Professional Documents
Culture Documents
Protein Classes PDF
Protein Classes PDF
Uploaded by
Mark HortizuelaOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Protein Classes PDF
Protein Classes PDF
Uploaded by
Mark HortizuelaCopyright:
Available Formats
Examples of Protein Structures
Protein types
Proteins fall into three general classes, based on their overall three-dimensional
structure and on their functional role: fibrous, membrane, and globular.
Fibrous proteins
Fibrous proteins tend to be long, narrow molecules. Fibrous proteins are used to
construct macroscopic structures, especially structures outside of cells. Fibrous
proteins tend to have a structural role, although some have more active functions as
well.
Examples:
The protein α-keratin is used to form hair, nails, and skin. α-Keratin is a coiled-coil
of α-helices ~300 residues long (corresponding to about 450 Å in length). α-Keratin
is formed from two α-helices wrapped around one another (the backbone shown
below is of a coiled-coil protein from PDB ID 3Q0X; this portion of the protein
probably resembles the keratin coiled-coil structure)), and two of those structures
wrapped around each other. Thus the keratin molecule contains a total of four
intertwined helices.
The helices in α-keratin are slightly distorted in order to allow the coiled-coil, but
otherwise are formed from torsion angles similar to those present in normal α-
helices. The approximate Φ / Ψ pair for the keratin helix is –64° / –42° compared to
a normal α-helix of –57° / –47°.
The keratin helices within the protein are maintained in their relative locations by
hydrogen bonds and disulfide bonds formed between side-chains of the helical
residues. Hair and nail α-keratin molecules have a higher disulfide bond content
than the form of keratin found in skin, and are tougher as a result.
High humidity allows facile rearrangement of hydrogen bonds, and can therefore
allow the keratin molecules in hair to change their relative positions somewhat.
Individuals with curly hair frequently find the degree of curling depends on the
humidity; they also usually find that hair shaped while wet tends to retain its
structure upon drying. A “permanent” results from reduction of the disulfide bonds
between the chains, followed by shaping the hair, and by then oxidizing the free
sulfhydryl groups to form new disulfide bonds that hold the hair in the new shape.
Copyright © 2000-2016 Mark Brandt, Ph.D. 46
Smaller coiled-coil structures are also found at the interaction interface between
some types of proteins that form less permanent complexes than keratin (especially
in some transcription factors such as leucine zipper proteins).
The multiple types of collagen together comprise the most common animal
protein (about 25% of the protein in animals is some form of collagen). Collagen is a
triple helix formed from three polypeptide chains. The individual polypeptides form
helices that are much more extended than an α-helix. A collagen helix contains 3.3
residues/turn compared to 3.6 residues/turn, but has a rise of 10 Å/turn, compared
to 5.4 Å for an α-helix. An α-helix has a rise per residue of 1.5 Å; in contrast, a
collagen helix has a rise of 3 Å per residue, and is therefore nearly as extended a
structure as a β-sheet (3.5 Å/residue). The collagen triple helix is ~14 Å in diameter,
compared to about 6 Å for an α-helix, due to the presence of three polypeptide
chains.
The extended collagen helix is the result of its Φ / Ψ pair of –51° / 153°. The Φ angle
is thus close to that of the α-helix, but the Ψ angle is quite different, and is close to
the maximal extension angle (180°). The collagen triple helix has three of these
extended helices wrapped around one another.13
Gly Ser
Gly Gly
HydroxyPro Pro
Pro Pro
Pro
Each chain in the collagen triple helix is ~1000 residues long. The collagen molecule
is therefore about 3000 Å = 0.3 µm long ([1000/3.3 residue/turn]•10 Å).
In collagen, the polypeptides have the typical sequence (Gly-X-Y)n, where X residues
are frequently proline and the Y residues are frequently hydroxyproline or
hydroxylysine. Glycine fits in the center of triple helix (it is the only residue that is
small enough to do so). Proline readily adopts the Φ / Ψ angles required by the triple
helix. Note the proline rings in the strands shown above.
