Professional Documents
Culture Documents
Organic Chemistry Laboratory: Report 7: Liquid-Liquid Extraction
Organic Chemistry Laboratory: Report 7: Liquid-Liquid Extraction
Uploaded by
Phú NguyễnOriginal Description:
Original Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Organic Chemistry Laboratory: Report 7: Liquid-Liquid Extraction
Organic Chemistry Laboratory: Report 7: Liquid-Liquid Extraction
Uploaded by
Phú NguyễnCopyright:
Available Formats
International University, Vietnam National University - HCMC
School of Biotechnology
Department of Biochemistry
ORGANIC CHEMISTRY LABORATORY
REPORT 7: LIQUID- LIQUID EXTRACTION
INSTRUCTOR: PhD. Hoàng Lê Sơn
TEACHING ASSISTANT: Nguyễn Thanh Phong
DATE OF SUBMISSION: 16-07-2019
Group member:
1. Trương Thị Ngọc Hằng - BTBCIU16041
2. Võ Minh Hạnh - BTBCIU16083
3. Nguyễn Hồng Anh Phương - BTBCIU16015
4. Nguyễn Tấn Phú – BTBCIU16057
Organic Chemistry Laboratory 1
International University, Vietnam National University - HCMC
School of Biotechnology
Department of Biochemistry
I. Abstract
The experiment was about using a liquid-liquid extraction technique to remove
tea-consisting caffeine and recrystallization to reform caffeine crystals. The main
principle is the compound's polarity and solubility. The solution is separated into two
layers and the layer required either top or bottom layer is collected. After this
experiment, about 0.03 g of caffeine out of 30.00 g of tea was acquired, and the
percentage yield of crystallization was 60.00%.
II. Introduction
Tea is one of the world's most widely used caffeinated drinks. The caffeine
(C8H10N4O2) discovered in tea is a bitter, white, crystalline methylxanthine and a
member of a compound class known as alkaloids. Caffeine also triggers increased
respiration and heart rate, as well as nervousness and insomnia. Although caffeine
has demonstrated physical dependence, it is also capable of enhancing alertness,
learning ability, and exercise efficiency (NCBI, 2013). Tea leaves, where caffeine is
discovered, also contain acidic tannins, undecomposed chlorophyll, cellulose, and
pigments. To remove caffeine from tea leaves, caffeine must be present as the free
foundation (Amrita, 2013). To do so, the acidic substances mentioned above must
stay water-soluble.
The extraction of liquid-liquid is the method for separating compounds in
different solvents based on their relative solubility. It is an extraction of a substance
from one liquid to another liquid phase. The easy-to-use technique of static
extraction is commonly used and very efficient in the field of extraction analysis.
A separate funnel, also known as a separation funnel that operates on the
notion of "like dissolves like" that we learned in chemistry engineering. This implies
that if a compound is polar, it can only be dissolved in a polar solvent. In accordance
with this logic, a non-polar compound can only be dissolved in a non-polar solvent.
There are two phases in a separate funnel, the aqueous layer is polar, and the organic
layer is non-polar. Since these two layers share a surface, any polar substances that
were originally in the solution will be taken into the polar (aqueous) layer and any
non-polar substances will be drawn into the non-polar (organic) layer. The greatest
danger when using a separate funnel is pressure build-up. Pressure accumulates
during blending when gas responses happen. This issue can be addressed readily by
regularly opening the stopper at the top of the funnel while blending. This should be
performed with the top of the funnel pointing away from the body. Hold the stopper
in position when shaking, or it may dislodge causing the fluids to spill. Simply keep
the stopper in place with one side to account for this. The solvent is the key to
effective liquid-liquid extraction separation. Less toxic (non-toxic), not to mix water,
solubility of component in solvent, proportion of component capable of extraction,
density, vapor pressure, viscosity, interfacial tension, etc. Liquid-liquid extraction has
Organic Chemistry Laboratory 2
International University, Vietnam National University - HCMC
School of Biotechnology
Department of Biochemistry
several applications in bioprocess technology: fermentation and algae broths,
removal of elevated boiling organics from sewage, removal of carboxylic acid,
protein separation and removal.
