Professional Documents
Culture Documents
Chemistry: Viteee - 2018 - Sample Questions
Chemistry: Viteee - 2018 - Sample Questions
Uploaded by
Biswajeet ChakrabortyCopyright:
Available Formats
You might also like
- 12 - Chemistry QP (Set-Ii)Document6 pages12 - Chemistry QP (Set-Ii)Shravan ZoneNo ratings yet
- Chemistry: Viteee - 2017 - Sample QuestionsDocument1 pageChemistry: Viteee - 2017 - Sample QuestionsgunjaguptaNo ratings yet
- Chem Practice Paper 3 QPDocument10 pagesChem Practice Paper 3 QPSANAJ BSNo ratings yet
- QWQWDocument6 pagesQWQWmaduenofurtawzib.c6.1.33No ratings yet
- Section A: Assertion: When Nacl Is Added To Water A Depression in Freezing Point Is ObservedDocument4 pagesSection A: Assertion: When Nacl Is Added To Water A Depression in Freezing Point Is Observedastlenathan15No ratings yet
- SET 2 Question PaperDocument8 pagesSET 2 Question PaperKrityapriya BhaumikNo ratings yet
- Q.P. Chem P B 1 2022 Set - 1Document6 pagesQ.P. Chem P B 1 2022 Set - 1AKNo ratings yet
- 2nd Pre Board 2023Document8 pages2nd Pre Board 2023chiragNo ratings yet
- Chemistry ExamDocument8 pagesChemistry ExamAnubrata SarkarNo ratings yet
- Xii-Cbse-Chemistrypreboardexam (26 02 2024)Document6 pagesXii-Cbse-Chemistrypreboardexam (26 02 2024)janakansenthil2010No ratings yet
- ChemistryDocument7 pagesChemistryrjakrithiNo ratings yet
- Chem Practice Paper 2 QPDocument10 pagesChem Practice Paper 2 QPSANAJ BSNo ratings yet
- Xii Chemistry - 1Document10 pagesXii Chemistry - 1M A T T H Y D E NNo ratings yet
- Class Xii Chemistry Mcqs and Assertion Reason Questions Feb 24Document43 pagesClass Xii Chemistry Mcqs and Assertion Reason Questions Feb 24Soumya PNo ratings yet
- Grade12Pre Boardexamination QPChemistryQPSET1Document7 pagesGrade12Pre Boardexamination QPChemistryQPSET1BigsmokeNo ratings yet
- Hydrocarbon 1Document6 pagesHydrocarbon 1VK CREATIONNo ratings yet
- QP 2479Document7 pagesQP 2479yashojayoneplusNo ratings yet
- Prepared by v. Aditya VardhanDocument6 pagesPrepared by v. Aditya Vardhankrishna kanthNo ratings yet
- Chemistry QP - PB XiiDocument5 pagesChemistry QP - PB XiiSnehaNo ratings yet
- KV Chan Set 4 QPDocument6 pagesKV Chan Set 4 QPdeepaNo ratings yet
- Chemistry Model Paper 5 (23-24) (2003 VERSION)Document4 pagesChemistry Model Paper 5 (23-24) (2003 VERSION)qzglsefafNo ratings yet
- 12 - Chemistry QP (Set-I)Document6 pages12 - Chemistry QP (Set-I)Shravan ZoneNo ratings yet
- Chemistry 1997 New PDFDocument6 pagesChemistry 1997 New PDFDeepak TholiaNo ratings yet
- CCC 2014 Solution EnglishDocument4 pagesCCC 2014 Solution EnglishXuNo ratings yet
- XIIth ChemistryDocument7 pagesXIIth ChemistryRiya MalikNo ratings yet
- Chemistry Model-2, Nov-2021Document14 pagesChemistry Model-2, Nov-2021Elsa HarryNo ratings yet
- Nta Abhyas Test-65 CDocument5 pagesNta Abhyas Test-65 CMIITY EDUNo ratings yet
- Chem 001Document22 pagesChem 001Yashveer RaiNo ratings yet
- Xii PB 1 2021 Aashima Set 1Document16 pagesXii PB 1 2021 Aashima Set 1HpNo ratings yet
- Sample Paper +2 Chemistry 18.01.23Document3 pagesSample Paper +2 Chemistry 18.01.23Raj Online ServicesNo ratings yet
- Xii Chem QPDocument6 pagesXii Chem QPnrusinghsamal2006No ratings yet
- Unit 1 Test 2 300920Document11 pagesUnit 1 Test 2 300920ibrahim ahmedNo ratings yet
- Set IDocument8 pagesSet IMagathi NarayananNo ratings yet
- MOCK Examination-XII Chemistry SET-IDocument11 pagesMOCK Examination-XII Chemistry SET-IsabatnirupamaNo ratings yet
- T3 Hs Ob MJVN DY4 Ru 2 NSIcDocument23 pagesT3 Hs Ob MJVN DY4 Ru 2 NSIcYashveer RaiNo ratings yet