In collagen, the helices are held in register by several factors. One is hydrogen
bonding involving hydroxyproline residues. The hydroxyl group is added to the
proline residues following the synthesis of the collagen molecule in a vitamin C-
dependent reaction. Some of the symptoms of scurvy (vitamin C deficiency) are due
to inhibition of proline hydroxylation in collagen. Another factor that stabilizes the
collagen structure is the result of covalent cross links formed between lysine,
hydroxylysine, and histidine residues within the protein.14
13 The drawing of collagen was created in VMD from PDB ID 3ADM; the structure was determined
using trimers of a 27-residue artificial model peptide rather than a full-length collagen molecule.
14 Since collagen molecules are much shorter than the macroscopic structures such as tendons and
ligaments that are produced largely from collagen, it is apparent that the larger structures require
Copyright © 2000-2016 Mark Brandt, Ph.D. 47
Membrane proteins
Membrane proteins comprise a unique class of proteins. For membrane proteins, a
significant region of the protein must be stable in a hydrophobic environment. This
is typically achieved by having non-polar side-chains on specific surface regions of
the protein. Because of this exposed hydrophobic surface, and because many
membrane proteins are destabilized by removal from the membrane, most
membrane proteins are difficult to work with. As a result, structural information is
available for only a relatively small number of these proteins, although new
techniques have allowed three-dimensional structure determination for increasing
numbers of membrane proteins in the last few years.
An example of a membrane protein is cytochrome c oxidase. Cytochrome c oxidase is
one of the more important membrane proteins: it is the protein that donates
electrons to oxygen in the electron transport chain. Cytochrome c oxidase is
therefore the primary oxygen-utilization enzyme in aerobic organisms. Its
importance stimulated the series of studies that resulted in the solution of its three-
dimensional structure.
The region of the cytochrome c oxidase protein that interacts with the membrane is
readily visible in the structural representation below.15 In the figure, yellow
residues are non-polar, light blue residues are polar, blue residues have basic side-
chains, and red residues have acidic side-chains. Note the band of non-polar
residues across the center of the protein. Note also that the region is predominantly
α-helical. This is because α-helices satisfy all of the hydrogen bonding requirements
of the peptide backbone, regardless of the polarity of the environment.
!"#$%&'$()*!*$+,-./)
interactions between collagen molecules. Some of these interactions are also mediated by side chain
hydrogen bonding and other links.
15 The structural representation for cytochrome c oxidase was created in SwissPDB Viewer from
PDB ID 2OCC (Yoshikawa, et al., Science 280: 1723-1729, 1998).
Copyright © 2000-2016 Mark Brandt, Ph.D. 48
Globular proteins
Globular proteins comprise the most varied type of proteins. Globular proteins are
soluble in aqueous solution. To achieve this, globular proteins generally have polar
residues on the surface and hydrophobic residues on the interior.
Globular proteins frequently have stable structures that lend themselves to
structure determination. This means that more three-dimensional structural
information is available for globular proteins than for all other classes of proteins
combined.
The structures of globular proteins are quite varied. Although early structural
determination studies suggested that proteins have large amounts of α-helix (as
was found for hemoglobin, shown below; hemoglobin is approximately 80% α-
helical), the helical content of proteins can vary dramatically. The structure of
human chorionic gonadotropin, for example, contains large amounts of β-sheet, but
only a single, short, α-helix.
Hemoglobin
(from PDB ID 1B86)
human
chorionic
gonadotropin
(hCG, from
PDB ID 1HCN)
The above structure for hCG shows a protein that is quite elongated, and is far from
spherical. However, hCG is considered a globular protein; the term “globular” refers
more to the property of solubility in aqueous solution than to the roughly spherical
shape of many proteins in this class (such as hemoglobin).
Copyright © 2000-2016 Mark Brandt, Ph.D. 49
Representations of protein structure
When structural information is available, the protein is often simply presented as a
snapshot of the three-dimensional structure. These snapshots can be of considerable
use, but often obscure important details. One way around the problem of presenting
complex three-dimensional information is to use computer programs that allow the
viewer to examine the structure from many perspectives. However, even with
computer graphics programs, the amount of information present in a three-
dimensional representation is too large to interpret readily. Most computer
visualization software packages have methods for simplifying the overall structure
(compare, for example, the representations of hCG shown below).
