Professional Documents
Culture Documents
Tea and Bone Health Steps Forward in Translational Nutrition
Tea and Bone Health Steps Forward in Translational Nutrition
Uploaded by
LandroCopyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Tea and Bone Health Steps Forward in Translational Nutrition
Tea and Bone Health Steps Forward in Translational Nutrition
Uploaded by
LandroCopyright:
Available Formats
Tea and bone health: steps forward in translational nutrition1–5
Chwan-Li Shen, Ming-Chien Chyu, and Jia-Sheng Wang
ABSTRACT of osteoporosis and related fractures can be reduced by modu-
Osteoporosis is a major health problem in the aging population world- lating these modifiable factors. Nutrition influences the risk of
wide. Cross-sectional and retrospective evidence indicates that tea osteoporosis and fractures through improving bone mineral
consumption may be a promising approach in mitigating bone loss density (BMD)6, microarchitecture, and strength (5). In the past
and in reducing risk of osteoporotic fractures among older adults. Tea decade, the awareness of complementary and alternative thera-
polyphenols enhance osteoblastogenesis and suppress osteoclasto- pies for the prevention and/or treatment of osteopenia and os-
genesis in vitro. Animal studies reveal that intake of tea polyphenols teoporosis, such as dietary supplements or functional foods, has
have pronounced positive effects on bone as shown by higher bone been increasing among consumers (6).
mass and trabecular bone volume, number, and thickness and lower The bone remodeling process involves both bone resorption
trabecular separation via increasing bone formation and inhibition of (osteoclastogenesis) and bone formation (osteoblastogenesis) (7).
bone resorption, resulting in greater bone strength. These osteoprotec- Bone is resorbed by osteoclast cells and then new bone is formed
tive effects appear to be mediated through antioxidant or antiinflam- by osteoblast cells. During bone remodeling, excessive osteo-
matory pathways along with their downstream signaling mechanisms. clastic activity and inadequate osteoblastic activity are the main
A short-term clinical trial of green tea polyphenols has translated the reasons for osteoporosis development.
findings from ovariectomized animals to postmenopausal osteopenic
women through evaluation of bioavailability, safety, bone turnover
markers, muscle strength, and quality of life. For future studies, pre-
clinical animal studies to optimize the dose of tea polyphenols for 1
From the Department of Pathology (C-LS and M-CC) and the Laura W
maximum osteoprotective efficacy and a follow-up short-term dose- Bush Institute for Women’s Health (C-LS), Texas Tech University Health
response trial in postmenopausal osteopenic women are necessary Sciences Center, Lubbock, TX; the Department of Mechanical Engineering
to inform the design of randomized controlled studies in at-risk pop- and the Graduate Healthcare Engineering Option, Texas Tech University,
ulations. Advanced imaging technology should also contribute to de- Lubbock, TX (M-CC); and the Department of Environmental Health Sci-
termining the effective dose of tea polyphenols in achieving better ence, University of Georgia, Athens, GA (J-SW).
2
bone mass, microarchitecture integrity, and bone strength, which Presented at the conference “Fifth International Scientific Symposium on
Tea and Human Health,” held at the US Department of Agriculture, Wash-
are critical steps for translating the putative benefit of tea consump-
ington, DC, 19 September 2012. The conference was organized by Jeffrey
tion in osteoporosis management into clinical practice and dietary
Blumberg, Tufts University, Boston, MA, and a Steering Committee includ-
guidelines. Am J Clin Nutr 2013;98(suppl):1694S–9S. ing representatives from each of the symposium cosponsors: the American
Cancer Society, the American College of Nutrition, the American Institute
for Cancer Research, the American Medical Women’s Association, the Amer-
INTRODUCTION
ican Society for Nutrition, and the Linus Pauling Institute. The symposium
Osteoporosis, a degenerative skeletal disorder, is characterized was underwritten by the Tea Council of the USA. Its contents are solely the
by low bone mass (osteopenia) and microarchitectural deterioration responsibility of the authors and do not necessarily represent the official views
of bone tissue that results in compromised bone strength and of the Tea Council of the USA or the cosponsoring organizations.
3
increased risk of fractures (1). Among different types of fractures, The contents of this manuscript are solely the responsibility of the au-
hip fracture is the most severe consequence of osteoporosis, leading thors and do not necessarily represent the official views of the National
Center for Complementary and Alternative Medicine or the NIH.
to reduced activities of daily living, lowered quality of life, and 4
Supported by NIH/National Center for Complementary and Alternative
increased mortality (2). As the population ages worldwide, the Medicine grants (R21AT003735 and U01AT006691), the Laura W Bush
increased life expectancy is accompanied with a higher preva- Institute for Women’s Health (C-LS), and NIH/National Cancer Institute
lence of osteoporosis in the elderly population, such that oste- grant CA90997 (J-SW).
5
oporosis has become a serious health concern in many countries Address reprint requests and correspondence to C-L Shen, Department of
(2, 3). For example, in the United States, it is estimated that by Pathology, 1A092, 3601 4th Street, Texas Tech University Health Sciences
2020, there will be .61 million women and men aged $50 y Center, Lubbock, TX 79430-9097. E-mail: leslie.shen@ttuhsc.edu.
6
affected by osteoporosis or low bone mass (3); by 2025, the cost Abbreviations used: BAP, bone-specific alkaline phosphatase; BMD,
bone mineral density; ECG, epicatechin-3-gallate; EGCG, epigallocate-
of osteoporosis-related expenses is projected to increase to $25.3
chin-3-gallate; GTP, green tea polyphenol; MAP, mitogen-activated protein;
billion (4). NF-kB, nuclear transcription factor kB; RANKL, receptor activator of nu-
The etiology of osteoporosis includes both nonmodifiable (ie, clear transcription factor kB ligand; ROS, reactive oxygen species; TRAP,
genetic, sex, and age) and modifiable (ie, nutrition, excessive al- tartrate-resistant acid phosphatase; 8-OHdG, 8-hydroxy-2#deoxyguanosine.
cohol, tobacco smoking, and physical inactivity) factors. The risk First published online October 30, 2013; doi: 10.3945/ajcn.113.058255.
1694S Am J Clin Nutr 2013;98(suppl):1694S–9S. Printed in USA. Ó 2013 American Society for Nutrition
Downloaded from https://academic.oup.com/ajcn/article-abstract/98/6/1694S/4577502
by guest
on 24 August 2018
TEA AND BONE HEALTH 1695S
Reactive oxygen species (ROS) can lead to damage to DNA, inhibits bone resorption via inhibition of IL-6 production (23) or
protein, and lipids. Oxidative stress results from high amounts of suppression of p44/p42 MAP kinase activation (24). EGCG sup-
ROS produced during normal cellular metabolism and from presses osteoclastic differentiation via downregulation of the re-
environmental stimuli perturbing the normal redox balance (8). ceptor activator of NF-kB ligand (RANKL)-induced expression
With regard to osteoblastogenesis, excessive oxidative stress of the nuclear transcription factor of activated T cells c1, thus
induces osteoblast and osteocyte apoptosis, decreases osteoblast blocking differentiation of monocytes into osteoclasts, followed
number via extracellular signal-regulated kinases and extracel- by decreased bone resorption (25, 26). EGCG has also been
lular signal-regulated kinase–dependent nuclear transcription shown to block RANKL signaling by suppressing RANKL-induced
factor kB (NF-kB) signaling pathways (9), and suppresses bone NF-kB transcription activity, a pathway essential to osteoclas-
formation rate via Wnt/b-catenin signaling pathways (10). On togenesis (27).
