng in phn dung dch CuCl2 (vi in cc tr) v phn ng n mn
in ho xy ra khi nhng hp kim Zn-Cu vo dung dch HCl c c im l:
A. Phn ng cc m c s tham gia ca kim loi hoc ion kim loi.
B. Phn ng cc dng u l s oxi ho Cl .
C. u sinh ra Cu cc m.
D. Phn ng xy ra lun km theo s pht sinh dng in.
Cu 4.Cu 37-A10-684: C cc pht biu sau:
1
Lu hunh, photpho u bc chy khi tip xc vi CrO3.
2
Ion Fe3+ c cu hnh electron vit gn l [Ar]3d5.
3
Bt nhm t bc chy khi tip xc vi kh clo.
4
Phn chua c cng thc l Na2SO4.Al2(SO4)3.24H2O.
Cc pht biu ng l:
A. 1, 3, 4. B. 2, 3, 4. C. 1, 2, 4. D. 1, 2, 3.
Cu 5.Cu 55-CD10-824: in phn dung dch CuSO4 vi anot bng ng (anot tan) v in phn
dung dch CuSO4 vi anot bng graphit (in cc tr) u c c im chung l
A. anot xy ra s oxi ho: Cu Cu2+ + 2e.
B. catot xy ra s kh: Cu2+ + 2e Cu.
C. catot xy ra s oxi ho: 2H2O + 2e 2OH + H2.
D. anot xy ra s kh: 2H2O O2 + 4H+ + 4e.
Cu 6.Cu 48-A11-318: Khi in phn dung dch NaCl (cc m bng st, cc dng bng than ch, c
mng ngn xp) th
A. cc m xy ra qu trnh kh H2O v cc dng xy ra qu trnh oxi ho ion Cl .
B. cc m xy ra qu trnh oxi ho H2O v cc dng xy ra qu trnh kh ion Cl .
C. cc m xy ra qu trnh kh ion Na+ v cc dng xy ra qu trnh oxi ho ion Cl .
D. cc dng xy ra qu trnh oxi ho ion Na+ v cc m xy ra qu trnh kh ion Cl .
Cu 7.Cu 49-CD13-415: in phn dung dch gm NaCl v HCl (in cc tr, mng ngn xp). Trong
qu trnh in phn, so vi dung dch ban u, gi tr pH ca dung dch thu c
A. tng ln. B. khng thay i. C. gim xung. D. tng ln sau gim xung.
VN 8: PHN NG NHIT LUYN
L THUYT
1. Khi nim
- L phn ng iu ch kim loi bng cc kh cc oxit kim loi nhit cao bng H2, CO, Al, C
2. Phn ng
CO CO2 (1)
toC
H2 + KL-O KL + H2O (2)
Al Al2O3 (3)
C hh CO, CO2 (4)
GV: Trn c Tun ( 01695 178 188) Trang: 10.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
iu kin:
- KL phi ng sau Al trong dy hot in ha ( ring CO, H2 khng kh c ZnO)
K, Ba, Ca, Na, Mg, Al, Zn, Fe....
Vd: CuO + CO Cu + CO2
MgO + CO khng xy ra.
- Ring phn ng (3) gi l phn ng nhit nhm ( phn ng ca Al vi oxit KL sau n nhit cao)
CU HI
Cu 1.Cu 23-A7-748: Cho lung kh H2 (d) qua hn hp cc oxit CuO, Fe2O3, ZnO, MgO nung
nhit cao. Sau phn ng hn hp rn cn li l:
A. Cu, FeO, ZnO, MgO. B. Cu, Fe, Zn, Mg. C. Cu, Fe, Zn, MgO. D. Cu, Fe, ZnO, MgO.
Cu 2.Cu 25-CD7-439: Phn ng ho hc xy ra trong trng hp no di y khng thuc loi phn
ng nhit nhm?
A. Al tc dng vi Fe3O4 nung nng. B. Al tc dng vi CuO nung nng.
C. Al tc dng vi Fe2O3 nung nng. D. Al tc dng vi axit H2SO4 c, nng.
Cu 3.Cu 36-CD11-259: Dy gm cc oxit u b Al kh nhit cao l:
A. FeO, MgO, CuO. B. PbO, K2O, SnO. C. Fe3O4, SnO, BaO. D. FeO, CuO, Cr2O3.
Cu 4.Cu 9-A12-296: Hn hp X gm Fe3O4 v Al c t l mol tng ng 1 : 3. Thc hin phn ng
nhit nhm X (khng c khng kh) n khi phn ng xy ra hon ton thu c hn hp gm
A. Al, Fe, Fe3O4 v Al2O3. B. Al2O3, Fe v Fe3O4.
C. Al2O3 v Fe. D. Al, Fe v Al2O3.
DNG 9: TNG HP CC TNH CHT CA MT S CHT V C THNG GP
L THUYT
I. PHN NG TO PHC CA NH3.
- NH3 c th to phc tan vi cation Cu2+, Zn2+, Ag+, Ni2+
TQ: M(OH)n + 2nNH3 [M(NH3)2n] (OH)n vi M l Cu, Zn, Ag.
VD: CuSO4 + 2NH3 + 2H2O Cu(OH)2 + (NH4)2SO4
Cu(OH)2 + 4NH3 [Cu(NH3)4] (OH)2
VD: AgCl + 2NH3 [Ag(NH3)2]Cl
II. PHN NG CA MUI AXIT ( HCO3-, HSO3-, HS- )
- Ion HCO3- , HSO3-, HS- c tnh lng tnh nn va tc dng vi dung dch axit, va tc dng vi
dung dch baz
HCO3- + H+ H2O + CO2
HCO3- + OH- CO32- + H2O
HCO3- + HSO4- H2O + CO2 + SO42-
III. PHN NG CA MUI HSO4-.
- Ion HSO4- l ion cha H ca axit mnh nn khc vi ion cha H ca axit yu nh HCO3-, HSO3-, HS-
- Ion HSO4- khng c tnh lng tnh, ch c tnh axit mnh nn phn ng ging nh axit H2SO4 long.
+ Tc dng vi HCO3-, HSO3-,
HSO4- + HCO3- SO42- + H2O + CO2
+ Tc dng vi ion Ba2+, Ca2+, Pb2+
HSO4- + Ba2+ BaSO4 + H+
IV. TC DNG VI HCl
1. Kim loi: cc kim loi ng trc nguyn t H trong dy hot ng ha hc ( K, Na,Mg.Pb)
n
M + nHCl MCln + H2
2
VD: Mg + 2HCl MgCl2 + H2
- Ring Cu nu c mt oxi s c phn ng vi HCl: 2Cu + 4HCl + O2 2CuCl2 + 2H2O
2. Phi kim: khng tc dng vi HCl
GV: Trn c Tun ( 01695 178 188) Trang: 11.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
3. Oxit baz v baz: tt c cc oxit baz v oxit baz u phn ng to mui ( ha tr khng i) v
H2O
M2On + 2nHCl 2MCln + nH2O
VD: CuO + 2HCl CuCl2 + H2O
Fe3O4 + 8HCl FeCl2 + 2FeCl3 + 4H2O
- Ring MnO2 tc dng vi HCl c theo phn ng: MnO2 + 4HCl MnCl2 + Cl2 + 2H2O
4. Mui: tt c cc mui ca axit yu v AgNO3, Pb(NO3)2 u phn ng vi HCl
VD: CaCO3 + 2HCl CaCl2 + CO2 + H2O
Ca(HCO3)2 + 2HCl CaCl2 + 2H2O + 2CO2
AgNO3 + HCl AgCl + HNO3
FeS + 2HCl FeCl2 + H2S ( lu CuS, PbS khng phn ng vi HCl)
FeS2 + 2HCl FeCl2 + H2S + S
- Ring cc mui giu oxi ca Mn, Cr tc dng vi HCl c to kh Cl2
VD: 2KMnO4 + 16HCl 2KCl + 2MnCl2 + 5Cl2 + 8H2O
V. TC DNG VI NaOH.
1. Kim loi:
- Nhm 1: cc kim loi phn ng vi H2O gm KLK v Ca, Sr, Ba. Cc kim loi nhm 1 s phn ng vi
H2O trong dung dch NaOH.
n
M + H2O M(OH)n + H2
2
VD: K tc dng vi dd NaOH s xy ra phn ng: K + H2O KOH + H2
- Nhm 2: cc kim loi Al, Zn, Be,Sn, Pb tc dng vi NaOH theo phn ng
n
M + (4-n) NaOH + (n 2) H2O Na4-nMO2 + H2
2
3
VD: Al + NaOH + H2O NaAlO2 + H2
2
Zn + 2NaOH Na2ZnO2 + H2
2. Phi kim: Cl2, Br2 phn ng vi NaOH.
- Clo phn ng vi dd NaOH nhit thng to nc giaven
Cl2 + 2NaOH NaCl + NaClO + H2O
- Clo phn ng vi dd NaOH nhit 100oC to mui clorat (ClO3-)
3Cl2 + 6KOH 5KCl + KClO3 + 3H2O
3. Oxit lng tnh v hidroxit lng tnh: Nh Al2O3, ZnO2, BeO, PbO, SnO, Cr2O3, Al(OH)3,
Zn(OH)2, Be(OH)2, Pb(OH)2, Sn(OH)2, Cr(OH)3
- Cc oxit lng tnh v hidroxit lng tnh u phn ng vi NaOH c ( vi dung dch NaOH th Cr2O3
khng phn ng) to mui v nc
VD: Al2O3 + 2NaOH 2NaAlO2 + H2O
ZnO + 2NaOH Na2ZnO2 + H2O
Al(OH)3 + NaOH NaAlO2 + 2H2O
Zn(OH)2 + 2NaOH Na2ZnO2 + 2H2O
Cc oxit, hidroxit ca kim loi ha tr III ( Cr) phn ng ging oxit, hidroxit ca nhm
Cc oxit, hidroxit ca kim loi ha tr II ( Be, Sn, Pb) phn ng ging oxit, hidroxit ca km.
4. Oxit axit ( CO2, SO2, NO2, N2O5, P2O5, SiO2)
-phn ng 1: Tc dng vi NaOH to mui trung ha v H2O
VD: CO2 + 2NaOH Na2CO3 + H2O
- phn ng 2: tc dng vi NaOH to mui axit ( vi cc oxit axit ca axit nhiu nc)
VD: CO2 + NaOH NaHCO3
Lu : - NO2 tc dng vi NaOH to 2 mui nh sau: 2NO2 + 2NaOH NaNO3 + NaNO2 + H2O
- SiO2 ch phn ng c vi NaOH c, khng phn ng vi NaOH long.
- Cc oxit CO, NO l oxit trung tnh khng tc dng vi NaOH
5. Axit: tt c cc axit u phn ng ( k c axit yu)
GV: Trn c Tun ( 01695 178 188) Trang: 12.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
- phn ng 1: Axit + NaOH Mui trung ha + H2O
VD: HCl + NaOH NaCl + H2O
H2SO4 + 2NaOH Na2SO4 + 2H2O
- Phn ng 2: Axit nhiu nc + NaOH Mui axit + H2O
VD: H3PO4 + NaOH NaH2PO4 +H2O
6. Mui amoni v dd mui ca kim loi c baz khng tan ( nh mui Mg 2+, Al3+.)
- phn ng 1: Mui amoni + NaOH Mui Na+ + NH3 + H2O
VD: NH4Cl + NaOH NaCl + NH3 + H2O
- Phn ng 2: Mui ca kim loi c baz khng tan + NaOH Mui Na+ + Baz
VD: MgCl2 + 2NaOH 2NaCl + Mg(OH)2
CU HI
Cu 1. Cu 55-A7-748: C 4 dung dch mui ring bit: CuCl2, ZnCl2, FeCl3, AlCl3. Nu thm dung
dch KOH (d) ri thm tip dung dch NH3 (d) vo 4 dung dch trn th s cht kt ta thu c l
A. 3. B. 2. C. 4. D. 1.
Cu 2. Cu 6-B07-285: Trong cc dung dch: HNO3, NaCl, Na2SO4, Ca(OH)2, KHSO4, Mg(NO3)2, dy
gm cc cht u tc dng c vi dung dch Ba(HCO3)2 l:
A. HNO3, NaCl, Na2SO4. B. HNO3, Ca(OH)2, KHSO4, Na2SO4.
C. NaCl, Na2SO4, Ca(OH)2. D. HNO3, Ca(OH)2, KHSO4, Mg(NO3)2.
Cu 3.Cu 48-CD7-439: Cho hn hp X gm Mg v Fe vo dung dch axit H2SO4 c, nng n khi
cc phn ng xy ra hon ton, thu c dung dch Y v mt phn Fe khng tan. Cht tan c trong dung
dch Y l
A. MgSO4. B. MgSO4 v Fe2(SO4)3.
C. MgSO4, Fe2(SO4)3 v FeSO4. D. MgSO4 v FeSO4.
Cu 4.Cu 38-B 07-285: Cho hn hp Fe, Cu phn ng vi dung dch HNO3 long. Sau khi phn ng
hon ton, thu c dung dch ch cha mt cht tan v kim loi d. Cht tan l
A. Cu(NO3)2. B. HNO3. C. Fe(NO3)2. D. Fe(NO3)3.
Cu 5. Cu 7-A8-329: Cho cc cht: Al, Al2O3, Al2(SO4)3, Zn(OH)2, NaHS, K2SO3, (NH4)2CO3. S
cht u phn ng c vi dung dch HCl, dung dch NaOH l
A. 6. B. 4. C. 5. D. 7.
Cu 6.Cu 18-A9-438: Dy gm cc cht u tc dng c vi dung dch HCl long l:
A. KNO3, CaCO3, Fe(OH)3. B. Mg(HCO3)2, HCOONa, CuO.
C. FeS, BaSO4, KOH. D. AgNO3, (NH4)2CO3, CuS.
Cu 7.Cu 27-CD9-956: Dy no sau y ch gm cc cht va tc dng c vi dung dch HCl, va
tc dng c vi dung dch AgNO3 ?
A. Zn, Cu, Mg. B. Al, Fe, CuO. C. Hg, Na, Ca. D. Fe, Ni, Sn.
Cu 8.Cu 36-A10-684: Cho cc cht: NaHCO3, CO, Al(OH)3, Fe(OH)3, HF, Cl2, NH4Cl. S cht tc
dng c vi dung dch NaOH long nhit thng l
A. 3. B. 4. C. 6. D. 5.
Cu 9.Cu 46-A10-684: Cc cht va tc dng c vi dung dch HCl va tc dng c vi dung dch
AgNO3 l:
A. CuO, Al, Mg. B. MgO, Na, Ba. C. Zn, Ni, Sn. D. Zn, Cu, Fe.
Cu 10.Cu 45-B10-937: Cho cc cp cht vi t l s mol tng ng nh sau:
(a) Fe3O4 v Cu 1:1); (b) Sn v Zn 2:1); (c) Zn v Cu 1:1);
(d) Fe2(SO4)3 v Cu 1:1); (e) FeCl2 v Cu 2:1); (g) FeCl3 v Cu 1:1).
S cp cht tan hon ton trong mt lng d dung dch HCl long nng l
A. 5. B. 4. C. 2. D. 3.
Cu 11.Cu 45-A11-318: Cho hn hp X gm Fe2O3, ZnO v Cu tc dng vi dung dch HCl (d) thu
c dung dch Y v phn khng tan Z. Cho Y tc dng vi dung dch NaOH (long, d) thu c kt ta
A. Fe(OH)3. B. Fe(OH)3 v Zn(OH)2.
GV: Trn c Tun ( 01695 178 188) Trang: 13.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
C. Fe(OH)2, Cu(OH)2 v Zn(OH)2. D. Fe(OH)2 v Cu(OH)2.
Cu 12.Cu 18-B11-846: Cho dy cc cht: SiO2, Cr(OH)3, CrO3, Zn(OH)2, NaHCO3, Al2O3. S cht
trong dy tc dng c vi dung dch NaOH (c, nng) l
A. 3. B. 4. C. 5. D. 6.
Cu 13.Cu 20-B11-846: Dy gm cc cht (hoc dung dch) u phn ng c vi dung dch FeCl2 l:
A. Bt Mg, dung dch BaCl2, dung dch HNO3. B. Kh Cl2, dung dch Na2CO3, dung dch HCl.
C. Kh Cl2, dung dch Na2S, dung dch HNO3. D. Bt Mg, dung dch NaNO3, dung dch HCl.
Cu 14.Cu 22-A12-296: Cho dy cc oxit: NO2, Cr2O3, SO2, CrO3, CO2, P2O5, Cl2O7, SiO2, CuO. C
bao nhiu oxit trong dy tc dng c vi dung dch NaOH long?
A. 7. B. 8. C. 6. D. 5.
Cu 15.Cu 10-A13-193: Dy cc cht u tc dng c vi dung dch Ba(HCO3)2 l:
A. HNO3, NaCl v Na2SO4. B. HNO3, Ca(OH)2 v KNO3.
C. NaCl, Na2SO4 v Ca(OH)2. D. HNO3, Ca(OH)2 v Na2SO4.
Cu 16. Cu 28-CD13-415: Dung dch H2SO4 long phn ng c vi tt c cc cht trong dy no sau
y?
A. CuO, NaCl, CuS. B. BaCl2, Na2CO3, FeS.
C. Al2O3, Ba(OH)2, Ag. D. FeCl3, MgO, Cu.
VN 10: CC CHT CNG TN TI TRONG MT HN HP
L THUYT
1. iu kin cng tn ti trong mt hn hp
- Cc cht cng tn ti trong hn hp trong mt iu kin cho trc khi v ch khi cc cht khng phn
ng vi nhau iu kin .
2. Cng tn ti trong hn hp kh
a. iu kin thng.
- Cc cp kh cng tn ti trong iu kin thng hay gp l
Cl2 v O2 Cl2 v CO2 Cl2 v SO3 Cl2 v O3
F2 v O2 F2 v CO2 F2 v SO3 F2 v O3
O2 v H2 O2 v CO2 O2 v SO2 O2 v N2
N2 v Cl2 N2 v HCl N2 v F2 N2 v H2S
.
- Cc cp kh khng cng tn ti trong cng mt hn hp iu kin thng l
F2 v H2 Cl2 v H2 H2S v O2 NH3 v Cl2
HI v O3 NH3 v HCl H2S v O3 NO v O2
b. iu kin un nng
- Cc cp kh khng cng tn ti trong iu kin un nng: ngoi cc cp khng tn ti iu kin
thng cn c thm
H2 v O2 SO2 v O2 ( khi c V2O5)
3. Cng tn ti trong dung dch
- Cc cp cht cng tn ti trong mt dung dch khi khng phn ng vi nhau
- Cc phn ng xy ra trong mt dung dch thng gp
a. Phn ng trao i:
* to : ( xem tnh tan ca mui)
* to : H+ + CO32-, HCO3-...
* axit baz: OH- + H+, HCO3-, HS-...
b. Phn ng oxi ha kh
* Fe(NO3)2 + AgNO3 Fe(NO3)3 + Ag
* 3Fe2+ + NO3- + 4H+ 3Fe3+ + NO + 2H2O
GV: Trn c Tun ( 01695 178 188) Trang: 14.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
* 2Fe3+ + 2I- 2Fe2+ + I2
* 2Fe3+ + 3S2- 2FeS + S
c. Phn ng thy phn.
CO32-, HCO3- CO2
Al3+ Al(OH)3 SO2
SO32-, HSO3-
Fe3+ + + H2O Fe(OH)3 + H2S + Mui
S2-, HS- Zn(OH)2
Zn 2+ AlO2-, ZnO22- Al(OH)3, Zn(OH)2
VD: 2FeCl3 + 3Na2CO3 + 3H2O 2Fe(OH)3 + 3CO2 + 6NaCl
CU HI
Cu 1. Cu 31-CD7-439: Cc kh c th cng tn ti trong mt hn hp l
A. Cl2 v O2. B. H2S v Cl2. C. NH3 v HCl. D. HI v O3.
Cu 2.Cu 5-CD9-956: Dy gm cc ion (khng k n s phn li ca nc) cng tn ti trong mt dung
dch l:
A. Ag+, Na+, NO3-, Cl- B. Al3+, NH4+, Br-, OH-
2+ + 2- 3-
C. Mg , K , SO4 , PO4 D. H+, Fe3+, NO3-, SO42-
Cu 3.Cu 25-A10-684: Hn hp kh no sau y khng tn ti nhit thng?
A. H2S v N2. B. Cl2 v O2. C. H2 v F2. D. CO v O2.
Cu 4.Cu 3-CD10-824: Dy gm cc ion cng tn ti trong mt dung dch l:
A. Na+, K+, OH-, HCO3- B. K+, Ba2+, OH-, Cl-
C. Al3+, PO43-, Cl-, Ba2+ D. Ca2+, Cl-, Na+, CO32-
Cu 5.Cu 20-CD13-415: Dy gm cc ion cng tn ti trong mt dung dch l:
A. K+, Ba2+, Cl- v NO3- B. K+, Mg2+, OH- v NO3-
C. Cu 2+; Mg2+ ; H+ v OH. D. Cl; Na+; NO- v Ag+.
VN 11: TNG HP CC HIN TNG PHN NG
L THUYT
- Cn lu trong mi chng v cht v c u c mt s hin tng, cc hin tng ny c gii thch
da vo phn ng oxi ha kh. Cc hin tng ny c ng dng lm cc bi tp nhn bit.
- Trong chng halogen c cc hin tng nh: tnh ty mu ca clo, mu kt ta ca AgX ( X l Cl, Br,
I), phn ng mu ca iot vi h tinh bt
- Trong chng oxi lu hunh c cc hin tng nh phn ng ca O3 vi Ag hoc dd KI,...
- Trong chng nit photpho c cc hin tng v cc phn ng ca HNO3, phn ng ca NH3 to phc,
hin tng ma chi
- Trong chng cacbon silic c cc hin tng v phn ng ca CO2 vi dung dch kim
- Trong phn kim loi c cc hin tng v phn ng ca NaOH vi cc dung dch mui, hin tng ca
kim loi tc dng vi dung dch mui, hin tng ca phn ng ca st (III)
CU HI
Cu 1.Cu 7-A7-748: Nh t t cho n d dung dch NaOH vo dung dch AlCl3. Hin tng xy ra l
A. ch c kt ta keo trng. B. khng c kt ta, c kh bay ln.
C. c kt ta keo trng, sau kt ta tan. D. c kt ta keo trng v c kh bay ln.
Cu 2.Cu 40-B9-148: Th nghim no sau y c kt ta sau phn ng?
A. Cho dung dch NaOH n d vo dung dch Cr(NO3)3.
B. Cho dung dch NH3 n d vo dung dch AlCl3.
C. Cho dung dch HCl n d vo dung dch NaAlO2 (hoc Na[Al(OH)4]).
D. Thi CO2 n d vo dung dch Ca(OH)2.
Cu 3.Cu 29-CD9-956: Cht kh X tan trong nc to ra mt dung dch lm chuyn mu qu tm thnh
v c th c dng lm cht ty mu. Kh X l
GV: Trn c Tun ( 01695 178 188) Trang: 15.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
A. NH3. B. CO2. C. SO2. D. O3.
Cu 4.Cu 10-CD10-824: Cht rn X phn ng vi dung dch HCl c dung dch Y. Cho t t dung
dch NH3 n d vo dung dch Y, ban u xut hin kt ta xanh, sau kt ta tan, thu c dung
dch mu xanh thm. Cht X l
A. CuO. B. Fe. C. FeO. D. Cu.
Cu 5.Cu 12-CD10-824: Nh t t dung dch NaOH n d vo dung dch X. Sau khi cc phn ng
xy ra hon ton ch thu c dung dch trong sut. Cht tan trong dung dch X l
A. CuSO4. B. AlCl3. C. Fe(NO3)3. D. Ca(HCO3) 2.
Cu 6.Cu 14-CD11-259: Tin hnh cc th nghim sau:
1
Sc kh H2S vo dung dch FeSO4;
2
Sc kh H2S vo dung dch CuSO4;
3
Sc kh CO2 (d) vo dung dch Na2SiO3;
4
Sc kh CO2 (d) vo dung dch Ca(OH)2;
5
Nh t t dung dch NH3 n d vo dung dch Al2(SO4)3;
6
Nh t t dung dch Ba(OH)2 n d vo dung dch Al2(SO4)3.
Sau khi cc phn ng xy ra hon ton, s th nghim thu c kt ta l
A. 5. B. 6. C. 3. D. 4.
Cu 7.Cu 60-CD11-259: Khi cho lng d dung dch KOH vo ng nghim ng dung dch kali
icromat, dung dch trong ng nghim
A. chuyn t mu da cam sang mu vng. B. chuyn t mu vng sang mu .
C. chuyn t mu da cam sang mu xanh lc. D. chuyn t mu vng sang mu da cam.
Cu 8.Cu 57-A11-318: Hin tng xy ra khi nh vi git dung dch H2SO4 vo dung dch Na2CrO4 l:
A. Dung dch chuyn t mu vng sang mu da cam.
B. Dung dch chuyn t mu da cam sang mu vng.
C. Dung dch chuyn t khng mu sang mu da cam.
D. Dung dch chuyn t mu vng sang khng mu.
Cu 9.Cu 52-B12-359: Mt mu kh thi c sc vo dung dch CuSO4, thy xut hin kt ta
mu en. Hin tng ny do cht no c trong kh thi gy ra?
A. H2S. B. NO2. C. SO2. D. CO2.
Cu 10.Cu 57-B12-359: Dung dch cht X khng lm i mu qu tm; dung dch cht Y lm qu tm
ha xanh. Trn ln hai dung dch trn thu c kt ta. Hai cht X v Y tng ng l
A. KNO3 v Na2CO3. B. Ba(NO3)2 v Na2CO3.
C. Na2SO4 v BaCl2. D. Ba(NO3)2 v K2SO4.
Cu 11. Cu 12-A13-193: Cht no sau y khng to kt ta khi cho vo dung dch AgNO3 ?
A. HCl. B. K3PO4. C. KBr. D. HNO3.
Cu 12. Cu 29-CD13-415: Dung dch no di y khi phn ng hon ton vi dung dch NaOH d, thu
c kt ta trng?
A. H2SO4. B. FeCl3. C. AlCl3. D. Ca(HCO3)2.
VN 12. D ON CC PHN NG V C
L THUYT
- Cc phn ng thng gp trong ha v c cc em cn nh k cng thc phn ng v iu kin
tng ng l
1. Phn ng ha hp
2. Phn ng phn hy
3. Phn ng th
4. Phn ng trao i
5. Phn ng oxi ha kh
6. Phn ng axit baz
7. Phn ng thy phn
GV: Trn c Tun ( 01695 178 188) Trang: 16.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
CU HI
Cu 1.Cu 23-CD7-439: Cho kh CO (d) i vo ng s nung nng ng hn hp X gm Al2O3, MgO,
Fe3O4, CuO thu c cht rn Y. Cho Y vo dung dch NaOH (d), khuy k, thy cn li phn khng
tan Z. Gi s cc phn ng xy ra hon ton. Phn khng tan Z gm
A. Mg, Fe, Cu. B. MgO, Fe3O4, Cu. C. MgO, Fe, Cu. D. Mg, Al, Fe, Cu.
Cu 2.Cu 24-CD7-439: Cho kim loi M tc dng vi Cl2 c mui X; cho kim loi M tc dng vi
dung dch HCl c mui Y. Nu cho kim loi M tc dng vi dung dch mui X ta cng c mui Y.
Kim loi M c th l
A. Fe. B. Al. C. Mg. D. Zn.
Cu 3.Cu 6-A8-329: Cho cc phn ng sau:
to to
(1) Cu(NO3)2 (2) NH4NO2
t o , Pt to
(3) NH3 + O2 (4) NH3 + Cl2
o o
t t
(5) NH4Cl (6) NH3 + CuO
Cc phn ng u to kh N2 l:
A. 1, 3, 4. B. 1, 2, 5. C. 2, 4, 6. D. 3, 5, 6.
Cu 4.Cu 22-A8-329: Cho Cu v dung dch H2SO4 long tc dng vi cht X (mt loi phn bn ha
hc), thy thot ra kh khng mu ha nu trong khng kh. Mt khc, khi X tc dng vi dung dch
NaOH th c kh mi khai thot ra. Cht X l
A. amophot. B. ure. C. natri nitrat. D. amoni nitrat.
Cu 5.Cu 24-B8-371: Hn hp rn X gm Al, Fe2O3 v Cu c s mol bng nhau. Hn hp X tan hon
ton trong dung dch
A. NH3(d). B. NaOH (d). C. HCl (d). D. AgNO3 (d).
Cu 6.Cu 35-B8-371: Cho cc phn ng sau:
to
H2S + O2 ( d) Kh X + H2O
o
t , Pt
NH3 + O2 Kh Y + H2O
NH4 HCO3 + HCl long Kh Z + NH4Cl + H2O
Cc kh X, Y, Z thu c ln lt l:
A. SO2, NO, CO2. B. SO3, N2, CO2. C. SO2, N2, NH3. D. SO3, NO, NH3.
Cu 7.Cu 49-B8-371: Cho cc phn ng:
to
(1) O3 + dung dch KI (2) F2 + H2O
o
t
(3) MnO2 + HCl c (4) Cl2 + dung dch H2S
Cc phn ng to ra n cht l:
A. 1, 2, 4. B. 2, 3, 4. C. 1, 2, 3. D. 1, 3, 4.
Cu 8.Cu 5-CD8-216: Trng hp khng xy ra phn ng ha hc l
to
A. 3O2 + 2H2S 2H2O + 2SO2 B. FeCl2 + H2S FeS + 2HCl
C. O3 + 2KI + H2O 2KOH + I2 + O2 D. Cl2 + 2NaOH NaCl + NaClO + H2O
Cu 9.Cu 10-CD8-216: Cho dy cc cht: NH4Cl, (NH4)2SO4, NaCl, MgCl2, FeCl2, AlCl3. S cht
trong dy tc dng vi lng d dung dch Ba(OH)2 to thnh kt ta l
A. 5. B. 4. C. 1. D. 3.
Cu 10.Cu 55-B8-371: Cho cc dung dch: HCl, NaOH c, NH3, KCl. S dung dch phn ng c
vi Cu(OH)2 l
A. 3. B. 1. C. 2. D. 4.
Cu 11.Cu 29-CD8-216: Kim loi M phn ng c vi: dung dch HCl, dung dch Cu(NO3)2, dung
dch HNO3 (c, ngui). Kim loi M l
A. Al. B. Zn. C. Fe. D. Ag.
GV: Trn c Tun ( 01695 178 188) Trang: 17.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
Cu 12.Cu 30-CD8-216: Cho dy cc cht: KOH, Ca(NO3)2, SO3, NaHSO4, Na2SO3, K2SO4. S cht
trong dy to thnh kt ta khi phn ng vi dung dch BaCl2 l
A. 4. B. 6. C. 3. D. 2.
Cu 13.Cu 41-CD8-216: Ha tan hon ton Fe3O4 trong dung dch H2SO4 long (d) c dung dch
X1. Cho lng d bt Fe vo dung dch X1 (trong iu kin khng c khng kh) n khi phn ng xy ra
hon ton, thu c dung dch X2 cha cht tan l
A. Fe2(SO4)3 v H2SO4. B. FeSO4. C. Fe2(SO4)3. D. FeSO4 v H2SO4.
Cu 14.Cu 47-CD8-216: Cp cht khng xy ra phn ng ho hc l
A. Cu + dung dch FeCl3. B. Fe + dung dch HCl.
C. Fe + dung dch FeCl3. D. Cu + dung dch FeCl2.
Cu 15. Cu 35-A9-438: Trng hp no sau y khng xy ra phn ng ho hc?
A. Sc kh H2S vo dung dch FeCl2. B. Cho Fe vo dung dch H2SO4 long, ngui.
C. Sc kh H2S vo dung dch CuCl2. D. Sc kh Cl2 vo dung dch FeCl2.
Cu 16.Cu 40-A9-438: Cho bn hn hp, mi hn hp gm hai cht rn c s mol bng nhau: Na2O
v Al2O3; Cu v FeCl3; BaCl2 v CuSO4; Ba v NaHCO3. S hn hp c th tan hon ton trong nc
(d) ch to ra dung dch l
A. 1. B. 2. C. 4. D. 3.
Cu 17.Cu 45-A9-438: C nm dung dch ng ring bit trong nm ng nghim: (NH4)2SO4, FeCl2,
Cr(NO3)3, K2CO3, Al(NO3)3. Cho dung dch Ba(OH)2 n d vo nm dung dch trn. Sau khi phn
ng kt thc, s ng nghim c kt ta l
A. 3. B. 5. C. 2. D. 4.
Cu 18.Cu 53-A9-438: Trng hp xy ra phn ng l
A. Cu + HCl (long) B. Cu + HCl (long) + O2
C. Cu + H2SO4 (long) D. Cu + Pb(NO3)2 (long)
Cu 19.Cu 4-B9-148: Ho tan m gam hn hp gm Al, Fe vo dung dch H2SO4 long (d). Sau khi
cc phn ng xy ra hon ton, thu c dung dch X. Cho dung dch Ba(OH)2 (d) vo dung dch X, thu
c kt ta Y. Nung Y trong khng kh n khi lng khng i, thu c cht rn Z l
A. hn hp gm BaSO4 v FeO. B. hn hp gm Al2O3 v Fe2O3.
C. hn hp gm BaSO4 v Fe2O3. D. Fe2O3.
Cu 20.Cu 28-B9-148: Cho cc phn ng ha hc sau:
1 2
(NH4)2SO4 + BaCl2 CuSO4 + Ba(NO3)2
3 4
Na2SO4 + BaCl2 H2SO4 + BaSO3
5 6
(NH4)2SO4 + Ba(OH)2 Fe2(SO4)3 + Ba(NO3)2
Cc phn ng u c cng mt phng trnh ion rt gn l:
A. 1, 2, 3, 6. B. 1, 3, 5, 6. C. 2, 3, 4, 6. D. 3, 4, 5, 6.
Cu 21.Cu 44-CD9-956: Ho tan hon ton mt lng bt Zn vo mt dung dch axit X. Sau phn ng
thu c dung dch Y v kh Z. Nh t t dung dch NaOH (d) vo Y, un nng thu c kh khng mu T.
Axit X l
A. H2SO4 c. B. H2SO4 long. C. HNO3. D. H3PO4.
Cu 22.Cu 26-A10-684: Cho 4 dung dch: H2SO4 long, AgNO3, CuSO4, AgF. Cht khng tc dng
c vi c 4 dung dch trn l
A. NH3. B. KOH. C. NaNO3. D. BaCl2.
Cu 23.Cu 2-B10-937: Cho dung dch Ba(HCO3 )2 ln lt vo cc dung dch: CaCl2, Ca(NO3)2,
NaOH, Na2CO3, KHSO4, Na2SO4, Ca(OH)2, H2SO4, HCl. S trng hp c to ra kt ta l
A. 6. B. 5. C. 7. D. 4.
Cu 24.Cu 4-CD10-824: Ho tan hn hp gm: K2O, BaO, Al2O3, Fe3O4 vo nc (d), thu c
dung dch X v cht rn Y. Sc kh CO2 n d vo dung dch X, sau khi cc phn ng xy ra hon
ton thu c kt ta l
A. K2CO3. B. BaCO3. C. Fe(OH)3. D. Al(OH) 3.
GV: Trn c Tun ( 01695 178 188) Trang: 18.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
Cu 25. Cu 18-CD10-824: Cho cc dung dch long: 1 FeCl3, 2 FeCl2, 3 H2SO4, 4 HNO3, 5 hn hp
gm HCl v NaNO3. Nhng dung dch phn ng c vi kim loi Cu l:
A. 1, 3, 4. B. 1, 4, 5. C. 1, 2, 3. D. 1, 3, 5.
Cu 26.Cu 41-CD10-824: Kim loi M c th c iu ch bng cch kh ion ca n trong oxit bi
kh H nhit cao. Mt khc, kim loi M kh c ion H+ trong dung dch axit long thnh H .
