Professional Documents
Culture Documents
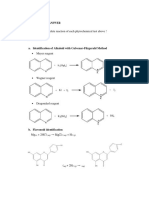
Vii. Result of The Experiment
Vii. Result of The Experiment
Uploaded by
ulfa afifah0 ratings0% found this document useful (0 votes)
34 views4 pages1. An experiment was conducted to create an iron hydroxide colloid by adding iron chloride to water, which turned the solution hazel colored, indicating the formation of an iron hydroxide colloid through condensation.
2. Starch flour was dispersed in water to create a colloid, seen by the white color of the filtrate. Adding iodine turned the solution purple, confirming the presence of a colloid.
3. When benzene and water were mixed, they separated into two immiscible layers due to their polar differences. Adding sodium oleate allowed the solutions to mix by forming an emulsion colloid.
Original Description:
Laporan Resmi Colloid
Original Title
lapsem coloid
Copyright
© © All Rights Reserved
Available Formats
DOCX, PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this Document1. An experiment was conducted to create an iron hydroxide colloid by adding iron chloride to water, which turned the solution hazel colored, indicating the formation of an iron hydroxide colloid through condensation.
2. Starch flour was dispersed in water to create a colloid, seen by the white color of the filtrate. Adding iodine turned the solution purple, confirming the presence of a colloid.
3. When benzene and water were mixed, they separated into two immiscible layers due to their polar differences. Adding sodium oleate allowed the solutions to mix by forming an emulsion colloid.
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
Download as docx, pdf, or txt
0 ratings0% found this document useful (0 votes)
34 views4 pagesVii. Result of The Experiment
Vii. Result of The Experiment
Uploaded by
ulfa afifah1. An experiment was conducted to create an iron hydroxide colloid by adding iron chloride to water, which turned the solution hazel colored, indicating the formation of an iron hydroxide colloid through condensation.
2. Starch flour was dispersed in water to create a colloid, seen by the white color of the filtrate. Adding iodine turned the solution purple, confirming the presence of a colloid.
3. When benzene and water were mixed, they separated into two immiscible layers due to their polar differences. Adding sodium oleate allowed the solutions to mix by forming an emulsion colloid.
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
Download as docx, pdf, or txt
You are on page 1of 4
VII.
RESULT OF THE EXPERIMENT
Num Experiment Procedure Result of Experiment Assumption/ Conclusion
. Reaction
Exp
Before After
1 - Aquades - FeCl3(aq) + Fe(OH)3 ia a
colorless After we add colloid that
3H2O(l) →
10 drop of inclued a kind a
FeCl3 became Fe(OH)3(aq) + sol. This
- FeCl3 :
Hazel colour experiment the
Yellow 3HCl(aq)
making of
colloids by
condensation
2 - Aquades : - After mix - The powder of To make a colloid
Colorless starch flour starch flour can be done with
- Starch flour : and aquades more refined dispersion.
White the colour Than filtrate A Dispersion is how
- Iod Solution become white after pound it to make colloids
: yellow (filtrate A) -the filtrate B is by breaking large
and more more turbid than particles into tiny
turbid in ( filtrate A , cause particles, one them
Filtrate B) we soft particle of by mechanical of
use refined starch flour still grinding
starch flour involved from
- After filtrate filteritation
B added iod - Filtrate B+ iod
solution solution → purple
became (base)
purple colour
3 - After mix - Benzene cant Benzena as non
- Benzena :
benzend with mix with polar solution
colorless
aquades the aquades and H2O as
- Aquades :
colorless solution - Benzena: polar, so the
- Na-Oleat :
became two nonpolar, solution can’t
colorless
- NaOH : layer, still 2 aquades : polar mix.
colorless
layer, top of - Na-oleat can Na-oleat hasten
layer (turbid) mix benzene separation and
and in below with aquades NaOH mix all
(colorless) - NaoH can mix solution.
added benzene if Na- To make a
NaOH, the Oleat fail mix colloid can be
layer was them done with
mixed be emulsiom
homogen
solution
4 - Aquades - Aquades+ The filtrate of Norit absorbs
colorless sugar : this solution is
impurities on
brown pale colorless, cause
- Sugar : dust - Sugar carbon adsorb sugar and norit
white solution+ the colour
absorbing the
norit : black
- Norit : black - After colour because
heated : be
the colour is
3 layer, on
surface and given norit can
bottom
be more clear .