the other hand, osteoblasts have been shown to upregulate an- Green tea’s bioactive components have also been shown to
tioxidant defenses such as glutathione peroxidase to stimulate benefit osteoblastogenesis by increasing osteoblastic survival,
transforming growth factor b, a growth factor involved in os- proliferation, differentiation, and mineralization. Observations
teoclast differentiation and survival (11). Oxidative stress ap- from in vitro studies support such stimulatory effects of EGCG
pears to enhance the differentiation and function of osteoclasts on osteoblastogenesis. For example, EGCG improves the survival
or directly contributes osteoclast-generated superoxide to bone of osteoblasts via suppression of TNF-a and IL-6 production (28).
degradation (12). A strong correlation exists between oxidative EGCG stimulates osteoblastogenesis by enhancing the mRNA
stress and bone loss during the development of osteoporosis (13, expression of Runt-related transcription factor-2–mediated os-
14). Because oxidative stress can contribute to bone loss, lowering teogenic genes and protein expression of alkaline phosphatase
its status via antioxidant mechanisms could be an important strat- activity (a biomarker of osoteoblastic differentiation) and even-
egy to mitigate or suppress osteoporosis-related bone loss. tually stimulating mineralization (29, 30). EGCG also increases
Tea, the dried leaves of the plant Camellia sinensis species of osteoblastic activity through Wnt signaling pathways, which mod-
the Theaceae family, is a popular beverage consumed all over ulate bone development and bone mass acquisition at all skeletal
the world with a production of 4.52 million tons in 2010, in- sites (31). EGCG upregulates prostaglandin F2a–stimulated vascu-
cluding black tea (78%) in Western countries, green tea (20%) in lar endothelial growth factor synthesis resulting from amplified
Asian countries, and oolong tea (2%) in southern China (15). activation of stress-activated protein kinase/c-Jun N-terminal ki-
Tea is an important source of dietary flavonoids, especially fla- nase in osteoblasts (23, 32). Furthermore, EGCG-induced sup-
vanols, and it has been associated with protecting against bone pression of p44/p42 MAP kinase activation plays a protective
loss and reducing risk of fractures. In this article, we briefly role against bone resorption by downregulating the synthesis of
summarize the state of knowledge regarding the effects of tea and IL-6, a potent bone resorptive agent in osteoblasts (24, 33).
its bioactive components on bone health and cover the findings Evidence from these in vitro studies suggests that tea poly-
from in vitro, animal, and human studies along with discussion of phenols may benefit bone health by enhancing osteoblastogenesis
relevant future directions in translational research. and suppressing osteoclastogenesis through modulation of growth
factors, cytokines, and chemokines and their receptors.
IN VITRO STUDIES
Theaflavins are the major polyphenols in black tea and are ANIMAL STUDIES
composed principally of theaflavin, theaflavin-3-gallate, thea- The effects of theaflavins and EGCG on in vitro osteogenesis
flavin-3#-gallate, and theaflavin-3,3#-gallate (16). Oka et al (17) have been confirmed in animal studies that used various bone-loss
recently showed that theaflavin-3,3-gallate inhibits the forma- models. For example, Das et al (34–36) found that black tea
tion and differentiation of osteoclasts via suppression of matrix extract has protective and restorative effects against ovariec-
metalloproteinases. Kim and Joo (18) reported that theaflavin tomy-induced bone loss in rats by decreasing active osteoclastic
inhibits LPS-induced IL-6, monocyte chemoattractant protein-1, number and bone turnover rate and increasing bone ash mineral
and intracellular adhesion molecule-1 expression in bone mar- content and strength. Karmakar et al (37) recently reported the
row–derived macrophages through the blockade of NF-kB and potential role of black tea extract as a protective agent against
mitogen-activated protein (MAP) kinase signaling pathways. nonalcoholic steatohepatitis-induced skeletal changes in bone
These findings support an antiinflammatory role of black tea on metabolism in rats.
bone health. In a rodent model, Shen et al (38, 39) used a mixture of GTPs
Compared with black tea, there are many more studies showing consisting of 480 mg EGCG, 160 mg ECG, 60 mg epicatechin,
a positive impact of green tea on bone health. Green tea poly- 103 mg epigallocatechin, and 30 mg catechin in 1000 mg GTPs
phenols (GTPs), including epigallocatechin-3-gallate (EGCG; the confirmed by HPLC–electrochemical detection and HPLC-UV
most abundant polyphenol in green tea), epigallocatechin, epi- analyses. GTP supplementation (0.1% and 0.5%, weight/volume)
catechin-3-gallate (ECG), epicatechin, and catechins (19). In vitro resulted in increased urinary epigallocatechin and epicatechin
studies have shown the antiresorptive properties of green tea’s concentrations in the sham and ovariectomized female rats in
bioactive components through increasing apoptosis of osteoclasts, a dose-dependent manner. The treatment also mitigated bone
suppression of osteoclast formation, and inhibition of bone re- loss (based on BMD measurement) and improved the integrity
sorption. For instance, EGCG enhances osteoclastic cell death of bone microstructure and quality. The doses of 0.1% and 0.5%
through the Fenton reaction (20) and caspase-3 activation (21). GTPs (equivalent to w1 and w4 cups green tea for human
EGCG suppresses osteoclast formation by inhibiting the expres- consumption/d, respectively) appear to be safe because no untoward
sion of matrix metalloproteinase-9 in osteoblasts (17, 22). EGCG changes were found in serum albumin, total protein, calcium,
Downloaded from https://academic.oup.com/ajcn/article-abstract/98/6/1694S/4577502
by guest
on 24 August 2018
1696S SHEN ET AL
phosphate, glucose, blood urea nitrogen, creatinine, total bili- antiresorptive agents (46, 47) and dietary supplements (48, 49)
rubin, alkaline phosphatase activity, creatinine phosphokinase, for mitigating BMD deterioration.
aspartate transferase, alanine transferase, globulin, amylase, and Biochemical markers of bone turnover have been shown to be
cholesterol. promising in predicting fractures in the elderly up to 2 y before
Similar findings of a protective effect by GTPs on bone health the event (50, 51). A higher bone resorption rate was strongly
have been observed in various bone-loss models, such as tes- associated with faster BMD loss (50, 51). Decreased vertebral
tosterone-deficiency–induced bone loss and microstructural de- BMD and increased bone turnover have approximately equal
terioration (40), chronic inflammation–induced bone loss and power in predicting the risk of bone loss and the osteoporosis-
microstructural deterioration (41, 42), and obesity-induced de- related fracture rate in postmenopausal women (50, 51). Therefore,
terioration of bone microstructure (43). In their animal models, depending on the duration of treatment, both BMD and bone
Shen et al (38–43) evaluated the bioavailability of GTPs in turnover biomarkers have been used clinically to monitor the
biofluids, bone efficacy, and potential mechanistic profiles and effects of treatments on bone health in humans.