- CCC 2014 PtA Answers ENDocument4 pagesCCC 2014 PtA Answers ENFahmi XiomiNo ratings yet
- Matrix Science Academy: Chemistry MHT CET L2 2022-23Document4 pagesMatrix Science Academy: Chemistry MHT CET L2 2022-23Light MayNo ratings yet
- PB Xii Chem 2023-24Document6 pagesPB Xii Chem 2023-24NehaNo ratings yet
- 12 Chemistry23 24 sp10Document14 pages12 Chemistry23 24 sp10Babur HussainNo ratings yet
- Kcet Mock Test Chemistry 1Document8 pagesKcet Mock Test Chemistry 1VikashNo ratings yet
- CHEMISTRY NewDocument11 pagesCHEMISTRY NewArnav KumarNo ratings yet
- Unit Test Sample Paper Grade 12 ChemistryDocument6 pagesUnit Test Sample Paper Grade 12 Chemistrymilonee lNo ratings yet
- Chem 1Document5 pagesChem 1kakajumaNo ratings yet
- Xii MotivationalDocument5 pagesXii MotivationalroobanNo ratings yet
- Common Chem 1Document11 pagesCommon Chem 1Fuafung Caleb YenwoluaNo ratings yet
- CHE102FF03PDocument5 pagesCHE102FF03PDhrumilParikhNo ratings yet
- Class Xii Pre Board Question Paper ChemistryDocument17 pagesClass Xii Pre Board Question Paper ChemistryJeremiah ShibuNo ratings yet
- Chemistry English Medium 11th Model Question Paper WWW tn11th inDocument5 pagesChemistry English Medium 11th Model Question Paper WWW tn11th inGRagaNo ratings yet
- Chem100 Midterm2021Document7 pagesChem100 Midterm2021atomtostNo ratings yet
- Some Basic Concept of Chemistry: Chapter - 01Document7 pagesSome Basic Concept of Chemistry: Chapter - 01MidhunNo ratings yet
- 12 Chemistry Q.p.set-3Document8 pages12 Chemistry Q.p.set-3Samarth ShrivastavaNo ratings yet
- Chemistry Practice Question Paper Class 12Document7 pagesChemistry Practice Question Paper Class 12tony starkNo ratings yet
- 2750IIT JEE Previous Years Question papersIITchemistryDocument6 pages2750IIT JEE Previous Years Question papersIITchemistrychandan yadavNo ratings yet
- Paper 2Document5 pagesPaper 2Tbsbi P.No ratings yet
- Kendriya Vidyalaya Sanghthan, Ahmedabad Region SAMPLE PAPER (2022-23) Chemistry Theory (043) MM:70 Time: 3 HoursDocument8 pagesKendriya Vidyalaya Sanghthan, Ahmedabad Region SAMPLE PAPER (2022-23) Chemistry Theory (043) MM:70 Time: 3 Hoursharsh.mahori09No ratings yet
- Practice Makes Perfect in Chemistry: Oxidation-ReductionFrom EverandPractice Makes Perfect in Chemistry: Oxidation-ReductionRating: 5 out of 5 stars5/5 (1)
Chemistry: Viteee - 2018 - Sample Questions
Chemistry: Viteee - 2018 - Sample Questions
Uploaded by
Biswajeet ChakrabortyOriginal Description:
Original Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Chemistry: Viteee - 2018 - Sample Questions
Chemistry: Viteee - 2018 - Sample Questions
Uploaded by
Biswajeet ChakrabortyCopyright:
Available Formats
VITEEE - 2018 - SAMPLE QUESTIONS
CHEMISTRY
1. The nucleus of an element contains 11 protons. Its valency would be
A) 0 B) 1 C) 2 D) 3
2. Identify the lanthanide which is obtained only by synthesis.
A) Lu B) Pm C) Pr D) Gd
3. Which one of the following compounds shows Frenkel defect?
A) ZnS B) CsCl C) FeO D) NaCl
4. A cylinder of cooking gas supplied by Indian Oil Corporation is assumed to contain 14 kg of butane (∆Hc for C4H10 = - 2600 kJ
mol-1). If a small family of three persons, requires 10,000 J of heat energy per day for cooking, the gas in the cylinder would
last for
A) 44 days B) 54 days C) 72 days D) 63 days
5. The molar conductivities of infinite dilution for sodium iodide, sodium acetate and aluminium acetate are 12.69, 9.10 and 24.52
S cm2 mol−1 respectively at 25 °C. What is the molar conductivity of AlI 3 at infinite dilution?