The advantages of this process include its effectiveness towards compounds
with high boiling points even at low concentrations, azeotropic mixtures (mixes with
points that, when boiled, have the same composition in vapor as initially, therefore
it cannot be modified by easy distillation) and heat-sensitive compounds such as
big organic molecules. The disadvantages are the high consumption of potentially
poisonous solvent, which also needs expensive machinery when reused in the
scheme. Thus, a proper choice of the solvent is needed in order to achieve a
sustainable process and generates difficulties for the large-scale sector to implement
the process.
III. Materials and chemicals
• Materials:
Some equipments were prepared, including one Erlenmeyer flask 250 mL, a
heating plate, a vacuum filter, one cylinder 10 mL and a cylinder 100 mL, a beaker
10 mL, a balance, and one separatory funnel 250 mL. Besides, the chemical reagent
was also prepared with the sample was dry tea leaf, methylene chloride, benzene,
and petroleum ether.
• Method:
The first step was the preparation of sample, tea solution was prepared by
boiling thirty grams of dry tea and 100 mL of water in ten minutes, after the tea
solution was filtered the binder by suction filtration. The liquid of caffeine in tea
was saved and kept it cool down, then the solution was transferred to the
separatory funnel 250 mL and then extracted three times with methylene chloride.
Methylene chloride allowed caffeine could be dissolved. When using the separatory
funnel containing solution did not shake the layers vigorously and use a swirling
motion. Moreover, the combination of methylene chloride extracts and evaporation
the extract to dryness on the steam bath made the crude caffeine, after transferring
the crude caffeine to a 50 mL beaker and added 5 mL of benzene, the heating it in
water bath. Benzene could dissolve chlorophyll in crude caffeine and also dissolve
caffeine. The important step to recrystallization was adding 10 ml of petroleum
ether, the product of recrystallization could be collected by suction filtration and
washed with 1 mL of petroleum ether which allowed to air-dry. Finally, the final
products were used for determining the melting boil.
Organic Chemistry Laboratory 3
International University, Vietnam National University - HCMC
School of Biotechnology
Department of Biochemistry
IV. Result
1. Extraction process:
Figure 1: Two separate solvent layers Figure 2: Methylene chloride extract
Figure 3: Crude caffeine after extraction process
2. Purification process:
Organic Chemistry Laboratory 4
International University, Vietnam National University - HCMC
School of Biotechnology
Department of Biochemistry
Figure 4: Caffeine crystal
Figure 5: Recovered crystal weight
Organic Chemistry Laboratory 5
International University, Vietnam National University - HCMC
School of Biotechnology
Department of Biochemistry
Figure 6: Caffeine’s melting point
V. Discussion
In the case of Caffeine extraction from tea powder, the organic solvent
dichloromethane is used because caffeine is more soluble in dichloromethane (140
mg/ml) than it is in water (22 mg/ml). The dichloromethane - caffeine mixture can
then be separated on the basis of the different densities of dichloromethane and
water because dichloromethane is much denser than water and insoluble in it. The
lower layer contained caffeine that diluted in methylene chloride.
Residual water is separated from dichloromethane by drain out the
dichloromethane through separating funnel, thus dichloromethane passed through
the funnel while polar solvents such as water is still remained in the funnel. Water
and dichloromethane is slightly soluble in each other. So, after separating the
solvents, residual water will remain the organic layer. Therefore, drying out the
solution to collect crude caffeine is necessity.
Recrystallize and determination melting point were performed to purify the
collected sample. In the recrystallize process, be careful to wash with petroleum
Organic Chemistry Laboratory 6
International University, Vietnam National University - HCMC
School of Biotechnology
Department of Biochemistry
ether so as not to dissolve any caffeine. When putting petroleum ether, crystal was
formed. After filtrating and dryness was completed, caffeine was done in
crystallization. The percentage of recovery caffeine crystal from crude caffeine was
60%, and melting point of caffeine crystal was 223.8 degree Celsius, which was a bit
lower compare to 225 Celsius degree, official melting point of caffeine. It was
because of the impurity crystal, the crystal might be contaminated, or the paper
powder of the filter paper was mixed in.