concluded that GTP supplementation improves BMD and Whereas a body of evidence from animal models suggests
strength of the femur; increases trabecular volume, thickness, a positive relation between tea intake and maintaining BMD,
and number and bone formation of proximal tibia; and sup- human studies of tea drinking and risk of osteoporosis and
presses trabecular separation and bone erosion at both the proxi- fracture have yielded inconsistent results. For instance, Hegarty
mal tibia and the tibia shaft, resulting in a larger net bone volume. et al (52) reported that the BMDs of postmenopausal women at
The osteoprotective role of GTPs appears to be mediated in part the lumbar spine, trochanter, and Ward’s triangle were higher in
via increases in antioxidant capacity (glutathione peroxidase tea drinkers than in non–tea drinkers after adjustment for BMI
activity), decreases in inflammation (TNF-a, cyclooxygenase-2, and age. Similarly, a positive association between tea drinking and
IL-6), and/or reduced oxidative stress DNA damage [8-hydroxy- BMD was also reported among postmenopausal women in the
2#deoxyguanosine (8-OHdG)]. United States (53), Canada (54), the United Kingdom (52), Aus-
Green tea extracts may act not only via antioxidant mecha- tralia (55), Denmark (56), and Japan (57) and among older Asian
nisms but also serve as a prooxidant at high doses. Iwaniec et al men and women (58). In a case-control study, Keramat et al (59)
(44) showed in growing male mice that green tea extracts at 1% reported that regular consumption of $7 cups tea/d was found to
and 2% in diets for 6 wk, which is equivalent to w7–14 servings/d be a significant protective factor for osteoporosis. On the other
for humans, is detrimental to bone growth during early bone hand, Hamdi Kara et al (60) reported that habitual tea drinking was
development, as shown by a suppressed rate of bone accumulation not found to be a significant factor for BMD in postmenopausal
resulting in shorter bone length, reduced total and cortical bone Turkish women (60). Furthermore, an inverse relation between tea
volume and thickness, and decreased bone mineral content of consumption and BMD was found in a study in pre- and peri-
femur (44). It is worth contrasting these results to those by Shen menopausal women (aged 50–60 y) in the United States (61).
et al (38–43), who used lower doses in adult animals, and worth Results from the MEDOS (Mediterranean Osteoporosis) Study
considering the impact of interventions during different stages of showed that tea drinking was associated with a 30% reduction in
bone metabolism, ie, modeling during growth compared with the risk of hip fractures in both women (62) and men (63) .50 y
remodeling during aging. of age. With the use of data from a longitudinal follow-up study,
Evidence from these animal studies suggests that moderate Chen et al (53) found that the current amount of tea consump-
intake of tea polyphenols may mitigate bone loss and micro- tion in the United States resulted in such a weak effect on BMD
structural deterioration and improve bone strength and quality. that it would be unlikely to have a significant impact on fracture
This action may be partly mediated via stimulating anabolic local risk among American postmenopausal women.
factors and systemic hormones, suppressing catabolic bone local The discrepancy among these results may be due to different
factors and systemic hormones, decreasing bone resorption rates, study designs, different populations, different tea categories (black,
and improving bone strength and mechanical quality. These results oolong, green, etc), incomplete adjustment of the confounding
of animal studies suggest a potential prophylactic role of tea effects of lifestyle characteristics, and different skeletal sites for
polyphenols in human bone health. measuring BMD. Therefore, to evaluate tea polyphenols on bone
health in humans, a translational approach in humans should
consider the following: 1) a population at high risk of osteo-
HUMAN STUDIES porosis, 2) objective and quantitative assessments of character-
In clinical application, dual-energy X-ray absorptiometry is izing tea consumption, 3) quantitative methods to assess tea
generally used to measure BMD at the spine, hip, and femoral bioavailability and bone treatment efficacy, 4) validated bio-
neck to predict fracture risk and to monitor the natural pro- markers of oxidative stress and inflammation to explore poten-
gression of diseases that affect BMD or to monitor the therapeutic tial mechanisms of action, and 5) blood and/or urine analyses to
response to osteoporosis-specific treatments 1–2 y after the in- monitor the safety of the intervention.
ception of the therapies (45). We translated the osteoprotective results of GTPs in ovari-
In addition to BMD, serum and urinary bone biochemical markers ectomized rats to a short-term clinical trial to evaluate the effects
of bone formation [eg, bone-specific alkaline phosphatase (BAP), of GTPs on bone turnover biomarkers in postmenopausal women
osteocalcin, and N-terminal propeptide of type I collagen] and with osteopenia (64). We assessed GTP bioavailability (65),
bone resorption [eg, tartrate-resistant acid phosphatase (TRAP), safety (66), and molecular mechanisms (65) and participants’
type I collagen cross-linked N- and C-telopeptides, deoxypridino- quality of life (66) and muscle strength (64). In addition to GTP
line, and pyridinoline] can provide additional information and an supplementation, the effects of tai chi (a mind-body exercise) as
indication of the effectiveness of certain therapies including another treatment on the same outcome variables were also
Downloaded from https://academic.oup.com/ajcn/article-abstract/98/6/1694S/4577502
by guest
on 24 August 2018
TEA AND BONE HEALTH 1697S
evaluated (64–67). In our study, 171 qualified postmenopausal may restore the balance and mitigate bone loss in the development
osteopenic women [mean 6 SD age: 57.5 6 6.9 y; BMI (in kg/m2): of osteoporosis. The evidence from in vitro and animal studies of
28.3 6 5.4] were randomly assigned to 1 of 4 groups: 1) placebo various bone-loss models strongly suggests that tea polyphenols,
(500 mg medicinal starch/d; n = 44); 2) GTP (500 mg GTP/d; especially GTPs, are effective in protecting against osteoporosis.
n = 47); 3) placebo + tai chi training at 60 min/session, 3 ses- The possible mechanisms of action for GTPs in bone protection
sions/wk (n = 42); and 4) GTP + tai chi training (n = 38) (64). are shown in Figure 1. The most widely recognized character-
On the basis of our laboratory analysis, the GTPs were 99.25% istics of GTPs in bone protection are their antioxidant activities,
pure and contained 46.5% EGCG, 21.25% ECG, 10% epicatechin, stemming from their ability to scavenge ROS. In our previous studies,
7.5% epigallocatechin, 9.5% gallocatechin-3-gallate, and 4.5% we showed that GTP supplementation suppressed the production
catechin. During the 6-mo intervention period, all participants of proinflammatory cytokines, inhibited oxidative stress damage,
were supplemented with 500 mg elemental calcium and 200 IU and enhanced antioxidant capacity in various bone-loss models
vitamin D (as cholecalciferol) daily. Bone treatment efficacy (38–43). Tea and its polyphenol components act by increasing bone
was assessed by measuring serum BAP and TRAP at baseline mass, trabecular bone volume, number, and thickness and reducing
and at 1, 3, and 6 mo. GTP bioavailability was assessed by mea- trabecular separation, as well as by suppressing bone resorption
suring serum and urine GTP components at baseline and at 1, 3, and enhancing bone formation, resulting in greater bone strength.
and 6 mo (65). Safety was monitored monthly by assessing serum However, there are some differences between animals and humans
liver enzymes (aspartate aminotransferase and alanine amino- in terms of bone fracture. For example, the animal studies focused
transferase), kidney function (urea nitrogen and creatinine), alkaline principally on long bones, instead of the spine and hip, the com-
phosphatase, total bilirubin, calcium, and inorganic phosphorus mon sites of serious and frequent bone fracture in humans. In
(66). Potential molecular mechanisms were assessed by mea- addition, animal studies are not able to evaluate antifracture ca-
suring urinary 8-OHdG at baseline and at 1, 3, and 6 mo (65). pacity, an important indicator for osteoporosis management.
Quality of life, assessed by using the Short Form-36 question-
naire (version 2) (66), and muscle strength, measured by using
the wall-sit test (64), were evaluated at baseline and at 3 and 6 mo.
After 6 mo of intervention, 150 subjects completed the study
with a 12% attrition rate. The compliance rates for placebo and
GTP supplementation were 89% and 89%, respectively (64). The
attendance rate for tai chi classes was 83%. Throughout the study
period, neither GTP supplementation nor tai chi affected liver,
kidney function, or any other blood variables (66). In addition, no
adverse events as a result of the study treatment were reported by
the participants (66). With regard to GTP bioavailability and
compliance, we found the following: 1) no GTP components in
serum or urine in any group at baseline, 2) no detectable GTPs
in the placebo and placebo + tai chi groups at any collection
time, and 3) higher serum EGCG and ECG as well as higher
urinary epicatechin and ECG concentrations after 1, 3, and 6 mo
in the GTP intervention groups (65).
