Professional Documents
Culture Documents
Enzymes
Enzymes
Uploaded by
Waseem El-HelwOriginal Description:
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Enzymes
Enzymes
Uploaded by
Waseem El-HelwCopyright:
Available Formats
lOMoARcPSD|2542479
Enzymes
BIOCHEMISTRY (Polytechnic University of the Philippines)
StuDocu is not sponsored or endorsed by any college or university
Downloaded by Jason Bolingo (jasonbolingo@gmail.com)
lOMoARcPSD|2542479
ENZYMES
• Is a compound, usually a protein, that acts as a catalyst for a biochemical reaction.
• Each cell in the human body contains thousands of different enzymes because almost every
reaction in a cell requires its own specific enzyme.
• Enzymes are not consumed during the reaction but merely help the reaction occur more rapidly.
• Enzymes undergo all the reactions of proteins including denaturation.
Activation energy
Enzyme structure
• Simple enzymes: is an enzyme composed only of protein ( amino acid chains)
• Conjugated enzymes: is an enzyme that has a non-protein part in addition to a protein part.
• Apoenzyme:
• Cofactor: it provides additional chemically reactive functional groups beside those
present in the amino acid side chains of apoenzyme.
• Coenzyme: is a small organic molecule that serves as a cofactor in a conjugated
enzymes.
Nomenclature and classification of enzymes
Downloaded by Jason Bolingo (jasonbolingo@gmail.com)
lOMoARcPSD|2542479
• Enzymes are commonly named by using a system that attempts to provide information about
the function (rather than the structure) of the enzyme.
• The typed of the reaction and the substrate identity are the focal points for the nomenclature of
an enzyme.
• Substrate: it is the reactant in an enzyme-catalyzed reaction. Substrate is a substance
upon which the enzymes ” acts ”.
Three important aspects of the enzyme-naming process are the following:
1. The suffix –ase identifies a substance as an enzyme.
2. The type of reaction catalyzed by an enzyme is often noted with a prefix. Oxidase, hydrolase.
3. The identity of the substrate is often noted in addition to the type of reaction. Infrequently, the
substrate but not the reaction type is given.( urease, lactase)
Classification of enzymes
• Oxidoreductase
• Transferase
• Hydrolase
• Lyase
• Isomerase
• Ligase
Oxidoreductase
Downloaded by Jason Bolingo (jasonbolingo@gmail.com)
lOMoARcPSD|2542479
1. Label the reaction to indicate which compound is oxidized and which compound is reduced.
2. Lactate dehydrogenase is the enzyme. What is the name of the term given to NAD + in this
reaction?
Transferase
Ligase
Downloaded by Jason Bolingo (jasonbolingo@gmail.com)
lOMoARcPSD|2542479
Hydrolase
Isomerase
Models of enzyme action
• Active site: is the relatively small part of an enzyme’s structure that is actually involved in the
catalysis.
Downloaded by Jason Bolingo (jasonbolingo@gmail.com)
lOMoARcPSD|2542479
• Enzyme-substrate complex: catalyst offer an alternative pathway with lower activation energy
through which a reaction can occur. An enzyme-substrate complex is the intermediate reaction
species that is formed when a substrate binds to the active site of an enzyme.
Lock-and-key model
Induce-fit model
Enzyme specificity
1. Absolute specificity: the enzyme will catalyze only one reaction.
2. Group specificity: the enzyme will act only on molecules that have specific functional group,
such as hydroxyl, amino or phosphate groups.
3. Linkage specificity: the enzyme will act on a particular type of chemical bond. (phosphatases)
4. Stereochemical specificity: the enzyme will act on a particular stereoisomer.
Factors that affect enzyme activity
• Temperature
• pH
• Substrate concentration
• Enzyme concentration
Downloaded by Jason Bolingo (jasonbolingo@gmail.com)
lOMoARcPSD|2542479
Temperature
• Higher temperatures means molecule are moving faster and colliding more frequently. This
concept applies to collision between substrate molecules and enzymes. As the temperature of
an enzymatically catalyzed reaction increases, so does the rate of the reaction.
