Professional Documents
Culture Documents
Formula Kimia
Formula Kimia
Uploaded by
Sara Syuib II0 ratings0% found this document useful (0 votes)
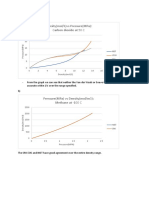
11 views2 pagesThe document provides concentration and volume information for various acids and salts that can be prepared in the lab. Specifically, it lists the grams or milliliters of chemical needed to make solutions of different molar concentrations for acids such as hydrochloric acid, sulfuric acid, and nitric acid as well as salts including sodium hydroxide, silver nitrate, and potassium iodide. The tables provide this information for solutions prepared in volumes of 250mL, 500mL, and 1000mL.
Original Description:
Original Title
formula kimia.xlsx
Copyright
© © All Rights Reserved
Available Formats
XLSX, PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentThe document provides concentration and volume information for various acids and salts that can be prepared in the lab. Specifically, it lists the grams or milliliters of chemical needed to make solutions of different molar concentrations for acids such as hydrochloric acid, sulfuric acid, and nitric acid as well as salts including sodium hydroxide, silver nitrate, and potassium iodide. The tables provide this information for solutions prepared in volumes of 250mL, 500mL, and 1000mL.
Copyright:
© All Rights Reserved
Available Formats
Download as XLSX, PDF, TXT or read online from Scribd
Download as xlsx, pdf, or txt
0 ratings0% found this document useful (0 votes)
11 views2 pagesFormula Kimia
Formula Kimia
Uploaded by
Sara Syuib IIThe document provides concentration and volume information for various acids and salts that can be prepared in the lab. Specifically, it lists the grams or milliliters of chemical needed to make solutions of different molar concentrations for acids such as hydrochloric acid, sulfuric acid, and nitric acid as well as salts including sodium hydroxide, silver nitrate, and potassium iodide. The tables provide this information for solutions prepared in volumes of 250mL, 500mL, and 1000mL.
Copyright:
© All Rights Reserved
Available Formats
Download as XLSX, PDF, TXT or read online from Scribd
Download as xlsx, pdf, or txt
You are on page 1of 2
Asid Hidroklorik (HCl) Natrium Hydroxide (NaOH)
250ml 500 ml 1000ml 250ml 500 ml 1000ml
0.1 8.6 0.25 1
0.2 43 0.1 4
1M 86 1M 40
2M 173 2M 80
5M 200
Asid Sulfurik (H2S04) Argentum/Silver Nitrate(AgNO3
250ml 500 ml 1000ml 250ml 500 ml 1000ml
0.1 5.6 0.1
0.5 2.8 0.05
1M 55
2M 112
Asid Nitrik (HN03) Plumbum (ii)Nitrate
250ml 500 ml 1000ml 250ml 500 ml 1000ml
0.1 6.4 0.02 6.62
1M 65 0.05 16.55
2M 128 0.1 33.1
0.25 82.75
0.5 165.5
1M 331
Kalium Iodida KI Iron/Ferum (ii) Sulfat
250ml 500 ml 1000ml 250ml 500 ml 1000ml
0.2M 16.5 0.5M 139
0.5M 83 *campur dengan 50 ml asid sulfurik pekat cegah hidrolisis
0.15M 24.9
Kuprum (ii) Sulfat Zinc Sulfate (ZnSO4)
250ml 500 ml 1000ml 250ml 500 ml 1000ml
0.1 25 0.5 143.5
0.5 125 1M 287
1M 250
You might also like
- ABMA Water GuideDocument1 pageABMA Water GuidesizmaruNo ratings yet
- Hinged 42mm2 PDFDocument120 pagesHinged 42mm2 PDFViswanathan Kannoor67% (3)
- STD PreparationDocument4 pagesSTD PreparationgesecNo ratings yet
- Data - Data Lab Chemical & MaterialDocument8 pagesData - Data Lab Chemical & MaterialmetalosoekNo ratings yet
- Perhi B2Document10 pagesPerhi B2Aldyan FaturohmanNo ratings yet