With regard to bone treatment efficacy, we observed the fol-
lowing: 1) elevation in BAP due to GTP intake (at 1 mo) and tai
chi exercise (at 3 mo) and 2) increases in the BAP:TRAP ratio
due to GTP intake (at 3 mo) and tai chi (at 6 mo) (64). Com-
pared with the placebo group, lower urinary 8-OHdG concen-
trations were found in the other 3 treatment groups at 3 and 6 mo
in a time-dependent manner (65). The tai chi interventions, but
not GTP intake alone, improved quality-of-life measures such as
role-emotional and mental health (66). Relative to the placebo
group, the 3 treatment groups showed improved muscle strength
at 6 mo (64). These results suggest that GTP supplementation at
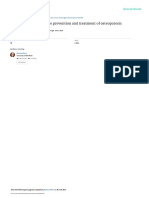
FIGURE 1. Possible mechanisms of action for green tea polyphenols in
500 mg daily (equivalent to w4–6 cups/d) and tai chi exercise bone protection. Green tea polyphenols increase antioxidant capacities and
for 6 mo are safe for postmenopausal women with osteopenia. concomitantly suppress oxidative stress and inflammation. Consequently,
GTP supplementation and tai chi exercise benefit bone remod- Runx2, Wnt, TGF-b, osterix, osteocalcin, and ALP are upregulated in oste-
eling starting from an early stage of intervention in association oblasts. RANKL, TRAP, and NF-kB activities are downregulated and cas-
pase-3 and Fenton reaction activities are upregulated in osteoclasts. These
with reduced oxidative stress and improved muscle strength. actions may enhance bone formation and mineralization and reduce bone
resorption and erosion, resulting in increased bone mass and bone mineral
density, increased bone strength, and reduced risk of bone fracture. ALP,
CONCLUSIONS AND FUTURE DIRECTIONS alkaline phosphatase; GPX, glutathione peroxidase; NF-kB, nuclear tran-
scription factor kB; PGE2, prostaglandin E2; RANKL, receptor activator
Osteoporosis results from a metabolic imbalance related to of nuclear transcription factor kB ligand; Runx2, Runt-related transcription
faster bone resorption than bone formation. The suppression of factor 2; SOD, superoxide dismutase; TGF-b, transforming growth factor b;
osteoclastogenesis and/or the enhancement of osteoblastogenesis TRAP, tartrate-resistant acid phosphatase; [, upregulated; Y, downregulated.
Downloaded from https://academic.oup.com/ajcn/article-abstract/98/6/1694S/4577502
by guest
on 24 August 2018
1698S SHEN ET AL
The evidence supporting BMD improvement and the anti- 9. Bai XC, Lu D, Bai J, Zheng H, Ke ZY, Li XM, Luo SQ. Oxidative
fracture effect of tea from longitudinal studies is quite limited. stress inhibits osteoblastic differentiation of bone cells by ERK and
NF-kappaB. Biochem Biophys Res Commun 2004;314:197–207.
Our 6-mo clinical trial showing that GTP favors bone formation 10. Manolagas SC. De-fense! De-fense! De-fense: scavenging H2O2 while
at an early-intervention stage, improves muscle strength, and making cholesterol. Endocrinology 2008;149:3264–6.
reduces oxidative stress in postmenopausal osteopenic women 11. Fuller K, Lean JM, Bayley KE, Wani MR, Chambers TJ. A role for
represents a first step toward translating the findings from animal TGF-b in osteoclast differentiation and survival. J Cell Sci 2000;113:
2445–53.
studies to human applications (64–67). However, future research 12. Garrett IR, Boyce BF, Oreffo RO, Bonewald L, Poser J, Mundy GR.
is necessary to better define the optimal dose of tea intake or Oxygen-derived free radicals stimulate osteoclastic bone resorption in
GTP supplementation and to examine the potential interactions rodent bone in vitro and in vivo. J Clin Invest 1990;85:632–9.
between tea polyphenols and other nutrients essential to bone 13. Syed FA, Ng AC. The pathophysiology of the aging skeleton. Curr
Osteoporos Rep 2010;8:235–40.
health, including calcium and vitamin D; and it should be of 14. Baek KH, Oh KW, Lee WY, Lee SS, Kim MK, Kwon HS, Rhee EJ,
sufficient duration to assess changes in BMD as well as bio- Han JH, Song KH, Cha BY, et al. Association of oxidative stress with
markers of bone turnover. postmenopausal osteoporosis and the effects of hydrogen peroxide on
To better understand the effects of tea on bone health in people osteoclast formation in human bone marrow cell cultures. Calcif Tissue
at high risk of osteoporosis and fracture, more basic research and Int 2010;87:226–35.
15. Food and Agriculture Organization of the United Nations–Production
human studies are warranted. For example, an animal study con- FAOSTAT. Medium-term prospects for agricultural commodities.
ducted to evaluate chronic toxicity, safety, and bone efficacy in Available from: http://www.fao.org/docrep/006/y5143e/y5143e00.htm
ovariectomized rats supplemented with different dosages of tea (cited 10 August 2010).
polyphenols administered in drinking water will be essential to 16. Balentine DA, Wiseman SA, Bouwens LC. The chemistry of tea fla-
vonoids. Crit Rev Food Sci Nutr 1997;37:693–704.
inform the design of new clinical trials. Similar short-term tea 17. Oka Y, Iwai S, Amano H, Irie Y, Yatomi K, Ryu K, Yamada S, Inagaki
polyphenol dose-response studies could then be conducted in K, Oguchi K. Tea polyphenols inhibit rat osteoclast formation and
postmenopausal osteopenic women to determine the optimal dose differentiation. J Pharmacol Sci 2012;118:55–64.
on the basis of relevant bone-related outcomes (ie, bone turnover 18. Kim S, Joo YE. Theaflavin inhibits LPS-induced IL-6, MCP-1, and
ICAM-1 expression in bone marrow-derived macrophages through the
markers) along with a safety evaluation. Both of these approaches blockade of NF-kB and MAPK signaling pathways. Chonnam Med J
are necessary to prepare for a large-scale, long-term, placebo- 2011;47:104–10.
controlled randomized trial with an optimal tea regimen. Such 19. Yang CS, Landau JM. Effects of tea consumption on nutrition and
a study should be designed to 1) assess compliance and bioactivity health. J Nutr 2000;130:2409–12.
20. Nakagawa H, Wachi M, Woo JT, Kato M, Kasai S, Takahashi F, Lee IS,
with appropriate biomarkers, 2) monitor safety, and 3) quantify Nagai K. Fenton reaction is primarily involved in a mechanism of
with sufficient power bone mass at different body sites and bone (-)-epigallocatechin-3-gallate to induce osteoclastic cell death. Bio-
microarchitecture (ie, volumetric BMD, stress-strain index, and chem Biophys Res Commun 2002;292:94–101.
bone geometry) by using imaging technology. On the basis of the 21. Hafeez BB, Ahmed S, Wang N, Gupta S, Zhang A, Haqqi TM. Green
available preliminary data, it also appears worthwhile to determine tea polyphenols-induced apoptosis in human osteosarcoma SAOS-2
cells involves a caspase-dependent mechanism with downregulation of
other outcomes that are relevant to bone health, such as mobility, nuclear factor-kappaB. Toxicol Appl Pharmacol 2006;216:11–9.
function, balance, and quality of life. 22. Yun JH, Pang EK, Kim CS, Yoo YJ, Cho KS, Chai JK, Kim CK, Choi
SH. Inhibitory effects of green tea polyphenol (-)-epigallocatechin
The authors’ responsibilities were as follows—C-LS: planned and drafted gallate on the expression of matrix metalloproteinase-9 and on the
the article; and M-CC and J-SW: critically reviewed and revised the article. formation of osteoclasts. J Periodontal Res 2004;39:300–7.