• Optimum temperature:
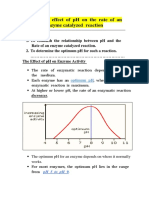
Ph
• Most enzyme exhibit maximum activity over a very narrow pH range.
• Optimum pH:
• How does gastric ulcer occur? What is the causative agent for this condition?
Substrate concentration
• When the concentration of an enzyme is kept constant and the concentration of substrate is
increased, the enzyme activity pattern is obtained. This activity pattern is called .
Enzyme concentration
• The greater the enzyme concentration, the greater the reaction rate.
Enzyme inhibition
• The rates of enzyme-catalyzed reactions can be decreased by a group of substances called
inhibitors.
• Enzyme inhibitor: is a substance that slows or stops the normal catalytic function of an enzyme
by binding to it.
• Reversible competitive inhibition, reversible noncompetitive inhibition and irreversible
inhibition.
Reversible competitive inhibition
• Competitive enzyme inhibitor: is a molecule that sufficiently resembles an enzyme substrate in
shape and charge distribution that it can compete with the substrate for occupancy of the
enzyme’s active site.
example: of competitive inhibitor
• Anti-histamines are competitive inhibitors of histidine decarboxylation, the enzymatic reaction
that converts histidine to histamine.
Reversible noncompetitive inhibition
• Noncompetitive enzyme inhibitor: is a molecule that decreases enzyme activity by binding to a
site on an enzyme other than the active site.
Downloaded by Jason Bolingo (jasonbolingo@gmail.com)
lOMoARcPSD|2542479
Example of noncompetitive inhibitor
• Metal ions (Pb2+, Ag2+ and Hg2+ )
• The binding sites for these ions are sulfhydryl (-SH) groups located away from the active site.
• Metal sulfide will affect the secondary and tertiary structure of the enzyme.
Irreversible inhibition
• Irreversible enzyme inhibitor: is a molecule that inactivates enzymes by forming a strong
covalent bond to an amino acid side-chain group at the enzyme’s active site.
Example of irreversible inhibitor
• Chemical warfare agents (nerve gases)
• Nerve agents (sarin, soman, cyclosarin, tabun)
• Vesicating or blistering agents (mustards, lewisite)
• Respratory agents (chlorine,phosgene, diphosgene)
• Cyanide
• Antimuscarinic agents (anticholinergic compounds)
• Opioid agents (opioid derivatives)
• Riot control agents ( pepper gas, cyanide)
• Vomiting agent (adamsite)
• Organophosphate insecticides
Regulation of enzyme activity
• A cell that continually produces large amounts of an enzyme for which substrate concentration
is always very low is wasting energy. The production of the enzyme needs to be “turned off”.
• A product of an enzyme-catalyzed reaction that is present in plentiful(more than needed)
amounts in a cell is a waste of energy if the enzyme continues to catalyze the reaction that
produces the product. The enzyme needs to be “turned off”.
“turned off” and “turned on” mechanism of enzymes
1. Feedback control associated with allosteric enzymes
2. Proteolytic enzymes and zymogens
3. Covalent modification
Feedback control associated with allosteric enzymes
Downloaded by Jason Bolingo (jasonbolingo@gmail.com)
lOMoARcPSD|2542479
• Allosteric enzymes: is an enzyme with two or more protein chains and two kinds of binding sites
( substrate and regulator)
• Many, but not all, enzymes responsible for regulating cellular processes are allosteric enzymes.
-Characteristics of Allosteric Enzyme-
• All allosteric enzymes have quaternary structure.
• All allosteric enzymes have two kinds of binding sites.
• Active and regulatory binding sites are distinct from each other in both location and
shape.
• Binding of a molecule at the regulatory site causes changes in the overall three
dimensional structure of the enzyme, including changes at the active site.
• Feedback control: is a process in which activation or inhibition of the first reaction in a reaction
sequence is controlled by a product of the reaction sequence.
• The product of each step is the substrate for the next enzyme.
Proteolytic enzymes and zymogen
• Proteolytic enzymes: is an enzyme that catalyzes the breaking of peptide bonds that maintain
the primary structure of a protein.
• Zymogen: is the inactive precursor of a proteolytic enzyme.
