Professional Documents
Culture Documents
MT IMT Notes Part 3
MT IMT Notes Part 3
Uploaded by
Angela ReyesOriginal Description:
Original Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
MT IMT Notes Part 3
MT IMT Notes Part 3
Uploaded by
Angela ReyesCopyright:
Available Formats
TOTAL QUALITY MANAGEMENT Measure the quantity of a particular substance in a sample
Measurements should be both accurate and precise
QUALITATIVE EXAMINATIONS METHODS
Quality Management Examinations that do not have numerical results:
o Growth or no growth
o Positive or Negative
Quality System o Reactive or Non-Reactive
o Color change
Quality SEMI-QUANTITATIVE EXAMINATION METHODS
Assurance
Results are expressed as an estimate of the measured
substance:
o “trace amount”, “moderate amount,” or “1+, 2+,
or 3+”
Quality Control o number of cells per microscopic field
o titers and dilutions in serologic tests
Quality Management: Policy, Objective, Commitment and Direction
Quality System: Organization, Structure, Responsibility, Accountability
Quality Assurance: Internal QA, External QA
Quality Control: Operational and Technical Activities on Fulfilling Quality
Requirements
TQM FRAMEWORK INVOLVES
Quality Laboratory Processes (QLP)
Quality Control (QC)
Quality Assessment (QA)
Quality Improvement (QI)
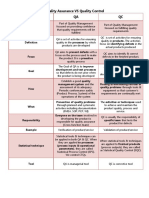
Quality Planning (QP) QUALITY ASSURANCE VS QUALITY CONTROL
Quality Goals Quality Assurance an overall management plan to guarantee
the integrity of data (The “System”)
QUALITY CONTROL AND QUALITY ASSURANCE Quality Control a series of analytical measurements to
assess the quality of the analytical data (The “Tools”)
DEFINITION (1)
Quality Control - QC refers to those measures that must be
included in each assay to verify that the test is working
properly.
Quality Assurance - QA is defined as the overall program
that ensures that the final results reported by the laboratory
are correct
"The aim of quality control is simply to ensure that the results
generated by the test are correct. However, quality
assurance is concerned with much more: that the right test is
carried out on the right specimen, and that the right result
and right interpretation is delivered to the right person at the
right time"
DEFINITION (2)
DEFINITON (QC) Quality Assessment - Quality assessment (also known as
Quality Control (QC) is part of quality management focused proficiency testing) is a means to determine the quality of the
on fulfilling quality requirements ISO 9000:2000 (3.4.10) results generated by the laboratory. It is usually an external
QC is examining “control” materials of known substances evaluation of the laboratory's performance. Internal quality
along with patient samples to monitor the accuracy and assessment programs can also be instituted. Quality
precision of the complete examination (analytic) process. assessment is a challenge to the effectiveness of the QA and
QC programs.
PURPOSE Quality Assessment may be external or internal. Examples of
The goal of QC is to detect errors and correct them before external programs includes NEQAS, HKMTAA and Q-probes
patients’ results are reported
VARIABLES THAT AFFECT THE QUALITY OF RESULTS
QUANTITATIVE EXAMINATIONS
The educational background and training of the laboratory WESTGARD RULES
personnel The formulation of Westgard rules were based on statistical
The condition of the specimens methods. Westgard rules are commonly used to analyses
The controls used in the test runs data in Shewhart control charts.
The interpretation of the results Westgard rules are used to define specific performance limits
The transcription of results for a particular assay and can be used to detect both random
The reporting of results and systematic errors.
There are six commonly used Westgard rules of which three
ERRORS IN MEASUREMENT are warning rules and the other three mandatory rules.
True value - this is an ideal concept which cannot be The violation of warning rules should trigger a review of test
achieved. procedures, reagent performance and equipment calibration.