C-LS received an honorarium and travel support from the Tea Council of the 23. Tokuda H, Takai S, Hanai Y, Matsushima-Nishiwaki R, Hosoi T,
USA for speaking at the Fifth International Scientific Symposium on Tea and Harada A, Ohta T, Kozawa O. (-)-Epigallocatechin gallate suppresses
Human Health and for preparing this article for publication. The authors endothelin-1-induced interleukin-6 synthesis in osteoblasts: inhibition
declared no competing financial interests. of p44/p42 MAP kinase activation. FEBS Lett 2007;581:1311–6.
24. Tokuda H, Takai S, Matsushima-Nishiwaki R, Akamatsu S, Hanai Y,
Hosoi T, Harada A, Ohta T, Kozawa O. (–)-Epigallocatechin gallate en-
hances prostaglandin F2alpha-induced VEGF synthesis via upregulating
REFERENCES SAPK/JNK activation in osteoblasts. J Cell Biochem 2007;100:1146–53.
1. NIH Consensus Development Panel on Osteoporosis Prevention, Di- 25. Morinobu A, Biao W, Tanaka S, Horiuchi M, Jun L, Tsuji G, Sakai Y,
agnosis, and Therapy. Osteoporosis prevention, diagnosis, and therapy. Kurosaka M, Kumagai S. (-)-Epigallocatechin-3-gallate suppresses
JAMA 2001;285:785–95. osteoclast differentiation and ameliorates experimental arthritis in
2. Holroyd C, Cooper C, Dennison E. Epidemiology of osteoporosis. Best mice. Arthritis Rheum 2008;58:2012–8.
Pract Res Clin Endocrinol Metab 2008;22:671–85. 26. Lee JH, Jin H, Shim HE, Kim HN, Ha H, Lee ZH. Epigallocatechin-3-gallate
3. Boonen S, Dejaeger E, Vanderschueren D, Venken K, Bogaerts A, inhibits osteoclastogenesis by down-regulating c-Fos expression and sup-
Verschueren S, Milisen K. Osteoporosis and osteoporotic fracture oc- pressing the nuclear factor-kappaB signal. Mol Pharmacol 2010;77:17–25.
currence and prevention in the elderly: a geriatric perspective. Best 27. Lin RW, Chen CH, Wang YH, Ho ML, Hung SH, Chen IS, Wang GJ.
Pract Res Clin Endocrinol Metab 2008;22:765–85. (-)-Epigallocatechin gallate inhibition of osteoclastic differentiation via
4. Burge R, Dawson-Hughes B, Solomon DH, Wong JB, King A, Tosteson NF-kappaB. Biochem Biophys Res Commun 2009;379:1033–7.
A. Incidence and economic burden of osteoporosis-related fractures in 28. Takai S, Matsushima-Nishiwaki R, Adachi S, Natsume H, Minamitani
the United States, 2005-2025. J Bone Miner Res 2007;22:465–75. C, Mizutani J, Otsuka T, Tokuda H, Kozawa O. (-)-Epigallocatechin
5. Ahmadieh H, Arabi A. Vitamins and bone health: beyond calcium and gallate reduces platelet-derived growth factor-BB-stimulated in-
vitamin D. Nutr Rev 2011;69:584–98. terleukin-6 synthesis in osteoblasts: suppression of SAPK/JNK. Me-
6. Barnes PM, Powell-Griner E, McFann K, Nahin RL. Complementary diators Inflamm 2008;2008:291808.
and alternative medicine use among adults: United States. Adv Data 29. Chen CH, Ho ML, Chang JK, Hung SH, Wang GJ. Green tea catechin
2004;343:1–19. enhances osteogenesis in a bone marrow mesenchymal stem cell line.
7. Matsuo K, Irie N. Osteoclast-osteoblast communication. Arch Bio- Osteoporos Int 2005;16:2039–45.
chem Biophys 2008;473:201–9. 30. Vali B, Rao LG, El-Sohemy A. Epigallocatechin-3-gallate increases the
8. Banfi G, Iorio EL, Corsi MM. Oxidative stress, free radicals and bone formation of mineralized bone nodules by human osteoblast-like cells.
remodeling. Clin Chem Lab Med 2008;46:1550–5. J Nutr Biochem 2007;18:341–7.
Downloaded from https://academic.oup.com/ajcn/article-abstract/98/6/1694S/4577502
by guest
on 24 August 2018
TEA AND BONE HEALTH 1699S
31. Rawadi G. Wnt signaling and potential applications in bone diseases. 50. Boonen S, Reginster JY, Kaufman JM, Lippuner K, Zanchetta J,
Curr Drug Targets 2008;9:581–90. Langdahl B, Rizzoli R, Lipschitz S, Dimai HP, Witvrouw R, et al.
32. Hayashi K, Takai S, Matsushima-Nishiwaki R, Hanai Y, Kato K, Fracture risk and zoledronic acid therapy in men with osteoporosis.
Tokuda H, Kozawa O. (-)-Epigallocatechin gallate reduces trans- N Engl J Med 2012;367:1714–23.
forming growth factor beta-stimulated HSP27 induction through the 51. Löfman O, Magnusson P, Toss G, Larsson L. Common biochemical
suppression of stress-activated protein kinase/c-Jun N-terminal kinase markers of bone turnover predict future bone loss: a 5-year follow-up
in osteoblasts. Life Sci 2008;82:1012–7. study. Clin Chim Acta 2005;356:67–75.
33. Tokuda H, Takai S, Hanai Y, Matsushima-Nishiwaki R, Yamauchi J, 52. Hegarty VM, May HM, Khaw KT. Tea drinking and bone mineral
Harada A, Hosoi T, Ohta T, Kozawa O. (-)-Epigallocatechin gallate density in older women. Am J Clin Nutr 2000;71:1003–7.
inhibits basic fibroblast growth factor-stimulated interleukin-6 syn- 53. Chen Z, Pettinger MB, Ritenbaugh C, LaCroix AZ, Robbins J, Caan
thesis in osteoblasts. Horm Metab Res 2008;40:674–8. BJ, Barad DH, Hakim IA. Habitual tea consumption and risk of os-
34. Das AS, Mukherjee M, Mitra C. Evidence for a prospective anti- teoporosis: a prospective study in the Women’s Health Initiative ob-
osteoporosis effect of black tea (Camellia Sinensis) extract in bilaterally servational cohort. Am J Epidemiol 2003;158:772–81.
ovariectomized rat model. Asia Pac J Clin Nutr 2004;13:210–6. 54. Hoover PA, Webber CE, Beaumont LF, Blake JM. Postmenopausal
35. Das AS, Das D, Mukherjee M, Mukherjee S, Mitra C. Phytoestrogenic bone mineral density: relationship to calcium intake, calcium absorp-
effects of black tea extract (Camellia sinensis) in an oophorectomized rat tion, residual estrogen, body composition, and physical activity. Can J
(Rattus norvegicus) model of osteoporosis. Life Sci 2005;77:3049–57. Physiol Pharmacol 1996;74:911–7.