2 2
Kim loi M l
A. Cu. B. Fe. C. Al. D. Mg.
Cu 27.Cu 48-CD11-259: Kh no sau y khng b oxi ho bi nc Gia-ven?
A. SO2. B. CO2. C. HCHO. D. H2S.
Cu 28.Cu 50-CD11-259: Cho hn hp X gm Cu, Ag, Fe, Al tc dng vi oxi d khi un nng c
cht rn Y. Cho Y vo dung dch HCl d, khuy k, sau ly dung dch thu c cho tc dng vi dung
dch NaOH long, d. Lc ly kt ta to thnh em nung trong khng kh n khi lng khng i thu
c cht rn Z. Bit cc phn ng xy ra hon ton. Thnh phn ca Z gm:
A. Fe2O3, CuO. B. Fe2O3, CuO, Ag. C. Fe2O3, Al2O3. D. Fe2O3, CuO, Ag2O.
Cu 29.Cu 10-A11-318: Trong cc th nghim sau:
1 2
Cho SiO2 tc dng vi axit HF. Cho kh SO2 tc dng vi kh H2S.
3 4
Cho kh NH3 tc dng vi CuO un nng. Cho CaOCl2 tc dng vi dung dch HCl c.
5 6
Cho Si n cht tc dng vi dung dch NaOH. Cho kh O3 tc dng vi Ag.
7
Cho dung dch NH4 Cl tc dng vi dung dch NaNO2 un nng.
S th nghim to ra n cht l
A. 4. B. 7. C. 6. D. 5.
Cu 30.Cu 17-A11-318: Thc hin cc th nghim sau:
1
t dy st trong kh clo.
2
t nng hn hp bt Fe v S (trong iu kin khng c oxi).
3
Cho FeO vo dung dch HNO3 (long, d).
4
Cho Fe vo dung dch Fe2(SO4)3.
5
Cho Fe vo dung dch H2SO4 (long, d).
C bao nhiu th nghim to ra mui st(II)?
A. 3. B. 2. C. 4. D. 1.
Cu 31.Cu 39-A11-318: Tin hnh cc th nghim sau:
1
Cho dung dch NaOH vo dung dch Ca(HCO3)2.
2
Cho dung dch HCl ti d vo dung dch NaAlO2 (hoc Na[Al(OH)4]).
3
Sc kh H2S vo dung dch FeCl2.
4
Sc kh NH3 ti d vo dung dch AlCl3.
5
Sc kh CO2 ti d vo dung dch NaAlO2 (hoc Na[Al(OH)4]).
6
Sc kh etilen vo dung dch KMnO4.
Sau khi cc phn ng kt thc, c bao nhiu th nghim thu c kt ta?
A. 6. B. 3. C. 5. D. 4.
Cu 32.Cu 11-B11-846: Thc hin cc th nghim sau:
(a) Nung NH4NO3 rn. (b) un nng NaCl tinh th vi dung dch H2SO4 (c).
(c) Sc kh Cl2 vo dung dch NaHCO3. (d) Sc kh CO2 vo dung dch Ca(OH)2 (d).
(e) Sc kh SO2 vo dung dch KMnO4. (g) Cho dung dch KHSO4 vo dung dch NaHCO3.
(h) Cho PbS vo dung dch HCl (long). (i) Cho Na2SO3 vo dung dch H2SO4 (d), un nng.
S th nghim sinh ra cht kh l
A. 6. B. 5. C. 4. D. 2.
Cu 33.Cu 58-B11-846: Thc hin cc th nghim sau:
(a) Nhit phn AgNO3. (b) Nung FeS2 trong khng kh.
GV: Trn c Tun ( 01695 178 188) Trang: 19.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
(c) Nhit phn KNO3. (d) Cho dung dch CuSO4 vo dung dch NH3 (d).
(e) Cho Fe vo dung dch CuSO4. (g) Cho Zn vo dung dch FeCl3 (d).
(h) Nung Ag2S trong khng kh. (i) Cho Ba vo dung dch CuSO4 (d).
S th nghim thu c kim loi sau khi cc phn ng kt thc l
A. 4. B. 3. C. 2. D. 5.
Cu 34.Cu 3-A12-296: Cho cc phn ng sau:
(a) H2S + SO2 (b) Na2S2O3 + dung dch H2SO4 (long)
to
(c) SiO2 + Mg tilemol1:2
(d) Al2O3 + dung dch NaOH
(e) Ag + O3 (g) SiO2 + dung dch HF
S phn ng to ra n cht l
A. 3. B. 6. C. 5. D. 4.
Cu 35. Cu 29-A12-296: Thc hin cc th nghim sau ( iu kin thng):
(a) Cho ng kim loi vo dung dch st(III) clorua.
(b) Sc kh hiro sunfua vo dung dch ng(II) sunfat.
(c) Cho dung dch bc nitrat vo dung dch st(III) clorua. (d) Cho bt lu hunh vo thy ngn.
S th nghim xy ra phn ng l
A. 3. B. 1. C. 4. D. 2
Cu 36.Cu 12-B12-359: Cho cc th nghim sau:
(a) t kh H2S trong O2 d; (b) Nhit phn KClO3 (xc tc MnO2);
(c) Dn kh F2 vo nc nng; (d) t P trong O2 d;
(e) Kh NH3 chy trong O2; (g) Dn kh CO2 vo dung dch Na2SiO3.
S th nghim to ra cht kh l
A. 5. B. 4. C. 2. D. 3.
Cu 37.Cu 58-B12-359: Trng hp no sau y to ra kim loi?
A. t FeS2 trong oxi d.
B. Nung hn hp qung apatit, x vn v than cc trong l ng.
C. t Ag2S trong oxi d.
D. Nung hn hp qung photphorit, ct v than cc trong l in.
Cu 38.Cu 2-CD12-169: Tin hnh cc th nghim sau:
1 2
Cho Zn vo dung dch AgNO3; Cho Fe vo dung dch Fe2(SO4)3;
3 4
Cho Na vo dung dch CuSO4; Dn kh CO (d) qua bt CuO nng.
Cc th nghim c to thnh kim loi l
A. 1 v 2. B. 1 v 4. C. 2 v 3. D. 3 v 4.
Cu 39.Cu 17-CD12-169: Cho Fe tc dng vi dung dch H2SO4 long to thnh kh X; nhit phn tinh th
KNO3 to thnh kh Y; cho tinh th KMnO4 tc dng vi dung dch HCl c to thnh kh Z. Cc kh X, Y v Z
ln lt l
A. SO2, O2 v Cl2. B. H2, NO2 v Cl2. C. H2, O2 v Cl2. D. Cl2, O2 v H2S.
Cu 40.Cu 27-CD12-169: Dung dch long (d) no sau y tc dng c vi kim loi st to thnh
mui st(III)?
A. H2SO4. B. HNO3. C. FeCl3. D. HCl.
Cu 41. Cu 58-CD12-169: Cho dy cc kim loi: Cu, Ni, Zn, Mg, Ba, Ag. S kim loi trong dy
phn ng c vi dung dch FeCl3 l
A. 6. B. 4. C. 3. D. 5.
Cu 42. Cu 20-A13-193: Thc hin cc th nghim sau:
(a) Cho dung dch HCl vo dung dch Fe(NO3)2. (b) Cho FeS vo dung dch HCl.
(c) Cho Si vo dung dch NaOH c. (d) Cho dung dch AgNO3 vo dung dch NaF.
(e) Cho Si vo bnh cha kh F2. (f) Sc kh SO2 vo dung dch H2S.
Trong cc th nghim trn, s th nghim c xy ra phn ng l
A. 5. B. 3. C. 6. D. 4.
Cu 43. Cu 21-A13-193: Kim loi st tc dng vi dung dch no sau y to ra mui st(II)?
GV: Trn c Tun ( 01695 178 188) Trang: 20.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
A. HNO3 c, nng, d. B. CuSO4. C. H2SO4 c, nng, d. D. MgSO4.
Cu 44. Cu 4-B13-279: Ha tan hon ton Fe3O4 trong dung dch H2SO4 long (d), thu c dung dch
X. Trong cc cht: NaOH, Cu, Fe(NO3)2, KMnO4, BaCl2, Cl2 v Al, s cht c kh nng phn ng c
vi dung dch X l
A. 4. B. 6. C. 5. D. 7.
Cu 45. Cu 43-B13-279: Thc hin cc th nghim sau:
(a) Cho Al vo dung dch HCl. (b) Cho Al vo dung dch AgNO3.
(c) Cho Na vo H2O. (d) Cho Ag vo dung dch H2SO4 long.
Trong cc th nghim trn, s th nghim xy ra phn ng l
A. 1. B. 2. C. 4. D. 3.
Cu 46. Cu 48-B13-279: Mt mu kh thi c cha CO2, NO2, N2 v SO2 c sc vo dung dch
Ca(OH)2 d. Trong bn kh , s kh b hp th l
A. 3. B. 4. C. 1. D. 2.
Cu 47. Cu 51-B13-279: Ha tan mt kh X vo nc, thu c dung dch Y. Cho t t dung dch Y n
d vo dung dch ZnSO4, ban u thy c kt ta trng, sau kt ta tan ra. Kh X l
A. NO2. B. HCl. C. SO2. D. NH3.
Cu 48. Cu 59-B13-279: Trng hp no sau y khng xy ra phn ng?
A. Au + HNO3 c B. Ag + O3
C. Sn + HNO3 long D. Ag + HNO3 c
Cu 49. Cu 23-CD13-415: Thc hin cc th nghim sau:
(a) Sc kh Cl2 vo dung dch NaOH nhit thng.
(b) Cho Fe3O4 vo dung dch HCl long (d).
(c) Cho Fe3O4 vo dung dch H2SO4 c, nng (d).
(d) Ha tan ht hn hp Cu v Fe2O3 (c s mol bng nhau) vo dung dch H2SO4 long (d).
Trong cc th nghim trn, sau phn ng, s th nghim to ra hai mui l
A. 1. B. 3. C. 2. D. 4.
Cu 50. Cu 54-CD13-415: Kim loi Ni u phn ng c vi cc dung dch no sau y?
A. NaCl, AlCl3. B. AgNO3, NaCl. C. CuSO4, AgNO3. D. MgSO4, CuSO4.
VN 13: LM KH KH
L THUYT
1. Cht lm kh:
- c tc dng ht m: H2SO4 c, dd kim, CuSO4, CaCl2, CaO, P2O5
- khng tc dng vi cht cn lm kh..
2. Kh cn lm kh.
H2, CO, CO2, SO2,SO3, H2S,O2, N2, NH3, NO2,Cl2, HCl, hidrocacbon.
3. Bng tm tt.
Dd kim, CaO H2SO4, P2O5 CaCl2 khan,CuSO4 khan
Kh lm H2, CO, O2, N2, NO, H2, CO2, SO2, O2, Tt c
kh c NH3, CxHy N2, NO, NO2, Cl2, HCl,
CxHy. Ch : vi CuSO4
khng lm kh c H2S,
NH3
Kh CO2, SO2, SO3, NO2, NH3.
khng lm Cl2, HCl, H2S
kh c Ch : H2SO4 khng
lm kh c H2S, SO3
cn P2O5 th lm kh c
GV: Trn c Tun ( 01695 178 188) Trang: 21.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
CU HI
Cu 1.Cu 4-CD7-439: C th dng NaOH ( th rn) lm kh cc cht kh
A. N2, NO2, CO2, CH4, H2. B. NH3, SO2, CO, Cl2.
C. NH3, O2, N2, CH4, H2. D. N2, Cl2, O2 , CO2, H2.
Cu 2.Cu 20-CD9-956: Cht dng lm kh kh Cl2 m l
A. CaO. B. dung dch H2SO4 m c. C. Na2SO3 khan. D. dung dch NaOH c.
VN 14: DY IN HA
L THUYT
1. Cp oxi ho - kh ca kim loi
- Nguyn t kim loi d nhng electron tr thnh ion kim loi, ngc li ion kim loi c th nhn
electron tr thnh nguyn t kim loi.
VD : Ag+ + 1e Ag Cu2+ + 2e Cu Fe2+ + 2e Fe
- Cc nguyn t kim loi (Ag, Cu, Fe,...) ng vai tr cht kh, cc ion kim loi (Ag+, Cu2+, Fe2+...) ng
vai tr cht oxi ho.
- Cht oxi ho v cht kh ca cng mt nguyn t kim loi to nn cp oxi ho - kh. Th d ta c cp
oxi ho - kh : Ag+/Ag ; Cu2+/Cu ; Fe2+/Fe.
Kt lun: Ni cp oxi ha kh l ni dng oxi ha trc dng kh sau, v chng ta ghi dng oxi ha trn
dng kh.
* Tng qut: Dng oxi ha
Dng kh.
2. So snh tnh cht ca cc cp oxi ho - kh
VD: So snh tnh cht ca hai cp oxi ho - kh Cu 2+/Cu v Ag+/Ag, thc nghim cho thy Cu tc dng
c vi dung dch mui Ag+ theo phng trnh ion rt gn :
Cu + 2Ag+ Cu 2+ + 2Ag
So snh : Ion Cu2+ khng oxi ho c Ag, trong khi Cu kh c ion Ag+. Nh vy, ion Cu2+ c
tnh oxi ho yu hn ion Ag+. Kim loi Cu c tnh kh mnh hn Ag.
- so snh cp oxi ha kh ta so snh tnh oxi ha ca dng oxi ha, tnh kh ca dng kh. M chiu
phn ng oxi ha kh l cht kh mnh phn ng vi cht oxi ha mnh to cht kh v cht oxi ha yu
hn.
+ tnh oxi ha: Cu2+ < Ag+
+ tnh kh: Cu > Ag
3. Dy in ho ca kim loi
Ngi ta so snh tnh cht ca nhiu cp oxi ho - kh v sp xp thnh dy in ho ca kim loi :
Tnh oxi ha ca ion kim loi tng dn
K+ Ba2+ Ca2+ Na+ Mg2+ Al3+ Zn2+ Fe2+ Ni2+ Sn2+ Pb2+ H+ Cu2+ Fe3+ Ag+
K Ba Ca Na Mg Al Zn Fe Ni Sn Pb H Cu Fe2+ Ag
Tnh kh ca kim loi gim dn
4. ngha ca dy in ho ca kim loi
ng dng 1: Xc nh th t u tin
Xc nh th t u tin phn ng ca cht kh, ca cht oxi ha.
Lu nu c hn hp nhiu cht oxi ha kh tc dng vi nhau th ta mi xt th t u tin.
Lut phn ng oxihoa kh.
Cht Mnh Cht yu
( p trc n ht) ( p tip )
ng dng 2: Quy tc
( Quy tc dng d on phn ng)
GV: Trn c Tun ( 01695 178 188) Trang: 22.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
Gi l quy tc v ta v ch l t c phn ng.
Tng qut:
Ox 1 Ox 2
Kh 1 Kh 2
=> phn ng:Ox 2 + Kh1 Ox 1 + Kh2 .
Dy in ho ca kim loi cho php d on chiu ca phn ng gia 2 cp oxi ho - kh theo quy tc
(anpha) : Phn ng gia 2 cp oxi ho - kh s xy ra theo chiu, cht oxi ho mnh nht s oxi ho cht
kh mnh nht, sinh ra cht oxi ho yu hn v cht kh yu hn.
CU HI
Cu 1.Cu 27-A7-748: Dy cc ion xp theo chiu gim dn tnh oxi ho l (bit trong dy in ha,
3+/ 2+ +
cp Fe Fe ng trc cp Ag /Ag):
A. Ag+, Cu2+, Fe3+, Fe2+. B. Fe3+, Ag+, Cu2+, Fe2+.
+ 3+ 2+ 2+
C. Ag , Fe , Cu , Fe . D. Fe3+, Cu2+, Ag+, Fe2+.
Cu 2.Cu 26-B07-285: Cho cc phn ng xy ra sau y:
AgNO3 + Fe(NO3)2 Fe(NO3)3 + Ag
Mn + 2HCl MnCl2 + H2
Dy cc ion c sp xp theo chiu tng dn tnh oxi ho l
A. Mn2+, H+, Fe3+, Ag+. B. Ag+, Fe3+, H+, Mn2+.
C. Ag+ , Mn2+ , H+, Fe3+. D. Mn2+, H+, Ag+, Fe3+.
Cu 3.Cu 40-CD7-439: Th t mt s cp oxi ho - kh trong dy in ho nh sau: Fe2+ /Fe; Cu 2+/Cu;
Fe3+/Fe2+ . Cp cht khng phn ng vi nhau l
A. Fe v dung dch FeCl3. B. dung dch FeCl2 v dung dch CuCl2.
C. Fe v dung dch CuCl2. D. Cu v dung dch FeCl3.
Cu 4.Cu 54-CD7-439: Cho cc ion kim loi: Zn2+, Sn2+, Ni2+, Fe2+, Pb 2+. Th t tnh oxi ho gim
dn l
A. Zn2+ > Sn2+ > Ni2+ > Fe2+ > Pb2+. B. Pb2+ > Sn2+ > Ni2+ > Fe2+ > Zn2+.
C. Sn2+ > Ni2+ > Zn2+ > Pb2+ > Fe2+. D. Pb2+ > Sn2+ > Fe2+ > Ni2+ > Zn2+.
Cu 5.Cu 4-A8-329: X l kim loi phn ng c vi dung dch H2SO4 long, Y l kim loi tc
dng c vi dung dch Fe(NO3)3. Hai kim loi X, Y ln lt l (bit th t trong dy th in ho:
Fe3+/Fe2+ ng trc Ag+/Ag)
A. Ag, Mg. B. Cu, Fe. C. Fe, Cu. D. Mg, Ag.
Cu 6.Cu 39-CD8-216: Cho hn hp bt Al, Fe vo dung dch cha Cu(NO3)2 v AgNO3. Sau khi cc
phn ng xy ra hon ton, thu c hn hp rn gm ba kim loi l:
A. Fe, Cu, Ag. B. Al, Cu, Ag. C. Al, Fe, Cu. D. Al, Fe, Ag.
Cu 7.Cu 1-A9-438: Cho hn hp gm Fe v Zn vo dung dch AgNO3 n khi cc phn ng xy ra
hon ton, thuc dung dch X gm hai mui v cht rn Y gm hai kim loi. Hai mui trong X l
A. Zn(NO3)2 v Fe(NO3)2. B. Fe(NO3)3 v Zn(NO3)2.
C. Fe(NO3)2 v AgNO3. D. AgNO3 v Zn(NO3)2.
Cu 8.Cu 58-CD9-956: Th t mt s cp oxi ho - kh trong dy in ho nh sau: Mg2+/Mg;
Fe2+/Fe; Cu2+ /Cu; Fe3+/Fe2+; Ag+ /Ag. Dy ch gm cc cht, ion tc dng c vi ion Fe3+ trong dung
dch l:
A. Mg, Fe, Cu. B. Mg, Fe2+, Ag. C. Fe, Cu, Ag+. D. Mg, Cu, Cu2+.
Cu 9.Cu 6-CD10-824: Cho bit th t t tri sang phi ca cc cp oxi ho - kh trong dy in
ho (dy th in cc chun) nh sau: Zn2+/Zn; Fe2+/Fe; Cu2+/Cu; Fe3+/Fe2+; Ag+ /Ag.
Cc kim loi v ion u phn ng c vi ion Fe2+ trong dung dch l:
GV: Trn c Tun ( 01695 178 188) Trang: 23.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
A. Zn, Ag+. B. Zn, Cu2+. C. Ag, Fe3+. D. Ag, Cu2+ .
Cu 10.Cu 44-CD11-259: Dy gm cc ion u oxi ha c kim loi Fe l
A. Fe3+, Cu2+, Ag+. B. Zn2+, Cu2+, Ag+. C. Cr2+, Au3+, Fe3+. D. Cr2+, Cu2+, Ag+.
Cu 11.Cu 58-A11-318: Cho cc phn ng sau: Fe + 2Fe(NO3)3 3Fe(NO3)2
AgNO3 + Fe(NO3)2 Fe(NO3)3 + Ag
Dy sp xp theo th t tng dn tnh oxi ho ca cc ion kim loi l:
A. Ag+, Fe3+ , Fe2+. B. Fe2+ , Ag+, Fe3+ . C. Fe2+, Fe3+, Ag+. D. Ag+, Fe2+ , Fe3+.
Cu 12.Cu 32-A12-296: Cho cc cp oxi ho - kh c sp xp theo chiu tng dn tnh oxi ho ca
dng oxi ha nh sau: Fe2+ /Fe, Cu 2+/Cu, Fe3+ /Fe2+. Pht biu no sau y l ng?
A. Fe2+ oxi ha c Cu thnh Cu2+. B. Cu2+ oxi ho c Fe2+ thnh Fe3+.
C. Fe3+ oxi ha c Cu thnh Cu2+. D. Cu kh c Fe3+ thnh Fe.
Cu 13.Cu 46-A12-296: Cho hn hp gm Fe v Mg vo dung dch AgNO3, khi cc phn ng xy ra
hon ton thu c dung dch X (gm hai mui) v cht rn Y (gm hai kim loi). Hai mui trong X l
A. Mg(NO3)2 v Fe(NO3)2. B. Fe(NO3)3 v Mg(NO3)2.
C. AgNO3 v Mg(NO3)2. D. Fe(NO3)2 v AgNO3.
Cu 14.Cu 35-CD -169: Cho dy cc ion: Fe2 +, Ni2+, Cu2+, Sn2+. Trong cng iu kin, ion c
12
tnh oxi ha mnh nht trong dy l
A. Sn2+. B. Cu2+. C. Fe2+. D. Ni2+.
Cu 15. Cu 24-A13-193: Cho bt Fe vo dung dch gm AgNO3 v Cu(NO3)2. Sau khi cc phn ng
xy ra hon ton, thu c dung dch X gm hai mui v cht rn Y gm hai kim loi. Hai mui trong
X v hai kim loi trong Y ln lt l:
A. Fe(NO3)2; Fe(NO3)3 v Cu; Ag. B. Cu(NO3)2; AgNO3 v Cu; Ag.
C. Cu(NO3)2; Fe(NO3)2 v Cu; Fe. D. Cu(NO3)2; Fe(NO3)2 v Ag; Cu.
Cu 16. Cu 44-A13-193: Cho cc cp oxi ha - kh c sp xp theo th t tng dn tnh oxi ha ca
cc ion kim loi: Al3+/Al; Fe2+/Fe; Sn2+/Sn; Cu 2+/Cu. Tin hnh cc th nghim sau:
(a) Cho st vo dung dch ng(II) sunfat.
(b) Cho ng vo dung dch nhm sunfat.
(c) Cho thic vo dung dch ng(II) sunfat.
(d) Cho thic vo dung dch st(II) sunfat.
Trong cc th nghim trn, nhng th nghim c xy ra phn ng l:
A. (a) v (b). B. (b) v (c). C. (a) v (c). D. (b) v (d).
VN 15: CHT OXI HA, CHT KH - S OXI HA, S KH
L THUYT
1. Khi nim
- Cht kh l cht nhng electron
- Cht oxi ha l cht nhn electron
- S kh l qu trnh nhn electron
- S oxi ha l s nhng electron.
=> Cht v s ngc nhau.
2. Cch xc nh cht oxi ha cht kh.
- Cn nh: Kh cho tng, O nhn gim
Ngha l cht kh cho electron s oxi ha tng, cht oxi ha nhn electron s oxi ha gim.
- xc nh c cht oxi ha cht kh ng ta da vo mt s kinh nghim sau:
* Cht va c tnh oxi ha kh l nhng cht:
- c nguyn t c s oxi ha trung gian nh FeO, SO2, Cl2
GV: Trn c Tun ( 01695 178 188) Trang: 24.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
- c ng thi nguyn t c soh thp v nguyn t c soh cao ( thng gp cc hp cht
ca halogen, NO3-) nh: HCl, NaCl, FeCl3, HNO3, NaNO3.
* Cht ch c tnh kh: l nhng cht ch c nguyn t c s oxi ha thp th hin tnh cht nh
H2S, NH3
* Cht ch c tnh oxi ha l nhng cht ch c nguyn t c s oxi ha cao th hin tnh cht nh
F2, O2, O3.
CU HI
Cu 1.Cu 35-CD8-216: Cho phn ng ha hc: Fe + CuSO4 FeSO4 + Cu.
Trong phn ng trn xy ra
A. s kh Fe2+ v s oxi ha Cu. B. s kh Fe2+ v s kh Cu 2+.
C. s oxi ha Fe v s oxi ha Cu. D. s oxi ha Fe v s kh Cu2+ .
Cu 2.Cu 23-B07-285: Khi cho Cu tc dng vi dung dch cha H2SO4 long v NaNO3, vai tr ca
NaNO3 trong phn ng l
A. cht xc tc. B. cht oxi ho. C. mi trng. D. cht kh.
2+
Cu 3.Cu 15-CD7-439: kh ion Cu trong dung dch CuSO4 c th dng kim loi
A. Ba. B. K. C. Fe. D. Na.
Cu 4.Cu 16-CD7-439: SO2 lun th hin tnh kh trong cc phn ng vi
A. O2, nc Br2, dung dch KMnO4. B. dung dch KOH, CaO, nc Br2.
C. dung dch NaOH, O2, dung dch KMnO4. D. H2S, O2, nc Br2.
Cu 5.Cu 19-CD -439: kh ion Fe3 trong dung dch thnh ion Fe2+ c th dng mt lng d
7
+
A. kim loi Cu. B. kim loi Ba. C. kim loi Ag. D. kim loi Mg.
Cu 6.Cu 35-A8-329: Cho cc phn ng sau:
4HCl + MnO2 MnCl2 + Cl2 + 2H2O. 2HCl + Fe FeCl2 + H2.
14HCl + K2Cr2O7 2KCl + 2CrCl3 + 3Cl2 + 7H2O. 6HCl + 2Al 2AlCl3 + 3H2.
16HCl + 2KMnO4 2KCl + 2MnCl2 + 5Cl2 + 8H2O.
S phn ng trong HCl th hin tnh oxi ha l
A. 3. B. 4. C. 2. D. 1.
Cu 7.Cu 23-A9-438: Cho dy cc cht v ion: Zn, S, FeO, SO2, N2, HCl, Cu2+, Cl-. S cht v ion c
c tnh oxi ha v tnh kh l
A. 6. B. 5. C. 7. D. 4.
Cu 8.Cu 27-B -371: Cho dy cc cht v ion: Cl , F , SO , Na , Ca2 , Fe2+, Al3+, Mn2+, S2-, Cl-.
8 2 2 2
+ +
S cht v ion trong dy u c tnh oxi ho v tnh kh l
A. 5. B. 6. C. 4. D. 3.
Cu 9.Cu 16-B9-148: Cho cc phn ng sau:
(a) 4HCl + PbO2 PbCl2 + Cl2 + 2H2O. (b) HCl + NH4HCO3 NH4Cl + CO2 + H2O.
(c) 2HCl + 2HNO3 2NO2 + Cl2 + 2H2O. (d) 2HCl + Zn ZnCl2 + H2.
S phn ng trong HCl th hin tnh kh l
A. 2. B. 3. C. 1. D. 4.
Cu 10.Cu 13-CD9-956: Trong cc cht: FeCl2, FeCl3, Fe(NO3)2, Fe(NO3)3, FeSO4, Fe2(SO4)3. S cht
c c tnh oxi ho v tnh kh l
A. 2. B. 4. C. 3. D. 5.
Cu 11.Cu 7-A10-684: Nung nng tng cp cht sau trong bnh kn: 1 Fe + S (r), 2 Fe2O3 + CO (k), 3 Au +
O2 (k), 4 Cu + Cu(NO3)2 (r), 5 Cu + KNO3 (r), 6 Al + NaCl (r). Cc trng hp xy ra phn ng oxi ho kim
loi l:
A. 1, 4, 5. B. 2, 3, 4. C. 1, 3, 6. D. 2, 5, 6.
GV: Trn c Tun ( 01695 178 188) Trang: 25.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
Cu 12.Cu 39-CD10-824: Nguyn t S ng vai tr va l cht kh, va l cht oxi ho trong phn
ng no sau y?
to
A. S + 2Na Na2S
o
t
B. S + 3F2 SF6
to
C. S + 6HNO3 c H2SO4 + 6NO2 + 2H2O
to
D. 4S + 6NaOH c 2Na2S + Na2S2O3 + 3H2O
Cu 13.Cu 31-A11-318: Cho dy cc cht v ion: Fe, Cl2, SO2, NO2, C, Al, Mg2+, Na+, Fe2+, Fe3+ . S
cht v ion va c tnh oxi ho, va c tnh kh l
A. 4. B. 6. C. 8. D. 5.
Cu 14.Cu 22-B11-846: Cho cc phn ng:
(a) Sn + HCl long (b) FeS + H2SO4 long
(c) MnO2 + HCl c (d) Cu + H2SO4 c
(e) Al + H2SO4 long ( g) FeSO4 + KmnO4 + H2SO4
+
S phn ng m H ng vai tr cht oxi ha l
A. 3. B. 5. C. 6. D. 2.
Cu 15. Cu 36-CD -169: Cho dy gm cc phn t v ion: Zn, S, FeO, SO , Fe2+, Cu 2+, HCl. Tng
12 2
s phn t v ion trong dy va c tnh oxi ha, va c tnh kh l
A. 5. B. 6. C. 7. D. 4.
Cu 16. Cu 4-A13-193: iu kin thch hp xy ra cc phn ng sau:
(a) 2C + Ca CaC2 (b) C + 2H2 CH4
( c) C + CO2 2CO ( d) 3C + 4Al Al4C3
Trong cc phn ng trn, tnh kh ca cacbon th hin phn ng
A. (a). B. (c). C. (d). D. (b).
Cu 17.Cu 45-B13-279: Cho phng trnh ha hc ca phn ng: 2Cr + 3Sn2+ 2Cr3+ + 3Sn.
Nhn xt no sau y v phn ng trn l ng?
A. Sn2+ l cht kh, Cr3+ l cht oxi ha. B. Cr l cht oxi ha, Sn2+ l cht kh.
C. Cr l cht kh, Sn2+ l cht oxi ha. D. Cr3+ l cht kh, Sn2+ l cht oxi ha.
Cu 18. Cu 26-CD13-415: Cho cc phng trnh phn ng sau:
(a) Fe + 2HCl FeCl2 + H2.
(b) Fe3O4 + 4H2SO4 Fe2(SO4)3 + FeSO4 + 4H2O.
(c) 2KMnO4 + 16HCl 2KCl + 2MnCl2 + 5Cl2 + 8H2O.
(d) FeS + H2SO4 FeSO4 + H2S.
(e) 2Al + 3H2SO4 Al2(SO4)3 + 3H2.
Trong cc phn ng trn, s phn ng m ion H+ ng vai tr cht oxi ha l
A. 1. B. 3. C. 2. D. 4.
VN 16: HON THNH PHN NG OXI HA KH
L THUYT
I. CN BNG PHN NG OXI HA KH
1. PHNG PHP THNG BNG ELECTRON
B1. Xc nh s oxi ho cc nguyn t. Tm ra nguyn t c s oxi ho thay i .
B2. Vit cc qu trnh lm thay i s oxi ho
Cht c oxi ho tng : Cht kh - ne s oxi ho tng
Cht c s oxi ho gim: Cht oxi ho + me s oxi ho gim
B3. Xc nh h s cn bng sao cho s e cho = s e nhn
GV: Trn c Tun ( 01695 178 188) Trang: 26.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
B4. a h s cn bng vo phng trnh, ng cht (Nn a h s vo bn phi ca pt trc) v kim
tra li theo trt t : kim loi phi kim hidro oxi
VD: Lp ptp oxh-k sau: Al + HNO3 Al(NO3)3 + N2O + H2O.
0 5 3 1
Al H N O3 Al ( NO3 ) 3 N 2 O H 2 O
0 3
8 Al Al 3e
5 1
3 2 N 2.4e 2 N
0 5 3 1
8 Al 30 H N O3 8 Al ( NO3 ) 3 3 N 2 O 15H 2 O
2. MT S V D VN DNG
DNG 1: PHNG TRNH PHN NG C MT CHT OXI HO V MT CHT KH
V d 1: Lp phng trnh ho hc sau theo phng php thng bng electron:
Fe2O3 + CO Fe + CO2
Bc 1: Xc nh s oxi ho ca nhng nguyn t c s oxi ho thay i.
Vn dng cc quy tc xc nh s oxi ho :
Fe+3 2O3 + C+2O Fe0 + C+4 O2
Bc 2: Vit qu trnh oxi ho v qu trnh kh, cn bng mi qu trnh.
Trc khi cn bng mi qu trnh thun tin cho cc phng trnh ta nn dng mt k xo l cn
bng s nguyn t thuc 2 v phng trnh sau nhn s lng cc nguyn t vi s electron nhng
hoc nhn.
2 Fe+3 + 2x 3e 2 Fe0
C+2 C+4 + 2e
Bc 3: Tm h s thch hp cho tng s electron cho cht kh nhng bng tng s electron m cht
oxi ho nhn
1 2 Fe+3 + 2x 3e 2 Fe0
3 C+2 C+4 + 2e
Bc 4: t h s ca cc oxi ho v cht kh vo s phn ng. Hon thnh phng trnh ho
hc
Fe2O3 + 3CO 2 Fe + 3CO2
V d 2: Lp phng trnh ho hc sau theo phng php thng bng electron:
MnO2 + HCl MnCl2 + Cl2 + H2O
Bc 1: Xc nh s oxi ho ca nhng nguyn t c s oxi ho thay i.
Vn dng cc quy tc xc nh s oxi ho :
Mn+4 O2 + HCl-1 Mn+2Cl2 + Cl02 + H2O
Bc 2: Vit qu trnh oxi ho v qu trnh kh, cn bng mi qu trnh.
Mn+4 + 2e Mn+2
-1
2 Cl Cl2 + 2e
Bc 3: Tm h s thch hp cho tng s electron cho cht kh nhng bng tng s electron m cht
oxi ho nhn
1 Mn+4 + 2e Mn+2
-1
1 2 Cl Cl2 + 2e
Bc 4: t h s ca cc oxi ho v cht kh vo s phn ng. Hon thnh phng trnh ho
hc
MnO2 + 4 HCl MnCl2 + Cl2 + 2H2O
V d 3: Lp phng trnh ho hc sau theo phng php thng bng electron:
Fe3 O4 + HNO3 long Fe(NO3)3 + NO + H2O
Bc 1: Xc nh s oxi ho ca nhng nguyn t c s oxi ho thay i.
Fe3+8/3O4 + HN+5O3 long Fe+3(NO3)3 + N+2O + H2O
Bc 2: Vit qu trnh oxi ho v qu trnh kh, cn bng mi qu trnh.
in trc Fe+8/3 v Fe+3 h s 3 trc khi cn bng mi qu trnh.
3Fe+8/3 + 3x(3- 8/3) e 3 Fe+3
GV: Trn c Tun ( 01695 178 188) Trang: 27.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
N+5 N+2 + 3e
Bc 3: Tm h s thch hp cho tng s electron cho cht kh nhng bng tng s electron m cht
oxi ho nhn
3 3Fe+8/3 + 3x(3- 8/3) e 3 Fe+3
1 N+5 N+2 + 3e
Bc 4: t h s ca cc oxi ho v cht kh vo s phn ng. Hon thnh phng trnh ho hc
3Fe3 O4 + 28HNO3 long 9 Fe(NO3)3 + NO + 14 H2O
V d 4 : Lp phng trnh ho hc sau theo phng php thng bng electron:
FeSO4 + K2Cr2O7 + H2SO4 Fe2(SO4)3 + K2SO4 + Cr2(SO4)3 + H2O
Bc 1: Xc nh s oxi ho ca nhng nguyn t c s oxi ho thay i.
Fe+2SO4 + K2Cr+62O7 + H2SO4 Fe+32(SO4)3 + K2SO4 + Cr+32(SO4)3 + H2O
Bc 2: Vit qu trnh oxi ho v qu trnh kh, cn bng mi qu trnh.
in trc Fe+2 v Fe+3 h s 2. in trc Cr+6 v Cr+3 h s 2 trc khi cn bng mi qu trnh.