Accepted true value - the value approximating the true value, The violation of mandatory rules should result in the rejection
the difference between the two values is negligible. of the results obtained with patients’ serum samples in that
Error - the discrepancy between the result of a measurement assay.
and the true (or accepted true value).
LEVEY JENNINGS CHART
RANDOM ERROR
An error which varies in an unpredictable manner, in
magnitude and sign, when a large number of measurements
of the same quantity are made under effectively identical
conditions.
Random errors create a characteristic spread of results for
any test method and cannot be accounted for by applying
corrections. Random errors are difficult to eliminate but
repetition reduces the influences of random errors.
Examples of random errors include errors in pipetting and
changes in incubation period. Random errors can be
minimized by training, supervision and adherence to
standard operating procedures.
SYSTEMATIC ERROR
An error which, in the course of a number of measurements WARNING RULES
of the same value of a given quantity, remains constant Warning 12SD: It is violated if the IQC value exceeds the
when measurements are made under the same conditions, mean by ±2SD. It is an event likely to occur normally in less
or varies according to a definite law when conditions change. than 5% of cases.
Systematic errors create a characteristic bias in the test Warning 22SD: It detects systematic errors and is violated
results and can be accounted for by applying a correction. when two consecutive IQC values exceed the mean on the
Systematic errors may be induced by factors such as same side of the mean by ±2SD.
variations in incubation temperature, blockage of plate Warning 41SD: It is violated if four consecutive IQC values
washer, change in the reagent batch or modifications in exceed the same limit (mean ±1SD) and this may indicate
testing method. the need to perform instrument maintenance or reagent
calibration.
SYSTEMATIC VS. RANDOM ERRORS
Systematic Error – Available error due to controllable
variables in a measurement
Random Errors – Unavailable errors that are always present
in any measurement. Impossible to eliminate
TRUE VALUE VS. MEASURED VALUE
True Value – The known, accepted value of a quantifiable
properly
Measured Value – The result of an individual’s measurement
of a quantifiable properly
ACCURACY VS. PRECISION
Accuracy – How well a measurement agrees with on an
accepted value
Precision – How well a series of measurements agree with
each other
MANDATORY RULES
Mandatory 13SD: It is violated when the IQC value exceeds TYPES OF ETHICS
the mean by 3SD. The assay run is regarded as out of General Ethics - presents truths about human acts from
control. which the general principles of morality is deduced
Mandatory R4SD: It is only applied when the IQC is tested in Special Ethics – involve the application of the principles of
duplicate. This rule is violated when the difference in SD general ethics in different departments of human activity both
between the duplicates exceeds 4SD. at the individual and social levels
Mandatory 10x: This rule is violated when the last 10 o Individual Ethics – concerned with GOD, self
consecutive IQC values are on the same side of the mean or and fellow human beings
target value. o Social Ethics – concerned with family, the state
and the world
o Professional Ethics
o Media Ethics
PROFESSIONAL ETHICS
A branch of moral science that deals with how and what a
professional should or should not to do in the workplace
It address the question “What should I do in this situation?”
Intended to bind professions more tightly together around a
shared standard values
Knowledge on professional ethics can guide staff in
analyzing assumptions and arriving at ethical decisions
OBJECTIVES OF PROFESSIONAL ETHICS
Perform duties and responsibilities objectively in accordance
with relevant standards and guidelines
Serve in a lawful and honest manner while maintaining high
standards of conduct and character and not engage in acts
discreditable to the profession.
FOLLOW-UP ACTION IN THE EVENT OF A VIOLATION
Maintain the privacy and confidentiality of information
There are three options as to the action to be taken in the
obtained in the course study unless disclosure is required by
event of a violation of a Westgard rule:
a legal authority. Such information shall not be used for
o Accept the test run in its entirety - this usually
personal benefit or released to inappropriate parties.
applies when only a warning rule is violated.