A) 35 S cm2 mol−1 B) 32 S cm2 mol−1 C) 28 S cm2 mol−1 D) 40 S cm2 mol−1
6. The number of structural isomers for the alcohols with the formula C 5H11OH is
A) 4 B) 6 C) 8 D) 10
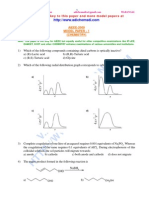
7. The best reagent to convert pent-3-en-2-ol into pent-3-en-2-one is
A) pyridinium chloro-chromate B) acidic dichromate C) acidic permanganate D) chromic anhydride in glacial acetic acid
8. The correct order of reactivity of PhMgBr with
A) I > II > III B) III > II > I C) II > III > I D) I > III > II
9. The product Z in the following reaction sequence is
A) CH3CN B) CH3OH C) CH3CONH2 D) CH3CH2OH
10. An unknown amine is treated with an excess of methyl iodide. Two equivalents of methyl iodide react with the amine.
The amine is treated with silver oxide and water, and then heated to 120 °C. The resulting products are trimethylamine and
ethylene. The unknown amine is
A) CH3CH2NHCH3 B) CH3CH2NH2 C) CH2 = CHNH2 D) CH2 = CHNHCH3
You might also like
- 12 - Chemistry QP (Set-Ii)Document6 pages12 - Chemistry QP (Set-Ii)Shravan ZoneNo ratings yet
- Chemistry: Viteee - 2017 - Sample QuestionsDocument1 pageChemistry: Viteee - 2017 - Sample QuestionsgunjaguptaNo ratings yet
- Chem Practice Paper 3 QPDocument10 pagesChem Practice Paper 3 QPSANAJ BSNo ratings yet
- QWQWDocument6 pagesQWQWmaduenofurtawzib.c6.1.33No ratings yet
- Section A: Assertion: When Nacl Is Added To Water A Depression in Freezing Point Is ObservedDocument4 pagesSection A: Assertion: When Nacl Is Added To Water A Depression in Freezing Point Is Observedastlenathan15No ratings yet
- SET 2 Question PaperDocument8 pagesSET 2 Question PaperKrityapriya BhaumikNo ratings yet
- Q.P. Chem P B 1 2022 Set - 1Document6 pagesQ.P. Chem P B 1 2022 Set - 1AKNo ratings yet
- 2nd Pre Board 2023Document8 pages2nd Pre Board 2023chiragNo ratings yet
- Chemistry ExamDocument8 pagesChemistry ExamAnubrata SarkarNo ratings yet
- Xii-Cbse-Chemistrypreboardexam (26 02 2024)Document6 pagesXii-Cbse-Chemistrypreboardexam (26 02 2024)janakansenthil2010No ratings yet
- ChemistryDocument7 pagesChemistryrjakrithiNo ratings yet
- Chem Practice Paper 2 QPDocument10 pagesChem Practice Paper 2 QPSANAJ BSNo ratings yet
- Xii Chemistry - 1Document10 pagesXii Chemistry - 1M A T T H Y D E NNo ratings yet
- Class Xii Chemistry Mcqs and Assertion Reason Questions Feb 24Document43 pagesClass Xii Chemistry Mcqs and Assertion Reason Questions Feb 24Soumya PNo ratings yet
- Grade12Pre Boardexamination QPChemistryQPSET1Document7 pagesGrade12Pre Boardexamination QPChemistryQPSET1BigsmokeNo ratings yet
- Hydrocarbon 1Document6 pagesHydrocarbon 1VK CREATIONNo ratings yet
- QP 2479Document7 pagesQP 2479yashojayoneplusNo ratings yet
- Prepared by v. Aditya VardhanDocument6 pagesPrepared by v. Aditya Vardhankrishna kanthNo ratings yet
- Chemistry QP - PB XiiDocument5 pagesChemistry QP - PB XiiSnehaNo ratings yet
- KV Chan Set 4 QPDocument6 pagesKV Chan Set 4 QPdeepaNo ratings yet
- Chemistry Model Paper 5 (23-24) (2003 VERSION)Document4 pagesChemistry Model Paper 5 (23-24) (2003 VERSION)qzglsefafNo ratings yet
- 12 - Chemistry QP (Set-I)Document6 pages12 - Chemistry QP (Set-I)Shravan ZoneNo ratings yet
- Chemistry 1997 New PDFDocument6 pagesChemistry 1997 New PDFDeepak TholiaNo ratings yet
