Professional Documents
Culture Documents
HP Dosing Calculation
HP Dosing Calculation
Uploaded by
Jegannathan Nambukumar0 ratings0% found this document useful (0 votes)
71 views1 pageThe document provides calculations for dosing chemicals in high pressure and low pressure boiler systems. It calculates the dosage of phosphates and trisodium phosphate needed for a high pressure boiler producing 10,000 kg/hr of steam, accounting for blowdown rate. It also calculates the dosage of hydrazine needed for a low pressure boiler with 10,000 kg/hr steam production to control oxygen levels, accounting for concentration of the dosing solution.
Original Description:
hp dosing and lp dosing calculation.
Original Title
hp lp
Copyright
© © All Rights Reserved
Available Formats
DOCX, PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentThe document provides calculations for dosing chemicals in high pressure and low pressure boiler systems. It calculates the dosage of phosphates and trisodium phosphate needed for a high pressure boiler producing 10,000 kg/hr of steam, accounting for blowdown rate. It also calculates the dosage of hydrazine needed for a low pressure boiler with 10,000 kg/hr steam production to control oxygen levels, accounting for concentration of the dosing solution.
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
Download as docx, pdf, or txt
0 ratings0% found this document useful (0 votes)
71 views1 pageHP Dosing Calculation
HP Dosing Calculation
Uploaded by
Jegannathan NambukumarThe document provides calculations for dosing chemicals in high pressure and low pressure boiler systems. It calculates the dosage of phosphates and trisodium phosphate needed for a high pressure boiler producing 10,000 kg/hr of steam, accounting for blowdown rate. It also calculates the dosage of hydrazine needed for a low pressure boiler with 10,000 kg/hr steam production to control oxygen levels, accounting for concentration of the dosing solution.
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
Download as docx, pdf, or txt
You are on page 1of 1
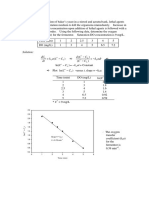
All dosage equation are based on C1Q1=C2Q2Where C= concentration of
chemical, Q= Flow rateOr, Chemical feed solution (mg/l) X feed pump
output(gallon/day) = Desired chemical dose(mg/l) X plantflow rate
HP dosing calculation
Steam tph=10000 kghTDS in feed water=.1TDS permitted in boiler= 25
ppmPercentage blow down = 100X.1/(25-0.1)=0.4%Actual blow down rate =
10000X 0.4/1000kgr=40kgrPPM PO4 dose=4ppmLoss of PO4 in
BD=4X40/1000g/h=0.16 gm/hrLoss of TSP in blowdown=0.16X4=0.64g/hrTSP
consumption/day=0.64X24/1000 kg/day=0.1536
LP dosing calculation
Steam tph=10000 kghO2 in feed water after deaeration @ 105
0
C=0.02ppmRequired oxygen level in feed water = 0.002 ppmOxygen to be
removed = 0.02
–
0.002 ppm =0.018N2H4 rqrd(30times) = 30 X 0.018 ppm=0.54 ppmResidual
N2H4 to be kept = 0.02 ppmTotal N2H4 rqrd = 0.54 + 0.02=0.56 ppmTotal
N2H4 rqrd in Kg/Kg of evaporation = 0.56 X 0.000001 kg/kg
= 0.00000056 kg/kgN2H4 rqrd/hr = 10000 X 0.00000056 = 0.0056 kg/hrConc. Of
N2H4 soln = 35%N2H4 to be dosed @ 35 % conc = .0056 X 100/35 kg/hr=0.016 kg
/hrN2H4 Consumption/day = 0.016 X 24 Kg/day = 0.384 kg/day
You might also like
- LP HP Dosing CalculationDocument1 pageLP HP Dosing Calculationvikas_be1750% (4)
- Design Capacity Calculation of 3X156 KPH Electro Chlorination SystemDocument2 pagesDesign Capacity Calculation of 3X156 KPH Electro Chlorination Systemshriyans5388100% (2)
- Regular Paper 2 - Set A: Section - Ii: Short Descriptive QuestionsDocument23 pagesRegular Paper 2 - Set A: Section - Ii: Short Descriptive QuestionsYogaAnandNo ratings yet
- Cell Kinetics and Fermenter Design 2Document17 pagesCell Kinetics and Fermenter Design 2rhia81% (16)
- LP HP Dosing Calculation - PDF - Physical Sciences - Chemical SubstancesDocument1 pageLP HP Dosing Calculation - PDF - Physical Sciences - Chemical SubstancesV U P RaoNo ratings yet
- Improvement of PH of RO Product Water: BasisDocument3 pagesImprovement of PH of RO Product Water: BasisPraneeth KoppineniNo ratings yet
- Water CalculationDocument6 pagesWater CalculationRamkiNo ratings yet
- Material Balance: Reactor: From The Above ReactionDocument3 pagesMaterial Balance: Reactor: From The Above ReactionHoucine BendaoudNo ratings yet
- Solved Problems in Chemical Process CalculationsDocument19 pagesSolved Problems in Chemical Process CalculationsPrincess Janine CatralNo ratings yet
- Pedia NotesDocument7 pagesPedia Noteseyakoy100% (4)
