Professional Documents
Culture Documents
Mind Mapping 1 Krisna Divayana (1913071010) PDF
Mind Mapping 1 Krisna Divayana (1913071010) PDF
Uploaded by
krisna divayana1050 ratings0% found this document useful (0 votes)
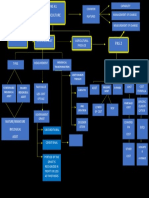
47 views1 pageThe document discusses factors that affect the rate of chemical reactions including temperature, concentration, surface area, and catalysts. It explains concepts from chemistry kinetics such as collision theory, activation energy, and transition states. Different types of reactions and their rates are also mentioned.
Original Description:
Original Title
MIND MAPPING 1 KRISNA DIVAYANA (1913071010).pdf
Copyright
© © All Rights Reserved
Available Formats
PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentThe document discusses factors that affect the rate of chemical reactions including temperature, concentration, surface area, and catalysts. It explains concepts from chemistry kinetics such as collision theory, activation energy, and transition states. Different types of reactions and their rates are also mentioned.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
Download as pdf or txt
0 ratings0% found this document useful (0 votes)
47 views1 pageMind Mapping 1 Krisna Divayana (1913071010) PDF
Mind Mapping 1 Krisna Divayana (1913071010) PDF
Uploaded by
krisna divayana105The document discusses factors that affect the rate of chemical reactions including temperature, concentration, surface area, and catalysts. It explains concepts from chemistry kinetics such as collision theory, activation energy, and transition states. Different types of reactions and their rates are also mentioned.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
Download as pdf or txt
You are on page 1of 1
CATA NATUR SURFACE
LYST E AREA pH THE AMOUNT OF REDUCTION OF
REACTION IN ONE TREATMENT
PRODUCT ADDED RATE
TYPE TIME
PRES PROPERTIES OF
SURE AND STATE OF BOND
SUBSTANCE
TEMPERATURE
RISES, CONCEPT
REACTION OF
RATE REACTION
INCREASED FACTOR RATE
TEMPERATURE AFFECTING
TEMPERATURE
DOWN, AVERAGE
REACTION CHEMISTRY
TYPE OF REACTION RATE
RATE DOWN KINETICS
REACTION
CONCENTRATI
ON RATE REAL ACTION
GREAT
CONCENTRAT
RATE
ION OF
REACTION,
THE LARGEST SMALL
TREATMENT THEORY THEORY OF
REACTION
RATE CONCENTRATI COLLISION OF THE
ON, THEORY REACTION TRANSITION
REASY SMALL STATE
REACTION RATE RATE
REACTION
THERE ARE
RATE WAS
SUBSTANCE
AFFECTED BY
NUMBER OF
WHICH HAVE I PUTU KRISNA DIVAYANA
VERY HIGH
POWDER PER
ENERGY SETIAWAN (1913071010)
UNITS
You might also like
- Alien RPG - Character Sheet - PrintableDocument1 pageAlien RPG - Character Sheet - PrintableAlejandro SablanNo ratings yet
- Thermocold AWC PROZONE Tech PDFDocument44 pagesThermocold AWC PROZONE Tech PDFAlexNo ratings yet
- OG OG Local Mass Transfer Coeff, Kya: Material Thermal ConductivityDocument2 pagesOG OG Local Mass Transfer Coeff, Kya: Material Thermal ConductivityJenny LlanesNo ratings yet
- Final Concept MapDocument1 pageFinal Concept MapEustina MumbireNo ratings yet
- Chemistry - Kadar TindakbalasDocument40 pagesChemistry - Kadar Tindakbalasrashifah100% (1)
- Factors Affecting Rate of Reaction Collision TheoryDocument1 pageFactors Affecting Rate of Reaction Collision TheoryMei XiangNo ratings yet