Ribbon diagram of the hCG backbone trace,
Licorice representation, showing all hCG color-coded by polypeptide chain (α chain in red,
polypeptide non-hydrogen atoms, color-coded by β chain in blue), with cysteine residues in licorice
atom type. (These diagrams were both created representation)
in VMD.)
Although the diagrams above show the three-dimensional structure accurately,
more stylized representations of structures are sometimes more useful. These allow
emphasis of important features, and usually present the information more clearly.
However, this may come at the cost of modifying the appearance of the actual
structure considerably. For example, the stylized diagram for the hCG structure
emphasizes the arrangement of β-structure in the protein at the cost of distorting
the structure considerably.16
,
α $%&$)
")&#'
β' β#
!%&!*
-
((&!%!
β" β! β$
)&#!
!"#$" '"&'* β" )*&(!
%&#&% ##&#(
'%#&(
'(&*)
, β#
'+&+(
α01232456 -
+$&)%
β#
,7β01232456
β"
β$ β! β" #$&$% )#*+
!%&!' "#&)"
)$&*% "'&#%
"+&!!%
'(#&&
'&#)"
.(#&!%%/
16 The secondary structure diagram was modified from information and drawings in Wu, et al.,
Structure 2: 545-558 (1994) and Lapthorn, et al., Nature 369: 455-461 (1994)
Copyright © 2000-2016 Mark Brandt, Ph.D. 50
Summary
Proteins can be classified according to their structural properties. This scheme
divides proteins into three main types: fibrous, membrane, and globular.
Fibrous proteins are long narrow molecules. Most are involved in forming
macroscopic structural elements, including keratin and collagen.
Membrane proteins typically have a hydrophobic region that interacts with the non-
polar interior of membranes. These proteins are much less well understood than
those of other classes, because they are difficult to purify and study. Membrane
spanning regions of membrane proteins are frequently α-helical, although some
exceptions have been observed.
Globular proteins are a diverse class of soluble proteins. Many of the most heavily
studied proteins are members of this class of proteins.
Copyright © 2000-2016 Mark Brandt, Ph.D. 51
You might also like
- Data Tables For Alien Gene AnalysisDocument3 pagesData Tables For Alien Gene Analysisapi-382372564100% (3)
- Prokaryotic DNA ReplicationDocument5 pagesProkaryotic DNA ReplicationMg H67% (6)
- Membrane BiochemistryDocument21 pagesMembrane BiochemistryAnna SafitriNo ratings yet
- Biosynthesis of CollagenDocument40 pagesBiosynthesis of CollagenNausheerNo ratings yet
- Collagen PDFDocument19 pagesCollagen PDFJaveria KhanNo ratings yet
- Collagen: The BiomaterialDocument5 pagesCollagen: The BiomaterialSandhik RoyNo ratings yet
- Protein Structures Continued Proteins of ECM Collagen of Connective TissueDocument37 pagesProtein Structures Continued Proteins of ECM Collagen of Connective TissueAytajNo ratings yet
- Biology Notes: Proteins: CollagenDocument5 pagesBiology Notes: Proteins: CollagenMuhammad AkramNo ratings yet
- RISHUDocument30 pagesRISHURishu Mittal100% (1)
- We Are Intechopen, The World'S Leading Publisher of Open Access Books Built by Scientists, For ScientistsDocument35 pagesWe Are Intechopen, The World'S Leading Publisher of Open Access Books Built by Scientists, For ScientistsCamila Flórez IdárragaNo ratings yet