- InputsDocument7 pagesInputsAnonymous GwUZTBNo ratings yet
- BEV2036 EV414 Exercises On Energy and Materials BalanceDocument4 pagesBEV2036 EV414 Exercises On Energy and Materials BalanceSam KhorNo ratings yet
- Pedia NotesDocument8 pagesPedia NotesQurrataini IbanezNo ratings yet
- Useful Formulas in Fluid TherapyDocument2 pagesUseful Formulas in Fluid TherapySharmela BrijmohanNo ratings yet
- Anaerobic Treatment of Municipal Wastewater (10,000 Liter/Day) With A Staged Anaerobic Fluidized Membrane Bioreactor (Saf-Mbr) SystemDocument11 pagesAnaerobic Treatment of Municipal Wastewater (10,000 Liter/Day) With A Staged Anaerobic Fluidized Membrane Bioreactor (Saf-Mbr) SystemAzam HussainNo ratings yet
- 1Minor1Cvl100 Sem1Document3 pages1Minor1Cvl100 Sem1Arnav TiwariNo ratings yet
- Jar Testing of Chemical DosagesDocument32 pagesJar Testing of Chemical DosagesRolando CortazarNo ratings yet
- 2015 CVL300 Tutorial 4 SolutionDocument7 pages2015 CVL300 Tutorial 4 SolutionAhmed Abuzour100% (2)
- Sample Problems of Chromium Hexa and CyanideDocument3 pagesSample Problems of Chromium Hexa and CyanideMichelle HutamaresNo ratings yet
- Dose The Right Amount of ChemicalsDocument3 pagesDose The Right Amount of ChemicalsrajachemNo ratings yet
- Chemical CalculationDocument4 pagesChemical CalculationKiệt Trần ThúcNo ratings yet
- Converting Occupational Exposure Limits From MGDocument6 pagesConverting Occupational Exposure Limits From MGmajedzNo ratings yet
- 1.0 Industrial - Waste - Water - Treatment-M. - SC - Student-2023-NewDocument63 pages1.0 Industrial - Waste - Water - Treatment-M. - SC - Student-2023-NewUday Kumar DasNo ratings yet
- SEPARATIONSDocument13 pagesSEPARATIONSR ZoNo ratings yet
- SBR Design PDFDocument7 pagesSBR Design PDFmkdholkiya100% (1)
- Methanolinjection - XLS: This Spreadsheet Calculates The Minimum Amount of Required Inhibotor Usage. InstructionsDocument3 pagesMethanolinjection - XLS: This Spreadsheet Calculates The Minimum Amount of Required Inhibotor Usage. Instructionsmath62210100% (1)
- 05-Chem Eng Tools 2016Document11 pages05-Chem Eng Tools 2016arif thoha bariklanaNo ratings yet
- Manufacture of CumeneDocument46 pagesManufacture of CumeneG Vamsee KrishnaNo ratings yet
- Engineering Thermodynamics - Open Systems ExercisesDocument1 pageEngineering Thermodynamics - Open Systems Exercisesloli XxxxNo ratings yet
- Unit III Trickling Filter DesignDocument1 pageUnit III Trickling Filter Designधीरज कुमारNo ratings yet
- Concentration Units: Chemicals in Liquid SampleDocument9 pagesConcentration Units: Chemicals in Liquid Samplethuaiyaalhinai0% (1)
- Activated Sludge Process Design2Document2 pagesActivated Sludge Process Design2Vipin YadavNo ratings yet
- HVAC Useful FormulasDocument1 pageHVAC Useful Formulasnareshbv749No ratings yet
- Presentation 1Document22 pagesPresentation 1Cherry ObiasNo ratings yet
- HW 3 BharatDocument3 pagesHW 3 BharatBharat MahajanNo ratings yet
- Exam PredictionDocument24 pagesExam PredictionTesfay K HaileNo ratings yet
- WRD Ot Pounds FormulaDocument21 pagesWRD Ot Pounds FormulaFredi Cari CarreraNo ratings yet
- Sample ProblemsDocument18 pagesSample ProblemsEggy ThreekingsNo ratings yet
- Feed:: PropyleneDocument7 pagesFeed:: PropyleneHamza AliNo ratings yet
- Nptel Ac in Courses 105104102 Lecture 2026 HTMDocument2 pagesNptel Ac in Courses 105104102 Lecture 2026 HTMGazi Mohammad ParvezNo ratings yet
- 05-Chem Eng Tools 2016Document11 pages05-Chem Eng Tools 2016Hazel Raditya MizumareruNo ratings yet
- Set3ans 10Document5 pagesSet3ans 10amalinaishahNo ratings yet
- Hydro-Flo Technologies, Inc.: Chemical Name Acid - Typical Project Name XX-XXXXDocument4 pagesHydro-Flo Technologies, Inc.: Chemical Name Acid - Typical Project Name XX-XXXXPutra TatorNo ratings yet
- CalculationsDocument14 pagesCalculationsPratyush BadrinarayanNo ratings yet
- Acr Hw03nckuDocument7 pagesAcr Hw03nckuBig-PhiLNo ratings yet
- Commonly Used HVAC Formulae and Conversions: Air SideDocument5 pagesCommonly Used HVAC Formulae and Conversions: Air SideWisam AnkahNo ratings yet
- Hvac FormulaeDocument5 pagesHvac FormulaeanishNo ratings yet
- Chem 1070 LabDocument9 pagesChem 1070 LabAcalaNo ratings yet
- ChE 195 Problem Set No. 2Document1 pageChE 195 Problem Set No. 2Jahz ChannelNo ratings yet
- Test 1 Sample QuestionDocument7 pagesTest 1 Sample QuestionAnonymous GsiB6EMGNo ratings yet
- MATERIAL BALANCE Distillation ColumnDocument9 pagesMATERIAL BALANCE Distillation ColumnRajeev Kumar DohareNo ratings yet