Maintain competency in their respective fields and agree to
o Reject the whole test run - this applies only
undertake only those activities which one can reasonably
when a mandatory rule is violated.
expect to complete with professional competence.
o Enlarge the greyzone and thus re-test range for
Perform tasks with dull confidence, absolute reliability and
that particular assay run - this option can be
accuracy
considered in the event of a violation of either a
Be dedicated to the use of clinical laboratory science to
warning or mandatory rule.
promote life and for the benefit of mankind
CODE OF ETHICS OF MEDICAL
TECHNOLOGIST IN THE PHILIPPINES
AS I ENTER INTO THE PRACTICE OF MEDICAL TECHNOLOGY I
SHALL:
Accept the responsibilities inherent to being a professional
Avoid associating or being identified with any enterprise of
questionable character
Work and act in a strict spirit of fairness to employer, clients,
contractors, employees and in a spirit of personal
helpfulness and fraternity toward other members of the
profession
Use only honorable means of competition for professional
employment or services and shall refrain form unfairly
injuring, directly or indirectly, the professional reputation,
projects or business of a fellow medical technologist
MEDICAL TECHNOLOGY ETHICS Accept employment from more than one employer only when
there is no conflict of interest
Are ethical problems associated with medical practices and Perform professional work in a manner that merits full
bioscience confidence and trust carried out with absolute reliability,
Ethical problems are brought about by the advances in accuracy, fairness and honesty
biotechnology and its power over life and death Review the professional work of other medical technologists,
A field of applied ethics that studies moral values and when requested, fairly and in confidence whether they are
judgements as they apply to medical technology subordinates or employees, authors of proposals for grants
Are as set of norms, values, principles that serve as or contracts, authors of technical papers or other publications
guidelines for medical technology practitioners in making or involved in litigation
decisions in clinical settings
Advance the profession by exchanging general information
and experience with fellow medical technologists and other
professionals and by contributing to the work of professional
organizations
Restrict my praises, criticisms, views and opinions within
constructive limits and shall not use the knowledge I know for
selfish ends
Treat any information I acquired about individuals in the
course of my work as strictly confidential, and may be
divulged only to authorized persons or entities or with
consent of the individual when necessary
Report any infractions of these principles of professional
conduct to the authorities responsible of enforcement of
applicable laws or regulations, or to the Ethics Committee of
the Philippine Association of Medical Technologists
As may be appropriate to these principles, I hereby
subscribe and pledge to conduct myself at all times in a
manner befitting the dignity of my profession.
MORAL PRINCIPLES
Autonomy – Patient has the right to refuse or approve
treatment
Beneficence – you have to do whats beneficial to patient
Non-maleficence – you should do not harm patients