- Structure and Function of Macromolecules: 2.2.1 Carbohydrates-The Energy GiversDocument12 pagesStructure and Function of Macromolecules: 2.2.1 Carbohydrates-The Energy GiversSteven BanksNo ratings yet
- 04ProAMO LicyayoDocument8 pages04ProAMO LicyayoMohamidin MamalapatNo ratings yet
- Proteins: Classification FunctionsDocument20 pagesProteins: Classification FunctionsGeo NalobNo ratings yet
- Structural of Red CellsDocument2 pagesStructural of Red CellsDrbee10No ratings yet
- Bchn213 (M-Campus) Test 5 Su 9 and 10 MemoDocument5 pagesBchn213 (M-Campus) Test 5 Su 9 and 10 Memozinhlez518No ratings yet
- Module 3 - Lecture 3 - LectureDocument8 pagesModule 3 - Lecture 3 - LectureNi CaoNo ratings yet
- Natural BiopolymersDocument65 pagesNatural BiopolymersBensen HsuNo ratings yet
- J Ijbiomac 2017 07 013Document17 pagesJ Ijbiomac 2017 07 013Isaac GoldNo ratings yet
- Biochemistry LN05-2Document12 pagesBiochemistry LN05-2Rahaf Al-muhtasebNo ratings yet
- Collagen StructureDocument33 pagesCollagen StructureSagar De'biomimicNo ratings yet
- 2.4, 7.3 Proteins Biology HLDocument37 pages2.4, 7.3 Proteins Biology HLmunir sehwai.No ratings yet
- BCCH 4Document59 pagesBCCH 4NG SIRNo ratings yet
- Protein StructureDocument17 pagesProtein Structureutpal mohanNo ratings yet
- QUITALIG Biochemistry Lecture Module 5 ActivityDocument5 pagesQUITALIG Biochemistry Lecture Module 5 ActivityAloysius QuitaligNo ratings yet
- Chapter 4 Lecture PptsDocument82 pagesChapter 4 Lecture PptsJota AlcuadradoNo ratings yet
- BiochemistryDocument130 pagesBiochemistryshiyntumNo ratings yet
- Lecture 4 PDFDocument18 pagesLecture 4 PDFNazir KhanNo ratings yet
- Amino Acids - 2 - NoDocument9 pagesAmino Acids - 2 - NoSunny Thakur17No ratings yet
- CHEM 151 (Chapter 4)Document4 pagesCHEM 151 (Chapter 4)Chantel Acevero100% (1)
- Protein Shape and Protein PurificationDocument60 pagesProtein Shape and Protein PurificationMilena De CresentNo ratings yet
- Chap. 4B The Three-Dimensional Structure of Proteins: TopicsDocument28 pagesChap. 4B The Three-Dimensional Structure of Proteins: Topicscatalina esanuNo ratings yet
- CollagenDocument79 pagesCollagenRobins Dhakal100% (4)
- Lec 9Document15 pagesLec 9FARHAN KAMALNo ratings yet
- The Three-Dimensional Structure of Proteins: Protein Structure and FunctionDocument26 pagesThe Three-Dimensional Structure of Proteins: Protein Structure and Function김경환No ratings yet
- ProteinsDocument44 pagesProteinsSusan SylvianingrumNo ratings yet
- Extra Cellular Matrix ADocument17 pagesExtra Cellular Matrix Aapi-301896945No ratings yet
- Structures and Functions of ProteinDocument54 pagesStructures and Functions of ProteinRollyRiksantoNo ratings yet
- Chapter 4-Jnu Biomembranes-1 - 複本Document48 pagesChapter 4-Jnu Biomembranes-1 - 複本Wai Kwong ChiuNo ratings yet
- Fibrous ProteinDocument16 pagesFibrous ProteinMatth N. ErejerNo ratings yet
- Biomolecules - Protein 3Document18 pagesBiomolecules - Protein 3NinjaTylerFortniteBlevins 2.0No ratings yet
- ProteinsDocument5 pagesProteinsnadjaNo ratings yet
- Materials: Collagen Based Materials in Cosmetic Applications: A ReviewDocument15 pagesMaterials: Collagen Based Materials in Cosmetic Applications: A ReviewMauricio Tarquino FloresNo ratings yet