Covalent modification of enzymes
• Covalent modification: is a process in which enzyme activity is altered by covalently modifying
the structure of the enzyme through attachment of a chemical group or removal of a chemical
group from a particular amino acid within the enzyme’s structure.
Downloaded by Jason Bolingo (jasonbolingo@gmail.com)
You might also like
- Enzymes - Amoeba SistersDocument2 pagesEnzymes - Amoeba Sistersrhdoiua67% (24)
- Topical Revision Notes Biology O Level PDFDocument140 pagesTopical Revision Notes Biology O Level PDFBilal Akram82% (22)
- Biology Matters GDocument2 pagesBiology Matters Gno one50% (2)
- EnzymesDocument9 pagesEnzymesMaria AnghelacheNo ratings yet
- ENZYMESDocument42 pagesENZYMESsanskritisingh4321No ratings yet
- Enzyme and Enzyme KineticsDocument61 pagesEnzyme and Enzyme KineticsNaiomiNo ratings yet
- Enzymes PresDocument34 pagesEnzymes PresRasmia SalamNo ratings yet
- C1 W14 EnzymesDocument49 pagesC1 W14 EnzymesJasmine Kaye CuizonNo ratings yet
- 20 Sayed Modinur Rahaman MicrobiologyDocument51 pages20 Sayed Modinur Rahaman Microbiologymuruganunofficial00No ratings yet
- ENZYMES Semester 1Document51 pagesENZYMES Semester 1asadullah2315No ratings yet
- 1380960835.1698lecture 4 & 5. EnzymesDocument30 pages1380960835.1698lecture 4 & 5. EnzymesKedir MohammedNo ratings yet
- Biochemistry - Enzyme-Protein Function and Metabolism 20180202Document78 pagesBiochemistry - Enzyme-Protein Function and Metabolism 20180202Artika Muliany TindaonNo ratings yet
- EnzymesDocument35 pagesEnzymesplayermessi778No ratings yet
- EnzymeDocument28 pagesEnzymeAmirah NazihahNo ratings yet
- Enzymes:: The Nature's CatalystsDocument51 pagesEnzymes:: The Nature's Catalystskrk100% (1)
- Enzyme Structure, Classification and Mechanism of ActionDocument31 pagesEnzyme Structure, Classification and Mechanism of ActionCathrine CruzNo ratings yet
- EnzymesDocument97 pagesEnzymesezechimaximillian2004No ratings yet
- Transport Mechanisms in EnzymesDocument17 pagesTransport Mechanisms in EnzymesBrasa Y. de AlmiraNo ratings yet
- EnzymesDocument18 pagesEnzymesMarie Veatrice Jacomille100% (1)
- 1FFF11B1BD16627EE05400144FEB5F70.pptDocument63 pages1FFF11B1BD16627EE05400144FEB5F70.pptNur AishaNo ratings yet
- EnzymesDocument99 pagesEnzymesSamuel AgulueNo ratings yet
- "Helper" Protein Molecules: EnzymesDocument7 pages"Helper" Protein Molecules: EnzymesMuhammad Junaid IqbalNo ratings yet
- EnzymesDocument18 pagesEnzymesKim TanNo ratings yet
- EnzymesDocument44 pagesEnzymesYumul, Jewel B.No ratings yet
- EnzymesDocument40 pagesEnzymesAmal100% (2)
- EnzymesDocument5 pagesEnzymeseyosiyas tekleweyinNo ratings yet
- Enzyme Kinetics and Applications (Part 1a: Kinetics of Enzyme Catalyzed Reactions)Document26 pagesEnzyme Kinetics and Applications (Part 1a: Kinetics of Enzyme Catalyzed Reactions)Nur AishaNo ratings yet
- Enzymes & Energetics - CTPWDocument42 pagesEnzymes & Energetics - CTPWjessicatieuuNo ratings yet
- Enzyme: An IntroductionDocument14 pagesEnzyme: An IntroductionVishnu SharmaNo ratings yet
- EnzymesDocument26 pagesEnzymesrabiullah.edu987No ratings yet