2Fe +2 + 2 x 1e 2 Fe+3
+6
2Cr 2Cr+3 + 2x3e
Bc 3: Tm h s thch hp cho tng s electron cho cht kh nhng bng tng s electron m cht
oxi ho nhn
3 2Fe +2 2 Fe+3 + 2 x 1e
+6
1 2 Cr + 2x3e 2Cr+3
Bc 4: t h s ca cc oxi ho v cht kh vo s phn ng. Hon thnh phng trnh ho hc
6FeSO4 + K2Cr2O7 + 7 H2SO4 3Fe2(SO4)3 + K2SO4 + Cr2(SO4)3 + 7H2O
V d 5:Lp phng trnh ho hc sau theo phng php thng bng electron:
Al + Fe3O4 Al2O3 + Fe
Bc 1: Xc nh s oxi ho ca nhng nguyn t c s oxi ho thay i.
Al0 + Fe3+8/3O4 Al2+3O3 + Fe0
Bc 2: Vit qu trnh oxi ho v qu trnh kh, cn bng mi qu trnh.
in trc Fe+8/3 v Fe0 h s 3. in trc Al0 v Al+3 h s 2 trc khi cn bng mi qu trnh.
3Fe +8/3 + 3 x 8/3e 3 Fe0
0
2 Al 2Al+3 + 2x3e
Bc 3: Tm h s thch hp cho tng s electron cho cht kh nhng bng tng s electron m cht
oxi ho nhn
3 3Fe +8/3 + 3 x 8/3e 3 Fe0
0
4 2 Al 2Al+3 + 2x3e
Bc 4: t h s ca cc oxi ho v cht kh vo s phn ng. Hon thnh phng trnh ho hc
8 Al + 3Fe3O4 4Al2O3 + 9Fe
V d 6:Lp phng trnh ho hc sau theo phng php thng bng electron:
Fe(OH)2 + O2 + H2O Fe(OH)3
Bc 1: Xc nh s oxi ho ca nhng nguyn t c s oxi ho thay i.
Fe+2(OH)2 + O02 + H2O Fe+3(O-2H)3
Bc 2: Vit qu trnh oxi ho v qu trnh kh, cn bng mi qu trnh.
in trc O-2 h s 2. trc khi cn bng mi qu trnh.
Fe +2 Fe+3 + 1e
0
O 2 + 2x2e 2O- 2
Bc 3: Tm h s thch hp cho tng s electron cho cht kh nhng bng tng s electron m cht
oxi ho nhn
4 Fe +2 Fe+3 + 1e
0
1 O 2 + 2x2e 2O- 2
Bc 4: t h s ca cc oxi ho v cht kh vo s phn ng. Hon thnh phng trnh ho
hc
4 Fe(OH)2 + O2 + 2 H2O 4 Fe(OH)3
V d 7:Lp phng trnh ho hc sau theo phng php thng bng electron:
GV: Trn c Tun ( 01695 178 188) Trang: 28.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
KClO4 + Al KCl + Al2O3
Bc 1: Xc nh s oxi ho ca nhng nguyn t c s oxi ho thay i.
KCl+7O4 + Al0 KCl-1 + Al+32O3
Bc 2: Vit qu trnh oxi ho v qu trnh kh, cn bng mi qu trnh.
in trc Al0 v Al+3 h s 2. trc khi cn bng mi qu trnh.
2Al 0 2Al+3 + 2x3e
+7
Cl + 8e Cl-
Bc 3: Tm h s thch hp cho tng s electron cho cht kh nhng bng tng s electron m cht
oxi ho nhn
4 2Al 0 2Al+3 + 2x3e
+7 -
3 Cl + 8e Cl
Bc 4: t h s ca cc oxi ho v cht kh vo s phn ng. Hon thnh phng trnh ho
hc
3 KCl+7O4 + 8 Al0 3 KCl-1 + 4 Al+32O3
Nh vy cn bng s nguyn t bng s ion hoc s ion bng s ion trc khi cn bng cc qu trnh
oxi ho v qu trnh kh gip ngi lm thun tin hn rt nhiu ln, cho kt qu nhanh hn v phc
tp hn.
DNG 2: PHNG TRNH PHN NG T OXI HO V T KH
V d 1:Lp phng trnh ho hc sau theo phng php thng bng electron:
Cl2 + NaOH NaCl + NaClO + H2O
Bc 1: Xc nh s oxi ho ca nhng nguyn t c s oxi ho thay i.
Cl02 + NaOH NaCl-1 + NaCl+1O + H2O
Bc 2: Vit qu trnh oxi ho v qu trnh kh, cn bng mi qu trnh.
in trc Cl- v Cl+ ca cc qu trnh h s 2 trc khi cn bng.
Cl02 + 2x1e 2Cl-
0
Cl 2 2Cl+ + 2x 1e
Bc 3: Tm h s thch hp cho tng s electron cho cht kh nhng bng tng s electron m cht
oxi ho nhn
1 Cl02 + 2x1e 2Cl-
0
1 Cl 2 2Cl+ + 2x 1e
Bc 4: t h s ca cc oxi ho v cht kh vo s phn ng. Hon thnh phng trnh ho
hc
2 Cl2 + 4 NaOH 2 NaCl + 2 NaClO + 2 H2O
Rt gn cc h s thu c phng trnh vi h s ti gin
Cl2 + 2 NaOH NaCl + NaClO + H2O
V d 2:Lp phng trnh ho hc sau theo phng php thng bng electron:
Cl2 + NaOH NaCl + NaClO3 + H2O
Bc 1: Xc nh s oxi ho ca nhng nguyn t c s oxi ho thay i.
Cl02 + NaOH NaCl-1 + NaCl+5O3 + H2O
Bc 2: Vit qu trnh oxi ho v qu trnh kh, cn bng mi qu trnh.
in trc Cl- v Cl+5 ca cc qu trnh h s 2 trc khi cn bng.
Cl02 + 2x1e 2Cl-
0
Cl 2 2Cl+5 + 2x 5e
Bc 3: Tm h s thch hp cho tng s electron cho cht kh nhng bng tng s electron m cht
oxi ho nhn
5 Cl02 + 2x1e 2Cl-
0 +5
1 Cl 2 2Cl + 2x 5e
Bc 4: t h s ca cc oxi ho v cht kh vo s phn ng. Hon thnh phng trnh ho
hc
6 Cl2 + 12 NaOH 10 NaCl + 2NaClO3 + 6 H2O
Rt gn cc h s thu c phng trnh vi h s ti gin
3 Cl2 + 6 NaOH 5 NaCl + NaClO + 3H2O
GV: Trn c Tun ( 01695 178 188) Trang: 29.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
DNG 3 : PHNG TRNH PHN NG C MT CHT OXI HO V HAI CHT KH
V d 1:Lp phng trnh ho hc sau theo phng php thng bng electron:
FeS2 + O2 Fe2O3 + SO2
Bc 1: Xc nh s oxi ho ca nhng nguyn t c s oxi ho thay i.
Fe+2S-12 + O02 Fe+32O-23 + S+4O-22
Bc 2: Vit qu trnh oxi ho v qu trnh kh, cn bng mi qu trnh.
Trc tin ta vit cc qu trnh oxi ho, tng hp cc qu trnh oxi ho sao cho l s nguyn ln cht
kh. Thm h s 2 vo trc Fe+2 v Fe+3 , thm h s 4 vo trc S-2 v S+4 c s nguyn ln FeS2
Qu trnh oxi ho:
2Fe+2 2 Fe+3 + 2x1e
-1
4S 4 S+4 + 4x 5e
2 FeS2 2 Fe+3 + 4 S+4 + 22e
Sau cn bng qu trnh kh:
in h s 2 vo trc O-2 :
O02 + 2x 2e 2 O-2
Tng hp 2 qu trnh oxi ho v qu trnh kh:
2 FeS2 2 Fe+3 + 4 S+4 + 22e
0
O 2 + 2x 2e 2 O-2
Bc 3: Tm h s thch hp cho tng s electron cho cht kh nhng bng tng s electron m cht
oxi ho nhn
2 2 FeS2 2 Fe+3 + 4 S+4 + 22e
0
11 O 2 + 2x 2e 2 O-2
Bc 4: t h s ca cc oxi ho v cht kh vo s phn ng. Hon thnh phng trnh ho
hc
4 FeS2 + 11 O2 2Fe2O3 + 8 SO2
V d 2:Lp phng trnh ho hc sau theo phng php thng bng electron:
Fe S2 + HNO3 Fe(NO3)3 + H2SO4 + NO2 + H2O
Bc 1: Xc nh s oxi ho ca nhng nguyn t c s oxi ho thay i.
Fe+2S-12 + HN+5O3 Fe+3(NO3)3 + H2S+6O4 + N+4O2 + H2O
Bc 2: Vit qu trnh oxi ho v qu trnh kh, cn bng mi qu trnh.
Trc tin ta vit cc qu trnh oxi ho, tng hp cc qu trnh oxi ho sao cho l s nguyn ln cht
kh. Thm h s 2 vo trc S-1 v S+6 , c s nguyn ln FeS2
Qu trnh oxi ho:
Fe+2 Fe+3 + 1e
-1
2S 2 S+6 + 2x 7e
FeS2 Fe+3 + 2 S+4 + 15e
Sau cn bng qu trnh kh:
N+5 + 1e N+4
Tng hp 2 qu trnh oxi ho v qu trnh kh:
FeS2 Fe+3 + 2 S+4 + 15e
N+5 + 1e N+4
Bc 3: Tm h s thch hp cho tng s electron cho cht kh nhng bng tng s electron m cht
oxi ho nhn
1 FeS2 Fe+3 + 2 S+4 + 15e
15 N+5 + 1e N+4
Bc 4: t h s ca cc oxi ho v cht kh vo s phn ng. Hon thnh phng trnh ho
hc
Fe S2 + 18 HNO3 Fe(NO3)3 + 2 H2SO4 + 15 NO2 + 7 H2O
DNG 4 : PHNG TRNH PHN NG C HAI CHT OXI HO V MT CHT KH
V d 1: Lp phng trnh ho hc sau theo phng php thng bng electron:
Fe + HNO3 Fe(NO3)3 + NO + NO2 + H2O ( t l NO:NO2=1:2)
Bc 1: Xc nh s oxi ho ca nhng nguyn t c s oxi ho thay i.
GV: Trn c Tun ( 01695 178 188) Trang: 30.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
Fe0 + HN+5O3 Fe+3(NO3)3 + N+2O + N+4O2 + H2O
Bc 2: Vit qu trnh oxi ho v qu trnh kh, cn bng mi qu trnh.
Trc tin ta vit cc qu trnh kh, tng hp cc qu trnh kh sao cho ng t l vi yu cu bi.
Thm h s 2 vo trc N+4
Qu trnh Kh:
N+5 + 3e N+2
+5
2N + 2x 1e 2 N+4
3N+5 + 5e N+2 + 2 N+4
Sau cn bng qu trnh oxi ho :
Fe0 Fe+3 + 3e
Tng hp 2 qu trnh oxi ho v qu trnh kh:
3N+5 + 5e N+2 + 2 N+4
0 +3
Fe Fe + 3e
Bc 3: Tm h s thch hp cho tng s electron cho cht kh nhng bng tng s electron m cht
oxi ho nhn
3 3N+5 + 5e N+2 + 2 N+4
0 +3
5 Fe Fe + 3e
Bc 4: t h s ca cc oxi ho v cht kh vo s phn ng. Hon thnh phng trnh ho
hc
5Fe + 24 HNO3 5Fe(NO3)3 +3NO + 6NO2 + 12H2O ( t l NO:NO2=1:2)
II. HON THNH PHN NG OXI HA KH
vit c cc phn ng oxi ha kh th chng ta cn bit mt s cht oxi ha v mt s cht kh
thng gp. Cht oxi ha sau khi b kh th to thnh cht kh lin hp (cht kh tng ng); Cng
nh cht kh sau khi b oxi ha th to thnh cht kh lin hp (cht kh tng ng). Ta phi bit
cc cht kh v cht oxi ha tng ng th mi vit c phn ng oxi ha kh.
1. CC CHT OXI HA THNG GP
a. Cc hp cht ca mangan: KMnO4, K2MnO4, MnO2 (MnO4-, MnO42-, MnO2)
- KMnO4, K2MnO4, MnO2 trong mi trng axit (H+) thng b kh thnh mui Mn2+
VD: 2KMnO4 + 10FeSO4 + 8H2SO4 2MnSO4 + 5Fe2(SO4)3 + K2SO4 + 8H2O
2KMnO4 + 5KNO2 + 3H2SO4 2MnSO4 + 5KNO3 + K2SO4 + 3H2O
K2MnO4 + 4FeSO4 + 4H2SO4 MnSO4 + 2Fe2(SO4)3 + K2SO4 + 4H2O
MnO2 + 4HCl() MnCl2 + Cl2 + 2H2O
MnO2 + 2FeSO4 + 2H2SO4MnSO4 + Fe2(SO4)3 + 2H2O
2KMnO4 + 10NaCl + 8H2SO4 2MnSO4 + 5Cl2 + K2SO 4 + 5Na2SO4 + 8H2O
- KMnO4 trong mi trng trung tnh (H2O) thng b kh thnh mangan ioxit (MnO2)
VD: 2KMnO4 + 4K2SO3 + H2O MnO2 + K2SO4 + KOH
2KMnO4 + 3MnSO4 + 2H2O 5MnO2 + K2SO4 + 2H2SO4
2KMnO4 + 3H2O2 2MnO2 + 3O2 + 2KOH + 2H2O
- KMnO4 trong mi trng baz (OH-) thng b kh to K2MnO4
VD: 2KMnO4 + K2SO3 + 2KOH 2K2MnO4 + K2SO4 + H2O
Lu :
- KMnO4 trong mi trng axit (thng l H2SO4) c tnh oxi ha rt mnh, nn n d b mt mu tm
bi nhiu cht kh nh: Fe2+; FeO; Fe3O4; SO2; SO32-; H2S; S2-; NaCl; HCl; KBr, HBr, HI; KI; Cl-; Br-; I-
; NO2-; Anken; Ankin; Ankaien; Aren ng ng benzen;
- KMnO4 c th ng vai tr cht oxi ha trong mi trng axit (H+), baz (OH-) hoc trung tnh (H2 O).
Cn K2MnO4, MnO2 ch c th ng vai tr cht oxi ha trong mi trng axit
b. Hp cht ca crom: K2Cr2O7; K2CrO4 (Cr2O72-; CrO42-)
- K2Cr2O7 (Kali icromat; Kali bicromat), K2CrO4 (Kali cromat) trong mi trng axit (H+) thng b
kh thnh mui crom (III) (Cr3+)
VD: K2Cr2O7 + 6FeSO4 + 7H2SO4 Cr2(SO4)3 + 3Fe2(SO4)3 + K2SO4 + 7H2O
GV: Trn c Tun ( 01695 178 188) Trang: 31.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
K2Cr2O7 + 3K2SO3 + 4H2SO4 Cr2(SO4)3 + 4K2SO4 + 4H2O
- Trong mi trng trung tnh, mui cromat (CrO42-) thng b kh to crom (III) hiroxit (Cr(OH)3)
VD: 2KCrO4 + 3(NH4)2S + 2H2O 2Cr(OH)3 + 3S + 6NH3 + 4KOH
c. Axit nitric (HNO3), mui nitrat trong mi trng axit (NO3-/H+)
- HNO3 m c thng b kh to kh mu nu nit ioxit NO2. Cc cht kh thng b HNO3 oxi
ha l: cc kim loi, cc oxit kim loi c s oxi ha trung gian (FeO, Fe3O4), mt s phi kim (C, S, P),
mt s hp cht ca phi kim c s oxi ha thp nht hay trung gian (H2S, SO2, SO32-, HI), mt s hp
cht ca kim loi trong kim loi c s oxi ha trung gian (Fe2+, Fe(OH)2)
VD: Fe + 6HNO3 (, nng) Fe(NO3)3 + 3NO2 + 3H2O
FeO + 4HNO3() Fe(NO3)3 + NO2 + 2H2O
Fe3O4 + 10HNO3() 3Fe(NO3)3 + NO2 + 5H2O
Fe(OH)2 + 4HNO3() Fe(NO3)3 + NO2 + 3H2O
C + 4HNO3() CO2 + 4NO2 + 2H2O
S + 6HNO3() H2SO4 + 6NO2 + 2H2O
P + 5HNO3() H3PO4 + 5NO2 + H2O
Al + 6HNO3(, nng) Al(NO3)3 + 3NO2 + 3H2O
- HNO3 long thng b kh thnh NO (kh nit oxit). Cc cht kh thng gp l: cc kim loi, cc
oxit kim loi hay hp cht kim loi c s oxi ha trung gian (FeO, Fe(OH)2, Fe3O4, Fe2+), mt s phi
kim (S, C, P), mt s hp cht ca phi kim trong phi kim c s oxi ho thp nht hoc c s oxi ha
trung gian (NO2-, SO3 ).
VD: 3Fe(OH)2 + 10HNO3(l) 3Fe(NO3)3 + NO + 8H2O
3FeO + 10HNO3(l) 3Fe(NO3)3 + NO + 5H2O
3Fe3O4 + 28HNO3(l) 9Fe(NO3)3 + NO + 14H2O
Cr + 4HNO3(l) Cr(NO3)3 + NO + 2H2O
3P + 5HNO3(l) + 2H2O 3H3PO4 + 5NO
- Mui nitrat trong mi trng axit (NO3-/H+) ging nh HNO3 long, nn n oxi ha c cc kim
loi to mui, NO3- b kh to kh NO, ng thi c s to nc (H2O)
VD: 3Cu + 2NaNO3 + 8HCl 3CuCl2 + 2NO + 2NaCl + 4H2O
3Cu + Cu(NO3)2 + 8HCl 4CuCl2 + 2NO + 4H2O
- Ba kim loi st (Fe), nhm (Al) v crom (Cr) khng b ha tan trong dung dch axit nitric m c
ngui (HNO3 , ngui) cng nh trong dung dch axit sunfuric m c ngui (H2SO4 , ngui) (b
th ng ha, b tr).
- Cc kim loi mnh nh magie (Mg), nhm (Al), km (Zn) khng nhng kh HNO3 to NO2,
NO, m c th to N2O, N2, NH4NO3. Dung dch HNO3 cng long th b kh to hp cht ca N hay
n cht ca N c s oxi ha cng thp.
VD: 8Al + 30HNO3(kh long) 8Al(NO3)3 + 3N2O + 15H2O
10Al + 36HNO3(rt long) 10Al(NO3)3 + 3N2 + 18H2O
8Al + 30HNO3(qu long) 8Al(NO3)3 + 3NH4NO3 + 9H2O
Lu : - thng bi tp khng vit r l kh long, rt long, qu long m ch vit long. Nu vit
long m to sn phm kh N2O, N2, NH4NO3 th ta vn vit phn ng bnh thng nh trn ch khng
c ni l khng th to ra N2O, N2, NH4NO3
- Mt kim loi tc dng dung dch HNO3 to cc kh khc nhau, tng qut mi kh ng vi mt phn ng
ring. Ch khi no bit t l s mol cc kh ny th mi vit chung cc kh trong cng mt phn ng vi t
l s mol kh tng ng.
d. Axit sunfuric m c nng, H2SO4(, nng)
- H2SO4(, nng) thng b kh to kh SO2. Cc cht kh thng tc dng vi H2SO4(, nng) l:
cc kim loi, cc hp cht ca kim loi s oxi ha trung gian (nh FeO, Fe3O4), mt s phi kim (nh
C, S, P), mt s hp cht ca phi kim (nh HI, HBr, H2S)
VD: 2Fe + 6H2SO4(, nng) Fe2(SO4)3 + 3SO2 + 6H2O
2FeO + 4H2SO4(, nng) Fe2(SO4)3 + SO2 + 4H2O
2Fe3O4 + 10H2SO4(, nng) 3Fe2(SO4)3 + SO2 + 10H2O
GV: Trn c Tun ( 01695 178 188) Trang: 32.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
Fe2O3 + 3H2SO4(, nng) Fe2(SO4)3 + 3H2O (phn ng trao i)
S + 2H2SO4(, nng) 3SO2 + 2H2O
C + 2H2SO4(, nng) CO2 + 2SO2 + 2H2O
2P + 5H2SO4(, nng) 2H3PO4 + 5SO2 +2H2O
2HBr + H2SO4(, nng) Br2 + SO2 + 2H2O
- Cc kim loi mnh nh Mg, Al, Zn khng nhng kh H2SO4 m c, nng thnh SO2 m cn
thnh S, H2S. H2SO4 m c nhng nu long bt th s b kh to lu hunh (S) hay hp cht ca
lu hunh c s oxi ha thp hn (H2S). Nguyn nhn ca tnh cht trn l do kim loi mnh nn d
cho in t ( H2SO4 nhn nhiu in t) v do H2SO4 t m c nn n khng oxi ha tip S, H2S.
VD: 2Al + 6H2SO4(, nng) Al2(SO4)3 + 3SO2 + 6H2O
8Al + 15H2SO4(hi c, nng) 4Al2(SO4)3 + 3H2S + 12H2O
2Al + 3H2SO4(long) Al2(SO4)3 + 3H2
- Khc vi HNO3, dung dch H2SO4 long l a xit thng thng (tc nhn oxi ha l H+), ch dung
dch H2SO4 m c, nng mi l axit c tnh oxi ha mnh (tc nhn oxi ha l SO42-). Trong khi
dung dch HNO3 k c m c ln long u l axit c tnh o xi ha mnh (tc nhn oxi ha l NO3-)
2. CC CHT KH THNG GP
a. Kim loi
- Tt c kim loi u l cht kh. Kim loi b kh to thnh hp cht ca kim loi trong kim loi c s
oxi ha dng. Phn ng no c kim loi tham gia th l phn ng oxi ha kh v kim loi lun lun
ng vai tr cht kh. Kim loi c th kh cc phi kim, axit thng thng, nc, axit c tnh oxi ha
mnh, mui ca kim loi yu hn, oxit ca kim loi yu hn, dung dch kim,
- Kim loi kh phi kim (F2, Cl2, Br2, I2, O2, S, N2, P, C, Si, H2) to mui hay oxit
VD: 2Fe + 3Cl2 2FeCl3
Fe + S FeS
3Fe + 2O2 Fe3O4
- Kim loi kh ion H+ ca axit thng thng, to mui v kh hiro.
Kim loi ng trc H trong dy th in ho kh c ion H+ ca axit thng thng to kh hiro (H2),
cn kim loi b oxi ho to mui: K Ca Na Mg Al Mn Zn Cr Fe Ni Sn Pb H Cu Ag Hg Pt Au
VD: Fe + H2SO4(l) FeSO4 + H2
2Al + 6HCl 2AlCl3 + 3H2
Cu + HCl khng phn ng
- Kim loi kim, kim th kh c nc nhit thng, to hiroxit kim loi v kh hiro.
Kim loi kim: Li, Na, K, Rb, Cs, Fr
Kim loi kim th: Ca, Sr, Ba, Ra
VD: Na+ H2O NaOH + H2
Ca + 2H2O Ca(OH)2 + H2
- Kim loi (tr vng, bch kim) kh c axit c tnh oxi ho mnh [HNO3, H2SO4(c, nng)] to
mui, kh NO2, NO hay SO2 v H2O.
- Kim loi mnh (tr kim loi kim, kim th) kh c ion kim loi yu hn trong dung dch mui.
K Ca Na Mg Al Mn Zn Cr Fe Ni Sn Pb H Cu Ag Hg Pt Au
- Cc kim loi c oxit lng tnh (tr Cr, gm Al, Zn, Be, Sn, Pb) kh c dung dch kim, to mui
v kh hiro.
b. Hp cht ca kim loi trong kim loi c s oxi ha trung gian, m thng gp l Fe(II) [nh
FeO, Fe(OH)2, FeSO4, FeCl2, Fe(NO3)2, Fe2+, FeS, FeS2], Fe3O4, Cr(II), Cu 2O. Cc cht kh ny b oxi
ha to thnh hp cht ca kim loi c s oxi ha cao hn.
VD: 2FeO + 1/2O2 Fe2O3
3FeO + 10HNO3(l) 3Fe(NO3)3 + NO + 5H2O
4Fe(OH)2 + O2 2Fe2O3 + 4H2O
3Fe(OH)2 + 10HNO3(l) 3Fe(NO3)3 + NO + 8H2O
10FeSO4 + 2KMnO4 + 8H2SO4 5Fe2(SO4)3 + 2MnSO4 + K2SO4 + 8H2O
FeCO3 + 4HNO3() Fe(NO3)3 + NO2 + CO2 + 2H2O
GV: Trn c Tun ( 01695 178 188) Trang: 33.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
FeS2 + 18HNO3() Fe(NO3)3 + 2H2SO4 + 15NO2 + 7H2O
2FeS2 + 14H2SO4(, nng) Fe2(SO4)3 + 15SO2 + 14H2O
c. Mt s phi kim, nh H2, C, S, P, Si, N2, Cl2. Cc phi kim ny b oxi ha to thnh hp cht ca phi
kim, trong phi kim c s oxi ha dng. Cc cht oxi ha thng dng oxi ha cc phi kim l oxit
kim loi, oxi, HNO3, H2SO4(c, nng).
d. Mt s hp cht ca phi kim, trong phi kim c s oxi ha trung gian, nh CO, NO, NO2, NO2,
SO2, SO32, Na2S2O3, FeS2, P2O3, C2H4, C2H2,Cc hp cht ny b oxi ha to thnh hp cht ca phi
kim trong phi kim c s oxi ha cao hn.
e. Cc hp cht ca phi kim, trong phi kim c s oxi ha thp nht (cc tiu), nh X (Cl, Br,
I, HCl, HBr, HI), S2, H2S, NH3, PH3, CH4, NaH, CaH2, Cc hp cht b oxi ha to phi kim n cht
hay hp cht ca phi kim c s oxi ha cao hn.
Lu : - Phn t no ch cn cha mt nguyn t c s oxi ha gim sau phn ng th c th kt lun
phn t l cht oxi ha; Cng phn t no ch cn cha mt nguyn t c s oxi ha tng sau phn ng
th c th kt lun phn t cht l cht kh.
- Nguyn t no c s oxi ha ti a (trong hp cht) nu tham gia phn ng oxi ha kh th nguyn t
ny ch c th ng vai tr cht oxi ha, v s oxi ha ca nguyn t ny ch c th gim, ch khng tng
c na.
VD: Fe3+ ; KMnO4 ; K2Cr2O7 ; HNO3 ; H2SO4(, nng) ; CuO ; H+ ; Ag+ ; Au3+ ; Zn2+
- Nguyn t no c s oxi ha thp nht (trong n cht kim loi, trong hp cht ca phi kim) nu tham
gia phn ng oxi ha kh th s ng vai tr cht kh, v s oxi ha ca nguyn t ny ch c th tng ch
khng gim c na.
VD: Tt c cc kim loi, nh: Na ; Mg ; Al ; Ag ; Hg ; Au ; Cc hp cht ca phi kim,
nh: X (F , Cl , Br , I) ; HCl ; HBr ; HI ; H2S ; S2 ; H ; NaH ; CaH2 ; NH3 ; PH3 ; CH4; SiH4 ; O2.
- Cn nguyn t no c s oxi ha trung gian (trong n cht phi kim, cc hp cht ca kim loi hay phi
kim trong kim loi hay phi kim c s oxi ha trung gian) nu tham gia phn ng oxi ha kh th ty
trng hp (ty theo tc cht m chng phn ng) m c th ng vai tr cht oxi ha hoc ng vai tr
cht kh.
VD: H2 ; C ; Si ; O2 ; S ; Cl2 ; Br2 ; I2 ; Fe2+ ; FeO ; Fe3O4 ; FeCl2 ; FeSO4 ; Cu 2O ; SO2 ;Na2S2O3 ; NO2.
- C phn t m trong phn t c cha c nguyn t c oxi ha cao nht ln nguyn t c s oxi ha thp
nht, do ty trng hp m phn t ny hoc l cht oxi ha hoc l cht kh hoc l cht trao i
(khng l cht oxi ha, khng l cht kh).
VD: HCl, H2S, KMnO4, K2Cr2O7, Fe2O3, KClO3
CU HI
Cu 1.Cu 22-A7-748: Tng h s (cc s nguyn, ti gin) ca tt c cc cht trong phng trnh
phn ng gia Cu vi dung dch HNO3 c, nng l
A. 10. B. 9. C. 8. D. 11.
Cu 2.Cu 4-B07-285: Trong phn ng t chy CuFeS2 to ra sn phm CuO, Fe2O3 v SO2 th mt phn
t CuFeS2 s
A. nhn 13 electron. B. nhn 12 electron.
C. nhng 13 electron. D. nhng 12 electron.
Cu 3.Cu 29-A 9-438: Cho phng trnh ho hc: Fe 3O4 + HNO3 Fe(NO3)3 + NxO y + H2O
Sau khi cn bng phng trnh ho hc trn vi h s ca cc cht l nhng s nguyn, ti gin th h s
ca HNO3 l
A. 23x - 9y. B. 45x - 18y. C. 13x - 9y. D. 46x - 18y.
Cu 4.Cu 17-A10-684: Thc hin cc th nghim sau:
(I) Sc kh SO2 vo dung dch KMnO4. (II) Sc kh SO2 vo dung dch H2S.
(III) Sc hn hp kh NO2 v O2 vo nc. (IV) Cho MnO2 vo dung dch HCl c, nng.
(V) Cho Fe2O3 vo dung dch H2SO4 c, nng. (VI) Cho SiO2 vo dung dch HF.
S th nghim c phn ng oxi ho - kh xy ra l
GV: Trn c Tun ( 01695 178 188) Trang: 34.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
A. 3. B. 6. C. 4. D. 5.
Cu 5.Cu 31-A7-748: Cho cc phn ng sau:
a) FeO + HNO3 (c, nng) b) FeS + H2SO4 (c, nng)
c) Al2O3 + HNO3 (c, nng) d) Cu + dung dch FeCl3
o
Ni ,t
e) CH3CHO + H2 f) glucoz + AgNO3 trong dung dch NH3
g) C2H4 + Br2 h) Glixerol + Cu(OH)2
Dy gm cc phn ng u thuc loi phn ng oxi ha - kh l:
A. a, b, c, d, e, h. B. a, b, c, d, e, g. C. a, b, d, e, f, h. D. a, b, d, e, f, g.
Cu 6.Cu 24-CD8-216: Cho dy cc cht: FeO, Fe(OH)2, FeSO4, Fe3O4, Fe2(SO4)3, Fe2O3. S cht
trong dy b oxi ha khi tc dng vi dung dch HNO3 c, nng l
A. 3. B. 5. C. 4 D. 6.
Cu 7.Cu 32-A7-748: Cho tng cht: Fe, FeO, Fe(OH)2, Fe(OH)3, Fe3O4, Fe2O3, Fe(NO3)2, Fe(NO3)3,
FeSO4, Fe2(SO4)3, FeCO3 ln lt phn ng vi HNO3 c, nng. S phn ng thuc loi phn ng oxi ho -
kh l
A. 5. B. 6. C. 7. D. 8.
Cu 8.Cu 47-A10-684: Trong phn ng: K2Cr2O7 + HCl CrCl3 + Cl2 + KCl + H2O
S phn t HCl ng vai tr cht kh bng k ln tng s phn t HCl tham gia phn ng. Gi tr
ca k l
A. 1/7. B. 4/7. C. 3/7. D. 3/14.
Cu 9.Cu 12-B10-937: Cho dung dch X cha KMnO4 v H2SO4 (long) ln lt vo cc dung dch:
FeCl2, FeSO4, CuSO4, MgSO4, H2S, HCl (c). S trng hp c xy ra phn ng oxi ho - kh l
A. 5. B. 4. C. 6. D. 3.
Cu 10.Cu 34-CD10-824: Cho phn ng:
Na2SO3 + KMnO4 + NaHSO4 Na2SO4 + MnSO4 + K2SO4 + H2 O.
Tng h s ca cc cht (l nhng s nguyn, ti gin) trong phng trnh phn ng l
A. 27. B. 47. C. 31. D. 23.
Cu 11.Cu 3-CD11-259: Dy gm cc kim loi u tc dng c vi dung dch HCl nhng khng tc
dng vi dung dch HNO3 c, ngui l:
A. Cu, Fe, Al. B. Fe, Al, Cr. C. Cu, Pb, Ag. D. Fe, Mg, Al.
Cu 12.Cu 15-CD11-259: Cho cc cht: KBr, S, SiO2, P, Na3PO4, FeO, Cu v Fe2O3. Trong cc cht
trn, s cht c th b oxi ha bi dung dch axit H2SO4 c, nng l
A. 4. B. 5. C. 7. D. 6.
Cu 13.Cu 13-B11-846: Thc hin cc th nghim vi hn hp bt gm Ag v Cu (hn hp X):
(a) Cho X vo bnh cha mt lng d kh O3 ( iu kin thng).
(b) Cho X vo mt lng d dung dch HNO3 (c).
(c) Cho X vo mt lng d dung dch HCl (khng c mt O2).
(d) Cho X vo mt lng d dung dch FeCl3.
Th nghim m Cu b oxi ho cn Ag khng b oxi ho l
A. (c). B. (a). C. (d). D. (b).
Cu 14.Cu 34-A12-296: Dy cht no sau y u th hin tnh oxi ha khi phn ng vi SO2 ?
A. Dung dch BaCl2, CaO, nc brom. B. Dung dch NaOH, O2, dung dch KMnO4.
C. O2, nc brom, dung dch KMnO4. D. H2S, O2, nc brom.
Cu 15. Cu 29-B12-359: Cho cc cht ring bit sau: FeSO4, AgNO3, Na2SO3, H2S, HI, Fe3O4, Fe2O3
tc dng vi dung dch H2SO4 c, nng. S trng hp xy ra phn ng oxi ho - kh l
A. 6. B. 3. C. 4. D. 5.
Cu 16.Cu 32-B12-359: Cho cc cht sau: FeCO3, Fe3O4, FeS, Fe(OH)2. Nu ho tan cng s mol mi
cht vo dung dch H2SO4 c, nng (d) th cht to ra s mol kh ln nht l
A. Fe3O4. B. Fe(OH)2. C. FeS. D. FeCO3.
Cu 17.Cu 42-B12-359: Cho phng trnh ha hc (vi a, b, c, d l cc h s):
GV: Trn c Tun ( 01695 178 188) Trang: 35.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
aFeSO4 + bCl2 cFe2(SO4)3 + dFeCl3
T l a : c l
A. 4 : 1. B. 3 : 2. C. 2 : 1. D. 3 : 1.
Cu 18.Cu 54-B12-359: Ho tan Au bng nc cng toan th sn phm kh l NO; ho tan Ag trong
dung dch HNO3 c th sn phm kh l NO2. s mol NO2 bng s mol NO th t l s mol Ag v Au
tng ng l
A. 1 : 2. B. 3 : 1. C. 1 : 1. D. 1 : 3.
Cu 19. Cu 29-CD12-169: Cho phn ng ha hc: Cl2+ KOH KCl + KClO3 + H2O
T l gia s nguyn t clo ng vai tr cht oxi ha v s nguyn t clo ng vai tr cht kh
trong phng trnh ha hc ca phn ng cho tng ng l
A. 1 : 5. B. 5 : 1. C. 3 : 1. D. 1 : 3.
Cu 20. Cu 5-A13-193: Tin hnh cc th nghim sau:
(a) Sc kh etilen vo dung dch KMnO4 long.
(b) Cho hi ancol etylic i qua bt CuO nung nng.
(c) Sc kh etilen vo dung dch Br2 trong CCl4.
(d) Cho dung dch glucoz vo dung dch AgNO3 trong NH3 d, un nng.
(e) Cho Fe2O3 vo dung dch H2SO4 c, nng.