- Pengukuran Berapa Pantas Tindak Balas Berlaku: Rate of ReactionDocument8 pagesPengukuran Berapa Pantas Tindak Balas Berlaku: Rate of ReactionFazza RudyNo ratings yet
- Interfacial TensionDocument7 pagesInterfacial TensionagilNo ratings yet
- Topics: Psychro. Evaporative, Cooling & Solids Drying Energy Resources Conversion & UtilizationDocument3 pagesTopics: Psychro. Evaporative, Cooling & Solids Drying Energy Resources Conversion & UtilizationErwin DoloresNo ratings yet
- Charger Menu Tree PDFDocument1 pageCharger Menu Tree PDFVeeramanikandan SenthilnathanNo ratings yet
- This Study Resource Was: Steam Nomo GraphDocument2 pagesThis Study Resource Was: Steam Nomo GraphAdelaida CruzNo ratings yet
- Dynamic Analysis FlowchartDocument1 pageDynamic Analysis FlowchartJarryd NortonNo ratings yet
- Dryer Powder Quality ComparisionDocument3 pagesDryer Powder Quality ComparisionRpsinghNo ratings yet
- Basic Acivalent Numbers of Oxidation: EquationDocument1 pageBasic Acivalent Numbers of Oxidation: Equationkrisna divayana105No ratings yet
- Chapter 4 Chemical KineticsDocument60 pagesChapter 4 Chemical KineticsYvan Nicholas B. JasminNo ratings yet
- 27 - Nadya Rahmasari - Mindmap Chapter 11 PDFDocument1 page27 - Nadya Rahmasari - Mindmap Chapter 11 PDFNadya RahmaNo ratings yet
- Nursyazmin - 205285 - Hakimi - 205433 - Lab Report 1 PDFDocument17 pagesNursyazmin - 205285 - Hakimi - 205433 - Lab Report 1 PDFMuhammad ArifNo ratings yet
- Rate of Chemical ReactionDocument22 pagesRate of Chemical Reactionkatrina freoNo ratings yet
- Soil Organic Carbon: Slope Climate ChangeDocument1 pageSoil Organic Carbon: Slope Climate Changejoydeepsarkar9046No ratings yet
- Phainorc Reviewr DraftDocument5 pagesPhainorc Reviewr Draftejeraalaysa54No ratings yet
- SPSS KurniatiDocument2 pagesSPSS KurniatiAbd. Wahid FIKNo ratings yet
- Mind Map On Is Matter Around Us Pure - MapDocument1 pageMind Map On Is Matter Around Us Pure - MapPiyush PawasaleNo ratings yet
- Chemical Kinetics: Recap of Early ClassesDocument72 pagesChemical Kinetics: Recap of Early ClassesRaashiNo ratings yet
- Daily Report - Granular Base Course: Compaction - Control Strip MethodDocument2 pagesDaily Report - Granular Base Course: Compaction - Control Strip MethodSami AjNo ratings yet
- Perry TabsDocument2 pagesPerry TabsJILLIAN DALUPONo ratings yet
- Topic 3.1Document1 pageTopic 3.1duneraoreedNo ratings yet
- Paper: Probing A Chemical Compass: Novel Variants of Low-Frequency Reaction Yield Detected Magnetic ResonanceDocument10 pagesPaper: Probing A Chemical Compass: Novel Variants of Low-Frequency Reaction Yield Detected Magnetic ResonanceDr. Amit Kumar JanaNo ratings yet
- Disclosure: Bearer Biological AssetDocument1 pageDisclosure: Bearer Biological Assetjhell dela cruzNo ratings yet
- Athirah Multazimah Binti Ali Z - Tee0817 4s1-5s1/09-10Document5 pagesAthirah Multazimah Binti Ali Z - Tee0817 4s1-5s1/09-10athir_ahNo ratings yet
- Increase in Temperature (Urban)Document1 pageIncrease in Temperature (Urban)KrutteruHeroNo ratings yet
- Chemical Kinetics 2023Document17 pagesChemical Kinetics 2023Rudra YadavNo ratings yet
- 100 Mental Models - MapsDocument5 pages100 Mental Models - Mapsronel velasquezNo ratings yet
- OpportunityDocument18 pagesOpportunityProvidences DevineNo ratings yet
- Our Easiest Device Yet: With-Patient TestingDocument12 pagesOur Easiest Device Yet: With-Patient TestingTony ChenNo ratings yet