- DNP Standard CurveDocument11 pagesDNP Standard CurveMoni JacobNo ratings yet
- Medias: Yeast Mannitol AgarDocument7 pagesMedias: Yeast Mannitol AgarKhadijaNo ratings yet
- STT Tên Hóa Chất Cthh Quy Cách Sl/Đvt: (Ghi Rõ Nồng Độ)Document2 pagesSTT Tên Hóa Chất Cthh Quy Cách Sl/Đvt: (Ghi Rõ Nồng Độ)price theshinNo ratings yet
- Chemical Consumption Format FRM-QCDocument7 pagesChemical Consumption Format FRM-QCVinay PalNo ratings yet
- IP-025 Reagents For COD-EnDocument2 pagesIP-025 Reagents For COD-EnkhuzaimahNo ratings yet
- Fed Batch 12 (5L) - 24 Nov 2022Document4 pagesFed Batch 12 (5L) - 24 Nov 2022Yolanda Winarny Eviphanie HutabaratNo ratings yet
- Seperation 2 GraphDocument3 pagesSeperation 2 GraphHamza SulehriNo ratings yet
- Hydroponic Nutrient Calulation SheetDocument6 pagesHydroponic Nutrient Calulation SheetravikirnnNo ratings yet
- Collection of Formulas For The Chemical, Electrochemical and Heat Colouring of MetalsDocument57 pagesCollection of Formulas For The Chemical, Electrochemical and Heat Colouring of MetalsOscar Del BarcoNo ratings yet
- CompoundsDocument1 pageCompoundsJian MinNo ratings yet
- ParameterDocument1 pageParameterlaki oxrNo ratings yet
- SP Heat Air and CLK PDFDocument1 pageSP Heat Air and CLK PDFDilnesa EjiguNo ratings yet
- List of Chemicals Needed in Biochemistry LaboratoryDocument3 pagesList of Chemicals Needed in Biochemistry LaboratoryMary Grace Ancheta RamosNo ratings yet
- Technical Data Sheet - AK34 Caustic Soda FlakesDocument1 pageTechnical Data Sheet - AK34 Caustic Soda FlakesArnoldo SanchezNo ratings yet
- Water QualityDocument1 pageWater QualityAgus Muhamad ZamilNo ratings yet
- Specification Guide: Chrome High Temp Fittings & FlangesDocument4 pagesSpecification Guide: Chrome High Temp Fittings & FlangesAdrian EspinosaNo ratings yet
- Tube Catalogue AnitaDocument64 pagesTube Catalogue AnitaChinnaraja GandhiNo ratings yet
- Chemical Compositions 1Document1 pageChemical Compositions 1Zahoor Hussain RanaNo ratings yet
- Copia de METODOS PREPARACION ESTANDAR PLASMADocument5 pagesCopia de METODOS PREPARACION ESTANDAR PLASMAjorgeNo ratings yet
- Concentration of NH4Cl (MM)Document3 pagesConcentration of NH4Cl (MM)Ali TarekNo ratings yet
- ASTM-Graded-Sand Datasheet PDFDocument1 pageASTM-Graded-Sand Datasheet PDFmalaya tripathy100% (1)
- Attention Sample Data: Lube Oil Analysis ReportDocument1 pageAttention Sample Data: Lube Oil Analysis ReportnurdinzaiNo ratings yet
- Attention Sample Data: Lube Oil Analysis ReportDocument1 pageAttention Sample Data: Lube Oil Analysis ReportnurdinzaiNo ratings yet
- Quantum Arc 6: Hobart Brothers LLC 101 Trade Square East Troy, OH 45373Document2 pagesQuantum Arc 6: Hobart Brothers LLC 101 Trade Square East Troy, OH 45373Gabor VatoNo ratings yet
- DNP Standard Curve - XLSX - Google SheetsDocument5 pagesDNP Standard Curve - XLSX - Google SheetsMoni JacobNo ratings yet
- Thermodynamics Exercise 2Document7 pagesThermodynamics Exercise 2Julia SatrianiNo ratings yet
- DSS 5 10Document3 pagesDSS 5 10kahivhenqcasinilloNo ratings yet
- Calculation For Standard PreparationDocument2 pagesCalculation For Standard Preparationjonahlyn.cuelloNo ratings yet
- Minimum Essential Medium Eagle (MEM) Alpha Modifications: M0644 M0894 M4526 M6199 M8042 M6074Document2 pagesMinimum Essential Medium Eagle (MEM) Alpha Modifications: M0644 M0894 M4526 M6199 M8042 M6074Aranza CamachoNo ratings yet