- Enzymes - BiochemistryDocument40 pagesEnzymes - Biochemistrysunil patelNo ratings yet
- Microbial Metabolism - Part 1Document85 pagesMicrobial Metabolism - Part 1Christine ElnasNo ratings yet
- Enzymes: D. F. PalacioDocument31 pagesEnzymes: D. F. PalacioCarlo AniagNo ratings yet
- Enzymes PresDocument57 pagesEnzymes Pres4tqpxmts7zNo ratings yet
- Chapter 20 EnzymesDocument24 pagesChapter 20 EnzymesAmy PhamNo ratings yet
- Enzymes L1 2023 2024Document33 pagesEnzymes L1 2023 20249829c7fchsNo ratings yet
- 1 Enzyme Regulation in Biochemical PathwaysDocument29 pages1 Enzyme Regulation in Biochemical PathwayshadeelNo ratings yet
- ENZYMESDocument39 pagesENZYMESDhaya VNo ratings yet
- Unit VIII - Enzymes - MCONDocument30 pagesUnit VIII - Enzymes - MCONikram ullah khanNo ratings yet
- Biomolecules and Cells:: Mr. Derrick Banda MSC, BSCDocument69 pagesBiomolecules and Cells:: Mr. Derrick Banda MSC, BSCAmon Sangulube100% (1)
- Biochemistry I BSC 211: Thomson Sanudi Basic Sciences DepartmentDocument17 pagesBiochemistry I BSC 211: Thomson Sanudi Basic Sciences DepartmentKelvin ChipezeniNo ratings yet
- Enzymes:: "Helper" Protein MoleculesDocument26 pagesEnzymes:: "Helper" Protein MoleculesAyesha JamilNo ratings yet
- Enzymes PPTDocument39 pagesEnzymes PPTsunil patelNo ratings yet
- Chapter 2: Diagnostic Enzymology: E+S Es E+PDocument67 pagesChapter 2: Diagnostic Enzymology: E+S Es E+PadamNo ratings yet
- Enzymes For BPT StudentsDocument68 pagesEnzymes For BPT Studentsilagernischit321No ratings yet
- ENZYMESDocument31 pagesENZYMESfmukuka12No ratings yet
- Enzyme Structure, Classification and Mechanism of ActionDocument35 pagesEnzyme Structure, Classification and Mechanism of ActionAnonymous cgKtuWzNo ratings yet
- Enzymes. RagaviDocument63 pagesEnzymes. Ragavikavi bharathiNo ratings yet
- EnzymesDocument53 pagesEnzymesMayuri DhanushwarNo ratings yet
- Enzymes 1Document54 pagesEnzymes 1Mayuri DhanushwarNo ratings yet
- Enzyme: Ayesha Shafi Pharm-D, (P.U.), M. Phil. Pharmaceutical Chemistry (P.U.)Document34 pagesEnzyme: Ayesha Shafi Pharm-D, (P.U.), M. Phil. Pharmaceutical Chemistry (P.U.)Shafaqat Ghani Shafaqat Ghani100% (2)
- ERT211 CHP 1-2 PDFDocument27 pagesERT211 CHP 1-2 PDFsobiyamaragathavelNo ratings yet
- Materi 2Document34 pagesMateri 2siti purnamaNo ratings yet
- Regulation of Enzyme ActivityDocument26 pagesRegulation of Enzyme ActivityWeird WorldNo ratings yet
- EnzymologyDocument47 pagesEnzymologyTererai Lalelani Masikati Hove100% (1)
- Clinical Chemistry II ENZYMESDocument5 pagesClinical Chemistry II ENZYMESEden MaeNo ratings yet
- 3 EnzymesDocument24 pages3 Enzymeskingofkingsmrphysioimtiyaz342No ratings yet
- Enzymes PPTDocument40 pagesEnzymes PPTJaisy PatelNo ratings yet
- EnzymesDocument46 pagesEnzymesHighlifeNo ratings yet
- Lecture 1 - The Kinetics of Enzyme Catalyzed ReactionsDocument18 pagesLecture 1 - The Kinetics of Enzyme Catalyzed ReactionsGenevive S. de VeraNo ratings yet
- Title: Lecture (9) Enzymes Present By: Associate Prof - Ingy BadawyDocument58 pagesTitle: Lecture (9) Enzymes Present By: Associate Prof - Ingy BadawyAbdo ElsaidNo ratings yet