VI. Conclusion
Caffeine is the world's most popular drug and can be found in many beverages
including tea. Caffeine was isolated from black tea leaves by using solid-liquid
extraction followed by liquid-liquid extraction. Therefore, solute in organic solvent,
caffeine was mix with methylene chloride which was the good organic solvent.
VII. Reference
• Organic Chemistry Laboratory Manual of the experiment 2. Ho Chi Minh:
International University, HCMC, 2018.
• vlab.amrita.edu,. (2011). Extraction of Caffeine from Tea. Retrieved 20 July 2019,
from vlab.amrita.edu
Organic Chemistry Laboratory 7
You might also like
- Solvent Extraction Lab ReportDocument11 pagesSolvent Extraction Lab ReportFaiz Farhan80% (5)
- Peptide HandbookDocument26 pagesPeptide HandbookKim HiềnNo ratings yet
- Protein Synthesis Questions HL ExtraDocument9 pagesProtein Synthesis Questions HL ExtraJohn OsborneNo ratings yet
- Experiment 3Document3 pagesExperiment 3BuiHopeNo ratings yet
- Liquid Liquid ExtractionDocument11 pagesLiquid Liquid Extractionvisha lkumar vermaNo ratings yet
- ORGANIC CHEMISTRY LABORATORY - Lab 2Document10 pagesORGANIC CHEMISTRY LABORATORY - Lab 2Phú NguyễnNo ratings yet
- Organic Chemistry Laboratory: Report 6: Reflux ReactionDocument7 pagesOrganic Chemistry Laboratory: Report 6: Reflux ReactionPhú NguyễnNo ratings yet
- 220CH1630 - M.tech ProjectDocument16 pages220CH1630 - M.tech ProjectMAHIMA BAIDNo ratings yet
- Organic Chemistry Laboratory: Report 4: Column ChromatographyDocument5 pagesOrganic Chemistry Laboratory: Report 4: Column ChromatographyPhú NguyễnNo ratings yet
- Comparison Between Single and Multiple Extraction of Caffeine From Dried Tea LeavesDocument3 pagesComparison Between Single and Multiple Extraction of Caffeine From Dried Tea LeavesKierby PecundoNo ratings yet
- Org Exp 45 PDFDocument15 pagesOrg Exp 45 PDFMardhia AtiqahNo ratings yet
- Organic Chemistry: Basra University College of Science and Technology Pharmacy DepartmentDocument10 pagesOrganic Chemistry: Basra University College of Science and Technology Pharmacy DepartmentcrtgyhujikNo ratings yet
- Organic Chemistry Laboratory: Report 3: Thin Layer ChromatographyDocument7 pagesOrganic Chemistry Laboratory: Report 3: Thin Layer ChromatographyPhú NguyễnNo ratings yet
- Single Extraction of Caffeine From Dried Tea LeavesDocument3 pagesSingle Extraction of Caffeine From Dried Tea LeavesAlyanna CacasNo ratings yet
- Experiment 2 Chem 33.1Document4 pagesExperiment 2 Chem 33.1Lucile BronzalNo ratings yet
- A192577 Report CaffeineDocument6 pagesA192577 Report CaffeineNOR AFIFAH BINTI MOHD AKHIARNo ratings yet
- Comparison Between Single and Multiple Extraction of Caffeine From Dried Tea LeavesDocument3 pagesComparison Between Single and Multiple Extraction of Caffeine From Dried Tea LeavesElizabeth Johnson33% (3)
- Lab ReportDocument3 pagesLab ReportKerby OmasasNo ratings yet
- Extraction of Caffeine From Tea Leaves With The Single Extraction MethodDocument3 pagesExtraction of Caffeine From Tea Leaves With The Single Extraction MethodMiguel Tan Casis100% (1)
- Caffeine ExtractionDocument9 pagesCaffeine ExtractionMehwish IqbalNo ratings yet