Justice – fairness / equality
Respect for dignity
Truthfulness and Honesty – dedication of Medical
Technologist to work
Stewardship - management or care of something, particularly
the kind that works.
VALUES OF A MEDICAL TECHNOLOGIST
Punctuality
Integrity
Commitment
Honesty
Loyalty
You might also like
- Blood Bank (2 Week) : Antibody ScreensDocument5 pagesBlood Bank (2 Week) : Antibody ScreensAngela Reyes100% (1)
- Chapter 1 - Part 1 Introduction To Quality AssuranceDocument25 pagesChapter 1 - Part 1 Introduction To Quality AssuranceLily ERc Peter100% (4)
- Amadeus TechRef Insurance Pricing 13.1 002Document68 pagesAmadeus TechRef Insurance Pricing 13.1 002Andrei GoguNo ratings yet
- Introduction To Participatory Assessment of Disaster Risk PDFDocument6 pagesIntroduction To Participatory Assessment of Disaster Risk PDFJune AlapaNo ratings yet
- Quality Assurance in The Clinical LaboratoryDocument1 pageQuality Assurance in The Clinical LaboratoryanaodtohanNo ratings yet
- Quality ControlDocument53 pagesQuality ControlMayHnin KhaingNo ratings yet
- Quality Assurance Quality Control in Laboratories A ReviewDocument6 pagesQuality Assurance Quality Control in Laboratories A ReviewAlexNo ratings yet
- Quality Control BookDocument122 pagesQuality Control Bookashh BukhariNo ratings yet
- 6 CCHM Trans LecDocument4 pages6 CCHM Trans LecCRUZ, ANNA MARIELLENo ratings yet
- Process Control - Introduction To Quality Control: Anas MaulanaDocument8 pagesProcess Control - Introduction To Quality Control: Anas MaulanaEprhy Adiansyah100% (1)
- LQMS 6 7 8 Quality ControlDocument28 pagesLQMS 6 7 8 Quality Controlrose_almonteNo ratings yet
- Module 7Document12 pagesModule 7hannieya1004No ratings yet
- Unit 8 Quality Assurance ManagementDocument10 pagesUnit 8 Quality Assurance Managementhii385156No ratings yet
- GLP 1 Qa Lab Measurement Results 20180131Document8 pagesGLP 1 Qa Lab Measurement Results 20180131Soumitra BhowmikNo ratings yet
- The Concept of QualityDocument4 pagesThe Concept of Qualitymzulqurnain imran100% (1)
- Process Control - Introduction To Quality Control: Anas MaulanaDocument8 pagesProcess Control - Introduction To Quality Control: Anas MaulanaAnas LaodeNo ratings yet
- MT PRT 2 qm.2Document2 pagesMT PRT 2 qm.2Astrum GlenNo ratings yet
- QC & Qa PDFDocument22 pagesQC & Qa PDFAmr El-BahnasawyNo ratings yet
- Quality ControlDocument12 pagesQuality ControlIshika AggarwalNo ratings yet
- Lesson 6 QUALITY ASSURANCE AND QUALITY CONTROLDocument3 pagesLesson 6 QUALITY ASSURANCE AND QUALITY CONTROLJanielle Medina Fajardo100% (1)
- QualityDocument2 pagesQualityAstrum GlenNo ratings yet
- QC and QADocument9 pagesQC and QAClarisse De GuzmanNo ratings yet
- Simonet 2005Document7 pagesSimonet 2005Frisca SoputraNo ratings yet
- QualityDocument38 pagesQualityserviceNo ratings yet
- Quality Assurance VS Quality Control Category QA QCDocument1 pageQuality Assurance VS Quality Control Category QA QCirc.indusNo ratings yet
- Swi QaDocument3 pagesSwi QapaulNo ratings yet
- 1-Introduction To QualityDocument4 pages1-Introduction To Qualityjp23061979No ratings yet
- QC 3Document32 pagesQC 3Fatma HamaadNo ratings yet
- QM001 Difference Between Quality Assurance and Quality ControlDocument2 pagesQM001 Difference Between Quality Assurance and Quality ControlSandeep KumarNo ratings yet