- Rate of Reaction - Freestate 2024 ComboDocument52 pagesRate of Reaction - Freestate 2024 ComboresegomohontiNo ratings yet
- Sacred and Terrible Fate - Character SheetDocument1 pageSacred and Terrible Fate - Character SheetturdipetraNo ratings yet
- 1-5 Eor IorDocument15 pages1-5 Eor IorAnusha AnuNo ratings yet
- Qasim Project Poster Final.Document1 pageQasim Project Poster Final.Qasim KhanNo ratings yet
- Factors Affecting Reaction Rates PDFDocument3 pagesFactors Affecting Reaction Rates PDFFarahNo ratings yet
- Perry TabDocument6 pagesPerry Tabjade brewNo ratings yet
- Gastroenteritis PathophysiologyDocument1 pageGastroenteritis PathophysiologyKeila RosalesNo ratings yet
- Huang2017 - Intrinsic Kinetics Study of Rosin Hydrogenation OnDocument9 pagesHuang2017 - Intrinsic Kinetics Study of Rosin Hydrogenation OnjgNo ratings yet
- t4 SC 1107 Aqa Gcse Chemistry Separate Science Unit 6 The Rate and Extent of Chemical Change KN Ver 7Document4 pagest4 SC 1107 Aqa Gcse Chemistry Separate Science Unit 6 The Rate and Extent of Chemical Change KN Ver 7Sarah KKCNo ratings yet
- Food Safety Plan in TransportingDocument3 pagesFood Safety Plan in TransportingLexi LeysaNo ratings yet
- Actybo Pest Control IncDocument2 pagesActybo Pest Control Inclemron oreiroNo ratings yet
- Disclosure: Biological TransformationDocument1 pageDisclosure: Biological Transformationjhell dela cruzNo ratings yet
- Trading Timing NotesDocument6 pagesTrading Timing NotesJoshua MendietaNo ratings yet
- R1MC Pedia RHD Drug Study Ni HolyDocument2 pagesR1MC Pedia RHD Drug Study Ni HolyHolyver TabarnillaNo ratings yet
- 100 Mental Models - MapsDocument5 pages100 Mental Models - MapsFriedrichNo ratings yet
- Visual Concept MappingDocument2 pagesVisual Concept MappingMaria Ellah C. BELASOTONo ratings yet
- Charsheet Humblewood FancyDocument1 pageCharsheet Humblewood FancyJordan WasemNo ratings yet
- Chemistry Syllabus For Grade 12S Term 1 of 2021Document9 pagesChemistry Syllabus For Grade 12S Term 1 of 2021Davies MasumbaNo ratings yet
- A3 HIRADC CREC 3 Power ElectricalDocument4 pagesA3 HIRADC CREC 3 Power Electricalhidanhaqqi1No ratings yet
- Fundamentals Measurement Diagnostics: Loudspeaker Cone VibrationDocument1 pageFundamentals Measurement Diagnostics: Loudspeaker Cone VibrationmerrickNo ratings yet
- 6.2. Reversible Reactions and Dynamic EquilibriumDocument1 page6.2. Reversible Reactions and Dynamic EquilibriumHarekrishna DasNo ratings yet
- Critical Care Concept Map 2Document1 pageCritical Care Concept Map 2nursing concept mapsNo ratings yet
- Basic Acivalent Numbers of Oxidation: EquationDocument1 pageBasic Acivalent Numbers of Oxidation: Equationkrisna divayana105No ratings yet
- Mind Map Stoikiometri 1Document1 pageMind Map Stoikiometri 1krisna divayana1050% (1)
- Mind Map Stoikiometri 1Document1 pageMind Map Stoikiometri 1krisna divayana105100% (1)
- Homogen: Suspension SolutionDocument1 pageHomogen: Suspension Solutionkrisna divayana105No ratings yet
- Muhammad Farrukh QAMAR - Written TestDocument43 pagesMuhammad Farrukh QAMAR - Written TestRana Muhammad Ashfaq KhanNo ratings yet
- Shane Hagaman Investigating Projectile Motion Lab Marble LauncherDocument5 pagesShane Hagaman Investigating Projectile Motion Lab Marble Launcherapi-550374396100% (1)
- 1 s2.0 S0260877415004203 MainDocument6 pages1 s2.0 S0260877415004203 MainMohsenNo ratings yet