Trong cc th nghim trn, s th nghim c xy ra phn ng oxi ha - kh l
A. 3. B. 4. C. 2. D. 5.
Cu 21. Cu 50-A13-193: Cho phng trnh phn ng aAl + bHNO3 cAl(NO3)3 + dNO + eH2O
T l a: b l
A. 1 : 3 B. 2 : 3 C. 2 : 5 D. 1 : 4
Cu 22. Cu 57: Cho phng trnh phn ng
aFeSO4 + bK2Cr2O7 + cH2SO4 dFe2(SO4)3 + eK2SO4 + fCr2(SO4)3 + gH2O
T l a : b l
A. 6 : 1. B. 2 : 3. C. 3 : 2. D. 1 : 6.
Cu 23. Cu 36-B13-279: Cho phn ng: FeO + HNO3 Fe(NO3)3 + NO + H2O.
Trong phng trnh ca phn ng trn, khi h s ca FeO l 3 th h s ca HNO3 l
A. 6. B. 8. C. 4. D. 10.
Cu 24. Cu 50-CD13-415: Cho cc phng trnh phn ng:
(a) 2Fe + 3Cl2 2FeCl3. (b) NaOH + HCl NaCl + H2O.
(c) Fe3O4 + 4CO 3Fe + 4CO2. (d) AgNO3 + NaCl AgCl + NaNO3.
Trong cc phn ng trn, s phn ng oxi ha - kh l
A. 3. B. 2. C. 4. D. 1.
VN 17: PHN LOI PHN NG HA HC
L THUYT
- Qu trnh bin i t cht ny thnh cht khc gi l phn ng ha hc. Cht ban u, b bin i trong
phn ng gi l cht tham gia (hay cht phn ng), cht mi sinh ra l sn phm. Phn ng ha hc c
ghi theo phng trnh ch nh sau:
Tn cc cht phn ng Tn cc sn phm
Nhng loi phn ng thng gp bao gm :
Phn ng ha hp: L phn ng ha hc trong ch c mt cht mi (sn phm) c to thnh t hai
hay nhiu cht ban u.
Phn ng phn hy: L phn ng ha hc trong mt cht sinh ra hai hay nhiu cht mi.
Phn ng oxi ha - kh: L phn ng ha hc trong xy ra ng thi s oxi ha v s kh.
Phn ng th
GV: Trn c Tun ( 01695 178 188) Trang: 36.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
Ngoi ra cn c cc phn ng khc nh phn ng trao i, phn ng ta nhit,phn ng trung ha,....
CU HI
Cu 1.Cu 45-B07-285: Cho 4 phn ng:
1
Fe + 2HCl FeCl2 + H2
2
2NaOH + (NH4)2SO4 Na2SO4 + 2NH3 + 2H2O
3
BaCl2 + Na2CO3 BaCO3 + 2NaCl
4
2NH3 + 2H2O + FeSO4 Fe(OH)2 + (NH4)2SO4
Cc phn ng thuc loi phn ng axit - baz l
A. 2, 4. B. 3, 4. C. 2, 3. D. 1, 2.
Cu 2.Cu 12-B8-371: Cho cc phn ng:
Ca(OH)2 + Cl2 CaOCl2 + H2O 2H2S + SO2 3S + 2H2O
2NO2 + 2NaOH NaNO3 + NaNO2 + H2O 4KClO3 KCl + 3KClO4
O3 O 2 + O
S phn ng oxi ha kh l:
A. 2. B. 3. C. 5. D. 4.
Cu 3.Cu 45-A12-296: Cho cc phn ng sau:
(a) FeS + 2HCl FeCl2 + H2S
(b) Na2S + 2HCl 2NaCl + H2S
(c) 2AlCl3 + 3Na2S + 6H2O 2Al(OH)3 + 3H2S + 6NaCl
(d) KHSO4 + KHS K2SO4 + H2S
(e) BaS + H2SO4 (long) BaSO4 + H2S
S phn ng c phng trnh ion rt gn S2- + 2H+ H2S l
A. 1. B. 3. C. 2. D. 4.
Cu 4.Cu 9-A13-193: Trong iu kin thch hp, xy ra cc phn ng sau:
(a) 2H2SO4 + C 2SO2 + CO2 + 2H2O
(b) H2SO4 + Fe(OH)2 FeSO4 + 2H2O
(c) 4H2SO4 + 2FeO Fe2(SO4)3 + SO2 + 4H2O
(d) 6H2SO4 + 2Fe Fe2(SO4)3 + 3SO2 + 6H2O
Trong cc phn ng trn, phn ng xy ra vi dung dch H2SO4 long l
A. (d). B. (a). C. (c). D. (b).
VN 18: QUNG V HP CHT THNG GP
L THUYT
1. Mt s qung thng gp
1.Qung photphorit. Ca3(PO4)2. 2. Qung apatit
3. Sinvinit: NaCl. KCl ( phn kali) 4. Magiezit: MgCO3
5. Canxit: CaCO3 6. olomit: CaCO3. MgCO3
7. Boxit: Al2O3.2H2O. 8. Mica: K2O. Al2O3.6SiO2.2H2O
9. t st: Al2O3.6SiO2.2H2O 10. fensfat: K2O. Al2O3.6SiO2
11. criolit: Na3AlF6. 12. mahetit: Fe3O4
13.hematit nu: Fe2O3.nH2O. 14. hematit : Fe2O3
15.xiderit: FeCO3 16.pirit st: FeS2
GV: Trn c Tun ( 01695 178 188) Trang: 37.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
17.florit CaF2. 18.Chancopirit ( pirit ng ) CuFeS2
2. Mt s hp cht thng gp
1. Phn chua: K2SO4. Al2(SO4)3. 24H2O 2. Thch cao sng CaSO4. 2H2O
3. Thch cao nung CaSO4.H2O 4. Thch cao khan CaSO4
5. Dim tiu KNO3 6. Dim sinh S
7. vi CaCO3 8. Vi sng CaO
9. Vi ti Ca(OH)2 dng c 10. Mui n NaCl
11. Xt NaOH 12. Potat KOH
13. Thch anh SiO2 14. Oleum H2SO4.nSO3
15. m ure (NH2)2CO 16. m 2 l NH4NO3
17. Supephotphat n Ca(H2PO4)2 + 2CaSO4 18. Supephotphat kp Ca(H2PO4)2
19. Amophot NH4H2PO4 v (NH4)2HPO4 20. Bt n: NaHCO3 ( lu : NH4 HCO3 l bt
khai)
21. Thy tinh thng: Na2O.CaO.6SiO2 22. Thy tinh kali: K2O.CaO.6SiO2
23. Thy tinh lng: Na2SiO3 v K2SiO3 2 24. Pha l: thy tinh cha nhiu PbO2
25. Silicagen ( cht ht m): H2SiO3 mt mt phn 26. thy tinh thch anh: cha nhiu SiO2
nc
CU HI
Cu 1.Cu 50-A8-329: Trong cc loi qung st, qung c hm lng st cao nht l
A. hematit . B. xierit. C. hematit nu. D. manhetit.
Cu 2.Cu 31-B8-371: Thnh phn chnh ca qung photphorit l
A. Ca(H2PO4)2. B. Ca3(PO4)2. C. NH4H2PO4. D. CaHPO4.
Cu 3.Cu 57-B9-148: Phn bn no sau y lm tng chua ca t?
A. KCl. B. NH4NO3. C. NaNO3. D. K2CO3.
Cu 4.Cu 42: Phn bn nitrophotka (NPK) l hn hp ca
A. (NH4)3PO4 v KNO3. B. (NH4)2HPO4 v KNO3.
C. NH4H2PO4 v KNO3. D. (NH4)2HPO4 v NaNO3
Cu 5.Cu 7-A11-318: Hp cht no ca canxi c dng c tng, b bt khi gy xng?
A. Thch cao sng ( CaSO4.2H2O) B. Thch cao nung ( CaSO4.H2O)
C. Vi sng ( CaO) D. vi ( CaCO3)
Cu 6.Cu 19-A11-318: Phn chua c dng trong ngnh cng nghip thuc da, cng nghip giy, cht
cm mu trong ngnh nhum vi, cht lm trong nc. Cng thc ho hc ca phn chua l
A. Li2SO4.Al2(SO4)3.24H2O. B. Na2SO4.Al2(SO4)3.24H2O.
C. K2SO4.Al2(SO4)3.24H2O. D. (NH4)2SO4.Al2(SO4)3.24H2O.
Cu 7.Cu 24-A11-318: Qung st manhetit c thnh phn chnh l
A. Fe2O3. B. FeCO3. C. Fe3O4. D. FeS2.
Cu 8.Cu 5-A12-296: Mt loi phn kali c thnh phn chnh l KCl (cn li l cc tp cht khng cha
kali) c sn xut t qung xinvinit c dinh dng 55%. Phn trm khi lng ca KCl trong loi
phn kali l
A. 95,51%. B. 65,75%. C. 87,18%. D. 88,52%.
Cu 9.Cu 6-A12-296: Qung no sau y giu st nht?
A. Xierit. B. Manhetit. C. Hematit . D. Pirit st.
Cu 10.Cu 12-CD12-169: Thnh phn chnh ca phn bn phc hp amophot l
A. Ca3(PO4)2 v (NH4)2HPO4. B. NH4NO3 v Ca(H2PO4)2.
GV: Trn c Tun ( 01695 178 188) Trang: 38.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
C. NH4H2PO4 v (NH4)2HPO4. D. NH4H2PO4 v Ca(H2PO4)2.
VN 19: V TR TRONG BNG TUN HON V CU HNH
L THUYT
I. XC NH V TR CA NGUYN T TRONG BNG H THNG TUN HAN
Xc nh chu k ta da vo cu hnh electron.
Nguyn t c n lp electron nguyn t chu k th n
Xc nh v tr phn nhm ta da vo dy nng lng.
Dy nng lng c electron cui cng sp xp trn orbitan s hoc orbitan p nguyn t phn
nhm chnh.
Dy nng lng c dng
ns1 phn nhm chnh nhm I hay phn nhm IA.
ns2 phn nhm chnh nhm II hay phn nhm IIA.
ns2 np1 phn nhm chnh nhm III hay phn nhm IIIA.
ns2 np2 phn nhm chnh nhm IVhay phn nhm IVA.
ns2 np3 phn nhm chnh nhm V hay phn nhm VA.
ns2 np4 phn nhm chnh nhm VI hay phn nhm VIA.
ns2 np5 phn nhm chnh nhm VII hay phn nhm VIIA.
ns2 np6 phn nhm chnh nhm VIII hay phn nhm VIIIA.
Dy nng lng c electron cui cng sp xp trn orbitan d nguyn t phn nhm ph.
Dy nng lng c dng:
ns1 (n-1)d10 phn nhm ph nhm I hay phn nhm IB.
ns2 (n-1)d10 phn nhm ph nhm II hay phn nhm IIB.
ns2 (n-1)d1 phn nhm ph nhm III hay phn nhm IIIB.
ns2 (n-1)d2 phn nhm ph nhm IV hay phn nhm IVB.
ns2 (n-1)d3 phn nhm ph nhm V hay phn nhm VB.
ns1 (n-1)d5 phn nhm ph nhm VI hay phn nhm VIB.
ns2 (n-1)d5 phn nhm ph nhm VII hay phn nhm VIIB.
ns2 (n-1)d6
ns2 (n-1)d7 phn nhm ph nhm VIII hay phn nhm VIIIB.
ns2 (n-1)d8
II. XC NH TNH KIM LOI, PHI KIM V KH HIM
(Ta da vo cu hnh electron)
Cu hnh electron lp ngai cng c:
1,2,3 electrton nguyn t l kim lai.
5,6, 7 electrton nguyn t l phi kim.
8 electrton nguyn t l kh him.
4 electron v Nu nguyn t chu k nh (CK1,2,3) l phi kim.
4 electron v Nu nguyn t chu k ln (CK4,5,6,7) l kim lai.
CU HI
Cu 1.Cu 26-A7-748: Anion X- v cation Y2+ u c cu hnh electron lp ngoi cng l 3s23p6. V
tr ca cc nguyn t trong bng tun hon cc nguyn t ha hc l:
A. X c s th t 17, chu k 4, nhm VIIA (phn nhm chnh nhm VII); Y c s th t 20, chu k 4,
nhm IIA (phn nhm chnh nhm II).
B. X c s th t 18, chu k 3, nhm VIA (phn nhm chnh nhm VI); Y c s th t 20, chu k
4, nhm IIA (phn nhm chnh nhm II).
C. X c s th t 18, chu k 3, nhm VIIA (phn nhm chnh nhm VII); Y c s th t 20, chu k 3,
nhm IIA (phn nhm chnh nhm II).
D. X c s th t 17, chu k 3, nhm VIIA (phn nhm chnh nhm VII); Y c s th t 20, chu k 4,
nhm IIA (phn nhm chnh nhm II).
GV: Trn c Tun ( 01695 178 188) Trang: 39.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
Cu 2. Cu 20-A9-438: Cu hnh electron ca ion X2+ l 1s22s22p63s23p63d6. Trong bng tun hon cc
nguyn t ho hc, nguyn t X thuc
A. chu k 4, nhm IIA. B. chu k 4, nhm VIIIB.
C. chu k 4, nhm VIIIA. D. chu k 3, nhm VIB.
Cu 3.Cu 27-CD11-259: Trong bng tun hon cc nguyn t ho hc, nguyn t X nhm IIA,
nguyn t Y nhm VA. Cng thc ca hp cht to thnh t 2 nguyn t trn c dng l
A. X2Y3. B. X2Y5. C. X3Y2. D. X5Y2.
Cu 4.Cu 18-A12-296: Phn trm khi lng ca nguyn t R trong hp cht kh vi hiro (R c s oxi
ha thp nht) v trong oxit cao nht tng ng l a% v b%, vi a : b = 11 : 4. Pht biu no sau y l
ng?
A. Phn t oxit cao nht ca R khng c cc.
B. Oxit cao nht ca R iu kin thng l cht rn.
C. Trong bng tun hon cc nguyn t ha hc, R thuc chu k 3.
D. Nguyn t R ( trng thi c bn) c 6 electron s.
Cu 5. Cu 19-A12-296: X v Y l hai nguyn t thuc cng mt chu k, hai nhm A lin tip. S proton
ca nguyn t Y nhiu hn s proton ca nguyn t X. Tng s ht proton trong nguyn t X v Y l
33. Nhn xt no sau y v X, Y l ng?
A. n cht X l cht kh iu kin thng.
B. m in ca X ln hn m in ca Y.
C. Lp ngoi cng ca nguyn t Y ( trng thi c bn) c 5 electron.
D. Phn lp ngoi cng ca nguyn t X ( trng thi c bn) c 4 electron.
Cu 6.Cu 20-B12-359: Nguyn t Y l phi kim thuc chu k 3, c cng thc oxit cao nht l YO3.
Nguyn t Y to vi kim loi M hp cht c cng thc MY, trong M chim 63,64% v khi lng.
Kim loi M l
A. Zn. B. Cu. C. Mg. D. Fe.
Cu 7.Cu 14-CD12-169: Nguyn t ca nguyn t X c tng s ht proton, ntron, electron l 52. Trong
ht nhn nguyn t X c s ht khng mang in nhiu hn s ht mang in l 1. V tr (chu k, nhm)
ca X trong bng tun hon cc nguyn t ha hc l
A. chu k 3, nhm VA. B. chu k 3, nhm VIIA.
C. chu k 2, nhm VIIA. D. chu k 2, nhm VA.
VN 20: S BIN I CC I LNG CA BNG TUN HON
L THUYT
1. Tnh kim loi, tnh phi kim ca n cht
- Tnh kim loi, tnh phi kim ca cc nguyn t nhm A bin i tun hon theo chiu tng ca in tch
ht nhn.
- Trong mi chu k, theo chiu tng ca in tch ht nhn, tnh kim loi ca cc nguyn t gim dn,
ng thi tnh phi kim tng dn.
- Trong mt nhm A, theo chiu tng ca in tch ht nhn tnh kim loi ca cc nguyn t tng dn,
ng thi tnh phi kim gim dn.
2. Bn knh
- Trong mt chu k, theo chiu tng ca in tch ht nhn, bn knh nguyn t gim dn
-Trong mt nhm A , theo chiu tng ca in tch ht nhn, bn knh nguyn t tng dn
- Bn knh ca M >> Mn+ v X < Xa-
- Bn knh ph thuc ln lt vo 3 yu t: s lp e >> in tch ht nhn >> s electron lp ngoi.
3. m in
- c trng cho kh nng ht electron ca nguyn t khi to thnh lin kt ha hc.
- Trong mt chu k theo chiu tng dn ca in tch ht nhn m in tng dn.
- Trong mt phn nhm theo chiu tng dn ca in tch ht nhn m in gim dn.
4. Tnh axit, tnh baz ca hp cht
GV: Trn c Tun ( 01695 178 188) Trang: 40.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
- Tnh axit - baz ca cc oxit v hiroxit tng ng ca cc nguyn t bin i tun hon theo chiu
tng ca in tch ht nhn nguyn t.
-Trong mt chu k, theo chiu tng ca in tch ht nhn, tnh baz ca oxit v hiroxit tng ng gim
dn, ng thi tnh axit ca chng tng dn.
-Trong mt nhm A, theo chiu tng ca in tch ht nhn, tnh baz ca cc oxit v hiroxit tng ng
tng dn, ng thi tnh axit ca chng gim dn.
CU HI
Cu 1.Cu 35-B07-285: Trong mt nhm A (phn nhm chnh), tr nhm VIIIA (phn nhm chnh
nhm VIII), theo chiu tng ca in tch ht nhn nguyn t th
A. tnh phi kim gim dn, bn knh nguyn t tng dn.
B. tnh kim loi tng dn, m in tng dn.
C. m in gim dn, tnh phi kim tng dn.
D. tnh kim loi tng dn, bn knh nguyn t gim dn.
Cu 2.Cu 21-CD7-439: Cho cc nguyn t M (Z = 11), X (Z = 17), Y (Z = 9) v R (Z = 19). m
in ca cc nguyn t tng dn theo th t
A. M < X < R < Y. B. M < X < Y < R. C. Y < M < X < R. D. R < M < X < Y.
Cu 3.Cu 42-A8-329: Bn knh nguyn t ca cc nguyn t: 3 Li, 8O, 9F, 11 Na c xp theo th t
tng dn t tri sang phi l
A. F, Na, O, Li. B. F, Li, O, Na. C. F, O, Li, Na. D. Li, Na, O, F.
Cu 4.Cu 21-B8-371: Dy cc nguyn t sp xp theo chiu tng dn tnh phi kim t tri sang phi l:
A. N, P, O, F. B. P, N, F, O. C. N, P, F, O. D. P, N, O, F.
Cu 5.Cu 3-B9-148: Cho cc nguyn t: K (Z = 19), N (Z = 7), Si (Z = 14), Mg (Z = 12). Dy gm cc
nguyn t c sp xp theo chiu gim dn bn knh nguyn t t tri sang phi l:
A. N, Si, Mg, K. B. K, Mg, Si, N. C. K, Mg, N, Si. D. Mg, K, Si, N.
Cu 6. Cu 14 -A10-684 : Cc nguyn t t Li n F, theo chiu tng ca in tch ht nhn th
A. bn knh nguyn t v m in u tng. B. bn knh nguyn t tng, m in gim.
C. bn knh nguyn t gim, m in tng. D. bn knh nguyn t v m in u gim.
Cu 7.Cu 1-CD10-824: Cc kim loi X, Y, Z c cu hnh electron nguyn t ln lt l: 1s22s22p 63s1;
1s22 s22p63 s2 ; 1s22s22p63s23p1. Dy gm cc kim loi xp theo chiu tng dn tnh kh t tri sang phi l:
A. Y, Z, X. B. Z, X, Y. C. X, Y, Z. D. Z, Y, X.
VN 21: LIN KT HA HC
L THUYT
I - KHI NIM V LIN KT HA HC
1. Khi nim v lin kt
- Lin kt ha hc l s kt hp gia cc nguyn t to thnh phn t hay tinh th bn vng hn.
- Khi c s chuyn cc nguyn t ring r thnh phn t hay tinh th tc l c lin kt ha hc th nguyn
t c xu hng t ti cu hnh electron bn vng ca kh him.
2.Quy tc bt t (8 electron)
- Ta bit, cc kh him hot ng ha hc rt km, chng tn ti trong t nhin di dng nguyn t t
do ring r, nguyn t ca chng khng lin kt vi nhau m to thnh phn t.
- Trong cc nguyn t kh him, nguyn t heli ch c 2 electron nn c 2 electron lp th nht cng
l lp ngoi cng, cn cc nguyn t kh him khc c 8 electron lp ngoi cng. Nh vy, cu hnh
vi 8 electron lp ngoi cng (hoc 2 electron i vi heli) l cu hnh electron vng bn.
Theo quy tc bt t (8 electron) th nguyn t ca cc nguyn t c khuynh hng lin kt vi cc
nguyn t khc t c cu hnh electron vng bn ca cc kh him vi 8 electron (hoc 2 i vi
GV: Trn c Tun ( 01695 178 188) Trang: 41.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
heli) lp ngoi cng.
- Vi quy tc bt t, ngi ta c th gii thch mt cch nh tnh s hnh thnh cc loi lin kt trong
phn t, c bit l cch vit cng thc cu to trong cc hp cht thng thng.
- V phn t l mt h phc tp nn trong nhiu trng hp quy tc bt t t ra khng y .
II. LIN KT ION V LIN KT CNG HA TR
1. So snh lin kt ion v lin kt cng ha tr
Loi lin kt Li n kt i on Li n kt cng h a tr
Nguyn nhn
hnh t hnh Cc nguyn t lin kt vi nhau c cu hnh electron bn vng ca kh him.
lin kt
L s dng chung c c electron (s
Lc ht tnh in gia cc ion mang dng chung theo kiu xen ph c c
Bn cht ca in tch tri du( cho v
lin kt AO)
nhn electron)
Xy r a gia 2 nguy n t ging nhau
Xy ra gia c c nguy n t khc hn v bn cht ha hc (th ng xy ra
iu kin ca nhau v bn cht ha hc (thng xy ra vi cc nhm IV, V, VI, VII)
lin kt gia kim lai in hnh v phi kim
in hnh).
c t nh Rt bn Bn
2. Hiu m in v lin kt ha hc
Hiu m in 0< < 0,4 0,4 < 1,7 1,7
Loi lin kt Cng ha tr kh ng cc Cng ha tr phn cc Ion
CU HI
Cu 1.Cu 41-A8-329: Hp cht trong phn t c lin kt ion l
A. NH4Cl. B. HCl. C. H2O. D. NH3.
Cu 2.Cu 26-CD8-216: Nguyn t ca nguyn t X c cu hnh electron 1s22s22p63s23p64s1, nguyn t
ca nguyn t Y c cu hnh electron 1s22s22p5. Lin kt ho hc gia nguyn t X v nguyn t Y
thuc loi lin kt
A. kim loi. B. cng ho tr. C. ion. D. cho nhn.
Cu 3.Cu 19-CD9-956: Dy gm cc cht trong phn t ch c lin kt cng ho tr phn cc l:
A. HCl, O3, H2S. B. O2, H2O, NH3. C. HF, Cl2, H2O. D. H2O, HF, H2S.
Cu 4.Cu 15-B10-937: Cc cht m phn t khng phn cc l:
A. NH3, Br2, C2H4. B. Cl2, CO2, C2H2. C. HBr, CO2, CH4. D. HCl, C2 H2, Br2.
Cu 5.Cu 33-CD10-824: Lin kt ho hc gia cc nguyn t trong phn t H2O l lin kt
A. cng ho tr khng phn cc. B. hiro.
C. cng ho tr phn cc. D. ion.
Cu 6.Cu 40-CD11-259: Mc phn cc ca lin kt ha hc trong cc phn t c sp xp theo
th t gim dn t tri sang phi l:
A. HI, HCl, HBr. B. HCl, HBr, HI. C. HI, HBr, HCl. D. HBr, HI, HCl
Cu 7.Cu 20-CD12-169: Cho dy cc cht: N2, H2, NH3, NaCl, HCl, H2O. S cht trong dy m phn t
ch cha lin kt cng ha tr khng cc l
A. 3. B. 4. C. 5. D. 2.
Cu 8.Cu 29-A13-193: Lin kt ha hc gia cc nguyn t trong phn t HCl thuc loi lin kt
A. cng ha tr c cc. B. hiro. C. cng ha tr khng cc. D. ion.
Cu 9.Cu 13-B13-279: Cho gi tr m in ca cc nguyn t: F = 3,98; O= 3,44; C= 2,55; H= 2,20;
Na = 0,93). Hp cht no sau y l hp cht ion?
GV: Trn c Tun ( 01695 178 188) Trang: 42.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
A. NaF. B. CO2. C. CH4. D. H2O.
Cu 10.Cu 39-CD13-415: Lin kt ha hc trong phn t Br2 thuc loi lin kt
A. hiro. B. cng ha tr c cc. C. ion. D. cng ha tr khng cc.
VN 22: CU HNH ELECTRON NGUYN T
L THUYT
CCH VIT CU HNH ELECTRON NGUYN T
1. Th t mc nng lng.
Cc electron trong nguyn t chim cc mc nng lng t thp n cao.
Thc nghim v l thuyt xc nh c th t ca cc lp v phn lp theo theo th t sau:
1s 2s 2p 3s 3p 4s 3d 4p 5s
Lu : Cch nh mc nng lng
* Da vo quy tc ziczac n gin ca Kletkopski
* Da vo s sau:
* Hoc s dng mt s cu ni vui:
* Hoc: sn, sn, phi sn, phi sn, i phi sn, i phi sn, fi i phi sn, fi i phi sn
s s ps ps dps dps fdps fdpf
Sau in s th t lp ln lt cho s (17), p(27), d(36), f(46)
Ta c 1s2s 2p3s 3p4s 3d 4p 5s 4d 5p 6s4f 5d 6p 7s 5f 6d 7p 6f
2. Cch vit cu hnh electron nguyn t.
Cu hnh electron ca nguyn t biu din s phn b electron trn cc phn lp thuc cc lp khc nhau.
Ngi ta quy c vit cu hnh electron nguyn t nh sau:
- S th t lp electron c ghi bng ch s (1, 2, 3).
- Phn lp c ghi bng ch ci thng s, p, d, f.
- S electron trong mt phn lp c ghi bng s pha bn phi ca phn lp (s2, p6), cc phn lp
khng c electron khng ghi.
Cch vit cu hnh electron nguyn t gm cc bc sau:
Bc 1: Xc nh s electron nguyn t.
Bc 2: Cc electron c phn b ln lt vo cc phn lp theo chiu tng ca nng lng trong
nguyn t (1s 2s 2p 3s 3p 4s 3d 4p 5s) v tun theo quy tc sau:
- phn lp s cha ti a 2 electron;
- phn lp p cha ti a 6 electron;
- phn lp d cha ti a 10 electron;
- phn lp f cha ti a 14 electron.
GV: Trn c Tun ( 01695 178 188) Trang: 43.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
Bc 3. Vit cu hnh electron biu din s phn b electron trn cc phn lp thuc cc lp khc nhau
(1s 2s 2p 3s 3p 4s 3d 4p 5s).
VD1: Vit cu hnh electron ca nguyn t O (Z=8):
1. Xc nh s electron: 8.
2. Cc electron phn b vo cc phn lp theo chiu tng dn ca nng lng trong nguyn t:
1s22s22p4.
3. Cu hnh electron l: 1s22s22p4.
VD2: Vit cu hnh electron ca nguyn t Fe (Z=26).
1. Xc nh s electron: 26.
2. Cc electron phn b vo cc phn lp theo chiu tng dn ca nng lng trong nguyn t:
1s22s22p63s23p64s23d6.
3. Vit cu hnh electron biu din s phn b electron trn cc phn lp thuc cc lp khc nhau:
1s22s22p63s23p63d64s2.
Hay vit gn l [Ar] 3d64s2.
VD3: cu hnh ca mt s nguyn t khc:
1H : 1s1
2
2He : 1s
3Li : 1s 2s1
2
2 2
4Be : 1s 2s
5B : 1s2 2s2 2p1
2 2 6 2 6 11
20K : 1s 2s 2p 3s 3p 4s .
- Nguyn t s l nhng nguyn t m nguyn t c electron cui cng c in vo phn lp s (V d
nguyn t H, Na, K...).
- Nguyn t p l nhng nguyn t m nguyn t c electron cui cng c in vo phn lp p (V d
nguyn t B, O, Cl...).
- Nguyn t d l nhng nguyn t m nguyn t c electron cui cng c in vo phn lp d (V d
nguyn t Fe, Cu, Cr...).
- Nguyn t f l nhng nguyn t m nguyn t c electron cui cng c in vo phn lp f (V d
nguyn t Ce, U, Pr...).
3. c im ca lp electron ngoi cng
- Lp ngoi cng: nsa => c enc = a ( a 2)
nsanpb => c enc = a + b ( a + b 8)
=> s electron lp ngoi cng ca nguyn t lun 8
- Cc nguyn t c 8 electron ngoi cng l kh him, chng khng tham gia vo cc phn ng ha hc.
- Cc nguyn t c 1, 2, 3 electron lp ngoi cng thng l nguyn t ca cc nguyn t kim loi.
- Cc nguyn t c 5, 6, 7 electron lp ngoi cng thng l nguyn t ca cc nguyn t phi kim.
- Cc nguyn t c 4 electron lp ngoi cng thng l nguyn t ca nguyn t kim loi hoc phi kim.
Nh vy, khi bit cu hnh electron ca nguyn t c th d on c loi nguyn t.
CU HI
Cu 1. Cu 34-A7-748: Dy gm cc ion X+, Y- v nguyn t Z u c cu hnh electron 1s22s22p6 l:
A. Na+, F-, Ne. B. Na+, Cl-, Ar. C. Li+, F-, Ne. D. K+, Cl-, Ar.
Cu 2.Cu 26-CD8-216: Nguyn t ca nguyn t X c cu hnh electron 1s22s22p63s23p64s1, nguyn t
ca nguyn t Y c cu hnh electron 1s22s22p5. Lin kt ho hc gia nguyn t X v nguyn t Y
thuc loi lin kt
A. kim loi. B. cng ho tr. C. ion. D. cho nhn.
Cu 3.Cu 26-CD9-956: Nguyn t ca nguyn t X c electron mc nng lng cao nht l 3p.
Nguyn t ca nguyn t Y cng c electron mc nng lng 3p v c mt electron lp ngoi cng.
Nguyn t X v Y c s electron hn km nhau l 2. Nguyn t X, Y ln lt l
A. phi kim v kim loi. B. kim loi v kh him.
C. kh him v kim loi. D. kim loi v kim loi.
GV: Trn c Tun ( 01695 178 188) Trang: 44.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
Cu 4.Cu 22-B10-937: Mt ion M3+ c tng s ht proton, ntron, electron l 79, trong s ht mang
in nhiu hn s ht khng mang in l 19. Cu hnh electron ca nguyn t M l
A. [Ar]3d54s1. B. [Ar]3d 64s2. C. [Ar]3d34s2. D. [Ar]3d64s1.
Cu 5.Cu 42-A11-318 : Cu hnh electron ca ion Cu2+ v Cr3+ ln lt l
A. [Ar]3d9 v [Ar]3d3. B. [Ar]3d 9 v [Ar]3d14s2.
7 2 1 2
C. [Ar]3d 4s v [Ar]3d 4s . D. [Ar]3d74s2 v [Ar]3d3.
Cu 6. Cu 11-A12-296: Nguyn t R to c cation R+. Cu hnh electron phn lp ngoi cng
ca R+ ( trng thi c bn) l 2p6. Tng s ht mang in trong nguyn t R l
A. 10. B. 11. C. 22. D. 23.
Cu 7.Cu 14-A13-193: trng thi c bn, cu hnh electron ca nguyn t Na (Z = 11) l
A. 1s22s22p 53s2. B. 1s22s22p63s1. C. 1s22s22p 63s2. D. 1s22s22p 43s1
Cu 8.Cu 32-B13-279: S proton v s ntron c trong mt nguyn t nhm (Al 2713 ) ln lt l
A. 13 v 14. B. 13 v 15. C. 12 v 14. D. 13 v 13.
Cu 9.Cu 7-CD13-415: trng thi c bn, nguyn t ca nguyn t X c 4 electron lp L (lp th
hai). S proton c trong nguyn t X l
A. 8. B. 5. C. 7. D. 6.
VN 23: TC PHN NG CN BNG HA HC
L THUYT
1. Tc phn ng
a. Khi nim v biu thc tc phn ng ha hc
- Tc phn ng l i lng c trng cho mc din ra nhanh hay chm ca phn ng ha hc,
c o bng bin thin nng ca mt trong cc cht tham gia hoc sn phm trong mt n v thi
gian.
- Biu thc tc trung bnh phn ng: Xt phn ng: aA + bB cC + dD (*)
Ti thi im t1: nng cht A l C1 (mol/lt)
Ti thi im t2: nng cht A l C2 (mol/lt)
C C2
Tc trung bnh ca phn ng c tnh theo cht A l: Vtb 1
t2 t1
- Th nguyn: mol/lt.s hoc mol/lt.pht
b. Cc yu t nh hng
- nh hng ca nng
Tc ca phn ng (*) c xc nh bi biu thc: v = k.[A]a.[B]b
Do : khi tng nng cht tham gia th tc phn ng tng ln.
- nh hng ca p sut (ch vi phn ng c cht kh tham gia): Khi tng p sut nng cht kh
tng nn tc phn ng tng
- nh hng ca nhit : Khi tng nhit th tc phn ng tng
Bng thc nghim ngi ta xc nh c rng: khi tng nhit thm 10 oC th tc phn ng tng
thm 2 4 ln. Gi tr = 2 4 c gi l h s nhit ca phn ng. Tr s ca c xc nh hon ton
v o
bng thc nghim. ( t C 10) . Nh vy nu mt phn ng xy ra nhit T1 vi tc v1, nhit
vto
T T
v2 2 1
T2 vi tc v2 (gi s: T 2 > T1) th: 10
v1
- nh hng ca din tch tip xc: din tch tip xc cng ln th tc phn ng cng tng
- nh hng ca xc tc: Cht xc tc lm tng tc phn ng, bn thn khng b bin i sau phn
ng
2. Cn bng ha hc
a. Khi nim cn bng ha hc, hng s cn bng ha hc
GV: Trn c Tun ( 01695 178 188) Trang: 45.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
- Cn bng ha hc l trng thi ca phn ng thun nghch khi tc phn ng thun bng tc phn
ng nghch. Cn bng ha hc l mt cn bng ng
- Xt phn ng: aA + bB cC + dD (**)
Mi cn bng ha hc c c trng bi mt hng s cn bng KC (hng s cn bng ha hc) c xc
[C]c .[D]d
nh bi biu thc: K C ( NNG CAO)
[A]a .[B]b
Ch : Hng s cn bng KC khng ph thuc vo nng ban u ca cc cht phn ng
Vi mi phn ng nht nh th KC ch ph thuc vo nhit
Trong cn bng c cht rn th nng cht rn khng c a vo biu thc ca KC
b. Cc yu t nh hng n cn bng ha hc
- nh hng ca nng : Khi tng hoc gim nng ca mt cht trong cn bng th cn bng chuyn
dch v pha lm gim hoc tng nng ca cht .
Ch : Trong h cn bng c cht rn ( dng nguyn cht) th vic tng hay gim khi lng cht
rn khng lm chuyn dch cn bng.
- nh hng ca p sut (cn bng c cht kh): Khi tng p sut chung ca h cn bng th cn bng
chuyn dch v pha to ra s mol kh t hn v ngc li.