- Ho - Basic Concepts of Qa & QCDocument13 pagesHo - Basic Concepts of Qa & QCtealyngripoNo ratings yet
- 6 C Intro QCDocument11 pages6 C Intro QCMayowa RichardNo ratings yet
- Process Control:: Introduction To Quality ControlDocument11 pagesProcess Control:: Introduction To Quality ControlThe Engineers EDGE, CoimbatoreNo ratings yet
- QA Vs QCDocument4 pagesQA Vs QCBalaNo ratings yet
- Quality Control in Clinical LaboratoryDocument98 pagesQuality Control in Clinical LaboratoryAnonymous S3wsIptcO100% (2)
- Analitik Kimyada Yöntem Geliştirme Ve Veri Analizi ÖdevDocument18 pagesAnalitik Kimyada Yöntem Geliştirme Ve Veri Analizi ÖdevYağmur SoysalNo ratings yet
- Laboratory ManagementDocument58 pagesLaboratory Management2C Lamsin, Estelle NerieNo ratings yet
- Mangesh and Mansi Quality Assurance UnitDocument27 pagesMangesh and Mansi Quality Assurance UnitSumant SainiNo ratings yet
- Qa QCDocument22 pagesQa QCAnnisa RahmatinaNo ratings yet
- Drug Quality ControlDocument24 pagesDrug Quality ControlmaqhaweNo ratings yet
- PMI & PME Laboratorium HematologiDocument76 pagesPMI & PME Laboratorium Hematologiibnunadhif haryantoNo ratings yet
- MLS Process ControlDocument49 pagesMLS Process ControlAqeela AbuidreesNo ratings yet
- L2 Introduction PDFDocument35 pagesL2 Introduction PDFMahbubur MasnunNo ratings yet
- QA ProgrammeDocument1 pageQA ProgrammeCristina ChamorroNo ratings yet
- Site Supervison CH 6Document14 pagesSite Supervison CH 6Tsegaye GirmaNo ratings yet
- Quality Control Vs AssuranceDocument1 pageQuality Control Vs AssuranceBani MarweinNo ratings yet
- QM MergedDocument76 pagesQM MergedSneha Giji SajiNo ratings yet
- Differentiate Between Quality ControlDocument4 pagesDifferentiate Between Quality ControlfayyazNo ratings yet
- Process Control:: Introduction To Quality ControlDocument11 pagesProcess Control:: Introduction To Quality ControlSaravana kumar NagarajanNo ratings yet
- QCR Chapter 1Document48 pagesQCR Chapter 1praveenkumarnitkkrNo ratings yet
- Quality Assurance & Lab MathDocument6 pagesQuality Assurance & Lab MathMica BernardoNo ratings yet
- Quality Managemnet System of The Histopahology LaboratoryDocument2 pagesQuality Managemnet System of The Histopahology LaboratoryOsannah Irish InsongNo ratings yet
- 6 C Intro QCDocument12 pages6 C Intro QCTakele OliNo ratings yet
- QC Exam QuestionDocument30 pagesQC Exam QuestionMd. Ahedul IslamNo ratings yet
- Iqc Midterms MolinaDocument4 pagesIqc Midterms MolinaJazyl TanNo ratings yet
- Lecture 8 Quality ControlDocument21 pagesLecture 8 Quality Controllight fragranceNo ratings yet
- محاضرة 1 جودةDocument29 pagesمحاضرة 1 جودةAmira HossamNo ratings yet
- Welding 4Document1 pageWelding 4Vijay KrishnanNo ratings yet
- Laboratory StandardsDocument10 pagesLaboratory StandardsStephen CubarNo ratings yet
- Special Communication Emerging Concepts of Quality Assurance in Clinical LaboratoriesDocument3 pagesSpecial Communication Emerging Concepts of Quality Assurance in Clinical LaboratoriesUlfat NiazyNo ratings yet
- Qavsqc: Quality Assurance Quality ControlDocument2 pagesQavsqc: Quality Assurance Quality ControlJayanath SACNo ratings yet
- Lesson 2.4 - Good Quality Control Laboratory PracticeDocument7 pagesLesson 2.4 - Good Quality Control Laboratory PracticeMark Harold GonzalesNo ratings yet