36. Das AS, Mukherjee M, Das D, Mitra C. Protective action of aqueous 55. Devine A, Hodgson JM, Dick IM, Prince RL. Tea drinking is associ-
black tea (Camellia sinensis) extract (BTE) against ovariectomy- ated with benefits on bone density in older women. Am J Clin Nutr
induced oxidative stress of mononuclear cells and its associated pro- 2007;86:1243–7.
gression of bone loss. Phytother Res 2009;23:1287–94. 56. Vestergaard P, Hermann AP, Gram J, Jensen LB, Eiken P, Abrahamsen
37. Karmakar S, Majumdar S, Maiti A, Choudhury M, Ghosh A, Das AS, B, Brot C, Kolthoff N, Sørensen OH, Beck Nielsen H, et al. Evaluation
Mitra C. Protective role of black tea extract against nonalcoholic of methods for prediction of bone mineral density by clinical and
steatohepatitis-induced skeletal dysfunction. J Osteoporos 2011;2011: biochemical variables in perimenopausal women. Maturitas 2001;40:
426863. 211–20.
38. Shen CL, Wang P, Guerrieri J, Yeh J, Wang JS. Protective effect of 57. Muraki S, Yamamoto S, Ishibashi H, Oka H, Yoshimura N, Kawaguchi
green tea polyphenols on bone loss in middle-aged female rats. Os- H, Nakamura K. Diet and lifestyle associated with increased bone
teoporos Int 2008;19:979–90. mineral density: cross-sectional study of Japanese elderly women at an
39. Shen CL, Yeh JK, Stoecker BJ, Chyu MC, Wang JS. Green tea poly- osteoporosis outpatient clinic. J Orthop Sci 2007;12:317–20.
phenols mitigate deterioration of bone microarchitecture in middle- 58. Wu CH, Yang YC, Yao WJ, Lu FH, Wu JS, Chang CJ. Epidemiological
aged female rats. Bone 2009;44:684–90. evidence of increased bone mineral density in habitual tea drinkers.
40. Shen CL, Cao JJ, Dagda RY, Tenner TE Jr, Chyu MC, Yeh JK. Sup- Arch Intern Med 2002;162:1001–6.
plementation with green tea polyphenols improves bone microstructure 59. Keramat A, Patwardhan B, Larijani B, Chopra A, Mithal A, Chakravarty
and quality in aged, orchidectomized rats. Calcif Tissue Int 2011;88: D, Adibi H, Khosravi A. The assessment of osteoporosis risk factors in
455–63. Iranian women compared with Indian women. BMC Musculoskelet
41. Shen CL, Yeh JK, Samathanam C, Cao JJ, Stoecker BJ, Dagda RY, Disord 2008;9:28.
Chyu MC, Dunn DM, Wang JS. Green tea polyphenols attenuate de- 60. Hamdi Kara I, Aydin S, Gemalmaz A, Aktürk Z, Yaman H, Bozdemir
terioration of bone microarchitecture in female rats with systemic N, Kurdak H, Sitmapinar K, Devran Sencar I, Bas xak O, et al. Habitual
chronic inflammation. Osteoporos Int 2011;22:327–37. tea drinking and bone mineral density in postmenopausal Turkish
42. Shen CL, Yeh JK, Cao JJ, Tatum OL, Dagda RY, Wang JS. Green tea women: Investigation of Prevalence of Postmenopausal Osteoporosis in
polyphenols mitigate bone loss of female rats in a chronic in- Turkey (IPPOT Study). Int J Vitam Nutr Res 2007;77(6):389–97
flammation-induced bone loss model. J Nutr Biochem 2010;21: [Published erratum appears in Int J Vitam Nutr Res 2008;78(3):166.]
968–74. 61. Hernández-Avila M, Stampfer MJ, Ravnikar VA, Willett WC, Schiff I,
43. Shen CL, Cao JJ, Dagda RY, Chanjaplammootil S, Lu C, Chyu MC, Francis M, Longcope C, McKinlay SM, Longcope C. Caffeine and
Gao W, Wang JS, Yeh JK. Green tea polyphenols benefits body com- other predictors of bone density among pre- and perimenopausal
position and improves bone quality in long-term high-fat diet-induced women. Epidemiology 1993;4:128–34.
obese rats. Nutr Res 2012;32:448–57. 62. Johnell O, Gullberg B, Kanis JA, Allander E, Elffors L, Dequeker J,
44. Iwaniec UT, Turner RT, Koo SI, Kaur R, Ho E, Wong CP, Bruno RS. Dilsen G, Gennari C, Lopes Vaz A, Lyritis G, et al. Risk factors for hip
Consumption of green tea extract results in osteopenia in growing male fracture in European women: the MEDOS Study. Mediterranean Os-
mice. J Nutr 2009;139:1914–9. teoporosis Study. J Bone Miner Res 1995;10:1802–15.
45. Melton LJ 3rd, Crowson CS, O’Fallon WM, Wahner HW, Riggis BL. 63. Kanis J, Johnell O, Gullberg B, Allander E, Elffors L, Ranstam J,
Relative contributions of bone density, bone turnover, and clinical risk Dequeker J, Dilsen G, Gennari C, Vaz AL, et al. Risk factors for hip
factors to long-tern fracture prediction. J Bone Miner Res 2003;18: fracture in men from southern Europe: the MEDOS Study. Mediter-
312–8. ranean Osteoporosis Study. Osteoporos Int 1999;9:45–54.
46. Hochberg MC, Greenspan S, Wasnich RD, Miller P, Thompson DE, 64. Shen CL, Chyu MC, Yeh JK, Zhang Y, Pence BC, Felton CK, Brismée
Ross PD. Changes in bone density and turnover explain the reductions JM, Arjmandi BH, Doctolero S, Wang JS. Effect of green tea and tai
in incidence of nonvertebral fractures that occur during treatment with chi on bone health in postmenopausal osteopenic women: a 6-month
antiresorptive agents. J Clin Endocrinol Metab 2002;87:1586–92. randomized placebo-controlled trial. Osteoporos Int 2012;23:1541–52.
47. Glover SJ, Eastell R, McCloskey EV, Rogers A, Garnero P, Lowery J, 65. Qian G, Xue K, Tang L, Wang F, Song X, Chyu MC, Pence BC, Shen
Belleli R, Wright TM, John MR. Rapid and robust response of bio- CL, Wang JS. Mitigation of oxidative damage by green tea polyphenols
chemical markers of bone formation to teriparatide therapy. Bone and tai chi exercise in postmenopausal women with osteopenia. PLoS
2009;45:1053–8. ONE 2012;7:e48090.
48. Grimnes G, Joakimsen R, Figenschau Y, Torjesen PA, Almås B, Jorde R. 66. Shen CL, Chyu MC, Pence BC, Yeh JK, Zhang Y, Felton CK, Doc-
The effect of high-dose vitamin D on bone mineral density and bone tolero S, Wang JS. Green tea polyphenols supplementation and tai chi
turnover markers in postmenopausal women with low bone mass—a ran- exercise for postmenopausal osteopenic women: safety and quality of
domized controlled 1-year trial. Osteoporos Int 2012;23:201–11. life report. BMC Complement Altern Med 2010;10:76.
49. Hooshmand S, Chai SC, Saadat RL, Payton ME, Brummel-Smith K, 67. Shen CL, Chyu MC, Yeh JK, Felton CK, Xu KT, Pence BC, Wang JS.
Arjmandi BH. Comparative effects of dried plum and dried apple on Green tea polyphenols and tai chi for bone health: designing a placebo-
bone in postmenopausal women. Br J Nutr 2011;106:923–30. controlled randomized trial. BMC Musculoskelet Disord 2009;10:110.