- Hemoglobin Is The One Protein Molecule That Knows by Everyone Which It Is One of The Most Importance and Plentiful Proteins of VertebratesDocument5 pagesHemoglobin Is The One Protein Molecule That Knows by Everyone Which It Is One of The Most Importance and Plentiful Proteins of VertebratesBilah BilahNo ratings yet
- 4 ProteinsDocument18 pages4 ProteinsjaymcornelioNo ratings yet
- Protein Structure and Folding LectureDocument18 pagesProtein Structure and Folding Lecturemaryscribd241No ratings yet
- Fibrous ProteinDocument40 pagesFibrous Proteinعبدالرحمن حاتمNo ratings yet
- CollagenDocument8 pagesCollagenkaditasookdeoNo ratings yet
- Online Practice Tests, Live Classes, Tutoring, Study Guides Q&A, Premium Content and MoreDocument15 pagesOnline Practice Tests, Live Classes, Tutoring, Study Guides Q&A, Premium Content and MoreYoAmoNYCNo ratings yet
- Application of 2 D-HPLC System For Plasma Protein SeparationDocument10 pagesApplication of 2 D-HPLC System For Plasma Protein Separationtrinhmy81No ratings yet
- Myoglobin: Myoglobin (Symbol MB or MB) Is An IronDocument8 pagesMyoglobin: Myoglobin (Symbol MB or MB) Is An IronNTA UGC-NETNo ratings yet
- Ripuseminar 171217093449Document52 pagesRipuseminar 171217093449Jun NguyễnNo ratings yet
- Protein: Globular and Fibrous ProteinDocument20 pagesProtein: Globular and Fibrous ProteinAndrewNo ratings yet
- 04model of Membranes - LecturesDocument11 pages04model of Membranes - LecturesmeliszaNo ratings yet
- ProteinsDocument9 pagesProteinsJada HartNo ratings yet
- Evaluation of Erythrocytes: Normal Ery Throcy TesDocument24 pagesEvaluation of Erythrocytes: Normal Ery Throcy Tesaixacamila3No ratings yet
- Protein Structure11Document5 pagesProtein Structure11gangmasterNo ratings yet
- Chapter 6 The Three Dimensional Structure of Proteins SummDocument48 pagesChapter 6 The Three Dimensional Structure of Proteins Summtania.delafuenteNo ratings yet
- CarbohidratosDocument8 pagesCarbohidratosElena OlmedoNo ratings yet
- Loops ProteinsDocument37 pagesLoops ProteinsSanjeev KumarNo ratings yet
- Peptides and Proteins: M.Prasad Naidu MSC Medical Biochemistry, PH.DDocument30 pagesPeptides and Proteins: M.Prasad Naidu MSC Medical Biochemistry, PH.DDr. M. Prasad NaiduNo ratings yet
- Mass Transport Answer Booklet 2017 6Document54 pagesMass Transport Answer Booklet 2017 6LucyNo ratings yet
- A-level Biology Revision: Cheeky Revision ShortcutsFrom EverandA-level Biology Revision: Cheeky Revision ShortcutsRating: 5 out of 5 stars5/5 (5)
- DM No. 667, S. 2022 - Resetting of The Schedule of Portfolio Day For SY 2022-2023Document3 pagesDM No. 667, S. 2022 - Resetting of The Schedule of Portfolio Day For SY 2022-2023LeneNo ratings yet
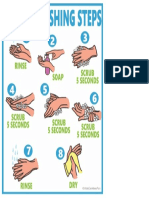
- Handwashing StepsDocument1 pageHandwashing StepsLeneNo ratings yet
- The Goose Voyage: Grade 5 Reading Comprehension WorksheetDocument4 pagesThe Goose Voyage: Grade 5 Reading Comprehension WorksheetLeneNo ratings yet
- WordsDocument1 pageWordsLeneNo ratings yet
- 5th Grade 5 Goose VoyageDocument5 pages5th Grade 5 Goose VoyageLeneNo ratings yet
- ActionDocument2 pagesActionLeneNo ratings yet
- Experiment 8 Acid-Base Titration: Pre - Lab QuestionsDocument14 pagesExperiment 8 Acid-Base Titration: Pre - Lab QuestionsLeneNo ratings yet
- SOP Preparing Sodium Hydroxide Solutions - 0Document3 pagesSOP Preparing Sodium Hydroxide Solutions - 0LeneNo ratings yet