Ch : Trong cn bng m tng s mol kh 2 v bng nhau th p sut khng nh hng n cn
bng
- nh hng ca nhit : Khi tng nhit th cn bng chuyn dch v pha phn ng thu nhit (H>0)
v ngc li khi gim nhit th cn bng chuyn dch v pha phn ng ta nhit (H<0)
CU HI
Cu 1. Cu 5-CD7-439: Cho phng trnh ho hc ca phn ng tng hp amoniac
t o , xt
N2 (k)+ 3H2 (k) 2NH3 (k)
Khi tng nng ca hiro ln 2 ln, tc phn ng thun
A. tng ln 8 ln. B. tng ln 6 ln. C. tng ln 2 ln. D. gim i 2 ln
Cu 2.Cu 38-A8-329: Cho cn bng ho hc: 2SO2 (k) + O2 (k) 2SO3 (k); phn ng thun l
phn ng ta nhit. Pht biu ng l:
A. Cn bng chuyn dch theo chiu nghch khi gim nng SO3.
B. Cn bng chuyn dch theo chiu thun khi gim p sut h phn ng.
C. Cn bng chuyn dch theo chiu nghch khi gim nng O2.
D. Cn bng chuyn dch theo chiu thun khi tng nhit .
t o , xt
Cu 3.Cu 32-B8-371: Cho cn bng ho hc: N2 (k) + 3H2 (k) 2NH3 (k); phn ng thun l
phn ng to nhit. Cn bng ho hc khng b chuyn dch khi
A. thay i nng N2. B. thm cht xc tc Fe.
C. thay i p sut ca h. D. thay i nhit .
Cu 4.Cu 21-CD8-216: Cho cc cn bng ho hc:
t o , xt
(1) N2 (k) + 3H2 (k) 2NH3 (k) (2)H2 (k) + I2 (k)
t o
2HI (k)
o
t , xt
(3) 2SO2 (k) + O2 (k)
2SO3 (k) (4) 2NO2 (k) N2O4 (k)
Khi thay i p sut nhng cn bng ha hc b chuyn dch l:
A. 1, 2, 3. B. 2, 3, 4. C. 1, 3, 4. D. 1, 2, 4.
Cu 5.Cu 56-CD8-216: Hng s cn bng ca phn ng xc nh ch ph thuc vo
A. nhit . B. p sut. C. cht xc tc. D. nng .
Cu 6.Cu 42-A9-438: Cho cn bng sau trong bnh kn: 2NO2 (k) N2O4 (k)
Nu khng mu
nu nht dn. Phn ng thun c
A. H < 0, phn ng ta nhit. B. H < 0, phn ng thu nhit.
C. H > 0, phn ng ta nhit. D. H > 0, phn ng thu nhit.
GV: Trn c Tun ( 01695 178 188) Trang: 46.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
Cu 7.Cu 8-B9-148: Cho cht xc tc MnO2 vo 100 ml dung dch H2O2, sau 60 giy thu c 33,6 ml
kh O2 ( ktc). Tc trung bnh ca phn ng (tnh theo H2O2) trong 60 giy trn l
A. 5,0.10 -4 mol/(l.s) B. 5,0.10 -5 mol/(l.s) C. 1,0.10 -3mol/(l.s) D. 2,5.10-4mol/(l.s)
Cu 8.Cu 18-CD9-956: Cho cc cn bng sau:
xt , t o
(1) 2SO3 (k) + O2 (k) 2SO3 (k)
o
xt ,t
(2) N2 (k) + 3H2 (k) 2NH3 (k)
o
xt , t
(3) CO2 (k) + H2 (k) CO (k) + H2O (k)
o
xt , t
(4) 2HI (k) H2 (k) + I2 (k)
Khi thay i p sut, nhm gm cc cn bng ho hc u khng b chuyn dch l
A. 1 v 2. B. 1 v 3. C. 3 v 4. D. 2 v 4.
Cu 9.Cu 45-CD9-956: Cho cn bng (trong bnh kn) sau:
xt ,t o
CO (k) + H2O (k) CO2 (k) + H2 (k)H < 0
Trong cc yu t: 1 tng nhit ; 2 thm mt lng hi nc; 3 thm mt lng H2; 4 tng
p sut chung ca h; 5 dng cht xc tc.
Dy gm cc yu t u lm thay i cn bng ca h l:
A. 1, 2, 4. B. 1, 4, 5. C. 2, 3, 4. D. 1, 2, 3.
Cu 10.Cu 53-CD9-956: Cho cc cn bng sau:
(1) H2 (k) + I2 (k) 2HI (k)
(2) H2 (k) + I2 (k) HI (k)
(3) HI (k) H2 (k) + I2 (k)
2HI (k)
(4) H2 (k) + I2 (r)
nhit xc nh, nu KC ca cn bng 1 bng 64 th KC bng 0,125 l ca cn bng
A. 3. B. 5. C. 4. D. 2.
Cu 11.Cu 23-A10-684: Cho cn bng: 2SO2 (k) + O2 (k) 2SO3 (k). Khi tng nhit th t khi
ca hn hp kh so vi H2 gim i. Pht biu ng khi ni v cn bng ny l:
A. Phn ng nghch to nhit, cn bng dch chuyn theo chiu thun khi tng nhit .
B. Phn ng thun to nhit, cn bng dch chuyn theo chiu nghch khi tng nhit .
C. Phn ng thun thu nhit, cn bng dch chuyn theo chiu nghch khi tng nhit .
D. Phn ng nghch thu nhit, cn bng dch chuyn theo chiu thun khi tng nhit .
Cu 12.Cu 58-A -684: Xt cn bng: N O (k) 2NO (k) 25oC. Khi chuyn dch sang mt trng
10 2 4 2
thi cn bng mi nu nng ca N2O4 tng ln 9 ln th nng ca NO2
A. tng 9 ln. B. tng 3 ln. C. gim 3 ln. D. tng 4,5 ln.
Cu 13.Cu 29-B10-937: Cho cc cn bng sau:
(I) 2HI (k) H2 (k) + I2 (k);
(II) CaCO3 (r) CaO (r) + CO2 (k);
(III) FeO (r) + CO (k) Fe (r) + CO2 (k);
(IV) 2SO2 (k) + O2 (k) 2SO3 (k).
Khi gim p sut ca h, s cn bng b chuyn dch theo chiu nghch l
A. 3. B. 4. C. 1. D. 2.
Cu 14.Cu 31-CD10-824: Cho cn bng ho hc: PCl5 (k) PCl3 (k) + Cl2 (k); H > 0.
Cn bng chuyn dch theo chiu thun khi
A. tng p sut ca h phn ng. B. tng nhit ca h phn ng.
C. thm PCl3 vo h phn ng. D. thm Cl2 vo h phn ng.
GV: Trn c Tun ( 01695 178 188) Trang: 47.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
Cu 15. Cu 42-CD10-824: Cho phn ng: Br2 + HCOOH 2HBr + CO2.
Nng ban u ca Br2 l a mol/lt, sau 50 giy nng Br2 cn li l 0,01 mol/lt. Tc trung
bnh ca phn ng trn tnh theo Br l 4.10-5 mol/(l.s). Gi tr ca a l
2
A. 0,018. B. 0,014. C. 0,012. D. 0,016.
Cu 16.Cu 6-CD11-259: Cho cn bng ha hc: N2 (k) + 3H2 (k) 2NH3 (k) H < 0
Cn bng trn chuyn dch theo chiu thun khi
A. tng nhit ca h phn ng. B. gim p sut ca h phn ng.
C. tng p sut ca h phn ng. D. thm cht xc tc vo h phn ng.
Cu 17.Cu 54 -CD11-259: Cho phn ng: H2 (k) + I2 (k) 2HI (k)
nhit 430C, hng s cn bng KC ca phn ng trn bng 53,96. un nng mt bnh kn
dung tch khng i 10 lt cha 4,0 gam H2 v 406,4 gam I2. Khi h phn ng t trng thi cn bng
430C, nng ca HI l
A. 0,275M. B. 0,225M. C. 0,151M. D. 0,320M.
Cu 18.Cu 27-A11-318: Cho cn bng ho hc: H2 (k) + I2 (k) 2HI (k); UH > 0.
Cn bng khng b chuyn dch khi
A. tng nhit ca h. B. gim nng HI.
C. gim p sut chung ca h. D. tng nng H2.
Cu 19.Cu 39-B11-846: Cho cn bng ho hc sau: 2SO2 (k) + O2 (k) 2SO3 (k); H < 0.
Cho cc bin php: 1 tng nhit , 2 tng p sut chung ca h phn ng, 3 h nhit , 4 dng thm cht
xc tc V2O5, 5 gim nng SO3, 6 gim p sut chung ca h phn ng. Nhng bin php no lm cn
bng trn chuyn dch theo chiu thun?
A. 2, 3, 5. B. 1, 2, 4. C. 1, 2, 4, 5. D. 2, 3, 4, 6.
Cu 20.Cu 60-B11-846: Cho 5,6 gam CO v 5,4 gam H2O vo mt bnh kn dung tch khng i 10
o
lt. Nung nng bnh mt thi gian 830 C h t n trng thi cn bng: CO (k) + H2O (k) CO2
(k) + H2 (k) (hng s cn bng KC = 1). Nng cn bng ca CO, H2O ln lt l
A. 0,008M v 0,018M. B. 0,018M v 0,008M.
C. 0,012M v 0,024M. D. 0,08M v 0,18M.
Cu 21.Cu 26-A -296: Xt phn ng phn hy N O trong dung mi CCl 45oC:
12 2 5 4
N2 O5 N 2 O4 + O 2
Ban u nng ca N2O5 l 2,33M, sau 184 giy nng ca N2O5 l 2,08M. Tc trung bnh ca
phn ng tnh theo N2O5 l
3 3
A. 2, 72.10 mol/(l.s). B. 1, 36.10 mol/(l.s).
3 4
C. 6,80.10 mol/(l.s). D. 6,80.10 mol/(l.s).
Cu 22.Cu 23-B12-359: Cho phn ng: N2 (k) + 3H2 (k) 2NH3 (k); H = 92 kJ. Hai bin
php u lm cn bng chuyn dch theo chiu thun l
A. gim nhit v gim p sut. B. tng nhit v tng p sut.
C. gim nhit v tng p sut. D. tng nhit v gim p sut.
Cu 23.Cu 23-CD12-169: Cho cn bng ha hc: CaCO3 (rn) CaO (rn) + CO2 (kh)
Bit phn ng thun l phn ng thu nhit. Tc ng no sau y vo h cn bng cn bng
cho chuyn dch theo chiu thun?
A. Tng nng kh CO2. B. Tng p sut.
C. Gim nhit . D. Tng nhit .
Cu 42 : Cho phn ng ha hc: Br2 + HCOOH 2HBr + CO2
Cu 24. -CD12-169
GV: Trn c Tun ( 01695 178 188) Trang: 48.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
Lc u nng ca HCOOH l 0,010 mol/l, sau 40 giy nng ca HCOOH l 0,008 mol/l.
Tc trung bnh ca phn ng trong khong thi gian 40 giy tnh theo HCOOH l
A. 5,0.10 -5 mol/(l.s). B. 2,5.10-5 mol/(l.s). C. 2,5.10-4 mol/(l.s). D. 2,0.10 -4 mol/(l.s).
Cu 25. Cu 32-A13-193: Cho cc cn bng ha hc sau:
2HI (k)
(a) H2 (k) + I2 (k)
(b) 2NO2 (k) N2O4 (k)
(c) 3H2 (k) + N2 (k) 2NH3 (k)
2SO2 (k)
(d) 2SO2 (k) + O2 (k)
nhit khng i, khi thay i p sut chung ca mi h cn bng, cn bng ha hc no
trn khng b chuyn dch?
A. (b). B. (a). C. (c). D. (d).
Cu 26. Cu 19-B13-279: Cho phng trnh ha hc ca phn ng: X + 2Y Z + T. thi im ban
u, nng ca cht X l 0,01 mol/l. Sau 20 giy, nng ca cht X l 0,008 mol/l. Tc trung bnh
ca phn ng tnh theo cht X trong khong thi gian trn l
A. 4,0.104 mol/(l.s). B. 1,0.10 4 mol/(l.s). C. 7,5.104 mol/(l.s). D. 5,0.104 mol/(l.s).
Cu 27.Cu 54-B13-279: Trong mt bnh kn c cn bng ha hc sau: 2NO2 (k) N2O4 (k).
T khi hi ca hn hp kh trong bnh so vi H2 nhit T1 bng 27,6 v nhit T2 bng
34,5. Bit T1 > T2. Pht biu no sau y v cn bng trn l ng?
A. Khi tng nhit , p sut chung ca h cn bng gim.
B. Khi gim nhit , p sut chung ca h cn bng tng.
C. Phn ng thun l phn ng ta nhit.
D. Phn ng nghch l phn ng ta nhit.
Cu 28.Cu 35-CD13-415: Trong bnh kn c h cn bng ha hc sau:
CO2 (k) + H2 (k) CO (k) + H2O (k) H > 0
(a) tng nhit ; (b) thm mt lng hi nc;
(c) gim p sut chung ca h; (d) dng cht xc tc;
(e) thm mt lng CO2.
Trong nhng tc ng trn, cc tc ng lm cn bng chuyn dch theo chiu thun l:
A. (a) v (e). B. (b), (c) v (d). C. (d) v (e). D. (a), (c) v (e).
VN 24: NHN BIT
L THUYT
- MT S THUC TH DNG NHN BIT CC CHT V C THNG DNG
Cht cn NB Thuc th Du hiu Phng trnh phn ng
Li Li cho ngn la
ta
K
K cho ngn la
Na tm
Ca Na cho ngn la
t chy
KIM LOI
Ba vng
Ca cho ngn la
da cam
Ba cho ngn la
vng lc
GV: Trn c Tun ( 01695 178 188) Trang: 49.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
Cht cn NB Thuc th Du hiu Phng trnh phn ng
n
H2O Dung dch + M + nH2O M(OH)n + H2
H2 2
(Vi Ca dd
c)
Be
Zn M +(4-n)OH- + (n-2)H2O
dd kim Tan H2 n
Al MO2n-4 + H2
2
Kloi t Mg Tan H2
Pb n
dd axit (HCl) (Pb c PbCl2 M + nHCl MCln + H2
2
mu trng)
HCl/H2SO4 Tan dung dch 2Cu + O2 + 4HCl
long c sc O2 mu xanh 2CuCl2 + 2H2O
KIM LOI
Cu
t trong O2 Mu mu t
2Cu + O2
0
en 2CuO
0
t
Tan NO2 mu Ag + 2HNO3
Ag HNO3/t0
nu AgNO3 + NO2 + H2O
I2 H tinh bt Mu xanh
S t trong O2 kh SO2 mi t
S + O2
0
hc SO2
0
t
4P + O2 2P2O5
t trong O2 v Dung dch to
P ha tan sn thnh lm qu P2O5 + 3H2O 2H3PO4
phm vo H2O tm
(Dung dch H3PO4 lm qu tm)
PHI KIM
0
t
C + O2 CO2
C t trong O2 CO2 lm c
nc vi trong CO2 + Ca(OH)2
CaCO3 + H2O
5Cl2 + Br2 + 6H2O
Nc Br2 Nht mu
10HCl + 2HBrO3
Cl2
dd KI + h tinh Khng mu Cl2 + 2KI 2KCl + I2
bt mu xanh I2
H tinh bt mu xanh
Tn m Tn m bng
chy
O2
Cu, t0 Cu mu t 0
KH V HI
2Cu + O2 2CuO
mu en
t,lm lnh Hi nc ngng t
2H2 + O2
0
t 2H2O
H2
0
CuO, t0 Ha t
CuO + H2 Cu + H2O
H2O (hi) CuSO4 khan Trng xanh CuSO4 + 5H2O CuSO4.5H2O
GV: Trn c Tun ( 01695 178 188) Trang: 50.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
Cht cn NB Thuc th Du hiu Phng trnh phn ng
0
t
CuO en CuO + CO Cu + CO2
CO + PdCl2 + H2O
dd PdCl2 Pd vng
Pd +2HCl + CO2
CO
t trong O2 t
2CO + O2
0
2CO2
ri dn sn
phm chy qua Dung dch nc CO2 + Ca(OH)2
dd nc vi vi trong vn c
trong CaCO3 + H2O
dd vi Dung dch nc CO2 + Ca(OH)2
CO2 trong vi trong vn c CaCO3 + H2O
SO2 + Br2 + 2H2O
nc Br2 Nht mu
H2SO4 + 2HBr
SO2
dd thuc 5SO2 + 2KMnO4 + 2H2O
Nht mu
tm 2H2SO4 + 2MnSO4 + K2SO4
BaCl2 + H2O + SO3
SO3 Dd BaCl2 BaSO4 trng
BaSO+ 2HCl
mi Trng thi
H2S Pb(NO3)2 +H2S
Dd Pb(NO3)2 PbS en
PbS + 2HNO3
Qu tm m Ha
HCl
NH3 Khi trng NH3 + HCl NH4Cl
Qu tm m Ha xanh
NH3
HCl Khi trng NH3 + HCl NH4Cl
KH V HI
NO Khng kh Ha nu 2NO + O2 2 NO2
Qu tim m Ha
NO2 Mu nu k0 0
11 C
Lm lnh 2NO2 N2O4
mu
N2 Que m chy Tt
Qu tm Ha
2HCl + CaCO3
CaCl2 + CO2 + H2O
DUNG DCH
Mui cacbonat;
Axit: HCl sunfit, sunfua, C kh CO2, SO2, 2HCl + CaSO3
kim loi ng H2S, H2 CaCl2 + SO2+ H2O
trc H
2HCl + FeS FeCl2 + H2S
2HCl + Zn ZnCl2 + H2
GV: Trn c Tun ( 01695 178 188) Trang: 51.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
Cht cn NB Thuc th Du hiu Phng trnh phn ng
0
t
Kh Cl2 mu 4HCl + MnO2
Axit HCl c MnO2 vng lc bay ln MnCl2 +Cl2 +2H2O
Qu tm Ho
H2SO4 + Na2CO3
Mui cacbonat; 2Na2SO4 + CO2 + H2O
Axit H2SO4 sunfit, sunfua,
kim loi ng C kh CO2, SO2, H2SO4 + CaSO3
long H2S, H2,
trc H
To kt ta trng. CaSO4 + SO2 + H2O
Dung dch
mui ca Ba. H2SO4 + FeS FeSO4 + H2S
H2SO4 + Zn ZnSO4 + H2
4HNO3() + Cu
Axit HNO3, Hu ht cc Cu(NO3)2 + 2NO + 2H2O
H2SO4 c kim loi (tr C kh thot ra
nng Au, Pt) Cu +2H2SO4(, nng)
CuSO4 + 2SO2 + 2H2O
Qu tm Ha xanh
Dung dch
Baz ( OH-) Dung dch Ha hng
phenolphtalein
SO42- Ba2+ trng BaSO4 BaCl2 + Na2SO4 BaSO4 + 2NaCl
-
Cl trng AgCl AgNO3 + NaCl AgCl+ NaNO3
Dd AgNO3 3AgNO3 + Na3PO4
PO43- vng Ag3PO4
Ag3PO4+ NaNO3
CaCO3 + 2HCl
CaCl2 + CO2 + H2O
CO32-, SO32- Dd axit CO2, SO2
CaSO3 + 2HCl
CaCl2 + SO2 + H2O
NaHCO3 + HCl
HCO3- Dd axit CO2
NaCl + CO2+ H2O
NaHSO3 + HCl
HSO3- Dd axit SO2
NaCl + SO2 + H2O
Dung dch Kt ta trng
Mg 2+
kim NaOH, Mg(OH)2 khng MgCl2 + 2KOH Mg(OH)2 + 2KCl
DUNG DCH
KOH tan trong kim
d
Cu2+ Kt ta xanh lam CuCl2 + 2NaOH Cu(OH)2 + 2NaCl
: Cu(OH)2
Fe2+ Kt ta trng FeCl2 + 2KOH Fe(OH)2 + 2KCl
xanh : Fe(OH)2
GV: Trn c Tun ( 01695 178 188) Trang: 52.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
Cht cn NB Thuc th Du hiu Phng trnh phn ng
Fe3+ Kt ta nu :
Fe(OH)3 FeCl3 + 3KOH Fe(OH)3 + 3KCl
Al3+ Kt ta keo trng AlCl3 + 3NaOH Al(OH)3 + 3NaCl
Al(OH)3 tan
trong kim d Al(OH)3 + NaOH NaAlO2 + 2H2O
Ngn la mu
Na+ vng
La n kh
K+ Ngn la mu
tm
Na2O, K2O, dd lm xanh
BaO, CaO H2O qu tm (CaO to Na2O + H2O 2NaOH
ra dung dch c)
P2O5 H2O dd lm qu P2O5 + 3H2O 2H3PO4
tm
SiO2 Dd HF tan to SiF4 SiO2 + 4HF SiF4 +2H2O
Al2O3 + 2NaOH 2NaAlO2 + H2O
Al2O3, ZnO kim dd khng mu
ZnO + 2NaOH Na2ZnO2 + H2O
CuO Axit dd mu xanh CuO + 2HCl CuCl2 + H2O
0
t
4HCl + MnO2
MnO2 HCl un nng Cl2 mu vng
MnCl2 +Cl2 +2H2O
Ag2O HCl un nng AgCl trng Ag2O + 2HCl 2AgCl + H2O
OXIT TH RN
FeO + 4HNO3
Fe(NO3)3 + NO2 + 2H2O
FeO, Fe3O4 HNO3 c NO2 mu nu
Fe3O4 + 10HNO3
3Fe(NO3)3 + NO2+ 5H2O
to dd mu Fe2O3 + 6HNO3
Fe2O3 HNO3 c nu , khng c
kh thot ra 2Fe(NO3)3 + 3H2O
Lu : Mt s dung dch mui lm chuyn mu qu tm:
- Dung dch mui cacbonat, sunfua, photphat, axetat ca kim loi kim lm qu tm xanh
- Dung dch mui (NH4)2SO4, NH4Cl, NH4NO3, AgNO3, AlCl3, Al(NO3)3, mui hirosunfat ca
kim loi kim lm qu tm ha .
CU HI
Cu 1.Cu 5--A7-748: nhn bit ba axit c, ngui: HCl, H2SO4, HNO3 ng ring bit trong ba l
b mt nhn, ta dng thuc th l
A. Al. B. Fe. C. CuO. D. Cu.
Cu 2.Cu 30-B07-285: C th phn bit 3 dung dch: KOH, HCl, H2SO4 (long) bng mt thuc th l
A. giy qu tm. B. Zn. C. Al. D. BaCO3.
Cu 3.Cu 32-CD9-956: Ch dng dung dch KOH phn bit c cc cht ring bit trong nhm no
sau y?
A. Mg, K, Na. B. Fe, Al2O3, Mg. C. Mg, Al2O3, Al. D. Zn, Al2O3, Al.
Cu 4.Cu 34-CD9-956: phn bit CO2 v SO2 ch cn dng thuc th l
GV: Trn c Tun ( 01695 178 188) Trang: 53.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
A. dung dch NaOH. B. nc brom. C. CaO. D. dung dch Ba(OH)2.
Cu 5.Cu 46-CD10-824: Thuc th dng phn bit dung dch NH4NO3 vi dung dch (NH4)2SO4
l
A. ng(II) oxit v dung dch HCl. B. kim loi Cu v dung dch HCl.
C. dung dch NaOH v dung dch HCl. D. ng(II) oxit v dung dch NaOH.
Cu 6. Cu 56 -CD10-824 : Thuc th dng phn bit 3 dung dch ring bit: NaCl, NaHSO4, HCl l
A. BaCO3. B. BaCl2. C. (NH4)2CO3. D. NH4 Cl.
Cu 7.Cu 17-CD11-259: nhn ra ion NO3 trong dung dch Ba(NO3)2, ngi ta un nng nh dung
dch vi
A. kim loi Cu. B. dung dch H2SO4 long.
C. kim loi Cu v dung dch Na2SO4. D. kim loi Cu v dung dch H2SO4 long.
Cu 8.Cu 35-CD11-259: C 4 ng nghim c nh s theo th t 1, 2, 3, 4. Mi ng nghim cha
mt trong cc dung dch AgNO3, ZnCl2, HI, Na2CO3. Bit rng:
- Dung dch trong ng nghim 2 v 3 tc dng c vi nhau sinh ra cht kh;
- Dung dch trong ng nghim 2 v 4 khng phn ng c vi nhau. Dung dch trong cc ng nghim 1,
2, 3, 4 ln lt l:
A. ZnCl2, HI, Na2CO3, AgNO3. B. ZnCl2, Na2CO3, HI, AgNO3.
C. AgNO3, HI, Na2CO3, ZnCl2. D. AgNO3, Na2CO3, HI, ZnCl2.
Cu 9.Cu 46-CD13-415: Thuc th no sau y dng phn bit kh H2S vi kh CO2?
A. Dung dch HCl. B. Dung dch Pb(NO3)2. C. Dung dch NaCl. D. Dung dch K2SO4.
Cu 10.Cu 59-CD13-415: Thuc th no di y phn bit c kh O2 vi kh O3 bng phng php
ha hc?
A. Dung dch H2SO4. B. Dung dch KI + h tinh bt.
C. Dung dch NaOH. D. Dung dch CuSO4.
VN 25: IU CH
L THUYT
1. iu ch kim loi.
Chia 2 loi.
Kim loi mnh: K, Ba,Ca, Na, Mg, Al. iu ch bng phng php in phn nng chy
* mui clorua: tr AlCl3 b thng hoa nhit cao.
* baz: tr Be(OH)2, Mg(OH)2, Al(OH)3 khng bn khi un nng
* oxit: ch dng iu ch Al.
Kim loi TB_Y. Mg tr i.
* Mui - tc dng vi kim loi mnh hn ( thy luyn )
- in phn dung dch
* Oxit: dng CO, H2, Al, C to cao kh ( nhit luyn )
2. iu ch cc phi kim v hp cht ca chng.
- Xem k v phn r cch iu ch trong phng th nghim v trong cng nghip
CU HI
Cu 1.Cu 19-A7-748: Trong phng th nghim, ngi ta thng iu ch clo bng cch
A. cho dung dch HCl c tc dng vi MnO2, un nng.
B. in phn dung dch NaCl c mng ngn.
C. in phn nng chy NaCl.
D. cho F2 y Cl2 ra khi dung dch NaCl.
Cu 2.Cu 39-A7-748: Trong phng th nghim, iu ch mt lng nh kh X tinh khit, ngi
ta un nng dung dch amoni nitrit bo ho. Kh X l
A. N2O. B. NO. C. NO2. D. N2.
Cu 3.Cu 50-A7-748: Dy gm cc kim loi c iu ch trong cng nghip bng phng php
in phn hp cht nng chy ca chng, l:
GV: Trn c Tun ( 01695 178 188) Trang: 54.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
A. Fe, Ca, Al. B. Na, Ca, Zn. C. Na, Cu, Al. D. Na, Ca, Al.
Cu 4.Cu 29-B07-285: Trong phng th nghim, ngi ta thng iu ch HNO3 t
A. NaNO2 v H2SO4 c. B. NaNO3 v H2SO4 c.
C. NH3 v O2. D. NaNO3 v HCl c.
Cu 5.Cu 14-CD7-439: Trong cng nghip, natri hiroxit c sn xut bng phng php
A. in phn dung dch NaNO3, khng c mng ngn in cc.
B. in phn NaCl nng chy.
C. in phn dung dch NaCl, khng c mng ngn in cc.
D. in phn dung dch NaCl, c mng ngn in cc.
Cu 6.Cu 14-A8-329: Trong phng th nghim, ngi ta iu ch oxi bng cch
A. in phn nc. B. nhit phn Cu(NO3)2.
C. nhit phn KClO3 c xc tc MnO2. D. chng ct phn on khng kh lng.
Cu 7.Cu 48-CD8-216: Hai kim loi c th c iu ch bng phng php in phn dung dch l
A. Al v Mg. B. Na v Fe. C. Cu v Ag. D. Mg v Zn.
Cu 8.Cu 11-A9-438: Dy cc kim loi u c th c iu ch bng phng php in phn dung
dch mui ca chng l:
A. Mg, Zn, Cu. B. Al, Fe, Cr. C. Fe, Cu, Ag. D. Ba, Ag, Au.
Cu 9.Cu 17-A9-438: Nu cho 1 mol mi cht: CaOCl2, KMnO4, K2Cr2O7, MnO2 ln lt phn ng
vi lng d dung dch HCl c, cht to ra lng kh Cl2 nhiu nht l
A. CaOCl2. B. KMnO4. C. K2Cr2O7. D. MnO2.
Cu 10.Cu 7-B9-148: Khi nhit phn hon ton 100 gam mi cht sau: KClO3 (xc tc MnO2),
KMnO4, KNO3 v AgNO3. Cht to ra lng O2 ln nht l
A. KClO3. B. KMnO4. C. KNO3. D. AgNO3.
Cu 11.Cu 26-B9-148: Thc hin cc th nghim sau:
(I) Cho dung dch NaCl vo dung dch KOH.
(II) Cho dung dch Na2CO3 vo dung dch Ca(OH)2.
(III) in phn dung dch NaCl vi in cc tr, c mng ngn.
(IV) Cho Cu(OH)2 vo dung dch NaNO3. (V) Sc kh NH3 vo dung dch Na2CO3.
(VI) Cho dung dch Na2SO4 vo dung dch Ba(OH)2.
Cc th nghim u iu ch c NaOH l:
A. II, III v VI. B. I, II v III. C. I, IV v V. D. II, V v VI.
Cu 12.Cu 8-A12-296: Dy cc kim loi u c th c iu ch bng phng php in phn dung
dch mui (vi in cc tr) l:
A. Ni, Cu, Ag. B. Ca, Zn, Cu. C. Li, Ag, Sn. D. Al, Fe, Cr
Cu 13.Cu 49-A13-193: Th nghim vi dung dch HNO3 thng sinh ra kh c NO2. hn ch kh
NO2 thot ra t ng nghim, ngi ta nt ng nghim bng:
(a) bng kh. (b) bng c tm nc.
(c) bng c tm nc vi. (d) bng c tm gim n.
Trong 4 bin php trn, bin php c hiu qu nht l
A. (d). B. (a). C. (c). D. (b).
VN 26: TCH TINH CH
L THUYT
- Dng 1 ha cht tch A ra khi hn hp.
=> tm cht m A khng phn ng, cn cht phn ng vi tt c cc cht cn li.
- Dng nhiu ha cht tch A ra khi hn hp.
=> Tm cht phn ng vi A m khng ( t ) phn ng vi cht cn li, cc cht tip theo s l
cht phc hi li A.
CU HI
GV: Trn c Tun ( 01695 178 188) Trang: 55.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
Cu 1.Cu 36-B07-285: thu c Al2O3 t hn hp Al2O3 v Fe2O3, ngi ta ln lt:
A. dng kh H2 nhit cao, dung dch NaOH (d).
B. dng kh CO nhit cao, dung dch HCl (d).
C. dng dung dch NaOH (d), dung dch HCl (d), ri nung nng.
D. dng dung dch NaOH (d), kh CO2 (d), ri nung nng.
Cu 2.Cu 25-B10-937: Phng php loi b tp cht HCl c ln trong kh H2S l: Cho hn hp kh
li t t qua mt lng d dung dch
A. AgNO3. B. NaOH. C. NaHS. D. Pb(NO3)2.
Cu 3.Cu 32-CD12-169: Kim loi no sau y iu ch c bng phng php thy luyn?
A. Mg. B. Ca. C. Cu. D. K.
Cu 4.Cu 43-CD12-169: loi b Al, Fe, CuO ra khi hn hp gm Ag, Al, Fe v CuO, c th dng
lng d dung dch no sau y?
A. Dung dch NaOH. B. Dung dch Fe(NO3)3. C. Dung dch HNO3. D. Dung dch HCl.
VN 27: S V C
L THUYT
- Lu : + Mi mi tn: mt phn ng
+ Mi k hiu cha bit: mt cht ha hc
- Cn nm c mi lin h gia tnh cht, cch iu ch cc cht.
CU HI
Cu 1.Cu 10-CD7-439: Cho s phn ng: NaCl (X) NaHCO3 (Y) NaNO3. X v Y c
th l
A. NaOH v NaClO. B. NaOH v Na2CO3.
C. NaClO3 v Na2CO3. D. Na2CO3 v NaClO.
Cu 2.Cu 30-A8-329: T hai mui X v Y thc hin cc phn ng sau:
to
X X1 + H2O X1 + H2O X2
X2 + Y X + Y1 + H2O X2 + 2Y X + Y2 + 2H2O
Hai mui X, Y tng ng l
A. BaCO3, Na2CO3. B. CaCO3, NaHSO4. C. MgCO3, NaHCO3. D. CaCO3, NaHCO3.
Cu 3.Cu 51-A8-329: Cho s chuyn ho qung ng thnh ng:
O2 , t o O2 , t o X ,t o
CuFeS2 X Y Cu
Hai cht X, Y ln lt l:
A. Cu 2O, CuO. B. CuS, CuO. C. Cu 2S, Cu2O. D. Cu 2S, CuO.
Cu 4.Cu 36-CD8-216: Cho s chuyn ho (mi mi tn l mt phng trnh phn ng):
ddX
NaOH Fe(OH)2 ddY
Fe2(SO4)3 ddZ
BaSO4
Cc dd (dung dch) X, Y, Z ln lt l:
A. FeCl3, H2SO4 (c, nng), Ba(NO3)2. B. FeCl3, H2SO4 (c, nng), BaCl2.
C. FeCl2, H2SO4 (c, nng), BaCl2. D. FeCl2, H2SO4 (long), Ba(NO3)2.
Cu 5. Cu 47-B9-148: Cho s chuyn ho gia cc hp cht ca crom:
KOH ( Cl2 KOH ) H 2 SO4 ( FeSO4 H 2 SO4 )
Cr(OH)3 X Y Z T
Cc cht X, Y, Z, T theo th t l:
A. K2CrO4; KCrO2; K2Cr2O7; Cr2(SO4)3. B. KCrO2; K2Cr2O7; K2CrO4; Cr2(SO4)3.
C. KCrO2; K2Cr2O7; K2CrO4; CrSO4. D. KCrO2; K2CrO4; K2Cr2O7; Cr2(SO4)3.
Cu 6.Cu 35-B10-937: Cho s chuyn ho:
KOH KOH
P2O5 X H 3 PO4
Y Z
Cc cht X, Y, Z ln lt l:
A. KH2PO4, K2HPO4, K3PO4. B. KH2PO4, K3PO4, K2HPO4.
GV: Trn c Tun ( 01695 178 188) Trang: 56.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
C. K3PO4, KH2PO4, K2HPO4. D. K3PO4, K2HPO4, KH2PO4.
Cu 7.Cu 54-B10-937: Cho s chuyn ho:
Fe3O4 + dung dch HI (d) X + Y + H2O.
Bit X v Y l sn phm cui cng ca qu trnh chuyn ho. Cc cht X v Y l
A. FeI3 v I2 B. Fe v I2 C. FeI2 v I2 D. FeI3 v I2
Cu 8.Cu 36-CD10-824: Cho s chuyn ho sau:
X Y Z
CaO CaCl2 Ca(NO3)2 CaCO3
Cng thc ca X, Y, Z ln lt l:
A. HCl, HNO3, Na2CO3. B. HCl, AgNO3, (NH4)2CO3.
C. Cl2, AgNO3, MgCO3. D. Cl2, HNO3, CO2.
Cu 9.Cu 11-B12-359: Cho s chuyn ho:
to COdu ,t o FeCl3 T
Fe(NO3)3 X Y Z Fe(NO3)3
Cc cht X v T ln lt l
A. FeO v NaNO3. B. FeO v AgNO3. C. Fe2O3 v Cu(NO3)2. D. Fe2O3 v AgNO3.
Cu 10.Cu 52-CD12-169: Cho s phn ng:
Cl2 du KOHdac Cl2
Cr X Y
Bit Y l hp cht ca crom. Hai cht X v Y ln lt l
A. CrCl2 v Cr(OH)3. B. CrCl3 v K2Cr2O7. C. CrCl3 v K2CrO4. D. CrCl2 v K2CrO4.
Cu 11.Cu 53 -A13-193: Cho s phn ng C r Cl2 du
X
dungdichNaOHdu
Y
Cht Y trong s trn l
A. Na[Cr(OH)4]. B. Na2Cr2O7. C. Cr(OH)2. D. Cr(OH)3.
Cu 12. Cu 8-B13-279: Cho s phn ng: Al2(SO4)3 X Y Al.