Downloaded from https://academic.oup.com/ajcn/article-abstract/98/6/1694S/4577502
by guest
on 24 August 2018
You might also like
- Science Quiz Bee SHS GroupDocument43 pagesScience Quiz Bee SHS GroupGie Apilado Ranay91% (11)
- ABO Blood Grouping Cbsebiology4uDocument14 pagesABO Blood Grouping Cbsebiology4uRahit BiswasNo ratings yet
- Osteoporosis Literature ReviewDocument5 pagesOsteoporosis Literature Reviewafmzinuvouzeny100% (1)
- American Journal of Clinical Nutrition 2008 87 (1) 175-180Document6 pagesAmerican Journal of Clinical Nutrition 2008 87 (1) 175-180Daniela Perez AyalaNo ratings yet
- Orchad Et Al 2012-Syst Review-Omega 3-OsteoporosisDocument14 pagesOrchad Et Al 2012-Syst Review-Omega 3-OsteoporosisSpySmpatiNo ratings yet
- Agm2 2 147 PDFDocument10 pagesAgm2 2 147 PDFAntonioNo ratings yet
- Fructus Ligustri Lucidi in Osteoporosis A Review oDocument18 pagesFructus Ligustri Lucidi in Osteoporosis A Review oJ BCNo ratings yet
- JJBMRDocument9 pagesJJBMRDeby AnditaNo ratings yet
- 10.1007@s40520 019 01191 WDocument7 pages10.1007@s40520 019 01191 WRodolfo BayardoNo ratings yet
- Eaton, Konner, Cordain 2010 AJCNDocument3 pagesEaton, Konner, Cordain 2010 AJCNBarnettLudwigNo ratings yet
- ProImmune F1 On Loss of Muscle Mass in AgingDocument11 pagesProImmune F1 On Loss of Muscle Mass in AgingBryan Edson M. YcoNo ratings yet
- Type 1 Diabetes and Osteoporosis A Review of LiteratureDocument8 pagesType 1 Diabetes and Osteoporosis A Review of LiteraturebqvlqjugfNo ratings yet
- 1042 FullDocument9 pages1042 Fulloki harisandiNo ratings yet
- Herbal Treatment For Osteoporosis: A Current Review: Ping Chung Leung, Wing Sum SiuDocument6 pagesHerbal Treatment For Osteoporosis: A Current Review: Ping Chung Leung, Wing Sum Siusiska khairNo ratings yet
- Stini-1990-American Journal of Physical AnthropologyDocument44 pagesStini-1990-American Journal of Physical AnthropologyIsolda Alanna RlNo ratings yet
- Original Communication Bone and Nutrition in Elderly Women: Protein, Energy, and Calcium As Main Determinants of Bone Mineral DensityDocument13 pagesOriginal Communication Bone and Nutrition in Elderly Women: Protein, Energy, and Calcium As Main Determinants of Bone Mineral DensitySyifa NashuhaNo ratings yet
- Dietary Calcium Intake, Vitamin D Status, and Bone Health in Postmenopausal Women in Rural PakistanDocument6 pagesDietary Calcium Intake, Vitamin D Status, and Bone Health in Postmenopausal Women in Rural PakistanAli HaiderNo ratings yet
- CGJ 18 29Document7 pagesCGJ 18 29ZenithaMeidaNo ratings yet
- The Aging of Skeletal Muscle and Potential Therapeutic Effects of Extracts From Edible and Inedible PlantsDocument19 pagesThe Aging of Skeletal Muscle and Potential Therapeutic Effects of Extracts From Edible and Inedible Plantsaaysha guptaNo ratings yet
- Osteoprotective Medicinal Plants Part 1Document18 pagesOsteoprotective Medicinal Plants Part 1Celine RoumierNo ratings yet
- Osteoarthritis Pain of The Knee-A Pilot StudyDocument9 pagesOsteoarthritis Pain of The Knee-A Pilot Studyana mariaNo ratings yet
- Am J Clin Nutr 2015 Paddon Jones 1339S 45SDocument7 pagesAm J Clin Nutr 2015 Paddon Jones 1339S 45SevaNo ratings yet
- Ijmm 36 3 792 PDFDocument9 pagesIjmm 36 3 792 PDFjljsjoostenNo ratings yet
- Comparative Effects of Dried Plum and dr-2Document8 pagesComparative Effects of Dried Plum and dr-2Radoslav MarkovicNo ratings yet
- Review Article: Combating Osteoarthritis Through Stem Cell Therapies by Rejuvenating Cartilage: A ReviewDocument13 pagesReview Article: Combating Osteoarthritis Through Stem Cell Therapies by Rejuvenating Cartilage: A ReviewAlvin GfNo ratings yet
- New England Journal Medicine: The ofDocument14 pagesNew England Journal Medicine: The ofJoevet T. TadlasNo ratings yet
- A Review On The Diseases and Treatments That Affect The Bone Physiology and StrengthDocument17 pagesA Review On The Diseases and Treatments That Affect The Bone Physiology and StrengthErnest Gabriel Cruz AdvinculaNo ratings yet
- Adverse Effects of Corticosteroids On BoneDocument6 pagesAdverse Effects of Corticosteroids On BoneDiego Rivas-VazquezNo ratings yet
- Vitamin D and Its Effects On Articular Cartilage and OsteoarthritisDocument8 pagesVitamin D and Its Effects On Articular Cartilage and OsteoarthritisMuhammad Bayu Zohari HutagalungNo ratings yet
- Hepatic Osteodystrophy MechanismDocument14 pagesHepatic Osteodystrophy MechanismM.miracleNo ratings yet
- OsteoblastDocument9 pagesOsteoblastPRATIK GANGULYNo ratings yet
- Nutrition and Osteoporosis: V.E. Bianchi, L.J. Dominguez, M. BarbagalloDocument10 pagesNutrition and Osteoporosis: V.E. Bianchi, L.J. Dominguez, M. BarbagalloMartha SimonaNo ratings yet
- 2023 - The Role of Nutrition in The Prevention of SarcopeniaDocument13 pages2023 - The Role of Nutrition in The Prevention of SarcopeniaAlex LimaNo ratings yet
- Does Obesity Protect Postmenopausal Women Against Osteoporosis?Document10 pagesDoes Obesity Protect Postmenopausal Women Against Osteoporosis?Indah MaulidawatiNo ratings yet
- Osteoporosis Prevention, Diagnosis, and Therapy: Consensus ConferenceDocument11 pagesOsteoporosis Prevention, Diagnosis, and Therapy: Consensus ConferenceCorin Boice TelloNo ratings yet
- Methionine Down-Regulates Tlr4/Myd88/Nf-Κb Signalling In Osteoclast Precursors To Reduce Bone Loss During OsteoporosisDocument15 pagesMethionine Down-Regulates Tlr4/Myd88/Nf-Κb Signalling In Osteoclast Precursors To Reduce Bone Loss During OsteoporosisLourdesNo ratings yet
- Nutritional Aspects of The Prevention and TreatmenDocument8 pagesNutritional Aspects of The Prevention and TreatmenSilviani FatmaNo ratings yet
- Antioxidants ARANDANO PDFDocument20 pagesAntioxidants ARANDANO PDFjairo perdomoNo ratings yet
- Chondroprotective Effects of The Combination ChondroitinDocument9 pagesChondroprotective Effects of The Combination Chondroitincursotplo2021No ratings yet
- JCM 11 04676 v2Document14 pagesJCM 11 04676 v2DeepaNo ratings yet
- 2018 Update On OsteoporosisDocument16 pages2018 Update On OsteoporosisManel PopescuNo ratings yet