- Report On SocDimsDocument41 pagesReport On SocDimsLeneNo ratings yet
- Mechanism of Enzyme ActionDocument10 pagesMechanism of Enzyme Actionsd2808No ratings yet
- Notes On Protein DenaturationDocument17 pagesNotes On Protein DenaturationKella OrtegaNo ratings yet
- Biochem Exam Practice QuestionsDocument12 pagesBiochem Exam Practice Questionsihack_101No ratings yet
- Price List WinSammering Pharma 2020Document1 pagePrice List WinSammering Pharma 2020shwE TVNo ratings yet
- Chart of Aminoacids Names and Abbreviations and CodonsDocument1 pageChart of Aminoacids Names and Abbreviations and Codonsstlcajun55No ratings yet
- 1 6 Monomer Drawing PracticeDocument4 pages1 6 Monomer Drawing PracticeSiddharth RajendranNo ratings yet
- Activity 5 From DNA To ProteinDocument4 pagesActivity 5 From DNA To ProteinKAREN SALLESNo ratings yet
- Himupro Protein PowderDocument1 pageHimupro Protein PowderNavi ThakurNo ratings yet
- Molecular Basis of InheritanceDocument55 pagesMolecular Basis of Inheritancearyan aggarwalNo ratings yet
- Worksheet On DNA and RNA AnswersDocument3 pagesWorksheet On DNA and RNA AnswersSelena CoronelNo ratings yet
- Chapter 2 LipidsDocument109 pagesChapter 2 LipidsTiffany Shane VallenteNo ratings yet
- DG Midterm #1 Spring 2021 From ULA ReviewDocument7 pagesDG Midterm #1 Spring 2021 From ULA ReviewRITIKA SURISETTYNo ratings yet
- Isoenzymes-Multienzyme Complex PDFDocument20 pagesIsoenzymes-Multienzyme Complex PDFintaliallitaNo ratings yet
- Gluconeogenesis AtfDocument5 pagesGluconeogenesis AtfDivyaa VisvalingamNo ratings yet
- DLL Grade 7 Video LessonDocument4 pagesDLL Grade 7 Video LessonJr CapanangNo ratings yet
- Lesson Plan Grade 8 - BiomuleculesDocument7 pagesLesson Plan Grade 8 - BiomuleculesRoxanne Reglos67% (3)
- Neptune Fish Concentrate Technical Data SheetDocument1 pageNeptune Fish Concentrate Technical Data SheetKatherine Leyla Anchayhua TorresNo ratings yet
- Insights On The Epigenetic MechanismsDocument12 pagesInsights On The Epigenetic MechanismsrafaellapordeusNo ratings yet
- Provon 395 SFL DTA - 0719-3 PDFDocument1 pageProvon 395 SFL DTA - 0719-3 PDFGomez AndreNo ratings yet
- Fatty Acids As Biocompounds: Their Role in Human Metabolism, Health and Disease - A Review. Part 1: Classification, Dietarysources and Biological FunctionsDocument14 pagesFatty Acids As Biocompounds: Their Role in Human Metabolism, Health and Disease - A Review. Part 1: Classification, Dietarysources and Biological FunctionsItan RuizNo ratings yet
- 14.respiration New BioHackDocument8 pages14.respiration New BioHackAnush KhillareNo ratings yet
- Plant Biochemistry 2 RevDocument59 pagesPlant Biochemistry 2 Revenrico andrionNo ratings yet
- Slip Test-5Document3 pagesSlip Test-5G.N. BhuvaneshwaranNo ratings yet
- Microbiology (Chapter 7: Microbial Genetics) : The Structure and Replication of GenomesDocument14 pagesMicrobiology (Chapter 7: Microbial Genetics) : The Structure and Replication of GenomesCarrie ANo ratings yet
- Harga Tayang Afifarma Non Jamkesnas 2023Document2 pagesHarga Tayang Afifarma Non Jamkesnas 2023Fadly AsriNo ratings yet
- Nucleic AcidDocument7 pagesNucleic AciddearbhupiNo ratings yet
- Ncert Line by Line Botany-Mbi Work SheetDocument17 pagesNcert Line by Line Botany-Mbi Work Sheetanusya548100% (2)