- (MAA 1.10) SYSTEMS OF LINEAR EQUATIONS - EcoDocument3 pages(MAA 1.10) SYSTEMS OF LINEAR EQUATIONS - EcoDAVIES KIPRONONo ratings yet
- Evaluation Exam 17: Kippap EducationDocument22 pagesEvaluation Exam 17: Kippap EducationMichael MercadoNo ratings yet
- CSB-F: Three-Phase Power Capacitor With Fuse ProtectionDocument2 pagesCSB-F: Three-Phase Power Capacitor With Fuse ProtectionAbdul RafaeNo ratings yet
- Dungs Mpa22Document112 pagesDungs Mpa22Piotr MNo ratings yet
- GATE Questions From Previous Papers: GP-CPC - PGP 01Document24 pagesGATE Questions From Previous Papers: GP-CPC - PGP 01shubhamNo ratings yet
- Molecular Computing PPT (Rachit N Ravneet)Document26 pagesMolecular Computing PPT (Rachit N Ravneet)Anit SachdevaNo ratings yet
- Cold-Formed Steel Structures To The AISI Specification (Civil and Environmental Engineering) by Gregory J. Hancock, Thomas Murray, Duane S. EllifritDocument411 pagesCold-Formed Steel Structures To The AISI Specification (Civil and Environmental Engineering) by Gregory J. Hancock, Thomas Murray, Duane S. EllifritDhiraj Roka100% (1)
- SolidWorks Sketch Relations SummaryDocument2 pagesSolidWorks Sketch Relations Summaryxitta00No ratings yet
- 1kz-Te Pedal TroubleshootDocument6 pages1kz-Te Pedal TroubleshootLance Ramai100% (9)
- Mpre CotDocument6 pagesMpre CotPrincess Joy SamoragaNo ratings yet
- Ushio Elevator Operation ManualDocument615 pagesUshio Elevator Operation Manual朝林檎No ratings yet
- Finall Structurall Report For PrintDocument66 pagesFinall Structurall Report For Printpoojitha100% (1)
- Chemistry Practical Part 1Document25 pagesChemistry Practical Part 1Hussain bohraNo ratings yet
- Lab05 - Wing FSI (Lab)Document64 pagesLab05 - Wing FSI (Lab)Guillermo Avila RiveraNo ratings yet
- Tutorial-12 Pre-Tutorial Assignment - Solution: Problem: LetDocument9 pagesTutorial-12 Pre-Tutorial Assignment - Solution: Problem: LetsknNo ratings yet
- Thermodynamics Practice Problems 3Document13 pagesThermodynamics Practice Problems 3Minh Trương PhúcNo ratings yet
- Physics - F4Document25 pagesPhysics - F4Nyomenda Jamhuri WilliamNo ratings yet
- Lecture 1 Why We Study Solid State PhysicsDocument19 pagesLecture 1 Why We Study Solid State Physicsريمكس REMIXNo ratings yet
- Electric Dipole Radiation From An Arbitrary SourceDocument4 pagesElectric Dipole Radiation From An Arbitrary SourceAlejandro GuzmánNo ratings yet
- Norbert Didden - On The Formation of Vortex Rings: Rolling-Up and Production of CirculationDocument16 pagesNorbert Didden - On The Formation of Vortex Rings: Rolling-Up and Production of CirculationJuaxmawNo ratings yet
- Applied Physics: Electric Charge Coulomb's LawDocument19 pagesApplied Physics: Electric Charge Coulomb's LawAhmadNo ratings yet
- Busce Rivkle GBDocument56 pagesBusce Rivkle GBVlad MarianNo ratings yet
- Sofar 11000 - 18000TL-LVDocument37 pagesSofar 11000 - 18000TL-LVbruno300kmNo ratings yet
- Weekly Learning Activity Sheets General Physics 1 Grade 12, Quarter 2, Week 4Document4 pagesWeekly Learning Activity Sheets General Physics 1 Grade 12, Quarter 2, Week 4Shekaina Faith Cuizon Lozada100% (2)
- Blok Design Receive Blok Design Cannel OptikDocument1 pageBlok Design Receive Blok Design Cannel Optikbpr platinmods4No ratings yet
- Astm E2847.21990Document17 pagesAstm E2847.21990Astrid Camila Riveros Mesa100% (7)