(carbon)
and in the
middle was
colorless
- After
filtered, the
filtrate :
colorless
You might also like
- The Comparison of Three Isomers of ButanolDocument5 pagesThe Comparison of Three Isomers of ButanolLilly0% (2)
- Using Pony in FlaskDocument90 pagesUsing Pony in FlaskPrivilege MudzingeNo ratings yet
- PleadingsDocument231 pagesPleadingspit1xNo ratings yet
- LMR (Last Minute Revision) Chemistry 10TH - 2023 PDFDocument236 pagesLMR (Last Minute Revision) Chemistry 10TH - 2023 PDFSneh BhalodiaNo ratings yet
- Salt Analysis: Step 1: Colour of Salt InferenceDocument5 pagesSalt Analysis: Step 1: Colour of Salt InferenceSarah DillardNo ratings yet
- AQA Chemistry All Chemical Tests TableDocument3 pagesAQA Chemistry All Chemical Tests TableEntertainer SidNo ratings yet
- Azoic ColorDocument4 pagesAzoic ColorMD saifu lislamNo ratings yet
- ReaksiDocument12 pagesReaksiLaila RoikhatulNo ratings yet
- Chem Lab WorkshopDocument10 pagesChem Lab Workshopdaffyjackson32No ratings yet
- ExperimentDocument16 pagesExperimentcloudx chimNo ratings yet
- Anion AnalysisDocument1 pageAnion Analysisaafaf.sdfddfaNo ratings yet
- Qualitative AnalysisDocument4 pagesQualitative AnalysisVictor OkosunNo ratings yet
- Unit AS 2: Further Physical and Inorganic Chemistry and Introduction To Organic Chemistry Content Paragraph Observation Gas TestsDocument12 pagesUnit AS 2: Further Physical and Inorganic Chemistry and Introduction To Organic Chemistry Content Paragraph Observation Gas TestsAnshu MovvaNo ratings yet
- 2022 Sec 3 Qa Notes StudentsDocument11 pages2022 Sec 3 Qa Notes Studentsapi-628191203No ratings yet
- Data Sheet EnglishDocument3 pagesData Sheet Englishhien nguyen quangNo ratings yet
- Viii. Observation Table No. Procedure Observation Result Hyphothesis/Reaction ConclusionDocument6 pagesViii. Observation Table No. Procedure Observation Result Hyphothesis/Reaction ConclusionUtari Ika CahyaniNo ratings yet
- Determination of Cation and Anion of The SubstancesDocument12 pagesDetermination of Cation and Anion of The SubstancesSunny02No ratings yet
- Laboratory Report CHM 153 (Inorganic Chemistry)Document9 pagesLaboratory Report CHM 153 (Inorganic Chemistry)Muhd Mirza Hizami100% (1)
- VII. Observation Result Experiment 1Document12 pagesVII. Observation Result Experiment 1Anggraini Nugroho PNo ratings yet
- Experiment Result Nu. Procedure Observationt Result Reaction Conclusion BeforeDocument6 pagesExperiment Result Nu. Procedure Observationt Result Reaction Conclusion BeforeAnggraini Nugroho PNo ratings yet
- Salt Analysis: Basic Radicals Group Determination Experiments Observation InferenceDocument5 pagesSalt Analysis: Basic Radicals Group Determination Experiments Observation InferenceFouzul Mobin KhanNo ratings yet
- Experiment 3: Inorganic Qualitative AnalysisDocument9 pagesExperiment 3: Inorganic Qualitative AnalysisNurfariha SafarNo ratings yet
- Reactions of Aldehydes and KetonesDocument9 pagesReactions of Aldehydes and Ketonesmoon star angelNo ratings yet
- Updated 2Document15 pagesUpdated 2Balaji BNo ratings yet