- Anti-In Ammatory Effects of Polyphenols in Arthritis: Mini-ReviewDocument7 pagesAnti-In Ammatory Effects of Polyphenols in Arthritis: Mini-ReviewAgung Gunawan PasannaiNo ratings yet
- Quality of Life Measurement in Bone Metastases A Literature ReviewDocument5 pagesQuality of Life Measurement in Bone Metastases A Literature ReviewiimytdcndNo ratings yet
- Collagen OsteoDocument16 pagesCollagen OsteoDouglas NoltieNo ratings yet
- J of Bone Mineral Res - 2009 - Babraj - Human Bone Collagen Synthesis Is A Rapid Nutritionally Modulated ProcessDocument8 pagesJ of Bone Mineral Res - 2009 - Babraj - Human Bone Collagen Synthesis Is A Rapid Nutritionally Modulated ProcessLjón BjörnNo ratings yet
- PIIS0002916523659611Document9 pagesPIIS0002916523659611Stefanus Prima NugrohoNo ratings yet
- Update Osteoporosis Management (Listo)Document18 pagesUpdate Osteoporosis Management (Listo)Jossie AcuñaNo ratings yet
- Gastritis 1Document5 pagesGastritis 1IttyNo ratings yet
- Thesis On Bone CancerDocument5 pagesThesis On Bone Cancerkvpyqegld100% (2)
- s00223 019 00581 6Document9 pagess00223 019 00581 6Dhruvi KansaraNo ratings yet
- Obsop Journal 1Document14 pagesObsop Journal 1Radditt GoberNo ratings yet
- Antioxidants 12 00362 v2Document4 pagesAntioxidants 12 00362 v2Paskah DianNo ratings yet
- Nandrolona Osteoporose RCTDocument6 pagesNandrolona Osteoporose RCTrodrigoNo ratings yet
- IJMR-156-640 - Arjun ShahuDocument9 pagesIJMR-156-640 - Arjun ShahuManohar ZateNo ratings yet
- Article ايبيDocument6 pagesArticle ايبيلمى الشهريNo ratings yet
- 2006 Simvastatin Decreases IL-6 and IL-8 Production in Epithelial CellsDocument5 pages2006 Simvastatin Decreases IL-6 and IL-8 Production in Epithelial CellsK KNo ratings yet
- Duque2021 Article OsteosarcopeniaAGeriatricGiantDocument4 pagesDuque2021 Article OsteosarcopeniaAGeriatricGiantVERONICA BARRERANo ratings yet
- Pharmaceuticals ProjectDocument18 pagesPharmaceuticals ProjectRakesh PradhanNo ratings yet
- Osteoporosis and Periodontal Disease: History and Physical ExaminationDocument15 pagesOsteoporosis and Periodontal Disease: History and Physical ExaminationYenniy IsmullahNo ratings yet
- Mecanismos de Dolor en OA - RevisiónDocument18 pagesMecanismos de Dolor en OA - Revisióncaps.sne911tguNo ratings yet
- Editorial: Secondary Osteoporosis: Endocrine and Metabolic Causes of Bone Mass DeteriorationDocument2 pagesEditorial: Secondary Osteoporosis: Endocrine and Metabolic Causes of Bone Mass DeteriorationAnonymous V5l8nmcSxbNo ratings yet
- A Practical Approach to Adolescent Bone Health: A Guide for the Primary Care ProviderFrom EverandA Practical Approach to Adolescent Bone Health: A Guide for the Primary Care ProviderSarah PittsNo ratings yet
- Jorth Et Al 2014 Metatranscriptomics of The Human Oral Microbiome During Health and DiseaseDocument10 pagesJorth Et Al 2014 Metatranscriptomics of The Human Oral Microbiome During Health and DiseasesznistvanNo ratings yet
- ELISA - Wikipedia, The Free EncyclopediaDocument8 pagesELISA - Wikipedia, The Free EncyclopediashivshankarbhaiNo ratings yet
- CHAPTER 2 BiologyDocument10 pagesCHAPTER 2 BiologyMohamad ShukriNo ratings yet
- Cointegrated VectorsDocument2 pagesCointegrated Vectorsdevikamurugan124206No ratings yet
- Xs Sports Whey Protein Anz PDFDocument3 pagesXs Sports Whey Protein Anz PDFHARSH GAUTAMNo ratings yet
- Gene TherapyDocument3 pagesGene TherapyGleeson Jay NiedoNo ratings yet
- Bacterial Genetics 2020Document67 pagesBacterial Genetics 2020Ntobi ThomasNo ratings yet
- Tecalco Cruz 2017 R3Document10 pagesTecalco Cruz 2017 R3Valentina Conde AndradeNo ratings yet
- MCAT2015 PreviewguideDocument153 pagesMCAT2015 PreviewguideMiao JingyaNo ratings yet
- (Lab) Cellular RespirationDocument4 pages(Lab) Cellular RespirationManuj KantNo ratings yet
- Ap Biology Portfolio 2019 - 2020 Ashrika DudejaDocument44 pagesAp Biology Portfolio 2019 - 2020 Ashrika Dudejaapi-520142437No ratings yet
- Vitamin D (Calcitriol)Document4 pagesVitamin D (Calcitriol)goremicNo ratings yet
- 1.6 Principles of Enzyme KineticsDocument9 pages1.6 Principles of Enzyme Kineticsiridescent brightwinNo ratings yet
- Nasal Physiology and Pathophysiology PDFDocument610 pagesNasal Physiology and Pathophysiology PDFRahat tanvirNo ratings yet
- DBT BET Question Paper 2010 With Answer KeyDocument24 pagesDBT BET Question Paper 2010 With Answer KeyAbhay Kumar100% (3)
- (Nutrition and Health) Connie M. Weaver, Robert P. Heaney - Calcium in Human Health-Humana Press (2005) - 21-32Document12 pages(Nutrition and Health) Connie M. Weaver, Robert P. Heaney - Calcium in Human Health-Humana Press (2005) - 21-32Felipe LegorretaNo ratings yet
- Molecular and CellularDocument10 pagesMolecular and CellularBAUTISTA TERRONES GILMERNo ratings yet
- The 12 Steps of Muscle ContractionsDocument1 pageThe 12 Steps of Muscle ContractionsgraceNo ratings yet
- Physicochemical Properties in Relation To Biological ActivityDocument11 pagesPhysicochemical Properties in Relation To Biological Activityjemms16No ratings yet
- Summary Fatty Alcohols LFRMIDocument2 pagesSummary Fatty Alcohols LFRMILuis Fernando Ramos Méndez IrisNo ratings yet
- HB-1193-006 HB PlasmidPurif 0723 WWDocument68 pagesHB-1193-006 HB PlasmidPurif 0723 WWDiana DiasNo ratings yet
- LEAP CELL DIVISION Week 1 BDocument5 pagesLEAP CELL DIVISION Week 1 BTHEA PAJULASNo ratings yet
- Gotaq g2 Green Master Mix ProtocolDocument2 pagesGotaq g2 Green Master Mix ProtocoljoyNo ratings yet
- Chapter 2: Diagnostic Enzymology: E+S Es E+PDocument72 pagesChapter 2: Diagnostic Enzymology: E+S Es E+PQananiisa Lammii Gammada100% (1)
- Genomic Tools For Crop ImprovementDocument41 pagesGenomic Tools For Crop ImprovementNeeru RedhuNo ratings yet
- Biotechnology Journal - 2008 - Zhou - Implementation of Advanced Technologies in Commercial Monoclonal Antibody ProductionDocument16 pagesBiotechnology Journal - 2008 - Zhou - Implementation of Advanced Technologies in Commercial Monoclonal Antibody ProductionBRUNA COELHO DE ANDRADENo ratings yet
- Dietrich, 2001 PDFDocument9 pagesDietrich, 2001 PDFargos1301No ratings yet
- Fetal Acid Base PhysiologyDocument2 pagesFetal Acid Base PhysiologyAde YonataNo ratings yet