- Enzymes and Vitamins CompressedDocument14 pagesEnzymes and Vitamins CompressedfredkaseydiNo ratings yet
- A-level Biology Revision: Cheeky Revision ShortcutsFrom EverandA-level Biology Revision: Cheeky Revision ShortcutsRating: 5 out of 5 stars5/5 (5)
- Factors Affecting Enzyme ActivityDocument29 pagesFactors Affecting Enzyme ActivityAngela Kim S. BernardinoNo ratings yet
- Elements Found in Living ThingsDocument13 pagesElements Found in Living ThingsmagiclcjNo ratings yet
- JHU Biochem - Exam 2 - 2013Document11 pagesJHU Biochem - Exam 2 - 2013Jenna ReynoldsNo ratings yet
- Lecture 5 - Enzymes Part 1Document15 pagesLecture 5 - Enzymes Part 1Fea Kristine PacquiaoNo ratings yet
- Caie As Biology 9700 Theory v4Document31 pagesCaie As Biology 9700 Theory v4USMAN JUNAID LAKHA NINo ratings yet
- Introduction To EnymesDocument34 pagesIntroduction To EnymesMeshal NoorNo ratings yet
- Effect of PH On Enzyme ActivityDocument12 pagesEffect of PH On Enzyme ActivityAb AbNo ratings yet
- Ch6 Application QuestionsDocument2 pagesCh6 Application QuestionsRandolphAPBIO100% (1)
- The Effect of Temperature On The Activity of The Enzyme AmylaseDocument12 pagesThe Effect of Temperature On The Activity of The Enzyme AmylaseNathaniel Hammo50% (2)
- Biochem Midterms Review Ni QTDocument17 pagesBiochem Midterms Review Ni QTAlyssa MacaraigNo ratings yet
- Chapter 2: Diagnostic Enzymology: E+S Es E+PDocument72 pagesChapter 2: Diagnostic Enzymology: E+S Es E+PQananiisa Lammii Gammada100% (1)
- 2.7.2 - Enzyme Active Site and Substrate Specificity - Biology LibreTextsDocument3 pages2.7.2 - Enzyme Active Site and Substrate Specificity - Biology LibreTextsjamil ahmedNo ratings yet
- 1ST SA BIOCHEMISTRY - AlmendrasDocument5 pages1ST SA BIOCHEMISTRY - AlmendrasCherry DagohoyNo ratings yet
- Senior12 Biology 1 Q1 - M8Document21 pagesSenior12 Biology 1 Q1 - M8RjasyNo ratings yet
- IA How Temperature Affects The Rate of Enzyme ActivityDocument16 pagesIA How Temperature Affects The Rate of Enzyme ActivityJohn Matthew Gonzales PrimiciasNo ratings yet
- Quiz BiochemistryDocument100 pagesQuiz BiochemistryMedShare88% (25)
- Cc2 Lec EnzymesDocument25 pagesCc2 Lec EnzymesJunea SeeNo ratings yet
- 22Document2 pages22Dipayan DasNo ratings yet
- Car Boxy Pep Tida SeDocument13 pagesCar Boxy Pep Tida SeJaisy PatelNo ratings yet
- Abcd. Proiect AdamDocument14 pagesAbcd. Proiect AdamSfiriac LauraNo ratings yet
- Compiled Quiz BiochemistryDocument38 pagesCompiled Quiz BiochemistryAdrianNo ratings yet
- Enzyme: Ayesha Shafi Pharm-D, (P.U.), M. Phil. Pharmaceutical Chemistry (P.U.)Document34 pagesEnzyme: Ayesha Shafi Pharm-D, (P.U.), M. Phil. Pharmaceutical Chemistry (P.U.)Shafaqat Ghani Shafaqat Ghani100% (2)
- 4.1 Enzyme Chemistry Part 1Document7 pages4.1 Enzyme Chemistry Part 1Geraldine Marie SalvoNo ratings yet
- Molecular DockingDocument64 pagesMolecular DockingQuty Papa KannaNo ratings yet
- Bachelor of Science in Medical Laboratory Science: Biochemistry LectureDocument16 pagesBachelor of Science in Medical Laboratory Science: Biochemistry LectureDCRUZNo ratings yet