- Chemistry Salt Analysis CheatsheetDocument5 pagesChemistry Salt Analysis CheatsheetYug VanviNo ratings yet
- Chapter 16 - SaltsDocument3 pagesChapter 16 - SaltsFaiza RizwanNo ratings yet
- Topic 9 NotesDocument3 pagesTopic 9 Notesmarin tamNo ratings yet
- Anion AnalysisDocument1 pageAnion AnalysisSarah LouieNo ratings yet
- Gain Familiarity With Some of The Acid-Base, Oxidation-Reduction and Complexion Reaction of The Elements of The First Transition Series.Document11 pagesGain Familiarity With Some of The Acid-Base, Oxidation-Reduction and Complexion Reaction of The Elements of The First Transition Series.FarahSyazwani100% (1)
- Exp 1 Colloids SiapDocument8 pagesExp 1 Colloids SiapFairuz Naim Z100% (1)
- Slides 2016 Qualitative Analysis UpdatedDocument58 pagesSlides 2016 Qualitative Analysis UpdatedsherineNo ratings yet
- 0 - Organic and Inorganic Tests For AS PDFDocument8 pages0 - Organic and Inorganic Tests For AS PDFAbed AymanNo ratings yet
- ss3 1st TermDocument45 pagesss3 1st TermVictor Okosun100% (4)
- Lab Report 7Document7 pagesLab Report 7Iena KasimNo ratings yet
- Inorganic Chemistry - Lab Report 2&3Document6 pagesInorganic Chemistry - Lab Report 2&3AlpNo ratings yet
- C12 AnalysisDocument21 pagesC12 AnalysiskhôiNo ratings yet
- Functional Group Analysis 1Document17 pagesFunctional Group Analysis 1حسين أحمد حسينNo ratings yet
- Scheme For The Analysis of Inorganic Mixtures2Document12 pagesScheme For The Analysis of Inorganic Mixtures2MinuNo ratings yet
- Cation Tests Form 4 KSSM Chemistry Chapter 7 Acid, Base and SaltDocument24 pagesCation Tests Form 4 KSSM Chemistry Chapter 7 Acid, Base and SaltEsther Randy100% (2)
- Salt AnalysisDocument8 pagesSalt AnalysisSyed Mohdammad AliNo ratings yet
- Chemistry Salt Analysis Cheatsheet: AnionsDocument4 pagesChemistry Salt Analysis Cheatsheet: AnionsAll About LifeNo ratings yet
- Scheme For Systematic Analysis of A Mixture Containing Two SaltsDocument10 pagesScheme For Systematic Analysis of A Mixture Containing Two SaltsMuhammad Shaheer JavedNo ratings yet
- Qualitative Analysis Guide Sheet For Chemistry PracticalDocument4 pagesQualitative Analysis Guide Sheet For Chemistry PracticalConnor EnglandNo ratings yet
- Chem Labnotebook Inorg Writeup XiDocument14 pagesChem Labnotebook Inorg Writeup Ximaitrayee.guharoyNo ratings yet
- Salt Analysis, ClassxiiDocument11 pagesSalt Analysis, ClassxiiDhairya KumarNo ratings yet
- Tutorial 02 A Qualitative Flowchart OKDocument30 pagesTutorial 02 A Qualitative Flowchart OKFiqran Ratu WaraNo ratings yet
- Chemistry Practical 2023 - XIIDocument19 pagesChemistry Practical 2023 - XIIPhsyics XINo ratings yet
- Imp. Practical Chem. KnowledgeDocument4 pagesImp. Practical Chem. KnowledgedebanivkashyapNo ratings yet
- Chemitry Lab - Identification of AnionsDocument3 pagesChemitry Lab - Identification of AnionsMiguel johnsonNo ratings yet
- Selina Solutions Concise Chemistry For Class 10 Chapter 4Document6 pagesSelina Solutions Concise Chemistry For Class 10 Chapter 4Akash SinghNo ratings yet