Trong s trn, mi mi tn l mt phn ng, cc cht X, Y ln lt l nhng cht no sau y?
A. NaAlO2 v Al(OH)3. B. Al(OH)3 v NaAlO2.
C. Al2O3 v Al(OH)3. D. Al(OH)3 v Al2O3.
VN 28: TNG HP CC PHT BIU TRONG HA V C
L THUYT
- Cc pht biu trong ha v c thng tp chung vo cc pht biu v vai tr ca cc cht trong phn
ng oxi ha kh, cc ng dng, trng thi tnh cht ca cc cht hoc tnh cht ca hp cht phc tp nh
st, crom, ng.
CU HI
Cu 1. Cu 46-A7-748: Mnh khng ng l:
A. Fe kh c Cu 2+ trong dung dch.
B. Fe3+ c tnh oxi ha mnh hn Cu 2+.
C. Fe2+ oxi ho c Cu.
D. Tnh oxi ha ca cc ion tng theo th t: Fe2+, H+, Cu2+, Ag+.
Cu 2. Cu 54-A7-748: Pht biu khng ng l:
A. Hp cht Cr(II) c tnh kh c trng cn hp cht Cr(VI) c tnh oxi ho mnh.
B. Cc hp cht CrO, Cr(OH)2 tc dng c vi dung dch HCl cn CrO3 tc dng c vi dung dch
NaOH.
C. Cc hp cht Cr2O3, Cr(OH)3, CrO, Cr(OH)2 u c tnh cht lng tnh.
D. Thm dung dch kim vo mui icromat, mui ny chuyn thnh mui cromat.
Cu 3.Cu 13-B8-371: Cho bit cc phn ng xy ra sau:
2FeBr2 + Br2 2FeBr3 2NaBr + Cl2 2NaCl + Br2
Pht biu ng l:
GV: Trn c Tun ( 01695 178 188) Trang: 57.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
A. Tnh oxi ha ca Cl2 mnh hn ca Fe3+. B. Tnh oxi ha ca Br2 mnh hn ca Cl2.
C. Tnh kh ca Br- mnh hn ca Fe2+. D. Tnh kh ca Cl- mnh hn ca Br -.
Cu 4.Cu 28-B8-371: Nguyn tc luyn thp t gang l:
A. Dng O2 oxi ho cc tp cht Si, P, S, Mn, trong gang thu c thp.
B. Tng thm hm lng cacbon trong gang thu c thp.
C. Dng CaO hoc CaCO3 kh tp cht Si, P, S, Mn, trong gang thu c thp.
D. Dng cht kh CO kh oxit st thnh st nhit cao.
Cu 5.Cu 52-CD8-216: Hai kim loi X, Y v cc dung dch mui clorua ca chng c cc phn ng ha
hc sau: X + 2YCl3 XCl2 + 2YCl2; Y + XCl2 YCl2 + X.
Pht biu ng l:
A. Ion Y2+ c tnh oxi ha mnh hn ion X2+ . B. Kim loi X kh c ion Y2+.
C. Kim loi X c tnh kh mnh hn kim loi Y. D. Ion Y3+ c tnh oxi ha mnh hn ion X2 +.
Cu 6.Cu 44-A9-438: Pht biu no sau y l ng?
A. Phn ur c cng thc l (NH4)2CO3.
B. Phn ln cung cp nit ho hp cho cy di dng ion nitrat (NO3-) v ion amoni (NH4 +).
C. Amophot l hn hp cc mui (NH4)2HPO4 v KNO3.
D. Phn hn hp cha nit, photpho, kali c gi chung l phn NPK.
Cu 7.Cu 33-B9-148: Pht biu no sau y l ng?
A. Photpho trng c cu trc tinh th nguyn t.
B. th rn, NaCl tn ti di dng tinh th phn t.
C. Nc thuc loi tinh th phn t.
D. Kim cng c cu trc tinh th phn t.
Cu 8.Cu 46-B9-148: ng dng no sau y khng phi ca ozon?
A. Ty trng tinh bt, du n. B. Cha su rng.
C. iu ch oxi trong phng th nghim. D. St trng nc sinh hot.
Cu 9.Cu 3-CD9-956: Nguyn tc chung c dng iu ch kim loi l
A. cho hp cht cha ion kim loi tc dng vi cht oxi ho.
B. kh ion kim loi trong hp cht thnh nguyn t kim loi.
C. oxi ho ion kim loi trong hp cht thnh nguyn t kim loi.
D. cho hp cht cha ion kim loi tc dng vi cht kh.
Cu 10.Cu 1-A10-684: Pht biu no sau y ng?
A. Cc kim loi: natri, bari, beri u tc dng vi nc nhit thng.
B. Kim loi xesi c dng ch to t bo quang in.
C. Theo chiu tng dn ca in tch ht nhn, cc kim loi kim th (t beri n bari) c nhit
nng chy gim dn.
D. Kim loi magie c kiu mng tinh th lp phng tm din.
Cu 11.Cu 12-A10-684: Pht biu khng ng l:
A. Trong cng nghip, photpho c sn xut bng cch nung hn hp qung photphorit, ct v than cc
1200oC trong l in.
B. Hiro sunfua b oxi ho bi nc clo nhit thng.
C. Tt c cc nguyn t halogen u c cc s oxi ho: -1, +1, +3, +5 v +7 trong cc hp cht.
D. Kim cng, than ch, fuleren l cc dng th hnh ca cacbon.
26
Cu 12.Cu 20-A10-684: Nhn nh no sau y ng khi ni v 3 nguyn t: 13 X , 55 26
26Y , 12 Z
A. X, Z l 2 ng v ca cng mt nguyn t ho hc.
B. X v Y c cng s ntron.
C. X, Y thuc cng mt nguyn t ho hc.
D. X v Z c cng s khi.
Cu 13.Cu 37-A10-684: C cc pht biu sau:
1
Lu hunh, photpho u bc chy khi tip xc vi CrO3.
GV: Trn c Tun ( 01695 178 188) Trang: 58.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
2
Ion Fe3+ c cu hnh electron vit gn l [Ar]3d5.
3
Bt nhm t bc chy khi tip xc vi kh clo.
4
Phn chua c cng thc l Na2SO4.Al2(SO4)3.24H2O. Cc pht biu ng l:
A. 1, 3, 4. B. 2, 3, 4. C. 1, 2, 4. D. 1, 2, 3.
Cu 14.Cu 45-A10-684: Cht c dng ty trng giy v bt giy trong cng nghip l
A. NO2. B. SO2. C. CO2. D. N2O.
Cu 15.Cu 18-B10-937: Pht biu no sau y khng ng?
A. m chy magie c th c dp tt bng ct kh.
B. Dung dch m c ca Na2SiO3 v K2SiO3 c gi l thy tinh lng.
C. Trong phng th nghim, N2 c iu ch bng cch un nng dung dch NH4NO2 bo ho.
D. CF2Cl2 b cm s dng do khi thi ra kh quyn th ph hy tng ozon.
Cu 16.Cu 23-B10-937: Pht biu no sau y khng ng?
A. Trong cc dung dch: HCl, H2SO4, H2S c cng nng 0,01M, dung dch H2S c pH ln nht.
B. Dung dch Na2CO3 lm phenolphtalein khng mu chuyn sang mu hng.
C. Nh dung dch NH3 t t ti d vo dung dch CuSO4, thu c kt ta xanh.
D. Nh dung dch NH3 t t ti d vo dung dch AlCl3, thu c kt ta trng.
Cu 17.Cu 27-B10-937: Pht biu no sau y khng ng khi so snh tnh cht ha hc ca nhm v
crom?
A. Nhm c tnh kh mnh hn crom.
B. Nhm v crom u bn trong khng kh v trong nc.
C. Nhm v crom u b th ng ha trong dung dch H2SO4 c ngui.
D. Nhm v crom u phn ng vi dung dch HCl theo cng t l v s mol.
Cu 18.Cu 56-B10-937: Pht biu no sau y khng ng?
A. Do Pb 2+/Pb ng trc 2H+/H trong dy in ho nn Pb d dng phn ng vi dung dch HCl long
2
ngui, gii phng kh H2.
B. Trong mi trng kim, mui Cr(III) c tnh kh v b cc cht oxi ho mnh chuyn thnh mui
Cr(VI).
C. Ag khng phn ng vi dung dch H2SO4 long nhng phn ng vi dung dch H2SO4 c nng.
D. CuO nung nng khi tc dng vi NH3 hoc CO, u thu c Cu.
Cu 19.Cu 2-CD10-824: Dy gm cc kim loi c cu to mng tinh th lp phng tm khi l:
A. Li, Na, K. B. Be, Mg, Ca. C. Li, Na, Ca. D. Na, K, Mg.
Cu 20.Cu 13-CD10-824: Pht biu no sau y ng?
A. Dung dch NaF phn ng vi dung dch AgNO3 sinh ra AgF kt ta.
B. Axit HBr c tnh axit yu hn axit HCl.
C. Iot c bn knh nguyn t ln hn brom.
D. Flo c tnh oxi ho yu hn clo.
Cu 21.Cu 50-CD10-824: Pht biu no sau y khng ng?
A. Ancol etylic bc chy khi tip xc vi CrO3.
B. Khi phn ng vi dung dch HCl, kim loi Cr b oxi ho thnh ion Cr2+.
C. Crom(VI) oxit l oxit baz.
D. Crom(III) oxit v crom(III) hiroxit u l cht c tnh lng tnh.
Cu 22.Cu 26-CD11-259: Pht biu no sau y khng ng?
A. Trong cc hp cht, ngoi s oxi ho -1, flo v clo cn c cc s oxi ho +1, +3, +5, +7.
B. Mui AgI khng tan trong nc, mui AgF tan trong nc.
C. Flo c tnh oxi ha mnh hn clo.
D. Dung dch HF ha tan c SiO2.
Cu 23.Cu 29-CD11-259: Dy gm cc kim loi u c cu to mng tinh th lp phng tm khi l:
A. Na, K, Ca, Ba. B. Na, K, Ca, Be. C. Li, Na, K, Mg. D. Li, Na, K, Rb.
Cu 24.Cu 32-A11-318: Pht biu no sau y l sai?
GV: Trn c Tun ( 01695 178 188) Trang: 59.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
A. m in ca brom ln hn m in ca iot.
B. Bn knh nguyn t ca clo ln hn bn knh nguyn t ca flo.
- -
C. Tnh kh ca ion Br ln hn tnh kh ca ion Cl
D. Tnh axit ca HF mnh hn tnh axit ca HCl.
Cu 25. Cu 36-A11-318: Khi so snh NH3 vi NH4+ , pht biu khng ng l:
A. Trong NH3 v NH4+, nit u c cng ha tr 3.
B. NH3 c tnh baz, NH4+ c tnh axit.
C. Trong NH3 v NH4+, nit u c s oxi ha 3.
D. Phn t NH v ion NH + u cha lin kt cng ha tr.
3 4
Cu 26.Cu 1-B11-846: Dy gm cc kim loi c cng kiu mng tinh th lp phng tm khi l:
A. Na, K, Ba. B. Li, Na, Mg. C. Na, K, Ca. D. Mg, Ca, Ba.
Cu 27.Cu 24-B11-846: Pht biu no sau y l sai?
A. Nhm bn trong mi trng khng kh v nc l do c mng oxit Al2O3 bn vng bo v.
B. Theo chiu tng dn ca in tch ht nhn, nhit nng chy ca kim loi kim gim dn.
C. nhit thng, tt c cc kim loi kim th u tc dng c vi nc.
D. Na2CO3 l nguyn liu quan trng trong cng nghip sn xut thu tinh.
Cu 28.Cu 27-B11-846: Pht biu no sau y l sai?
A. Trong tinh th nguyn t, cc nguyn t lin kt vi nhau bng lin kt cng ho tr.
B. Tinh th nc , tinh th iot u thuc loi tinh th phn t.
C. Trong tinh th NaCl, xung quanh mi ion u c 6 ion ngc du gn nht.
D. Tt c cc tinh th phn t u kh nng chy v kh bay hi.
Cu 29.Cu 44-B11-846: Pht biu no sau y l sai?
A. Ch (Pb) c ng dng ch to thit b ngn cn tia phng x.
B. Nhm l kim loi dn in tt hn vng.
C. Trong y hc, ZnO c dng lm thuc gim au dy thn kinh, cha bnh eczema, bnh nga.
D. Thic c th dng ph ln b mt ca st chng g.
Cu 30.Cu 43-A12-296: Nhn xt no sau y khng ng?
A. Vt dng lm bng nhm v crom u bn trong khng kh v nc v c mng oxit bo v.
B. Crom l kim loi cng nht trong tt c cc kim loi.
C. Nhm v crom u b th ng ha bi HNO3 c, ngui.
D. Nhm v crom u phn ng vi dung dch HCl theo cng t l s mol.
Cu 31.Cu 56-A12-296: Nhn xt no sau y khng ng?
A. BaSO4 v BaCrO4 hu nh khng tan trong nc.
B. Al(OH)3 v Cr(OH)3 u l hiroxit lng tnh v c tnh kh.
C. SO3 v CrO3 u l oxit axit.
D. Fe(OH)2 v Cr(OH)2 u l baz v c tnh kh.
Cu 32.Cu 1-B12-359: Pht biu no sau y l ng?
A. Tt c cc phn ng ca lu hunh vi kim loi u cn un nng.
B. Trong cng nghip, nhm c sn xut t qung olomit.
C. Ca(OH)2 c dng lm mt tnh cng vnh cu ca nc.
D. CrO3 tc dng vi nc to ra hn hp axit.
Cu 33.Cu 3-B12-359: Khi ni v kim loi kim, pht biu no sau y l sai?
A. Cc kim loi kim c mu trng bc v c nh kim.
B. Trong t nhin, cc kim loi kim ch tn ti dng hp cht.
C. T Li n Cs kh nng phn ng vi nc gim dn.
D. Kim loi kim c nhit nng chy v nhit si thp.
Cu 34.Cu 14-B12-359: Pht biu no sau y l sai?
A. Nguyn t kim loi thng c 1, 2 hoc 3 electron lp ngoi cng.
B. Cc nhm A bao gm cc nguyn t s v nguyn t p.
C. Trong mt chu k, bn knh nguyn t kim loi nh hn bn knh nguyn t phi kim.
D. Cc kim loi thng c nh kim do cc electron t do phn x nh sng nhn thy c.
GV: Trn c Tun ( 01695 178 188) Trang: 60.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
Cu 35.Cu 17-B12-359: Pht biu no sau y l ng?
A. Hn hp FeS v CuS tan c ht trong dung dch HCl d.
B. Thi khng kh qua than nung , thu c kh than t.
C. Photpho d bc chy trong khng kh iu kin thng.
D. Dung dch hn hp HCl v KNO3 ho tan c bt ng.
Cu 36.Cu 48-B12-359: Pht biu no sau y l sai?
A. Clo c dng dit trng nc trong h thng cung cp nc sch.
B. Amoniac c dng iu ch nhin liu cho tn la.
C. Lu hunh ioxit c dng lm cht chng nm mc.
D. Ozon trong khng kh l nguyn nhn chnh gy ra s bin i kh hu.
Cu 37. Cu 49-B12-359: Pht biu no sau y l sai?
A. Cr(OH)3 tan trong dung dch NaOH.
B. Trong mi trng axit, Zn kh Cr3+ thnh Cr.
C. Photpho bc chy khi tip xc vi CrO3.
D. Trong mi trng kim, Br2 oxi ha CrO2- thnh CrO42-
Cu 38. Cu 3-CD12-169: Pht biu no sau y l ng?
A. Trong hp cht, tt c cc kim loi kim u c s oxi ha +1.
B. Tt c cc kim loi nhm IIA u c mng tinh th lp phng tm khi.
C. Tt c cc hiroxit ca kim loi nhm IIA u d tan trong nc.
D. Trong nhm IA, tnh kh ca cc kim loi gim dn t Li n Cs.
Cu 39. Cu 42-A13-193: Cho cc pht biu sau:
(a) Trong bng tun hon cc nguyn t ha hc, crom thuc chu k 4, nhm VIB.
(b) Cc oxit ca crom u l oxit baz.
(c) Trong cc hp cht, s oxi ha cao nht ca crom l +6.
(d) Trong cc phn ng ha hc, hp cht crom(III) ch ng vai tr cht oxi ha.
(e) Khi phn ng vi kh Cl2 d, crom to ra hp cht crom(III).
Trong cc pht biu trn, nhng pht biu ng l:
A. (b), (c) v (e). B. (a), (c) v (e). C. (b), (d) v (e). D. (a), (b) v (e).
Cu 40. Cu 2-B13-279: Pht biu no sau y l ng?
A. Thnh phn chnh ca supephotphat kp gm hai mui Ca(H2PO4)2 v CaSO4.
B. Ur c cng thc l (NH2)2CO.
C. Supephotphat n ch c Ca(H2PO4)2.
D. Phn ln cung cp nit cho cy trng.
Cu 41. Cu 5-B13-279: Cho cc pht biu sau:
(a) Trong cc phn ng ha hc, flo ch th hin tnh oxi ha.
(b) Axit flohiric l axit yu.
(c) Dung dch NaF long c dng lm thuc chng su rng.
(d) Trong hp cht, cc halogen (F, Cl, Br, I) u c s oxi ha: -1, +1, +3, +5 v +7.
(e) Tnh kh ca cc ion halogenua tng dn theo th t: F, Cl, Br, I.
Trong cc pht biu trn, s pht biu ng l
A. 3. B. 5. C. 2. D. 4.
Cu 42. Cu 5-CD13-415: Pht biu no sau y khng ng?
A. Kim loi Fe phn ng vi dung dch HCl to ra mui st(II).
B. Dung dch FeCl3 phn ng c vi kim loi Fe.
C. Trong cc phn ng ha hc, ion Fe2+ ch th hin tnh kh.
D. Kim loi Fe khng tan trong dung dch H2SO4 c, ngui.
Cu 43. Cu 24-CD13-415: Pht biu no di y khng ng?
A. SiO2 l oxit axit.
B. t chy hon ton CH4 bng oxi, thu c CO2 v H2O.
C. Sc kh CO2 vo dung dch Ca(OH)2 d, dung dch b vn c.
D. SiO2 tan tt trong dung dch HCl.
GV: Trn c Tun ( 01695 178 188) Trang: 61.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
Cu 44. Cu 40-CD13-415: Pht biu no sau y khng ng?
A. Kim loi Al tan c trong dung dch HNO3 c, ngui.
B. Al(OH)3 phn ng c vi dung dch HCl v dung dch KOH.
C. Trong cng nghip, kim loi Al c iu ch bng phng php in phn Al2O3 nng chy.
D. Trong cc phn ng ha hc, kim loi Al ch ng vai tr cht kh.
Cu 45. Cu 41-CD13-415: Pht biu no di y khng ng?
A. Nguyn tc chung iu ch kim loi l kh ion kim loi thnh nguyn t kim loi.
B. n mn ha hc pht sinh dng in.
C. Tnh cht ha hc c trng ca kim loi l tnh kh.
D. Bn cht ca n mn kim loi l qu trnh oxi ha - kh.
Cu 46. Cu 53-CD13-415: Pht biu no sau y khng ng?
A. Kim loi Cu phn ng c vi dung dch hn hp KNO3 v HCl.
B. Cr(OH)2 l hiroxit lng tnh.
C. Cu(OH)2 tan c trong dung dch NH3.
D. Kh NH3 kh c CuO nung nng.
VN 29: HA HC VI KINH T, X HI V MI TRNG
L THUYT
I. HA HC V KINH T
1. Nng lng v nhin liu
a. Nng lng v nhin liu c vai tr quan trng nh th no i vi s pht trin kinh t?
- Cc ngun nng lng chnh l: Mt Tri, thc phm, g gi, nc, du m, kh t nhin, than , cc
cht c phn ng ht nhn,...
- C nhiu dng nng lng khc nhau: ng nng, nhit nng, ha nng, in nng, quang nng, th
nng,... T dng nng lng ny c th bin i sang dng nng lng khc
- Nhin liu khi b t chy sinh ra nng lng (nhit nng). Hin nay ngun cung cp nhin liu ch yu
l than, du m v kh t nhin. Cc dng nhin liu ny c gi l nhin liu ha thch, c trong v
Tri t.
* Mi qu trnh hot ng ca con ngi u cn nng lng v nhin liu. Nng lng v nhin liu cn
cho s pht trin cc ngnh kinh t: nng nghip, cng nghip, xy dng, giao thng vn ti, ng
nghip,... Nhn loi khng th tn ti v pht trin nu thiu nng lng.
b. Vn v nng lng v nhin liu ang t ra cho nhn loi hin nay l g?
- Cng vi s pht trin cc ngnh kinh t, nhu cu v nng lng v nhin liu ngy cng tng. Trong
khi cc ngun nng lng, nhin liu ha thch nh du m, than , kh t nhin... khng phi l v
tn m c gii hn v ngy cng cn kit do b khai thc qu nhiu.
- Ngi ta d on rng, mt vi trm nm na cc ngun nhin liu ha thch trn Tri t s cn kit
do con ngi khai thc ngy cng nhiu s dng cng nh lm vt phm, hng ha xut khu.
- Khai thc v s dng nng lng ha thch cn l mt trong nhng nguyn nhn ch yu gy nn
nhim mi trng v lm thay i kh hu ton cu.
c. Ha hc gp phn gii quyt vn nng lng v nhin liu nh th no trong hin ti v
tng lai?
- Nhn loi ang gii quyt vn thiu nng lng v khan him nhin liu do tiu th qu nhiu theo
hng nng cao tnh hiu qu trong vic sn xut v s dng nng lng, m bo s pht trin bn vng.
Ha hc nghin cu gp phn sn xut v s dng ngun nhin liu thin nhin nh than, du m.
- Sn xut etanol t ng, sn thay th xng, ch bin du thc vt (da, c,...) thay cho du iezen
trong cc trong cc ng c t trong.
- Sn xut ra cht thay cho xng t ngun nguyn liu v tn l khng kh v nc.
- Nng lng c sn sinh trong cc l phn ng ht nhn c s dng cho mc ch ha bnh. Ha
hc gip xc nh c s khoa hc ca quy trnh k thut to ra vt liu c bit xy l phn ng ht
nhn, gip qu trnh lm sch nguyn liu urani,... s dng trong cc nh my in nguyn t.
GV: Trn c Tun ( 01695 178 188) Trang: 62.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
- Ha hc cng gp phn to ra vt liu chuyn dng ch to pin Mt Tri, ch to thit b, my mc
thch hp khai thc, s dng hiu qu nhng ngun nng lng sch c tim nng to ln khc t thin
nhin: Nng lng thy in; nng lng gi; nng lng Mt Tri; nng lng a nhit; nng lng
thy triu,...
- Trong cng nghip ha hc, ngi ta s dng cc ngun nhin liu, nng lng mi mt cch khoa
hc v tit kim.
- Ha hc gip to ra v s dng ngun nng lng in ha trong pin in ha hoc acquy. Acquy
kh v acquy ch axit l loi c dng ph bin nht hin nay.
2 Vt liu
a. Vai tr ca vt liu i vi s pht trin kinh t
- Trong lch s pht trin ca nhn loi s dng nhiu loi vt liu khc nhau.
- S pht trin ca cc vt liu mi gp phn to ra s pht trin cho nhng ngnh kinh t mi nhn
ca nhn loi.
b. Vn v vt liu ang t ra cho nhn loi l g?
- Cng vi s pht trin ca cc ngnh kinh t v khoa hc k thut, nhu cu ca nhn loi v vt liu mi
vi nhng tnh nng vt l v ha hc, sinh hc mi ngy cng cao.
- Ngoi nhng vt liu t nhin, nhu cu v vt liu nhn to ngy cng a dng, phong ph p ng
nhu cu ngy cng cao v vt liu ca cc ngnh kinh t quc dn.
c. Ha hc gp phn gii quyt vn v vt liu nh th no?
- Ha hc v ang gp phn to nn cc loi vt liu mi cho nhn loi. Cc nh ha hc nghin
cu c cc cht ha hc lm nguyn liu ban u, nhng iu kin c bit, nhng cht xc tc v c
v hu c to ra nhng vt liu c tnh nng ring, c bit phc v cho cc ngnh kinh t, y hc, cng
ngh sinh hc, khoa hc v tr,...
* V liu v c: Ngnh sn xut ha hc v c to ra nhiu loi vt liu c s dng trong cng nghip
v i sng.
Th d: Luyn kim en v luyn kim mu sn xut ra cc kim loi: vng, nhm, st, thp, ng, titan v
hp kim nh uyra,...
Cng nghip silicat sn xut ra ngch, ngi, xi mng, thy tinh, gm, s,...
Cng nghip ha cht sn xut ra cc ha cht c bn nh HCl,H2SO4,HNO3,NH3,NaOH,... lm nguyn
liu sn xut phn bn, thuc tr su.
* Vt liu hu c: Nhiu loi vt liu hu c c sn xut bng con ng ha hc. Th d: Sn tng
hp, nha, cht do, PVC, cao su tng hp, t, si tng hp.
* Vt liu mi: Ngy nay, ha hc cng vi ngnh khoa hc vt liu nghin cu to nn mt s loi vt
liu mi c tnh nng c bit: Trng lng siu nh, siu dn in, siu bn, siu nh,... gip pht trin
cc ngnh cng nghip in t, nng lng ht nhn, y t,... Th d:
- Vt liu nano (cn gi l vt liu nanomet) l loi vt liu c to nn t nhng ht c kch thc c
nanomet. Vt liu nano c rn siu cao, siu do v nhiu tnh nng c bit m vt liu thng khng
c c.
- Vt liu quang in t c siu dn nhit cao c dng trong sinh hc, y hc, in t,...
- Vt liu compozit c tnh nng bn, chc khng b axit hoc kim v mt s ha cht ph hy.
II. HA HC V X HI
1 - HA HC V VN LNG THC, THC PHM
a.Vai tr ca lng thc, thc phm i vi i sng ca con ngi
- Lng thc, thc phm p ng nhu cu dinh dng, cung cp nng lng cho con ngi sng v hot
ng.
- m bo duy tr s sng th lng thc, thc phm trong khu phn n hng ngy cn m bo y
theo mt t l thch hp cc cht bt (cacbohirat), cht m (protein), cht bo (lipit), vitamin, cht
khong v cc cht vi lng.
- n khng nng lng hoc thiu cht dinh dng s lm cho c th hot ng khng hiu qu, sc
khe yu, chm pht trin tr tu,... c bit l i vi cc ph n ang mang thai v tr em. Th d: Nu
thiu iot s gy km tr nh , thiu vitamin A s gy bnh kh mt dn n m la, thiu st dn n bnh
thiu mu
GV: Trn c Tun ( 01695 178 188) Trang: 63.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
b. Vn v lng thc, thc phm ang t ra cho nhn loi hin nay
- Nhn loi ang ng trc thch thc ln v lng thc, thc phm. Dn s th gii ngy cng tng
nht l nhng nc ang pht trin dn n nhu cu v lng thc v thc phm ngy cng tng ln. ---
- Trong khi , nhu cu v lng thc, thc phm c cht lng cao m bo n ngon, chng bnh bo
ph mt s nc pht trin li ang c t ra. c tnh c khong 15% dn s cc nc mc bnh
bo ph.
- Ngoi ra, din tch trng trt ngy cng b thu hp do b th ha, do kh hu tri t nng ln v thin
tai (ma, bo, l lt,...) ngy cng khc nghit dn n gim sn lng lng thc.
c. Ha hc gp phn gii quyt vn lng thc, thc phm cho nhn loi nh th no?
- gii quyt vn lng thc, thc phm cho nhn loi, ha hc gp phn nghin cu v sn xut
cc cht ha hc c tc dng bo v, pht trin thc vt, ng vt gip tng sn lng, cht lng v bo
qun tt hn. Th d:
- Sn xut cc loi phn bn ha hc c tc dng tng nng sut cy trng nh: Phn m, phn ln, phn
kali, phn hn hp, phn phc hp, phn vi lng,...
- Tng hp ha cht c tc dng dit tr c di to iu kin cho cy lng thc pht trin.
- Tng hp ha cht dit nm bnh,... bo v cy lng thc trnh c dch bnh nh: Etirimol,
benoxyl, ng sunfat,...
- Sn xut nhng ha cht bo qun lng thc v thc phm, lm chm s pht trin ca vi khun gy
hi cho lng thc, thc phm.
- Nghin cu ch bin thc n tng hp tng sn lng chn nui gia sc, gia cm, thy sn.
Ch bin thc phm nhn to hoc ch bin thc phm theo cng ngh ha hc.
Ha hc gip thay th ngun nguyn liu lm lng thc, thc phm dng trong cng nghip ha hc
bng nguyn liu phi lng thc, phi thc phm. Th d:
- Thay th tinh bt bng hp cht hirocacbon sn xut ancol etylic; thay th vic sn xut x phng
git t cht bo bng sn xut bt git tng hp.
- Sn xut glucoz t nhng cht thi nh v bo, mn ca, rm r,...
- Tng hp cht bo nhn to (b magarin) t axit stearic v glixerol, s chuyn ha du (cht bo lng)
thnh b, m (cht bo rn),....
- Ch bin protein t protein t nhin.
Cng vi ngnh cng ngh sinh hc, ha hc gp phn to nn nhng cht ha hc gip to nn nhng
ging mi c nng sut cao hn.
Ha hc gp phn to nn nhng thc phm ring dnh cho nhng ngi mc bnh khc nhau. Th d:
Thc phm dnh ho nhng ngi n king nh bnh, sa, ng,...
Ngnh ha thc phm cng ch bin c nhiu loi sn phm lm tng tnh thm m v hp dn ca
thc phm. Th d: Ch bin hp to nn v ngon v bo qun tt nhng thc phm cho con ngi;
Mt s loi hng liu, ph gia thc phm lm cho thc phm thm hp dn bi mu sc, mi thm
nhng vn m bo v sinh an ton thc phm. Hin nay sn xut c 200 cht ph gia cho thc
phm.
2 - HA HC V VN MAY MC
a. Vai tr ca may mc i vi i sng con ngi
- Cng vi nhu cu n, th may mc l mt trong nhng nhu cu thit yu ca con ngi gip nhn loi
tn ti v pht trin.
b. Vn may mc ang t ra cho nhn loi hin nay
- Dn s th gii gia tng khng ngng, v vy t si t nhin nh bng, gai,... khng th p ng nhu
cu may mc v s lng cng nh cht lng.
c. Ha hc gp phn gii quyt vn may mc ca nhn loi nh th no?
- Ha hc gp phn sn xut ra t, si ha hc tha mn nhu cu may mc cho nhn loi. T ha hc
(gm t nhn to v t tng hp) so vi t t nhin (si bng, si gai, si tm) c nhiu u im ni bt:
dai, n hi, t thm nc, mm mi, nh, xp, p v r tin,... Nguyn liu sn xut t nhn to l
nhng polime c sn trong t nhin nh xenluloz (c trong bng, gai, g, tre, na,...). T xenluloz, ch
bin bng con ng ha hc thu c t visco, t axetat.
- Nguyn liu sn xut t tng hp l nhng polime khng c sn trong t nhin m do con ngi tng
GV: Trn c Tun ( 01695 178 188) Trang: 64.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
hp bng phng php ha hc nh t nilon, t capron, t poliaxrylat,...
- Cc loi t si ha hc c tng hp hon ton trong nh my (t nguyn liu ban u n sn phm
cui cng) nn dnh ra c nhiu t ai cho trng trt v chn nui gia sc.
- Ha hc gp phn sn xut ra nhiu loi phm nhum to nn mu sc khc nhau ph hp vi nhu cu
thm m ca con ngi.
- Ngoi ra, cng ngh ha hc to ra cc vt liu c bn ch to cc thit b chuyn dng trong cc
nh my dt v trong ngnh dt may gip to ra nhng loi vi a dng, phong ph p ng nhu cu may
mc ngy cng cao.
3- HA HC V VN SC KHE CON NGI
bo v sc khe con ngi, phng chng bnh tt v cc t nn x hi, ha hc gp phn quan
trng trong lnh vc dc phm v vn v cht gy nghin ma ty.
a. Dc phm
- sinh tn v pht trin, t xa, con ngi bit dng c, cy, con,... trc tip hoc gin tip ch
bin lm thuc cha bnh.
- Tuy nhin, ngun dc phm t nhin khng th p ng cha tr nhng bnh him ngho, bnh do
virut,...
- Ha hc cng gp phn to ra nhng loi thuc c tr c tc dng tr bnh nhanh, mnh, hiu qu,...
nhng loi thuc b tng cng sc khe cho con ngi.
V thuc cha bnh:
- Ha hc gp phn nghin cu thnh phn ha hc ca mt s dc liu t nhin nh cy, con gip
pht hin c nhiu loi dc liu c ngun gc t nhin. T cc dc liu ban u chit sut c
nhng cht c khi lng v nng cao lm thuc cha bnh. Ngoi ra ngnh Ha Dc nghin
cu v sn xut ra nhiu loi thuc cha bnh cho con ngi t cc cht ha hc.
V thuc b dng c th
Cc loi vitamin ring l nh A, B, C, D,... cc loi thuc b tng hp,... c thnh phn chnh l cc cht
ha hc c tng hp bng con ng ha hc hoc c chit sut t dc liu t nhin gip
tng cng cc vitamin v mt s cht vi lng cho c th phng v chng bnh tt cho c th.
b. Cht gy nghin, cht ma ty v cch phng chng ma ty
- Ma ty gm nhng cht b cm dng nh thuc phin, cn sa, heroin, cocain, mt s thuc c dng
theo ch dn ca thy thuc nh moocphin, seduxen,...
- Ma ty cn c ch bin tinh vi di dng nhng vin thuc tn dc khng d g pht hin c.
- Ma ty c th di dng bt trng dng ht, vin nn ung v c bit di dng dung dch dng
tim chch trc tip vo mch mu.
Ma ty d dng no khi a vo c th con ngi u c th lm thay i mt hay nhiu chc nng
sinh l.
- Ma ty c tc dng c ch, gim au, kch thch mnh m gy o gic cho ngi dng. Nhiu t im
sn nhy dng loi ma ty tng hp cn gi l thuc lc lm ngi dng b kch thch dn n khng lm
ch c bn thn.
- Nghin ma ty s dn n ri lon tm, sinh l, nh ri lon tiu ha, ri lon chc nng thn kinh, ri
lon tun hon, h hp. Tim chch ma ty c th gy try tim mch d dn n t vong.
Hin nay, nn nghin ma ty ngy cng gia tng c bit trong gii tr.
- Ha hc nghin cu lm r thnh phn ha hc ca nhng cht ma ty t nhin, ma ty nhn to v
cc tc dng sinh l ca chng. T s dng chng nh mt loi thuc cha bnh hoc ngn chn tc
hi ca cc cht gy nghin,...
- Do , phng nga cht gy nghin ma ty, khng c dng thuc cha bnh qu liu ch nh ca
bc s, khng s dng thuc khi khng bit r tnh nng tc dng ca n v lun ni KHNG vi ma ty.
III. HA HC V MI TRNG
1 - NHIM MI TRNG
a. nhim mi trng
- nhim mi trng khng kh l hin tng lm cho khng kh sch thay i thnh phn, c nguy c
gy tc hi n thc vt, ng vt, sc khe con ngi v mi trng xung quanh.
- Khng kh sch thng gm 78% kh nit, 21% kh oxi v mt lng nh kh cacbonic v hi nc,...
GV: Trn c Tun ( 01695 178 188) Trang: 65.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
Khng kh b nhim thng c cha qu mc cho php nng cc kh CO2,CH4 v mt s kh c
khc, th d CO,NH3,SO2,HCl,... mt s vi khun gy bnh,...
b. nhim nc
- nhim nc l hin tng lm thay i thnh phn tnh cht ca nc gy bt li cho mi trng
nc, phn ln do cc hot ng khc nhau ca con ngi gy nn.