- Quality Control & Quality Assurance in Hematology and Hemostasis TestingDocument22 pagesQuality Control & Quality Assurance in Hematology and Hemostasis TestingBenjamin Lopez Carreras100% (2)
- Assignment#1 - 3a - Group 7Document20 pagesAssignment#1 - 3a - Group 7Angela ReyesNo ratings yet
- Tumor Markers: Blood Group AntigenDocument5 pagesTumor Markers: Blood Group AntigenAngela ReyesNo ratings yet
- MYCODocument59 pagesMYCOAngela ReyesNo ratings yet
- Hema 2Document35 pagesHema 2Angela ReyesNo ratings yet
- HEMA-1 Up To PG 30.-1-31Document31 pagesHEMA-1 Up To PG 30.-1-31Angela ReyesNo ratings yet
- Urine Sediment ConstituentsDocument12 pagesUrine Sediment ConstituentsAngela ReyesNo ratings yet
- Hema DagdagDocument1 pageHema DagdagAngela ReyesNo ratings yet
- Aubf Lec (2 Week) Physical Examination of Urine: Color, Clarity Specific GravityDocument38 pagesAubf Lec (2 Week) Physical Examination of Urine: Color, Clarity Specific GravityAngela ReyesNo ratings yet
- Finals - VirologyDocument14 pagesFinals - VirologyAngela ReyesNo ratings yet
- Pedigree Analysis WorksheetDocument3 pagesPedigree Analysis WorksheetAngela ReyesNo ratings yet
- Ilovepdf MergedDocument56 pagesIlovepdf MergedAngela ReyesNo ratings yet
- Coagulation Screening Procedures:: RD THDocument2 pagesCoagulation Screening Procedures:: RD THAngela ReyesNo ratings yet
- Phagocyte System.: Innate/non-Specific Immune SystemDocument6 pagesPhagocyte System.: Innate/non-Specific Immune SystemAngela ReyesNo ratings yet
- Ilovepdf MergedDocument31 pagesIlovepdf MergedAngela ReyesNo ratings yet
- Midterms - MycologyDocument17 pagesMidterms - MycologyAngela ReyesNo ratings yet
- Myco LQDocument37 pagesMyco LQAngela ReyesNo ratings yet
- Finals - ImmunohematologyDocument20 pagesFinals - ImmunohematologyAngela ReyesNo ratings yet
- Microbiology: T. VaginalisDocument78 pagesMicrobiology: T. VaginalisAngela ReyesNo ratings yet
- 1 Hema LecDocument5 pages1 Hema LecAngela ReyesNo ratings yet
- 1 Hema LecDocument5 pages1 Hema LecAngela ReyesNo ratings yet
- Biochemical Tests: JAPAY, Princess Agatha Mei R. BS Medical Technology 3ADocument5 pagesBiochemical Tests: JAPAY, Princess Agatha Mei R. BS Medical Technology 3AAngela ReyesNo ratings yet
- Blue. 1 or 2 HoursDocument5 pagesBlue. 1 or 2 HoursAngela ReyesNo ratings yet
- Cclec MidtermsDocument65 pagesCclec MidtermsAngela ReyesNo ratings yet
- Histopath Lec (Module 1) : Iintroduction To PathologyDocument17 pagesHistopath Lec (Module 1) : Iintroduction To PathologyAngela ReyesNo ratings yet
- Biochem Test 1.2Document9 pagesBiochem Test 1.2Angela ReyesNo ratings yet
- Histopath Lec (Module 2) : Cellular Adaptations, Cellular Injury and Cell DeathDocument5 pagesHistopath Lec (Module 2) : Cellular Adaptations, Cellular Injury and Cell DeathAngela ReyesNo ratings yet
- BLOOD COLLECTION Part 1 Hema LabDocument7 pagesBLOOD COLLECTION Part 1 Hema LabAngela ReyesNo ratings yet
- 5TH Week Hematocrit DeterminationDocument24 pages5TH Week Hematocrit DeterminationAngela ReyesNo ratings yet
- Histopath Lab (Module 2) : Receiving, Accessioning and Gross Examination of Tissue SamplesDocument5 pagesHistopath Lab (Module 2) : Receiving, Accessioning and Gross Examination of Tissue SamplesAngela ReyesNo ratings yet
- Iec Iso 17025 - 2017Document39 pagesIec Iso 17025 - 2017ngradhapyarisinghaNo ratings yet