- Chem 305 Lab 2Document7 pagesChem 305 Lab 2Gobe JamNo ratings yet
- 9 - Practical ChemistryDocument2 pages9 - Practical ChemistrysamnoobplayzNo ratings yet
- SALTANALYSISLABMANUAL 9owo8xhodwx68kh7Document13 pagesSALTANALYSISLABMANUAL 9owo8xhodwx68kh7Lalith AdityaNo ratings yet
- Experiment 1: Chemical ReactionsDocument11 pagesExperiment 1: Chemical ReactionsAmphotorite QuynhNo ratings yet
- Chemistry Salt Analysis CheatsheetDocument4 pagesChemistry Salt Analysis CheatsheetyashvpNo ratings yet
- Chemistry Salt Analysis CheatsheetDocument4 pagesChemistry Salt Analysis CheatsheetSumit Dhall74% (50)
- Lampiran Perhitungan Vit CDocument3 pagesLampiran Perhitungan Vit Culfa afifahNo ratings yet
- Lesson PlanDocument10 pagesLesson Planulfa afifahNo ratings yet
- Skenario InkuiriDocument2 pagesSkenario Inkuiriulfa afifahNo ratings yet
- A. Experiment TitleDocument11 pagesA. Experiment Titleulfa afifahNo ratings yet
- A. Experiment Title B. Day/ Date of Experiment A.M. C. Finished The Experiment: Friday, 8 A.M. D. Experiment PurposeDocument1 pageA. Experiment Title B. Day/ Date of Experiment A.M. C. Finished The Experiment: Friday, 8 A.M. D. Experiment Purposeulfa afifahNo ratings yet
- Question Answer FitokimiaDocument6 pagesQuestion Answer Fitokimiaulfa afifahNo ratings yet
- Metal Properties (Electropositive) and Nonmetallic Properties (Electronegative)Document7 pagesMetal Properties (Electropositive) and Nonmetallic Properties (Electronegative)ulfa afifahNo ratings yet
- Calculation 1. Making 0.1M HCL SolutionDocument2 pagesCalculation 1. Making 0.1M HCL Solutionulfa afifahNo ratings yet
- Sitasi Online - Zotero - PKU 17 - Ulfa Nur Afifah - 17030194033Document22 pagesSitasi Online - Zotero - PKU 17 - Ulfa Nur Afifah - 17030194033ulfa afifahNo ratings yet
- Documentation No Steps Picture Tools and MaterialsDocument5 pagesDocumentation No Steps Picture Tools and Materialsulfa afifahNo ratings yet
- Isolasi JaheDocument4 pagesIsolasi Jaheulfa afifahNo ratings yet
- Alur Dan Bahan KFDocument2 pagesAlur Dan Bahan KFulfa afifahNo ratings yet
- Lesson Plan Reaction RateDocument11 pagesLesson Plan Reaction Rateulfa afifahNo ratings yet
- F. Tools and Materials A. ToolsDocument6 pagesF. Tools and Materials A. Toolsulfa afifahNo ratings yet
- Coll OidDocument4 pagesColl Oidulfa afifahNo ratings yet
- Alur Pembuatan Larutan ORLABDocument3 pagesAlur Pembuatan Larutan ORLABulfa afifahNo ratings yet
- Mortgage Loan Disclosure Statement - GFEDocument3 pagesMortgage Loan Disclosure Statement - GFEafncorpNo ratings yet
- PAPS 1001: Computer Information Systems Environment - Stand-Alone Personal ComputersDocument8 pagesPAPS 1001: Computer Information Systems Environment - Stand-Alone Personal ComputersJomaica AmadorNo ratings yet
- Edinzon Fernando Yangua-Calva, A088 190 231 (BIA Nov. 28, 2011)Document5 pagesEdinzon Fernando Yangua-Calva, A088 190 231 (BIA Nov. 28, 2011)Immigrant & Refugee Appellate Center, LLCNo ratings yet