- Nc sch khng cha cc cht nhim bn, vi khun gy bnh v cc cht ha hc lm nh hng n
sc khe ca con ngi. Nc sch nht l nc ct trong thnh phn ch l H2O. Ngoi ra, nc sch
cn c quy nh v thnh phn gii hn ca mt s ion, mt s ion kim loi nng, mt s cht thi
nng di mc cho php ca T chc Y t th gii.
- Nc nhim thng c cha cc cht thi hu c, cc vi sinh vt gy bnh, cc cht dinh dng thc
vt, cc ha cht hu c tng hp, cc ha cht v c, cc cht phng x, cht c ha hc,...
c. nhim mi trng t
- nhim t l tt c cc hin tng, cc qu trnh lm nhim bn t, thay i tnh cht l, ha t nhin
ca t do cc tc nhn gy nhim, dn n lm gim ph ca t.
- t sch khng cha cc cht nhim bn, mt s cht ha hc, nu c ch t nng di mc quy
nh.
- t b nhim c cha mt s c t, cht c hi cho cy trng vt qu nng c quy nh.
* Sn xut ha hc l mt trong nhng ngun gy nhim mi trng do kh thi, cht thi rn, nc thi
c chc nhng cht c hi cho con ngi v sinh vt.
Tc hi ca mi trng b nhim (khng kh, t, nc) gy suy gim sc khe ca con ngi, gy thay
i kh hu ton cu, lm dit vong mt s loi sinh vt,... Th d nh hin tng thng tng ozon, hiu
ng nh knh, ma axit,... l hu qu ca nhim mi trng.
2 - HA HC V VN BO V MI TRNG TRONG I SNG SN XUT V HC
TP HA HC
nhim mi trng ang xy ra trn quy m ton cu, gy nh hng ln n cuc sng trn Tri t.
Hin tng tri t b nng ln do hiu ng nh knh, hin tng nhiu cht c hi c trong khng kh,
nc sng, bin, trong t,... lm cho mi trng ca hu ht cc nc b nhim. Do vn bo
v mi trng l vn chung ca ton nhn loi.
Ha hc c nhng ng gp g trong vn bo v mi trng sng ?
a. Nhn bit mi trng b nhim bng phng php ha hc
C th nhn thy c mi trng b nhim bng cch no ?
* Quan st
- Ta c th nhn thy mi trng b nhim qua mi, mu sc,...
- Cn c vo mi v tc dng sinh l c trng ca mt s kh ta d dng nhn ra khng kh b nhim.
* Xc nh cht nhim bng cc thuc kh
Th d: xc nh trong nc c cc cht v ion (gc axit hoc cc ion kim loi) ta cn c nhng thuc
th hoc n nhng ni c th xc nh c thnh phn ca nc, xc nh: Cc ion kim loi nng
(hm lng l bao nhiu?) ; Nng ca mt s ion Ca2+,Mg2+ gy nn cng ca nc; pH ca
nc.
* Xc nh bng cc dung c o
Th d: Dng nhit k xc nh nhit ca nc; dng sc k xc nh cc ion kim loi hoc cc
ion khc; dng my o pH xc nh pH ca t, nc,...
b. Vai tr ca Ha hc trong vic x l cht nhim
- X l cht nhim trong i sng, sn xut nng nghip v cng nghip nh th no?
- Nguyn tc chung ca vic x l cht nhim bng phng php ha hc l: C nhiu bin php x l
khc nhau cn c vo thc trng nhim, l x l nhim t, nc, khng kh da trn c s khoa
hc c kt hp vi khoa hc vt l v sinh hc.
- Phng php chung nht l loi b cht thi c hi bng cch s dng cht ha hc khc c phn ng
vi cht c hi, to thnh cht t c hi hn dng rn, kh hoc dung dch. Hoc c th c lp cht c
hi trong nhng dng c c bit, ngn chn khng cho cht c hi thm nhp vo mi trng t, nc,
khng kh gu nhim mi trng.
Sau y l mt s trng hp c th :
GV: Trn c Tun ( 01695 178 188) Trang: 66.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
+ X l nc thi
Khi pht hin nhim nhng ni c cht thi ca nh my, x nghip, cn c nhng xut c quan c
trch nhim x l.
+ X l kh thi
+ X l cht thi trong qu trnh hc tp ha hc
Vi mt s cht thi sau th nghim trn lp hoc sau bi thc hnh, ta c th thc hin theo cc bc
sau:
- Phn loi ha cht thi xem chng thuc loi no trong s cc cht hc.
- Cn c vo tnh cht ha hc ca mi cht x l cho ph hp.
Th d:
- Nu l cc cht c tnh axit th thng dng nc vi d trung ha.
- Nu l kh c c th dng cht hp th l than hot tnh hoc cht rn, hoc dung dch hp th
chng, to nn cht khng c hoc t c hi hn.
- Nu l cc ion kim loi, ion SO42- ..., c th dng nc vi d kt ta chng v thu gom li dng
rn v tip tc x l.
- Nu l ion cc kim loi qu th cn x l thu gom ti s dng.
CU HI
Cu 1.Cu 55-CD7-439: T l s ngi cht v bnh phi do ht thuc l gp hng chc ln s ngi
khng ht thuc l. Cht gy nghin v gy ung th c trong thuc l l
A. moocphin. B. cafein. C. aspirin. D. nicotin.
Cu 2.Cu 54-A8-329: Tc nhn ch yu gy ma axit l
A. SO2 v NO2. B. CH4 v NH3. C. CO v CH4. D. CO v CO2.
Cu 3.Cu 51-B8-371: Hi thu ngn rt c, bi vy khi lm v nhit k thu ngn th cht bt c
dng rc ln thu ngn ri gom li l
A. vi sng. B. mui n. C. lu hunh. D. ct.
Cu 4.Cu 59-A9-438: Dy gm cc cht v thuc u c th gy nghin cho con ngi l
A. ampixilin, erythromixin, cafein. B. penixilin, paradol, cocain.
C. cocain, seduxen, cafein. D. heroin, seduxen, erythromixin.
Cu 5.Cu 51-A10-684: Trong s cc ngun nng lng: 1 thy in, 2 gi, 3 mt tri, 4 ho thch; nhng
ngun nng lng sch l:
A. 2, 3, 4. B. 1, 2, 4. C. 1, 3, 4. D. 1, 2, 3.
Cu 6.Cu 44-B10-937: Cho mt s nhn nh v nguyn nhn gy nhim mi trng khng kh nh
sau:
1
Do hot ng ca ni la.
2
Do kh thi cng nghip, kh thi sinh hot.
3
Do kh thi t cc phng tin giao thng.
4
Do kh sinh ra t qu trnh quang hp ca cy xanh.
5
Do nng cao ca cc ion kim loi: Pb2+, Hg2+, Mn2+, Cu2+ trong cc ngun nc.
Nhng nhn nh ng l:
A. 2, 3, 5. B. 2, 3, 4. C. 1, 2, 3. D. 1, 2, 4.
Cu 7.Cu 59-B10-937: nh gi s nhim kim loi nng trong nc thi ca mt nh my, ngi ta
ly mt t nc, c c ri thm dung dch Na2S vo thy xut hin kt ta mu vng. Hin tng trn
chng t nc thi b nhim bi ion
A. Cd 2+. B. Fe2+ . C. Cu 2+. D. Pb2+.
Cu 8.Cu 59-CD11-259: Dn mu kh thi ca mt nh my qua dung dch Pb(NO3)2 d th thy
xut hin kt ta mu en. Hin tng chng t trong kh thi nh my c kh no sau y?
A. NH3. B. CO2. C. SO2. D. H2S.
Cu 9.Cu 47-A11-318: Nhm nhng cht kh (hoc hi) no di y u gy hiu ng nh knh khi
nng ca chng trong kh quyn vt qu tiu chun cho php?
A. CO2 v O2. B. CO2 v CH4. C. CH4 v H2O. D. N2 v CO.
Cu 10.Cu 56-A11-318: Khng kh trong phng th nghim b nhim bi kh clo. kh c, c th
GV: Trn c Tun ( 01695 178 188) Trang: 67.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
xt vo khng kh dung dch no sau y?
A. Dung dch NaOH. B. Dung dch NH3.
C. Dung dch NaCl. D. Dung dch H2SO4 long.
Cu 11.Cu 57-A12-296: Cho cc pht biu sau:
(a) Kh CO2 gy ra hin tng hiu ng nh knh.
(b) Kh SO2 gy ra hin tng ma axit.
(c) Khi c thi ra kh quyn, freon (ch yu l CFCl3 v CF2Cl2) ph hy tng ozon.
(d) Moocphin v cocain l cc cht ma ty.
S pht biu ng l
A. 3. B. 1. C. 2. D. 4.
Cu 12.Cu 52-A13-193: Cho cc pht biu sau:
(a) x l thy ngn ri vi, ngi ta c th dng bt lu hunh.
(b) Khi thot vo kh quyn, freon ph hy tng ozon.
(c) Trong kh quyn, nng CO2 vt qu tiu chun cho php gy ra hiu ng nh knh.
(d) Trong kh quyn, nng NO2 v SO2 vt qu tiu chun cho php gy ra hin tng ma axit.
Trong cc pht biu trn, s pht biu ng l
A. 2. B. 1. C. 4. D. 3.
P N THAM KHO
VN 1: CHT LNG TNH
Cu 1 2 3 4 5 6 7
A B A B B C A B
VN 2: MI TRNG CA DUNG DCH MUI
Cu 1 2 3 4 5
A D D D C D
VN 3: CC CHT PHN NG VI NC NHIT THNG
Cu 1 2 3
A D B A
VN 4: NC CNG
CU 1 2 3 4 5
A B B B C A
VN 5: N MN KIM LOI
CU 1 2 3 4 5 6 7 8 9 10 11 12 13
A C D B A B B C A D B D A B
VN 6: PHN NG NHIT PHN
CU 1 2 3 4
A C B A A
VN 7: PHN NG IN PHN
CU 1 2 3 4 5 6 7
A A D A D B A A
GV: Trn c Tun ( 01695 178 188) Trang: 68.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
VN 8: PHN NG NHIT LUYN
CU 1 2 3 4
A D D D D
VN 9: TNG HP TNH CHT CA MT S CHT V C THNG GP
CU 1 2 3 4 5 6 7 8
A D B D C C B D D
CU 9 10 11 12 13 14 15 16
A C D D D C C D B
VN 10: CC CHT CNG TN TI TRONG MT HN HP
CU 1 2 3 4 5
A A D C B A
VN 11: TNG HP CC HIN TNG PHN NG
CU 1 2 3 4 5 6 7 8 9 10 11 12
A C B C A B D A A A B D D
VN 12: D ON CC PHN NG V C
CU 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15
A C A C S C A C B D A B A B D A
CU 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30
A A A B C A C C A D B B B A C A
CU 31 32 33 34 35 36 37 38 39 40 41 42 43 44 45
A D A B D C B C B C B D A B D D
CU 46 47 48 49 50
A A D A B C
VN 13: LM KH KH
CU 1 2
A C B
VN 14: DY IN HA
CU 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16
A C A B B C A A A A A C C A B D C
VN 15: CHT OXI HA, CHT KH, S OXI HA, S KH
CU 1 2 3 4 5 6 7 8 9
A D B C A A C B C A
CU 10 11 12 13 14 15 16 17 18
A D A D D D A B C C
VN 16: HON THNH PHN NG OXI HA KH
CU 1 2 3 4 5 6 7 8 9 10 11 12
A A C D C D C C C B A B B
CU 13 14 15 16 17 18 19 20 21 22 23 24
A C C C C D C B B D A D B
VN 17: PHN LOI PHN NG HA HC
CU 1 2 3 4
GV: Trn c Tun ( 01695 178 188) Trang: 69.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
A A D A D
VN 18: QUNG V HP CHT THNG GP
CU 1 2 3 4 5 6 7 8 9 10
A D B B B B C C C B C
VN 19: V TR TRONG BNG TUN HON V CU HNH
CU 1 2 3 4 5 6 7
A D B C A D D B
VN 20: S BIN I CC I LNG CA BNG TUN HON
CU 1 2 3 4 5 6 7
A A D C D B C D
VN 21: LIN KT HA HC
CU 1 2 3 4 5 6 7 8 9 10
A A C D B C B D A A D
VN 22: CU HNH ELECTRON NGUYN T
CU 1 2 3 4 5 6 7 8 9
A A C A B A C B A D
VN 23: TC PHN NG CN BNG HA HC
CU 1 2 3 4 5 6 7 8 9 10 11 12 13 14
A A C B C A A A C D A B B C B
CU 15 16 17 18 19 20 21 22 23 24 25 26 27 28
A C C A C A A B C D A B B C A
VN 24: NHN BIT
CU 1 2 3 4 5 6 7 8 9 10
A D D C B B A D C B B
VN 25: IU CH
CU 1 2 3 4 5 6 7 8 9 10 11 12 13
A A D D B D C C C C A A A C
VN 26: TCH TINH CH
CU 1 2 3 4
A D C C D
VN 27: S V C
CU 1 2 3 4 5 6 7 8 9 10 11 12
A B D C C D C C B D C A D
VN 28: TNG HP CC PHT BIU TRONG HA V C
CU 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16
A C C A A D D C C B B C D D B A C
CU 17 18 19 20 21 22 23 24 25 26 27 28 29 30 31 32
A D A A C C A D D D A C D B D B D
CU 33 34 35 36 37 38 39 40 41 42 43 44 45 46
A C C D D B A B B D C D A B B
GV: Trn c Tun ( 01695 178 188) Trang: 70.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
VN 29: HA HC VI KINH T, X HI V MI TRNG
CU 1 2 3 4 5 6 7 8 9 10 11 12
A D A C C D C A D B B D C
GV: Trn c Tun ( 01695 178 188) Trang: 71.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
PH LC
TM TT HA HC V C
PHN 1.PHI KIM 3.Phng php in phn:
dpnc
A. HALOGEN ( NHM VII A ) 2NaCl 2Na + Cl2
I.Mt s tnh cht chung ca nhm 2NaCl+H2O dpdd
Cl2 +H2 + 2NaOH
mnx
FLO CLO BROM IOT
III. Axit Clohiric: L mt Axit mnh
1, K F Cl Br I 1.Tnh cht ha hc
hiu *Vi kim loi (trc Hiro) mui + H2
2, 19 35,5 80 127 2HCl + Zn ZnCl2 + H2
KLNT
3,in 9 17 35 53 *Vi Oxit Baz, baz mui + nc
tch Z 2HCl + CuO CuCl2 + H2O
4, Cu 2s22p5 3s23p5 4s24p5 5s25p5 2HCl + Cu(OH)2 CuCl2 + H2O
hnh e *Vi mui:
ho tr HCl + AgNO3 AgCl(trng) + HNO3
5, I2 Cl2 Br2 I2
CTPT
* HCl c c tnh kh khi tc dng vi cc
6, Kh, lc Kh, vng lng, rn, tm cht oxi ha mnh nh MnO2, KClO3,
Trng nht lc nu than KMnO4
thi MnO2 + 4HCl MnCl2 + Cl2 + 2H2O
mu 2KMnO4 + 16HCl 2KCl + 2MnCl2 + 5Cl2 +
7, -188 -34- +59 +185 8H2O
si
8, Axit Khng HClO HBrO HIO
Lu : - Tnh axit ca HCl < HBr < HI
c oxi HClO2 - - - Tnh kh ca HCl < HBr < HI
HClO3 HBrO3 HIO3 - HF l axit yu, c phn ng ring vi SiO2
HClO4 - HIO4 *c bit dng Axt HF v ln thu tinh
9, 4.0 3.0 2.8 2.6 4HF + SiO2 SiF4( tan) + H2O
m in
2.iu ch:
II. Tnh cht ha hc ca cc halogen *Tng hp:
1. Vi kim loi mui Halogenua H2 + X2 2HX
nX2 + 2M 2MXn *Dng H2SO4 c:
(n: S oxi ho cao nht ca M) H2SO4() + NaCl NaHSO4 + HCl
2Fe + 3Cl2 = 2FeCl3 H2SO4() + 2NaCl Na2SO4 + 2HCl
2.Vi hir Hiro halogenua Phng php ny ch dng iu ch c HF,
H2 + X2 2 HX ( cn xem k iu kin) HCl khng dng iu ch c HBr, HI v HBr,
Vi F2 phn ng xy ra ngay trong ti, to thp HI c tnh kh mnh s phn ng oxi ha kh vi
Cl2 phn ng khi c nh sng, Br2 phn ng khi un H2SO4 c.
nng, I2 phn ng nhit cao v l phn ng --------------o0o---------------
thun nghch. B. OXI-LU HUNH ( NHM VI A )
3.Vi H2O I.Mt s tnh cht
F2 + H2O 2HF + O2 OXI LU SELEN TELU
HX + HXO ( X: Cl,Br) HUNH
X2 + H2O 1.K hiu O S Se Te
- Nc Clo c tnh oxi ho mnh nn c dng 2.KLNT 16 32 79 127,6
st khun, ty ra 3.in 8 16 34 52
- I2 khng phn ng vi H2O tch Z
II. iu ch 4.Cu 2s22p4 3s23p4 4s24p4 5s25p4
hnh e ho
1,Dng HX tc dng vi cht oxi ha mnh: tr
to
HX+MnO2 MnX2 + X2 + 2H2O K2Cr2 O7 5.CTCT O2 S Se Te
+ 14HCl 2CrCl3 + 3Cl2 + 7H2O + 2KCl 2KMnO4 + 6.Trng Kh rnvng rn rn
16HCl 2KCl+2MnO2 + 5HCl +8H2 O thi
2,Dng hot ng: 7.Axit c - H2SO4 H2SeO4 H2 TeO4
Oxi - H2SO3 H2SeO3 H2 TeO3
Cl2 + 2 HBr Br2 + 2 HCl 8. n 3,5 2,5 2,4 2,1
Br2 + 2 NaI I2 + 2NaBr in
GV: Trn c Tun ( 01695 178 188) Trang: 72.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
tialuadien
II.OXI 3O2 2O3
1.Tnh cht ha hc V.Hirsunfua H2S1.Tnh cht vt l: Cht kh khng
* Vi H2 mu, mi trng thi, c, d tan trong nc axit
to sunfuahiric
2H2 + O2 2H2O
* Vi cc kim loi (tr Ag, Au, Pt) 2.Tnh cht ha hc
3Fe + 2O2 Fe3O4 *Vi nhit :
to
2Cu + O2 2CuO(en) H2S H2 + S
* Vi phi kim( tr F2,Cl2, Br2, I2) *Vi Oxi
3000o C to
N2 + O2 2NO 2H2S +3O 2SO2 + 2H2O ( d O2)
o o
t t
S + O2 SO2 2H2S + O2 2S + 2H2O ( thiu O2)
*Vi cht khc: *Tnh kh :
to to
CH4 + 2O2 CO2 + 2H2O H2S + Cl2 2HCl + S
o to
2CO + O2 t
2CO2 H2S + H2SO4() SO2 + 2H2O + S
4Fe3O4 + O2 to
6Fe2O3 3.iu ch:
to
2.iu ch: H2 + S H2S
a. Chng ct phn on khng kh lng. FeS + 2HCl H2S + FeCl2
b. Nhit phn cc mui giu oxi VI. Anhirit sunfur SO2: O=SO
2KClO3 to
2KCl + O2 1.Tnh cht vt l: Kh khng mu, mi hc tan
o
t
trong nc Axit sunfur
2KMnO4 K2MnO4 + MnO2 + O2 2.Tnh cht ha hc:
c. in phn H2O ( c pha H+ hoc OH- ) a. Tnh oxi ho:
dpddaxithoacbazo
H2O 2H2 + O2 SO2 + Mg to
2MgO + S
d. in phn oxit kim loi to
dpnc SO2 + H2 2H2O + S
2Al2O3 4Al + 3O2
to
III. Lu hunh SO2 + 2H2S 2H2O + 3S
1.Tnh cht ha hc: to thng lu hunh hot b. Tnh kh:
o
ng km. 2SO2 + O2
V2O5 ,450 C
2SO3
* Vi kim loi ( tr Au, Ag, Pt ) mui sunfua.
SO2 + 2H2O +Cl2 H2SO4 + 2HCl
to
Fe + S FeS(en) 5SO2 +2KMnO4 +2H2 O2MnSO4 +2KHSO4+H2
to SO4
Cu + S CuS (en)
* Vi Hir c. Tnh oxit axit:
S + H2 to SO2 + H2 O H2 SO3
H2S (mi trng thi)
3. iu ch:
* Vi phi kim ( tr N2,I2 ) sunfua to
to S + O2 SO2
C + 2S CS2
to
o
t 2H2SO4() + S 3SO2 + 2H2O
5S + 2P P2S5 o
t
*Vi axit c tnh oxi ha mnh 4FeS2 + 11O2 8SO2 + 2Fe2O3
to to
2H2SO4 + S 3SO2 + 2H2O Cu + 2H2SO4() CuSO4 + SO2 +
6HNO3 + S to 2H2O
H2SO4 + 6NO2+2H2O
VII.Axit sunfuric: H2SO4
2.iu ch:
1.Tnh cht vt l : H2SO4 khan l cht lng,
Khai thc t qung
khng mu, snh nh du, khng bay hi, khng
H2S + Cl2 2HCl + S mi v, tan tt, trong nc to nhiu nhit.
2H2S + SO2 2H2O + 3S 2.Tnh cht ha hc: L axit mnh
IV. OZN O3 *Lm qu tm
1.Tnh cht ha hoc: C tnh oxi ho mnh hn Oxi *Tc dng vi baz, oxit baz, vi mui.
O3 + 2 Ag Ag2O + O2 *Tc dng vi KL ng trc H, gii phng
2KI(trng)+ O3+ H2O 2KOH+I2(nu)+O2 H2.
( Nhn bit Ozn) a. H2SO4 m c :
2.iu ch:
GV: Trn c Tun ( 01695 178 188) Trang: 73.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
*B phn tch: ( khng gii thiu nguyn t BITMUT Bi)
to II.NIT: N
H2SO4 SO3 + H2O
*Ho nc: 1.Tnh cht ha hc:
C12H22O11 + H2SO4 C + H2SO4.nH2O
*C tnh oxi ho mnh: *Vi Oxi:
+Vi phi kim : C,S,P CO2, SO2, P2O5+Vi kim 3000 C o
N2 + O2 2NO
loi mui , khng gii phng kh hir.
*Vi H2:
Nhit thng: Khng phn ng vi Al,Fe,Cr. o
xt , t
N2 + 3H2 2NH3
un nng: Tc dng hu ht vi cc kim loi (tr
Au,Pt) *Vi kim loi in hnh ( hot ng mnh)
o o
t t
H2SO4() + Cu CuSO4 + SO2 + 2H2O N2 + 3Mg Mg3N2 (Maginitrua)
t
6 H2SO4()+2Al
o
Al2(SO)4+SO2+ 6H2O ( Mg3N2 + 6H2O 3Mg(OH)3 + NH3 )
Vi kim loi kh mnh ( Kim, kim th, Al,Zn) 2.iu ch: Chng ct phn on KK lng
o
t
c th cho SO2, S, H2S. NH4NO2 N2 + 2H2O
o o
t t
H2SO4() + 3Zn 3ZnSO4 + S + 4H2O 2NH4NO2 2N2 + O2 + 4H2O
o o
t t
H2SO4() + 4Zn 4ZnSO4+ H2S + 4H2O (NH4)Cr2O7 N2 + Cr2O3 + 4H2O
3.Sn xut H2SO4 III. Cc oxit ca oxi
*iu ch SO2: NO v NO2
t o CTPT NO NO2
4FeS2 + 11O2 8SO2 + 2Fe2 O3
Tnh Kh khng mu, c rt Kh nu, hc c tan nhiu
o
t
S + O2 SO2 cht t tan trong H2O trong H2O
*Oxi ho SO2 SO3: vt l
V O ,450o C Tnh Khng tc dng vi L Oxit axit
2SO2 + O2 2 5
2SO3
cht H2O *2NO2+H2O 2HNO3+NO
*To ra H2SO4 t SO3 :
Ho Axit, kim l oxit *4NO2+2H2O+O24HNO3
SO3 + H2O H2SO4
--------------o0o--------------- hc khng to mui *2NO2+ 2NaOH NaNO3
C. NIT- PHT PHO (NHM VA) + NaNO2 + H2O
I. Mt s tnh cht: iu 3000 C o
*Cu+4HNO3()Cu(NO3)2
NIT PHT ASEN STIBI
*N2+ O2
PHO ch + 2NO + 2H2O
1.K hiu N P As Sb 2NO
2.KLNT 14 31 75 122
*3Cu+8HNO3(l)
3.in 7 15 33 51
tch Z Cu(NO3)2+2NO+4H2O
4.Cu 2s22p4 3s23p4 4s24p4 5s25p4
hnh e ho N2 O 5
tr Rn trng tan nhiu trong H2O , to thng hoa 32,3oC
5.CTCT N2 P As Sb
6.Trng Kh Rn , rn rn * Tnh cht ha hc
thi khng trng
mu L oxit axit
7.Axit c HNO 3 H3PO4 H3AsO4 H3AsO4 *N2O5 + H2O 2HNO3
Oxi HNO 2
8. m 3,0 2,1 2,0 1,9 *N2O5 + 2NaOH 2NaNO3 + H2O
in
* iu ch
GV: Trn c Tun ( 01695 178 188) Trang: 74.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
P2O5 hutnuoc
2HNO3 N2O5 + H2O b.Tnh oxi ho mnh.
*Vi kim loi (tr Au,Pt) mui c s oxi ho
IV.Amoniac NH3
cao.
1.Tnh cht vt l: Kh khng mu, mi khai, xc,
HNO3() + M M(NO3)n + NO2 + H2O
tam tt trong nc.
HNO3(l) + M M(NO3)n + (c th :
2.Tnh cht ha hc:
t o NO,N2,N2O,NH4NO3) + H2O
* Hu: 2NH3 N2 + 3H2 V d:
*Vi axit: *4Mg + 10HNO3(l) 4Mg(NO3)2 + N2O +
NH3 + HCl NH4Cl 5H2O
*Vi H2O: *4Zn(NO3)2+10HNO3(l) 4Zn(NO3)2+NH4NO3
NH3 + H2O NH+4 + OH-
*Tnh kh: + 3H2O
o
t
4NH3 + 5O2 4NO + 6H2O HNO3(c,ngui) khng phn ng Al, Fe
o
t
2NH3 + 3Cl2 N2 + 6HCl *Ch : Au, Pt ch c th tan trong nc cng
o
t
2NH3 + 3CuO N2 + 3Cu + 3H2O toan (HCl + HNO3 )
3.iu ch: Au + 3HCl + HNO3 AuCl3 + NO+ 2H2O
o
t
*Dung dch NH3 NH3 *Vi phi kim:
o o
t t
*NH4Cl + NaOH NaCl + NH3 + H2O *4HNO3() + C CO2 + 4NO2 +2 H2O
o o
xt ,t * t
*N2 + H2 2NH3
6HNO3() + S H2SO4 +6NO2 + 2H2O
o
t
V.Dung dch NH3- Mui Amoni *4HNO3() + P H3PO4 +5NO2 + H2O
1.Dung dch NH3: Ho xanh qu tm. 3.iu ch:
o
t
*KNO3 + H2SO4() KHSO4 + HNO3
*Vi axit mui:
*NH3 NO NO2 HNO3
NH3 + H+ + SO2- + 2-
4 2NH4 + SO4 Pt ,t o
*4NH3 + 5O2 4NO + 6H2O
*Vi dung dch mui: 2NO + O2 2NO2
FeSO4+2NH3+ 2H2O Fe(OH)2 + (NH4)2SO4 3NO2 + H2O 2HNO3 + NO
*Lu : Vi cc dung dch mui cha Cu2+, Zn2+, Hoc: 4NO2 + O2 + H2O 4HNO3
Ag+ c th to phc cht, tan. VI.Mui NITRAT
CuCl2 + 2NH3 + 2H2O Cu(OH)2 + 2NH4Cl 1.Tnh cht ha hc:
Cu(OH)2 + 4NH3 [Cu(NH3)4] 2+ + OH- Tinh th khng mu d tan ( Phn m)
( Xanh thm) 2.Tnh cht ha hc:
2.Mui Amni: Nhit phn phn phn tch theio 3 kiu:
a.Tnh cht vt l: Tinh th, khng mu, v mn, a, M(NO3) t o
M(NO2)n + O2
d tan. M trc Mg
b Tnh cht ha hc: t o
b,M(NO3) M2On + NO2 + O2
*Tnh cht chung ca mui
t o Mg ( t Mg Cu)
*Hu: NH4Cl NH3 + HCl t o
o
c, M(NO3)n M + NO2+ O2
t
NH4NO3 N2O + 2H2O M ng sau Cu
VI. Axit NITRIC HNO3
1.Tnh cht vt l: L cht lng khng mu, mi VII. PHT PHO V HP CHT
1.Pht pho
hc, tan tt tos= 86oC v phn hu:
o
a. Tnh cht vt l:
t
4HNO3 2H2O + 4NO2 + O2
2.Tnh cht ha hc:
a.Tnh axit: ( nh axit thng thng)
GV: Trn c Tun ( 01695 178 188) Trang: 75.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
P (trng) P () D. CACBON - SILIC
-Rn, tinh th -Bt sm I.Mt s tnh cht
D=1,8; tonc=44oC tos = 281oC D= 2,3 NHM CACBON SILICGECM THIC CH
-khng tan trong H2O. Tan
VA ANI
trong CS2, C 2H2, te Khng tan trong H2O v
-Rt c, d gy bng nng. trong CS2 K hiu C Si Ge Sn Pb
V vy phi ht sc cn thn KLNT 12 28 72,6 118,7 207
khi dng P trng. Khng c inh 6 14 32 50 82
-Khng bn, t bc chy to tch Z
thng, lu, bin chm Cu 2s22p2 3s23p 4s24p2 5s25p2 6s26p2
hnh e
thnh . Bn to thng, bc ho tr
chy 240oC. >250oC Trng Rn rn rn rn rn
khng c kk P trng thi
m 2,5 1,8 1,8 1,8 1,8
b.Tnh cht ha hc: P (trng, ) in
*Vi cc cht oxi ho: *Cc bon c 3 dng th hnh; kim cng
4P(t) +3O2 2P2O5 + ln quang. ( rt cng), than ch ( dn in), Cc bon v nh
o
4P(t) + 5O2 t
2P2O5 + ln quang hnh ( than, m hng) c kh nng hp th tt.
2P(t) + 5Cl2 2PCl5 Mi pht hin gn y C60, dng tri bng( hnh
t o cu).
3P() + 5HNO3 + H2O 3H3PO4 + 5NO
- Silic c th dng tinh th (mu xm, dn,
*Vi cht kh: hot tnh thp) hay dng v nh hnh ( bt nu,
t o kh hot ng).
2P(t) + 3H2 2PH3 Phtphuahir
II.TNH CHT HA HC CA C V Si
(PH3 : Phtphin mi c thi rt c)
o
1.Vi n cht.
t
2P(t) +3Mg Mg3P2 *Kim loi ( nhit cao > tnngchy).
o
t to
2P(t) + 3Zn Zn3P2 ( thuc chut) Ca + 2C CaC2 (Canxicacbua)
o
Mui phtphua d b thu phn. 2Mg + Si t
Mg2Si ( Magi xilixua)
Zn3P2 +6H2O 3Zn(HO)3 + PH3 *Vi H:
c.iu ch: C + H2 to
CH4 (Mtan)
o
t
Ca3(PO4)2 +3SiO2+5C 3CaSiO3 +5CO2+P( hi) Si + H2 to
SiH4 ( Silan)
2.Hp cht ca P *Vi Oxi:
a.Anhirit photphoric P2O5: L cht bt trng, C + O2 to
CO2
khng mi, khng c, ht nc mnh o
t
C + CO2 2CO
*L Oxit axit: to
P2O5 + H2O 2HPO3 (Axitmetaphotphoric) Si + O2 SiO2
HPO4 + H2O H3PO4 (Axitphotphoric) *Vi nhau:
to
b.Axit photphoric H3PO4: Cht rn, khng mu, tan Si + C SiC
tt. 2.Vi hp cht:
*L mt axit trung bnh (3 ln axit) to 3 mui. V *Vi H2O:
to
d: NH4 + H3PO4 SP H2O + C CO + H2
o
t
NH4 H2PO4 : Amoni_ihirophtpht. hay 2H2O + C CO2 + 2H2
(NH4)2HPO4 : Amni_hirphtphat *Vi Axit:
(NH4)3PO4 : Amni_phtphat. C + 2H2SO4(cnng) CO2 + 2SO2+ 2H2O
Tu thuc vo t l mol cc cht tham gia p C + 4HNO3(cnng) CO2 + 4NO2 + H2O
Si khng tc dng vi Axit to thng.
*iu ch: Ca3(PO4)2 + 3H2SO4(c,d) 2H3PO4 *Vi baz: Ch Si tc dng.
to
+3CaSO4( t tan) Si + 2KOH + H2O K2SiO3 + H2
----------- *C l cht kh tng i mnh nhit
cao:
to
CO2 + C 2CO
GV: Trn c Tun ( 01695 178 188) Trang: 76.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
C + CuO t o
Cu + CO b phn hu khi un nng.
to
t
C + CaO
o
CaC2 + CO MgCO3 MgO + CO2
to *Mui cacbonat axit d b phn hu:
C + 4KNO3 CO2 + 2K2O + 4NO2 to
2NaHCO3 Na2CO3 + CO2 + H2O
III.HP CHT CA CACBON. *Trung ho axit:
III.1. Oxit: 2HCl + K2CO3 2KCl + H2O + CO2
1.Cacbonmonoxit CO: HCl + KHCO3 KCl + H2O + CO2
a,L cht kh mnh. *B thu phn to dung dch c tnh kim.
to
*CuO + CO Cu + CO2 Na2CO3 + H2O NaHCO3 + NaOH
o
t
* Fe2O3 + 3CO 2Fe + 3CO2 (qua 3 giai NaHCO3 + H2O NaOH + CO2 + H2O
on) Fe2O3Fe3O4FeOFe *Ch : NaHCO3 l mui tan, tan t hn
*CO + H2O + PdCl2 Pd + 2HCl + CO2 Na2CO3 v kt ta trong dung dch NH4Cl bo
(Dng Phn ng ny rt nhy, nhn bit CO, ho;
lm xanh thm dd PdCl2 ) NaCl + NH4HCO3 NaHCO3 + NH4Cl
to (Dung dchbo ho)
*CO + O2 2CO2 + 135Kcal
b.Phn ng kt hp: IV.HP CHT CA Si:
CO + Cl2 COCl2 ( phosgen) IV.1.Silicioxit SiO2 : Cht rn khng mu c
to trong thch anh, ct trng.
3CO +Cr Cr(CO)3 (Cacbonyl Crm)
*Khng tan, khng tc dng vi nc v axit
c.iu ch kh than:
( tr axit Flohiric).
*Kh than kh:
to
SiO2 + 4HF SiF4 + 2H2O
C + O2 CO2 H > 0 *Tc dng vi baz nhit cao.
to to
C + CO2 2CO H < 0 SiO2 + 2NaOH Na2SiO3 + H2O
*Kh than t: IV.2.Silan SiH4 : l kh khng bn, t bc
to
C + O2 CO2 + Q chy trong khng kh:
H2O + C
o
t
CO + H2 -Q SiH4 + O2 SiO2 + 2H2O
*c bit: IV.3.Axit silicic H2SiO3 v mui Silicat:
to
1,H2SiO3 l axit rt yu ( yu hn H2CO3), to
CO + NaOH HCOONa kt ta keo trong nc v b nhit phn:
III.2.Kh cacbonic CO2: o
t
*Kh khng mu, ho lng khi nn n 60atm, H2SiO3 SiO2 + H2O
lm lnh to tuyt cacbonic ( nc kh). 2.Mui Silicat:
*L oxt axit tc dng vi baz v oxit baz *Dung dch c ca Na2SiO3 hay K2SiO3 gi
CO2 + CaO CaCO3 l thu tinh lng, dng tm vo vi, g l
CO2 + Ca(OH)2 CaCO3 + H2O cho chng khng chy, dng ch to keo dn
2CO2 + Ca(OH)2 Ca(HCO3)2 thu tinh
*B nhit phn hu tocao
to
-----------
CO2 2CO + O2
*Tc dng vi cht kh mnh tocao:
to
CO2 + 2Mg 2MgO + C
to
CO2 + C 2CO
o
t
CO2 + H2 CO + H2O
III.3Axit cacbonic v mui cacbonat:
a,H2CO3 l axit yu, khng bn
( ch lm qu tn hi hng) ch tc dng vi baz
mnh.
b,Mui cacbonat (trung tnh v axit).