- Glass Bubbles K S Series PDFDocument4 pagesGlass Bubbles K S Series PDFLinheris D.A SitompulNo ratings yet
- 30 - 08 - Formato Control de Inventario GPBDocument1 page30 - 08 - Formato Control de Inventario GPBReizo Medina GonzalesNo ratings yet
- Ion Exchange CalculationDocument3 pagesIon Exchange CalculationAfra AlethianaNo ratings yet
- Density (Mol/L) Vs Pressure (Mpa) : Carbon Dioxide at 50 CDocument6 pagesDensity (Mol/L) Vs Pressure (Mpa) : Carbon Dioxide at 50 CbobNo ratings yet
- BCA Standard CurveDocument4 pagesBCA Standard CurveJavier AlemanNo ratings yet
- Characteristics of Boiler Feedwater - LenntechDocument1 pageCharacteristics of Boiler Feedwater - LenntechjagjitNo ratings yet
- Gráficas de Mediciones de ConductividadDocument6 pagesGráficas de Mediciones de ConductividadSantiago Perez Maria FernandaNo ratings yet
- Stream Table Production of Ferric Chloride SolutionDocument2 pagesStream Table Production of Ferric Chloride Solutionsyafiq izzuddin bin sapriNo ratings yet
- Alloy 200/201: Commercially Pure Nickel (99.6%) With Good Mechanical Properties and Excellent Corrosion ResistanceDocument4 pagesAlloy 200/201: Commercially Pure Nickel (99.6%) With Good Mechanical Properties and Excellent Corrosion ResistanceRohit KarhadeNo ratings yet
- Description Qty Available 3 1 1 3 1 1 2 1 2 1 1 3 3 1 1 1 1 1 1 3 5 8 1 1 1 1 5 1 1 1 2 1 1 1 1Document2 pagesDescription Qty Available 3 1 1 3 1 1 2 1 2 1 1 3 3 1 1 1 1 1 1 3 5 8 1 1 1 1 5 1 1 1 2 1 1 1 1ongcchemist gd chitraNo ratings yet
- Merck (250-288)Document40 pagesMerck (250-288)mariyamsNo ratings yet
- Methyl Blue Spiritus: 500 ML 1000 MLDocument6 pagesMethyl Blue Spiritus: 500 ML 1000 MLMaxi GrahaNo ratings yet
- Exp 4result Discussion For FaDocument4 pagesExp 4result Discussion For FanasuhaNo ratings yet
- How Much Quantity of Chemical Reuqired For Chilled Water SystemDocument3 pagesHow Much Quantity of Chemical Reuqired For Chilled Water SystemRaju Ksn100% (1)
- Bio 100 Ex2b FlowchartDocument3 pagesBio 100 Ex2b Flowchartmikoticaro0910No ratings yet
- Konversi Volume Ke Berat Kantong DarahDocument5 pagesKonversi Volume Ke Berat Kantong Darahwahyu ariNo ratings yet
- Norris-Steel-Sucker-Rods - Pony-Rods - P001-V02-100908 - 5Document1 pageNorris-Steel-Sucker-Rods - Pony-Rods - P001-V02-100908 - 5Mohamed GhareebNo ratings yet
- 2 5415610794232517386Document4 pages2 5415610794232517386eman hussainNo ratings yet
- Sample Number Sample Time Elapsed Time Sodium Hydroxide Concentaion (Mol/dm ) Ethyl Acetate Concentraio N (Mol/dm )Document5 pagesSample Number Sample Time Elapsed Time Sodium Hydroxide Concentaion (Mol/dm ) Ethyl Acetate Concentraio N (Mol/dm )Kelly Sheine SisonNo ratings yet
- Sample Number Sample Time Elapsed Time Sodium Hydroxide Concentaion (Mol/dm ) Ethyl Acetate Concentraio N (Mol/dm )Document5 pagesSample Number Sample Time Elapsed Time Sodium Hydroxide Concentaion (Mol/dm ) Ethyl Acetate Concentraio N (Mol/dm )Kelly Sheine SisonNo ratings yet
- Curva PNF LunesDocument7 pagesCurva PNF Lunesgina vargasNo ratings yet
- Silica/Sand Specifications: Particle Size Analysis and PropertiesDocument1 pageSilica/Sand Specifications: Particle Size Analysis and PropertiesforuzzNo ratings yet
- Across Pro DS EN 80774703 - CDocument2 pagesAcross Pro DS EN 80774703 - CZoimar iseaNo ratings yet