- Soliswatt Poly Module 310 330WDocument2 pagesSoliswatt Poly Module 310 330Wyu wongNo ratings yet
- Difference Betwn Gross Salary, CTCDocument8 pagesDifference Betwn Gross Salary, CTCabdulyunus_amirNo ratings yet
- Experiment 13 PPDocument3 pagesExperiment 13 PPMohit Patil (PC)No ratings yet
- Chapter 6 CompleteDocument30 pagesChapter 6 Completeduniya t vNo ratings yet
- Projects Synopsis: 1. Surendra Singh (08esmec104) 2. Surender Singh (08esmec102)Document7 pagesProjects Synopsis: 1. Surendra Singh (08esmec104) 2. Surender Singh (08esmec102)Hamza HebNo ratings yet
- Computer MCQ FOR BANK PO & CLERK EXAMDocument26 pagesComputer MCQ FOR BANK PO & CLERK EXAMShubhajit Nandi80% (5)
- Fundamentals of Information Systems PDFDocument164 pagesFundamentals of Information Systems PDFharshithaNo ratings yet
- Admixtures 2Document34 pagesAdmixtures 2MudduKrishna shettyNo ratings yet
- EVALUATING OTHERS DRAFT PowerPoint PresentationDocument16 pagesEVALUATING OTHERS DRAFT PowerPoint PresentationtrixiaagelzNo ratings yet
- BM60112 - CFFA - FP 2021 - v1.0Document2 pagesBM60112 - CFFA - FP 2021 - v1.0MousumiNo ratings yet
- Case Digests in Statutory Construction Philippine Law StudentDocument58 pagesCase Digests in Statutory Construction Philippine Law StudentJoshua Emmanuel100% (5)
- Central Bank v. Tolentino: 125 SCRA 122Document15 pagesCentral Bank v. Tolentino: 125 SCRA 122alyza burdeosNo ratings yet
- BRM v3-1 Taxonomy 20130615Document52 pagesBRM v3-1 Taxonomy 20130615kevinzhang2022No ratings yet
- Rulloda Vs ComelecDocument4 pagesRulloda Vs ComelecZarah MaglinesNo ratings yet
- Business Agreement: Between M/S METROPOLIS HEALTHCARE LIMITED, Having Its RegisteredDocument4 pagesBusiness Agreement: Between M/S METROPOLIS HEALTHCARE LIMITED, Having Its RegisterednimeshnarnaNo ratings yet
- PGDM Admissions Brochure Chennai 2021 23Document32 pagesPGDM Admissions Brochure Chennai 2021 23shahulNo ratings yet
- Time Travel: Colonization vs. Globalization: GAC008 Assessment Event 4: Academic Research EssayDocument4 pagesTime Travel: Colonization vs. Globalization: GAC008 Assessment Event 4: Academic Research EssayMiguel BrionesNo ratings yet
- Trends in Food Science & Technology: John Spink, David L. Ortega, Chen Chen, Felicia WuDocument6 pagesTrends in Food Science & Technology: John Spink, David L. Ortega, Chen Chen, Felicia WuDubravka UžarNo ratings yet
- Rad-Studio-Feature-Matrix 10.4Document25 pagesRad-Studio-Feature-Matrix 10.4endickhkNo ratings yet
- 8.building Analysis ModelDocument64 pages8.building Analysis ModeltierSargeNo ratings yet
- Business PlanDocument16 pagesBusiness Planareolajoshua13No ratings yet
- G120XA CatalogDocument88 pagesG120XA CatalogYogeshPatelNo ratings yet
- Sand Moulded Castings Defects, Causes, Remedies - Review PaperDocument5 pagesSand Moulded Castings Defects, Causes, Remedies - Review PapersaranyaNo ratings yet
- # Installation Guide PDFDocument1 page# Installation Guide PDFscarletNo ratings yet
- Inflection Points and Bar Cut OffDocument8 pagesInflection Points and Bar Cut Offjohn sorianoNo ratings yet