- Bioetika Didalam Ilmu Kedokteran Blok 1 UMSUDocument25 pagesBioetika Didalam Ilmu Kedokteran Blok 1 UMSUMuhammad Hafiz MuflihNo ratings yet
- Ststistical Treatment of DataDocument3 pagesStstistical Treatment of DatavhineNo ratings yet
- Katana Viewer A Pi Getting Started GuideDocument30 pagesKatana Viewer A Pi Getting Started GuiderendermanuserNo ratings yet
- CV Jing Jiawen 1Document2 pagesCV Jing Jiawen 1api-437518283No ratings yet
- Driver ServerDocument10 pagesDriver ServerGeraldoadriNo ratings yet
- How To Setup Network Once Ntos 7Document2 pagesHow To Setup Network Once Ntos 7mrlamb711No ratings yet
- Ahlada Group of CompaniesDocument1 pageAhlada Group of CompaniesAhlada Group of CompaniesNo ratings yet
- "IKIGAI - Find Your Purpose For Being": DescriptionDocument3 pages"IKIGAI - Find Your Purpose For Being": DescriptionashindeNo ratings yet
- AFCAT Model Question Paper-IIDocument24 pagesAFCAT Model Question Paper-IIShubham RajNo ratings yet
- Heritage Amruth-Senior HealthDocument31 pagesHeritage Amruth-Senior HealthSubhasreeNo ratings yet
- Modular Quiz 28Document2 pagesModular Quiz 28Justin GarciaNo ratings yet
- 2e Starter Language Focus ReferenceDocument14 pages2e Starter Language Focus ReferenceCami AdornoNo ratings yet
- Can Dy/dx Be Treated As A Fraction?Document3 pagesCan Dy/dx Be Treated As A Fraction?Teresa Villena GonzálezNo ratings yet
- PI Troubleshooting Tips - How To Tune PI Synchronous Scenarios - Process Integration - SCN Wiki PDFDocument3 pagesPI Troubleshooting Tips - How To Tune PI Synchronous Scenarios - Process Integration - SCN Wiki PDFSujith KumarNo ratings yet
- Comparison Between Engineering Branches: Cse and ItDocument12 pagesComparison Between Engineering Branches: Cse and ItRajesh MachineniNo ratings yet
- Simulation of Gas-Solid Flow & Design Modifications of Cement Plant CyclonesDocument8 pagesSimulation of Gas-Solid Flow & Design Modifications of Cement Plant CyclonesNael100% (1)
- Benedek Internally-Directed-Attention-In-Creative-CognitionDocument15 pagesBenedek Internally-Directed-Attention-In-Creative-CognitionMario VillenaNo ratings yet
- CRT 3Document7 pagesCRT 3Dickson phiriNo ratings yet
- ASLP Newsletter (Issue 2, June 13) PDFDocument26 pagesASLP Newsletter (Issue 2, June 13) PDFAslp LibrariansNo ratings yet
- How John F. Kennedy Changed Decision Making For Us AllDocument4 pagesHow John F. Kennedy Changed Decision Making For Us Allutkarsh bhargavaNo ratings yet
- Configuring Roaming User Profiles - Group Policy PDFDocument7 pagesConfiguring Roaming User Profiles - Group Policy PDFEmilio MartinezNo ratings yet
- Unit 1 Introduction To Microeconomics - DT-PostedDocument63 pagesUnit 1 Introduction To Microeconomics - DT-PostedKereth WhittleNo ratings yet
- Lorenzo's Oil-Why Research Is Important?Document4 pagesLorenzo's Oil-Why Research Is Important?R2589770% (1)
- Appendix To ISSAI 5000 EDocument4 pagesAppendix To ISSAI 5000 EAli HabibNo ratings yet
- Curex KG 1 1819Document13 pagesCurex KG 1 1819nunikNo ratings yet
- 6 Tips For Turning Vendors Into Strategic Partners: Tip SheetDocument1 page6 Tips For Turning Vendors Into Strategic Partners: Tip SheetBhanu SreenivasNo ratings yet
- CITY FLEUVE Ecology KalDocument77 pagesCITY FLEUVE Ecology Kalkashjustin100% (1)