- CCC 2014 Solution EnglishDocument4 pagesCCC 2014 Solution EnglishXuNo ratings yet
- XIIth ChemistryDocument7 pagesXIIth ChemistryRiya MalikNo ratings yet
- Chemistry Model-2, Nov-2021Document14 pagesChemistry Model-2, Nov-2021Elsa HarryNo ratings yet
- Nta Abhyas Test-65 CDocument5 pagesNta Abhyas Test-65 CMIITY EDUNo ratings yet
- Chem 001Document22 pagesChem 001Yashveer RaiNo ratings yet
- Xii PB 1 2021 Aashima Set 1Document16 pagesXii PB 1 2021 Aashima Set 1HpNo ratings yet
- Sample Paper +2 Chemistry 18.01.23Document3 pagesSample Paper +2 Chemistry 18.01.23Raj Online ServicesNo ratings yet
- Xii Chem QPDocument6 pagesXii Chem QPnrusinghsamal2006No ratings yet
- Unit 1 Test 2 300920Document11 pagesUnit 1 Test 2 300920ibrahim ahmedNo ratings yet
- Set IDocument8 pagesSet IMagathi NarayananNo ratings yet
- MOCK Examination-XII Chemistry SET-IDocument11 pagesMOCK Examination-XII Chemistry SET-IsabatnirupamaNo ratings yet
- T3 Hs Ob MJVN DY4 Ru 2 NSIcDocument23 pagesT3 Hs Ob MJVN DY4 Ru 2 NSIcYashveer RaiNo ratings yet
- CCC 2014 PtA Answers ENDocument4 pagesCCC 2014 PtA Answers ENFahmi XiomiNo ratings yet
- Matrix Science Academy: Chemistry MHT CET L2 2022-23Document4 pagesMatrix Science Academy: Chemistry MHT CET L2 2022-23Light MayNo ratings yet
- PB Xii Chem 2023-24Document6 pagesPB Xii Chem 2023-24NehaNo ratings yet
- 12 Chemistry23 24 sp10Document14 pages12 Chemistry23 24 sp10Babur HussainNo ratings yet
- Kcet Mock Test Chemistry 1Document8 pagesKcet Mock Test Chemistry 1VikashNo ratings yet
- CHEMISTRY NewDocument11 pagesCHEMISTRY NewArnav KumarNo ratings yet
- Unit Test Sample Paper Grade 12 ChemistryDocument6 pagesUnit Test Sample Paper Grade 12 Chemistrymilonee lNo ratings yet
- Chem 1Document5 pagesChem 1kakajumaNo ratings yet
- Xii MotivationalDocument5 pagesXii MotivationalroobanNo ratings yet
- Common Chem 1Document11 pagesCommon Chem 1Fuafung Caleb YenwoluaNo ratings yet
- CHE102FF03PDocument5 pagesCHE102FF03PDhrumilParikhNo ratings yet
- Class Xii Pre Board Question Paper ChemistryDocument17 pagesClass Xii Pre Board Question Paper ChemistryJeremiah ShibuNo ratings yet
- Chemistry English Medium 11th Model Question Paper WWW tn11th inDocument5 pagesChemistry English Medium 11th Model Question Paper WWW tn11th inGRagaNo ratings yet
- Chem100 Midterm2021Document7 pagesChem100 Midterm2021atomtostNo ratings yet
- Some Basic Concept of Chemistry: Chapter - 01Document7 pagesSome Basic Concept of Chemistry: Chapter - 01MidhunNo ratings yet
- 12 Chemistry Q.p.set-3Document8 pages12 Chemistry Q.p.set-3Samarth ShrivastavaNo ratings yet
- Chemistry Practice Question Paper Class 12Document7 pagesChemistry Practice Question Paper Class 12tony starkNo ratings yet
- 2750IIT JEE Previous Years Question papersIITchemistryDocument6 pages2750IIT JEE Previous Years Question papersIITchemistrychandan yadavNo ratings yet
- Paper 2Document5 pagesPaper 2Tbsbi P.No ratings yet
- Kendriya Vidyalaya Sanghthan, Ahmedabad Region SAMPLE PAPER (2022-23) Chemistry Theory (043) MM:70 Time: 3 HoursDocument8 pagesKendriya Vidyalaya Sanghthan, Ahmedabad Region SAMPLE PAPER (2022-23) Chemistry Theory (043) MM:70 Time: 3 Hoursharsh.mahori09No ratings yet
- Practice Makes Perfect in Chemistry: Oxidation-ReductionFrom EverandPractice Makes Perfect in Chemistry: Oxidation-ReductionRating: 5 out of 5 stars5/5 (1)