*Mui cacbonat trung ho ca kim loi kim u
bn vng vi nhit, cc mui cacbonat khc
GV: Trn c Tun ( 01695 178 188) Trang: 77.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
PHN 2. KIM LOI Liti Natri Kali Rubidi Cesi
A. I CNG V KIM LOI 1,K Li Na K Rb Cs
hiu
I.Cu to nguyn t.:
Cu (He)2s1 (ne)3s1 (Ar)4s1 (Kr)5s1 (Xe)6s1
*C t e lp ngoi cng ( n 3). hnh
*Bn knh nguyn t ln hn so vi phi kim cng e
chu k. m 1 0,9 0,8 0,8 0,7
*in tch ht nhn tng i ln cho nn kim loi in
BKNT 1,55 1,89 2,36 2,48 2,68
c tnh kh: M Mn+ + ne
(Ao)
II.Ho tnh:
2.Tnh cht ha hc: Tnh kh M M+ + 1e
1.Vi Oxi Oxit baz
K Ba Ca Na Mg Zn G Fe Ni Sn Pb Ag Pt Au
a.Vi phi kim: M + O2 M2O
(H) Cu Hg b.Vi H2O: 2M + H2O 2M(OH) + H2
-Phn ng mnh Phn ng khi nung Khng phn ng K Ba Ca Na Mg Al Mn Zn Cr
-t: chy sng t: khng chy Fe
phn ng C k Phn ng Phn ng
2.Vi Cl2: Tt c u tc dng MCln khng iu Phc tp to nhit cao
kin to Al(OH)3 ( 200--
3.Vi H2O 500O,
hyroxit v *100oC nn dng
kh H2 Mg(OH)2 li ngay. Hi nc)
Kim loi kim v Ca, Sr, Ba tc dng c vi H2 Coi To kim
nc nhit thng to dung dch kim + H2 khng loi Oxit v
4.Vi dung dch axit: * 200OC phn ng kh H2
a, M tr Pb + Axit thng thng mui + MgO + H2
H2.
b, M ( tr Au, Pt) + axit oxi ho mnh Mui, c.Vi axit: 2M + 2HCl 2MCl + 2H2
khng gi phng H2 . d.Vi dung dch mui:Tc dng vi nc trc.
5.Vi dung dch mui: Tr K, Na, Ca, Ba) cc 2M + H2O 2M(OH) + H2
kim loi ng trc y kim loi ng sau ra khi NaOH + CuSO4 Cu(OH)2+ Na2SO4
mui ca n. 3.iu ch:
dpnc
III.Dy in ho ca kim loi 2MCl 2M + Cl2
Tnh oxi ho tng 1
Li+ K+ Ba2+ Ca2+ Na+ Mg2+ Al3+ Mn2+ Zn+ Cr3+ Fe2+ Ni2+
2MOHpnc 2M + O2 + H2O (hi)
2
Li K Ba Ca Na Mg Al Mn Zn Cr Fe Ni 4.Mt s hp cht ca Natri.
Tnh kh gim a.Natrihiroxit NaOH: L Baz mnh.
Tnh oxi ho tng 2NaOH + CO2 Na2CO3 + H2O
Sn2+ Pb2+ H+ Cu2+ Hg2+ Ag+ Hg2+ Pt2+ Au3+
Sn Pb H Cu Hg Ag Hg Pt Au nNaOH : nCO2 2 : to mui trung tnh
Tnh kh gim nNaOH : nCO2 = 1:Mui Axit
*Da vo dy in ho xt chiu phn ng: NaOH + CO2 NaHCO3
*Cht oxi ho mnh nht s oxi ho cht kh mnh 1< nNaOH : nCO2 < 2: C 2 mui
nht, sinh ra cht oxi ho yu hn v cht kh yu
hn. *iu ch:
dpdd
Cu2+ + Zn Cu 2+ + Zn2+ 2NaCl + 2H2O
mnx
2NaOH + H2+Cl2
OXI KH KH OXI Na2CO3 + Ca(OH)2 2NaOH + CaCO3
mnh mnh yu yu b.Natrihircacbonat NaHCO3:
Ch : 2Fe3+ + Cu 2Fe2+ + Cu 2+ *Nhit phn:
to
2FeCl3 + Cu 2FeCl2 + CuCl2 2NaHCO3 Na2CO2 + CO2 + H2O
*Thu phn:
----------- NaHCO3 + H2O NaOH + H2CO3
B. KIM LOI KIM -KIM TH-NHM Lng tnh:
I.Kim loi kim (nhm IA) NaHCO3 + HCl NaCl + CO2 + H2O
1.Tnh cht vt l: NaHOC3 + NaOH Na2CO3 + H2O
GV: Trn c Tun ( 01695 178 188) Trang: 78.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
c.Natri cacbonat Na 2CO3 (x a).
*Thu phn: *Phn ng c bit:
Na2CO3 + H2O NaHCO3 + NaOH
o
t
CO2- + H2O HCO3 + OH - - CaO + 3C CaC2 + CO
3
*iu ch: Phng php Solvay. o
t
CO2 + H2O + NH3 NH4HCO3 *iu ch: CaCO3 CaO + CO2
NH4 HCO3 + NaCl NaHCO3 + NH4Cl
2NaHCO3 to
Na2CO3 + CO2 + H2O b.Canxihiroxit Ca(OH)2: ( Vi ti).
II.Kim loi nhm IIA ( kim th)
*Ca(OH)2 l cht rn mu trng, t tan.
1.Tnh cht vt l:
Beri Magi Canxi Stronti Bari
K hiu Be Mg Ca Ba
*Dung dch Ca(OH)2 gi l nc vi trong, tinh
Cu hinh (He)2s2 (ne)3s2 (Ar)4s2 (Kr)5s2 (Xe)6s2 baz yu hn NaOH.
e
m 1,5 1,2 1,0 1,0 0,9 *Phn ng c bit: iu ch Clorua vi.
in
2.Tnh cht ha hc: 2Ca(OH)2 + 2Cl2 CaCl2+ Ca(ClO)2 + 2H2O.
M M2+ + 2e ( kh mnh)
a.Vi oxi v cc phi kim: *iu ch:
2M + O2 2MO dpdd
CaCl2 + H2O
mn
H2 + Ca(OH)2 + 2H2O
M + H2 MH2 ( Hirua kim loi)
M + Cl2 MCl2
o
CaCl2 + 2NaOH Ca(OH)2 + 2NaCl
t
M + S MS
o
t
3M + N2 M 3 N2 CaO + H2O Ca(OH)2
o
t
3M + 2P M 3P2 c.Canxicacbonat CaCO3
b.Vi dung dch axit:
*Vi axit thng thng mui + H2 *Phn ng c bit:
*Vi HNO3,H2SO4() Mui khng gii phng H2.
(1)
c.Vi H2O ( tr Be) :
CaCO3+ H2O+ CO2 Ca(HCO3)2(tan)
(2)
Mg + H2O (hi) MgO + H2
Chiu (1) gii thch s xm thc ca nc ma.
M + 2H2O M(OH)2 + H2
Chiu (2) Gii thch s to thnh thch nh
d.Vi dung dch baz: Ch c Be tc dng to trong hang ng, cn vi trong m.
mui tan.
*iu ch:
Be + 2NaOH NaBeO2 (Natriberilat) + H2
Ca(OH)2 + CO2 CaCO3 + H2O
3.iu ch:
Ca(OH)2 + Ca(HCO3)2 2CaCO3 + 2H2O
MX2 dpnc
M + X2 5.Nc cng:
a.nh ngha:
4.Mt s hp cht ca Canxi Ca: Nc cng l nc cha nhiu ion Ca2+,Mg2+
*Nc cng tm thi: Cha Ca(HCO3)2,
a.Canxi oxit CaO: L oxit baz ( cn gi l vi Mg(HCO3)2.
sng). *Nc cng vnh cu: Cha MCl2, MSO4
( M : Ca, Mg).
* Nc cng ton phn: Cha c 2 loi trn.
GV: Trn c Tun ( 01695 178 188) Trang: 79.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
Cch lm mm nc cng: C. CRM -ST - NG
*Dng ho cht lm kt ta cc ion Ca2+, Mg2+, hoc I,Crm Cr:
un si. Cu hnh e: 1s22s22p63s23p63d44s2.
*Trao i ion: Dng nha ionit. 1.Tnh cht vt l: Trng bc, rt cng Sx thp
III, NHM. 2.Tnh cht ha hc:
Cr Cr2+ + 2e ( ho tr II)
1.Tnh cht ha hc: Kh mnh:
Cr Cr3+ + 3e (ho tr III)
Al Al3+ + 3e a.Vi oxi v Clo
a.Vi oxi v cc phi kim: to
t o 4Cr + 3O2 Cr2O3
4Al + O2 2Al2O3 to
o
2Cr + 3Cl2 3CrCl3
t
4Al + 3C Al4C3 b.Vi H2O:
o
t to
2Al + 3S Al2S3 2Cr + 3H2O Cr2O3 + H2
2Al + N2
o
t
2AlN c.Vi dung dch axit:
Cr + 2HCl CrCl2 + H2
b.Vi H2O :
4Cr + 12HCl + O2 4CrCl3+ 2H2O+ 4H2
2Al + 6H2O 2Al(OH)3 + 3H3 d.Vi dd Kim:
Phn ng dng li v to Al(OH)3 khng tan. Cr + NaOH + NaNO3 Na2CrO4 + 3NaNO2 +
c.Vi kim NatriAluminat. H2O
2Al + 2NaOH + 2H2O 2NaAlO2 + 3H2 3.Hp cht ca Crm:
Chnh xc hn: a.Crm (III) oxit Cr2O3:
2Al + 2NaOH + 6H2O 2Na[Al(OH)4] + 3H2 *L oxit lng tnh:
(Natritetrahirxaluminat) Cr2O3 + 6HCl 2CrCl2 + 3H2O
Cr2O3 + 2NaOH NaCrO2 + H2O
d.Vi dung dch axit: Nh cc kim loi khc.
*iu ch:
e.Vi oxit km hot ng- to
Phn ng nhit Nhm: (NH4)2Cr2O7 CrO3 + N2 + 4H2O
o
t
Fe2O3 + 2Al to
Al2O3 + Fe + Q Na2Cr2O7 + 2C Cr2O3 + Na2CO3 + CO
o
t
t o K2Cr2O7 + S Cr2O3 + K2SO4
Cr2O3 + 2Al Al2O3 + Cr
o
b.Crm (III) hiroxit Cr(OH)3 (xanh)
t
3CuO + 2Al Al2O3 + Cu *L hidroxit lng tnh:
2,iu ch: Cr(OH)3 + 3HCl CrCl3 + H2O
2Al2O3 dpnc
4Al + O2 Cr(OH)3 + NaOH NaCrO2 + 2H2O
3.Hp cht ca Nhm : *B oxi ho:
2NaCrO3+3Br2+8NaOH 2Na2CrO4+6NaBr
a.Nhm oxit Al2O3 : L hp cht lng tnh.
+4H2O
Al2O3 + 6HCl AlCl3 + 3H2O *B nhit phn:
Al2O3 + 2NaOH NaAlO2 + 2H2O to
2Cr(OH)3 Cr2O3 + H2O
HAlO2.H2O ( axit aluminic)
c.Crm (VI) oxit CrO3 ( rn, sm) rt c.
b. Nhm hidroxit Al(OH)3: l hp cht lng tnh *L oxit axit :
Al(OH)3 + 3HCl AlCl3 + 3H2O CrO3 + H2O H2CrO4 ( axit Crmic)
Al(OH)3 + NaOH NaAlO2 + 2H2O 2NaOH + CrO3 Na2CrO4 + H2O
- khng bn vi nhit *L cht oxi ho mnh:
to
2Al(OH)3 Al2O3 + 3H2O 4CrO3 to
2Cr2O3 + O2
c. Phn nhm d.Kali bi crmat K2Cr2O7( da cam)
- Phn chua c cng thc K2SO4.Al2(SO4)3.24H2O to
Nu thay ion K+ bng ion Li+, Na+, NH4+ ta c cc *4K2Cr2O7 4K2CrO4 + 2Cr2O3 + 3O2
mui kp khc c tn chung l phn nhm (khng K2Cr2O7 + 14HCl 2KCl +2CrCl3 +3Cl3 + 7H2O
gi l phn chua) II. ST 5626Fe
1s22s22p63s23p63d64s2
----------- 1.Tnh cht ha hc; Trng xm, do, nhim t.
2.Tnh cht ha hc:
a.Vi oxi v cc phi kim.
GV: Trn c Tun ( 01695 178 188) Trang: 80.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
o
3Fe +2O2 t
Fe3O4 *iu ch:
t o 3Fe2O3 + CO t 2Fe3O4 + CO2
2Fe + 3Cl2 2FeCl3 c.St (III) oxit Fe2O3 : Rn nu, khng tan.
o
t
Fe + S FeS *L oxit baz: Tc dng vi axit mui
2Fe + C t o
Fe3C ( xe men tit) st(III).
*B kh bi H2, CO, Al Fe
b.Vi H2O :
o o
*iu ch:
t 570 C
3Fe + 4H2O Fe3O4 + 4H2 2Fe(OH)3 t Fe2O3 + 3H2O
o o
t 570 C
Fe + H2O FeO + H2 d. St (II), (II) hiroxit
2Fe + 1,5O2 + nH2O Fe2O3.nH2O(d) Fe(OH)2 Fe(OH)3
Rn, trng xanh Rn nu
2Fe + 2O2 + nH2O Fe3O4.nH2O (thiu)
L nhng baz khng tan:
c.Vi dung dch axit:
4Fe(OH)2 + O2 + H2O 4Fe(OH)3
*Nh cc kim loi khc st (II) + H2 e.Mui St (II), (III)
*c bit: *Mui st (II) c tnh kh
Fe + 2HNO3 longlnh Fe(NO3)2 + H2 2FeCl2 + Cl2 2FeCl3
4Fe + 10HNO3 longlnh 4Fe(NO3)2 +N2O + 5H2O 3Fe(NO3)2+4HNO3 3Fe(NO3)3+NO + 2H2O
Fe + 4HNO3 longnng Fe(NO3)3 + NO + 2H2O FeSO4 + H2SO4 n Fe(SO4)3 + SO2 + 2H2O
8Fe + 30HNO3rtlong 8Fe(Fe(NO3)3 + 3NH4NO3 10FeSO4 + 2KMnO4 +8H2O 5Fe2(SO4) +
+ 9H2O K2SO4 + 2MnSO4 + 8H2O (dng phn ng ngy
t o nh lng st)
2Fe + H2SO4 Fe2(SO4)3 + 3SO2+ 6H2O 6FeSO4 + K2Cr2O7 + 7H2SO4 3Fe2(SO4)3 +
d.Vi mui: ( Mui kim loi yu hn) K2SO4 + Cr2(SO)3 + 7H2O
Fe + CuSO4 FeSO4 + Cu *Mui st III c tnh oxi ho:
3.iu ch: 2FeCl3 + Cu 2FeCl2 + CuCl2
dpdd
FeCl2 Fe + Cl2 2FeCl3 + 2KI 2FeCl2 + 2KCl + I2
dpdd 1 2FeCl3 + H2S 2FeCl2 + 2HCl + S
FeSO4 + H2O Fe+ O2 + H2SO4 5.Sn xut gang thp:
2
t o a.Cc phn ng xy ra trong l luyn gang ( l
FeSO4 + Mg Fe + MgSO4
o
cao).
t
FeO + H2 Fe + H2O *Than cc chy:
t o
to
Fe3O4 + 4CO Fe + 4CO2 C + O2 CO2 + Q
o
t
4.Hp cht ca St CO2 + C 2CO - Q
a.St (II) oxit FeO (rn en) khng tan *CO kh Fe2O3 Fe
*L oxit baz. to
3Fe2O3 + CO Fe3O4 + CO2
*B kh bi CO, H2, Al .. Fe Fe3O4 + CO t o
2FeO + CO2
*B Oxi ho: to
t o FeO + CO Fe + CO2
FeO + O2 2Fe2O3 *Sau :
3FeO + 10 HNO3 long 3Fe(NO3)3 + NO + 5H2O Fe + C to
Fe3C + CO2
*iu ch: t o
to
3Fe + 2CO Fe3C (Fe3C: xementit)
Fe3O4 + CO
FeO + CO2 *Ch : V trong nguyn liu c tp cht l oxit
o
t
Fe(CO2)2 FeO + CO2 + CO SiO2, MnO, P2O5.. nn:
to
b.St t oxit Fe2O3 ( hay FeO.Fe2O3) rn, en, SiO2 + C Si + 2CO
o
khng tan, nhim t. P2O5 + 5C t
2P + CO
*L oxit baz Nh vy St nng chy c ho tan mt lng nh
Fe3O4 + 8HCl FeCl2 + FeCl3 + 4H2O C, (< 4% ) Si, P ,S gi l gang.
*B kh bi: CO, H2, Al Fe *Cht chy tc dng vi cc tp cht qung)
*B oxi ho : ni ln trn mt gang nng chy.
to
3Fe3O4 + 28HNO3 9Fe(NO3)3 + NO+ 14H2O CaCO3 CaO + CO2
GV: Trn c Tun ( 01695 178 188) Trang: 81.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
o o
t t
CaO + SiO2 CaSiO3 *CuFeS2 + 2O2 + SiO2 Cu + FeSiO2 +
b.Cc phn ng xy ra trong l luyn thp. SO2
Oxi ho cc tp cht c trong gang ( C, Si, P, Mn) (Cancopirit)
o
t
Si + O2 SiO2 *Fe + CuSO4 Cu + FeSO4
dpdd
t
2Mn + O2 MnO2
o
*CuCl2 Cu + Cl2
t o 3.Hp cht ca ng:
C + O2 CO2
a.ng (I) oxit Cu2O ( mu )
Sau :
o
*Vi oxit axit:
t
2Fe + O2 FeO Cu2O + H2SO4 CuSO4 + Cu + H2O
t
FeO + SiO2 FeSiO2
o
X thp *Vi axit:
t o Cu2O + HCl 2CuCl2 + H2O
MnO + SiO2 MnSiO3 *Vi Cu2S:
P, S it b loi do phn ng: 2Cu2O + S 4Cu + SO2
o
t
S + O2 SO2 *iu ch:
to
4P + 5O2 t
P2 O5
o
4Cu + O2 2Cu2O
o
t
Do nn chn gang t S, P luyn thp. 4CuO 2Cu2O + O2
III, NG_6429Cu b. ng (I) clorua: CuCl rn trng, khng tan
1s 2s 2p 3s 3p63d104s1
2 2 6 2 *D phn hu:
to
1.Tnh cht ha hc: 2CuCl CuCl2 + Cu
Tnh kh yu: *D b oxi ho:
Cu Cu+ + 1e 4CuCl + O2 + 4HCl 4CuCl2 + 2H2O
*To phc vi dung dch NH3:
Cu Cu2+ + 2e
CuCl + 2NH3 [Cu(NH3)2] Cl
*Vi oxi:
to
c.ng (II) oxit CuO (rn, en, khng tan)
2Cu + O2 2CuO (en)
*B kh bi Al, H2, CO, C, NH3 to caoCu
1 to
3CuO + 2NH3 3Cu + N2 + 3H2O
2Cu + O2 Cu2O ( ) *L oxit baz ( Baz theo Bronsted)
2
CuO + 2H+ Cu 2+ + H2O
*Vi Clo:
*iu ch:
Cu + Cl2 CuCl2 ( mu hung) to
t o Cu(OH)2 CuO + H2O
Cu + CuCl2 2CuCl ( mu trng)
d.ng (II) hiroxit Cu(OH)2 mu xanh lam
*Vi S: to
o
t
*Km bn: CuO + H2O
Cu + S CuS(en) *L baz:
*Vi axit c tnh oxi ho mnh mui , khng c *To phc:
H2 Cu(OH)2 + 4NH3 [Cu(NH3)4](OH)2
o
Cu + 2H2SO4 () t
CuSO4 + SO2 + H2O Xanh m
4Cu + 10 HNO3(rt long) Cu(NO3)2 + N2O + e.Cc mui ng (II) u c, dung dch c
mu xanh lam ca Cu2+ b hirat ho
5H2O
[Cu(H2O)]2+ . Cho phn ng to phc
3Cu + 8HNO3( long) 3Cu(NO3)2 + 2NO+ 4H2O [Cu(H2O)]Cl2
o
t
Cu + 4HNO3(c) Cu(NO3)2 + NO2 + 2H2O -----------
*Vi dung dch mui:
Cu + Hg(NO3)2 Cu(NO3)2 + Hg D. CC KIM LOI KHC
119
2.iu ch: I. THIC 50 Sn:
t o *Sn l kim loi mu trng xm nh bc, rt mm,
2Cu + C 2Cu + CO2
o
c 2 dng th hnh: Thic trng v thic xm.
t
CuS + O2 2CuO + SO2 * Tnh cht ha hc
(Cancozin)
GV: Trn c Tun ( 01695 178 188) Trang: 82.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
Sn + O2 SnO2 AgCl + 2NH3 Ag(NH3)2Cl
Sn + 2S SnS2 AgBr + 2Na2S2O3 Na3[Ag(S2O3)]+NaBr
Sn + HCl SnCl2 + H2 Ag2S + 4KCN 2K[Ag(CN)2] + K2S
Sn + 4HCl + O2 SnCl4 + 2H2O IV.CH 207 82 Pb
Sn + 4HNO3 H2SnO3 + 4NO + H2O 1.Tnh cht ha hc:
( axit metastanics) to
Sn + 2KOH + 2H2O K2 [Sn(OH)4] +H2 2Pb + O2 PbO
to
(Sn + O2 + KOH K2SnO3 + H2O) Pb + S PbS
II, THU NGN Hg 3Pb + 8HNO3 3Pb(NO3)2+2NO+ 4H2O
1.Tnh cht ha hc: Pb + H2SO4(long) PbSO4 + H2
*Phn ng vi O2 khi ung nng: Pb + 2HCl PbCl2 + H2
2Hg + O2 2HgO ( Hai phn ng ny chm dn dn v dng li v
*Hg khng tc dng vi axit HCl,H2SO4(l) to cht khng tan)
*Vi HNO3: 2.Hp cht ca CH:
Hg + 4HNO3 Hg(NO3)2 + 2NO2 + 2H2O a.Ch (II) oxit PbO: Bt vng hay , khng tan
Vi H2SO4 : trong nc, tan trong axit , baz.
Hg + 2H2SO4 HgSO4 + SO2 + 2H2O PbO + 2HNO3 Pb(NO3)2 + H2O
Hg + HgCl2 Hg2Cl2 PbO + 2NOH + H2O Na2[Pb(OH)4]
2.iu ch: PbO(nngchy) + 2NaOH NaPbO2 + H2O
to
HgS + O2 Hg + SO2 b.Ch (II) hiroxit Pb(OH)2: Cht rn mu
3.Cc hp cht ca thu Ngn. trng, tan trong axit, baz.
* HgO: rn, mu hoc vng, khng tan khng Pb(OH)2 + 2HNO3 Pb(NO2)2 + 2H2O
tc dng vi H2O. Tan trong axit. Khi nng b phn Pb(OH)2 + 2NaOH Na[Pb(OH)4]
tch. o
t
Pb(OH)2 + 2NaOH Na2PbO2 + 2H2O
to
2HgO 2Hg + O2 c.Ch (IV) oxit PbO2: Cht mu da ln, kh tan
*Hg(OH)2: khng bn, rt d b phn hu: trong axit, tan trong baz kim.:
to
Hg(OH)2 HgO + H2O PbO2(nu si) + 2H2SO4() Pb(SO4)2 + 2H2O
*Mui sunfat, nitrat, clorua ca Hg2+ u tan PbO2 + 2NaOH + H2O Na2[Pb(OH)6](tan)
nhiu trong H2O PbO2 + 2NaOH(nngchy) Na2PbO3 + H2O
III. BC 108 47Ag PbO2 + 2MnSO4 + 3H2SO4 2HMnO4 +
*l kim loi mu trng bc, dn nhit dn nhit rt 5PbSO4 + 2H2O
tt.
1.Ho tnh: V.KM 65 30Zn
*Khng trc tip tc dng vi Oxi. I.Tnh cht ha hc: Kim loi hot ng kh
*Tc dng trc tip vi Halogen: mnh:
to
2Ag + Cl2 AgCl ( km bn) -Trong khng kh ph 1 lp ZnO mng.
Khng tc dng vi dung dch HCl, H2SO4(long) -t nng:
Ch tc dng vi H2SO4 (c), HNO3: to
2Zn( bt) + O2 2ZnO
to
2Ag + 2H2SO4 () Ag2SO4 + SO2 + H2O Zn + Cl2 ZnCl 2
to to
Ag + HNO3() AgNO3 + NO2+ H2O Zn + S ZnS
2.Hp cht ca Bc . to
Zn + H2O ZnO + H2
a.Bc oxit Ag2O: Rt t tan trong nc, tan tt Zn + 2HCl ZnCl2 + H2
trong dd NH3 Zn + H2SO4(l) ZnSO4 + H2
Ag2O 4NH3 + H2O 2 [Ag(NH3)2] OH to
3Zn + 4H2SO4 ZnSO4 + S + 4H2O
b.Mui Bc: AgF, AgNO3, AgClO3,AgClO4 : Tan
tt trong nc 4Zn + H2SO4() 4ZnSO4 + H2S+ 4H2O
Ag2SO4, CH3COOAg t tan. Zn + 2NaOH Na2ZnO2 + H2
AgCl, AgBr, AgI khng tan trong nc nhng ( Natri zincat)
tan trong ung dch NH3 v dung dch thiosunfat
Na2SiO3.
GV: Trn c Tun ( 01695 178 188) Trang: 83.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
2.iu ch: b.Mangan (IV) oxit MnO2 mu en:
1,Kh ZnO bi cacbon nhit cao: -L cht oxi ho mnh trong mi trng axit:
to 2FeSO4 + MnO2 +2H2SO4 Fe2(SO4)3 +
ZnO + C Zn + CO
2,in phn dung dch ZnSO4, Zn kim loi bn vo MnSO4 + 2H2O
Catot. MnO2 + 4HCl MnCl2 + Cl2 + 2H2O
3,Hp cht ca Zn: c.Anhdrit Pemanganic Mn2O7 : en lc (lng)
a. KmOxit ZnO: -L cht Oxi ho cc k mnh:
- Cht bt trng rt t tan trong H2O, kh bn vi Mn2O7 + H2O 2HMnO4 (axit Pemanganic)
nhit. d.Mangan (II) hidroxit Mn(OH)2:
-L Oxit lng tnh: -Vi oxit axit :
ZnO + 2HCl ZnCl2 + H2O Mn(OH)2 + SO3 MnSO4 + H2O
ZnO + NaOH NaZnO2 + H2O -Vi axit:
b.Km hiroxit Zn(OH)2 : Cht bt mu trng. Mn(OH)2 + H2SO4 MnSO4 + 2H2O
-L hiroxit lng tnh: -Vi oxi ca khng kh:
Zn(OH)2 + 2HCl ZnCl2 + H2O Mn(OH)2 + O2 + H2O 2Mn(OH)4
Zn + 2NaOH NaZnO2 + H2O Mangan (IV) hiroxit c mu nu.
-B nhit phn e.Mui clorua: MnCl2
to -Tinh th nht, tan trong nc.
Zn(OH)2 ZnO + H2O
-Vi baz kim:
-Vi dung dch NH3 : Zn(OH)2 tan do phn t NH3
MnCl2 + 2NaOH Mn(OH)2 + 2NaCl
kt hp bng lin kt cho nhn vi ion Zn2+ to ra
g.Mui Pemanganat: MnO4-: KMnO4
phc [Zn(NH3)4 ]2+: -Tinh th mu tm co anh kim.
Zn(OH)2 + 4NH3 [Zn(NH3)4]2+ + 2OH- -L cht Oxi ho mnh v ty theo mi trng m
c.Mui clorua: ZnCl2 : mc oxi ho khc nhau.
+Cht bt trng rt ho nc * Trong mi trng axit: Mn+7 Mn+2
+D tan trong nc.: 3K2SO4+2KMnO4+3H2SO4 3K2SO4 2MnSO4 +
ZnCl2 + 2NaOH(va) Zn(OH)2 + 2NaCl 3H2O
d.Mui phtphua ( P3-): * Trong mi trng trung : Mn+7 Mn+4 :
Zn3P2 (Km phtphua): 3K2SO4 +KMnO4 + H2O 3K2SO4 + MnO2 +
+Tinh th mu nu xm rt c 2KOH
+Vi axit * Trong mi trng kim: Mn+7 Mn+6:
Zn3P2 + 6HCl 3ZnCl2 + 3PH3 K2SO4+2KMnO4 +2KOH K2SO4 +2K2MnO4
2 2 6 2 6 5 2
IV.MANGAN 55 25Mn : 1s 2s 2p 3s 3p 3d 4s + H2O
1.Ho tnh: Kim loi hot ng mnh hn Zn nhng
km hn Al. -----------
-Trong khng kh : ph lp mng MnO2
to
-t nng : 2Mn + O2 2MnO
o
t
Mn + Cl2 MnCl2
o
t
Mn + S MnS
o
t
Mn + H2O Mn(OH)2 + H2
Mn + 2HCl MnCl2 + H2
Mn + H2SO4(long) MnSO4 + H2
Mn + H2SO4(c) MnSO4 + SO2 + 2H2O
3Mn + 8HNO3 3Mn(NO3)2 + NO + H2O
Mn + 4HNO3 Mn(NO3)2 + 2NO2 + 2H2O
2.iu ch: Phn ng nhit nhm:
to
3MnO + 2Al Al2O3 + 3Mn
3.Hp cht ca Mn.
a.Mangan (II) oxit MnO: Mu xanh lc.
-Vi axit: MnO + 2HCl MnCl2 + H2O
GV: Trn c Tun ( 01695 178 188) Trang: 84.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
PH LC 2: BNG TNH TAN CA MT S AXIT BAZ MUI
Cation
Anion + + + + 2+ + 2+ 2+ 2+
Li Na K NH4 Cu Ag Mg Ca Sr Ba2+ Zn 2+ Hg2+ Al3+ Sn2+ Pb2+ Bi3+ Cr3+ Mn2+ Fe3+ Fe 2+
Cl- T T T T T K T T T T T T T T I - T T T T
Br- T T T T T K T T T T T I T T I - T T T T
I- T T T T - K T T T T T K T T K - T K - T
NO3- T T T T T T T T T T T T T - T T T T T T
CH3COO- T T T T T T T T T T T T T - T - - T - T
S2- T T T T K K - T T T K K - K K K - K K K
SO3 2- T T T T K K K K K K K K - - K K - K - K
SO4 2- T T T T T I T K K K T - T T K - T T T T
CO32- I T T T - K K K K K K - - - K K - K - K
SiO32- T T T - - - K K K K K - K - K - - K K K
CrO42- T T T T K K T I I K K K - - K K T K - -
PO43- K T T T K K K K K K K K K K K K K K K K
OH- T T T T K - K I I T K - K K K K K K K K
Lu
T: cht d tan *Mui:
I : cht t tan ( tan nh hn 1g/100g nc) +Tt c mui Nitrat (NO3-) ,axetat (CH3COO-) v amoni
(NH4+) u tan
K: cht thc t khng tan ( tan nh hn 0,01 +Tt c mui clorua (Cl-) u tan.tr PbCl2 v AgCl
g/100g nc) +Tt c mui sunfat (SO4 2-) u tan tr PbSO4, BaSO4. Cn
- : cht khng tn ti hoc b nc thy phn lu CaSO4 t tan nhng vn xem nh tan tt
+Tt c mui Sunfua (S2-) u ko tan tr IA, Ca,Ba v NH4+
Tan +C 2 trng hp cn c bit ch :
*Bazo: bazo ca IA v Ca, Ba *Mui cacbonat:
*Axit: HCl ,H2SO 4,HNO3,H3PO 4, c bit l CH3COOH -CO3 2- : Ch tan nhm IA v NH4+
*Mui Photphat: Do ci ny phn li ba nc nn c ba loi
mui:
bit axit no mnh axit no yu cn nh -PO43- : Ch tan nhm IA v NH4+
- Axit ko c Oxi th HCl v HBr, HI mnh trong -HPO42- : Ch tan nhm IA v NH4+
HCl<HBr<HI , cn li yu ht -H2PO4- : Tt c u tan.
- Axit c Oxi th ly s nguyn t Oxi tr i s nguyn t H * Nhng mui ko tan to bi bazo v axit yu d b thy
trong phn t . Nu hiu ln hn bng 2 l axit mnh. Nh phn trong nc to bazo v axit ban u
hn l axit yu VD: Cho dd FeCl2 vo dd Na2CO3.
VD: H2SO4 C hiu s O v H l 2 mnh Hin tng
HClO4 c hiu s O v H l 3 mnh - Fe(OH)2 kt ta trng xanh
HClO c hiu s O v H l 0 yu - C kh CO2 bay ra
*Axit mnh ko ng ngha vi tnh OXH mnh Nguyn nhn : Ban u to FeCO3, nhng do ci ny l mui
to bi bazo v axit yu nn b thy phn trong nc to 2 th
trn
PH LC 3: DY IN HA
T tri sang phi, tnh oxi ha ca ion kim loi tng dn, tnh kh ca kim loi gim dn
GV: Trn c Tun ( 01695 178 188) Trang: 85.
TUYN TP NHNG VN L THUYT THNG GP TRONG HA V C
GV: Trn c Tun ( 01695 178 188) Trang: 86.
You might also like
- kĩ thuật khăn trải bànDocument6 pageskĩ thuật khăn trải bànLê Quang VũNo ratings yet
- Chuyen Lam Son Thanh Hoa Lan 2Document7 pagesChuyen Lam Son Thanh Hoa Lan 2Lê Quang VũNo ratings yet
- Kĩ Thuật Kwl Và KwlhDocument2 pagesKĩ Thuật Kwl Và KwlhLê Quang VũNo ratings yet
- De-Thi-Giua-Ki-2-Hoa-9-De-4 (1) 12421643Document6 pagesDe-Thi-Giua-Ki-2-Hoa-9-De-4 (1) 12421643Lê Quang VũNo ratings yet
- 62471.Phục lục 1 hoá học 8 cv 5512Document7 pages62471.Phục lục 1 hoá học 8 cv 5512Lê Quang VũNo ratings yet
- Trac Nghiem Ly Thuyet Su Dien Li Acid Va Base Hoa Lop 11 HayDocument4 pagesTrac Nghiem Ly Thuyet Su Dien Li Acid Va Base Hoa Lop 11 HayLê Quang VũNo ratings yet
- De Thi HK1 Hoa 11 So GD Bac Ninh 23 24Document3 pagesDe Thi HK1 Hoa 11 So GD Bac Ninh 23 24Lê Quang VũNo ratings yet
- De Cuong On Tap Giua HK2 Hoa 11 KNTT HayDocument5 pagesDe Cuong On Tap Giua HK2 Hoa 11 KNTT HayLê Quang VũNo ratings yet
- De Cuong On Thi TN (Co Giai)Document185 pagesDe Cuong On Thi TN (Co Giai)phatdangquang60% (5)
- 100 de Tu On Luyen Hoa Hoc THCSDocument172 pages100 de Tu On Luyen Hoa Hoc THCSLê Quang VũNo ratings yet