- Experiment 2 - ExtractionDocument3 pagesExperiment 2 - ExtractionIson DyNo ratings yet
- Extraction Polyphenol Review 16pDocument16 pagesExtraction Polyphenol Review 16pHồng Thắng100% (1)
- Isolation of Caffeine From Dried Green Tea Leaves Through Multiple Liquid-Liquid ExtractionDocument6 pagesIsolation of Caffeine From Dried Green Tea Leaves Through Multiple Liquid-Liquid ExtractionSebastian MoronNo ratings yet
- Experiment With The First ClassDocument2 pagesExperiment With The First ClassaizatNo ratings yet
- Plant BiotechnologyDocument25 pagesPlant Biotechnologygrace meunierNo ratings yet
- Exp 2 Chemlab Formal ReportDocument3 pagesExp 2 Chemlab Formal ReportHarvey Mher RarangNo ratings yet
- Trịnh Huy Cường Đặng Nguyễn Minh Khôi: Natural ProductsDocument47 pagesTrịnh Huy Cường Đặng Nguyễn Minh Khôi: Natural ProductsAndré Luiz Meleiro PortoNo ratings yet
- Derivatization To Make Compound VolatileDocument5 pagesDerivatization To Make Compound VolatileshahnawazNo ratings yet
- NUTRACEUTICALS LABORATORY Lab 2Document10 pagesNUTRACEUTICALS LABORATORY Lab 2Phú NguyễnNo ratings yet
- Separation of Caffeine and Catechin Compounds FromDocument6 pagesSeparation of Caffeine and Catechin Compounds FromAssis GomesNo ratings yet
- A. Title of ExperimentDocument18 pagesA. Title of ExperimentRafidah AmaliaNo ratings yet
- Extracción Fenólicos 2014Document7 pagesExtracción Fenólicos 2014Kathia Acosta JaimeNo ratings yet
- Report 3 OfficiallyDocument7 pagesReport 3 OfficiallyLinhNguyeNo ratings yet
- Formal Report ExtractionDocument5 pagesFormal Report ExtractionPhilina PasicolanNo ratings yet
- Single ExtractionDocument3 pagesSingle Extractioncarlyzza021412No ratings yet
- 3 Practice 2 Extraction TechniqueDocument26 pages3 Practice 2 Extraction TechniqueNguyen Ngoc Lam VyNo ratings yet
- 2020 VietnameseDocument7 pages2020 VietnameseLAURANo ratings yet
- Medicinal Chemistry PpotDocument12 pagesMedicinal Chemistry PpotAkira SinhaNo ratings yet
- Extraction of Caffeine From Tea Leaves With The Single Extraction MethodDocument3 pagesExtraction of Caffeine From Tea Leaves With The Single Extraction MethodKierby PecundoNo ratings yet
- Laboratory Report 2 - Canillas, CDocument5 pagesLaboratory Report 2 - Canillas, CColline Faye CanillasNo ratings yet
- Optimization of Sequential Supercritical Fluid Extraction (Sfe) of Caffeine & Catechins From Green TeaDocument14 pagesOptimization of Sequential Supercritical Fluid Extraction (Sfe) of Caffeine & Catechins From Green TeaBharath ChandranNo ratings yet
- Analysis of Phenolic Content and Antioxidant Activity of Cocoa Pod Husk PDFDocument6 pagesAnalysis of Phenolic Content and Antioxidant Activity of Cocoa Pod Husk PDFTài ColeNo ratings yet
- 2023 Efficient Extraction, Excellent Activity, and Microencapsulation of Flavonoids From Moringa Oleifera Leaves Extracted by Deep Eutectic SolventDocument15 pages2023 Efficient Extraction, Excellent Activity, and Microencapsulation of Flavonoids From Moringa Oleifera Leaves Extracted by Deep Eutectic SolventOlíviaGomesNo ratings yet
- 10 1016@j Cogsc 2017 03 010Document5 pages10 1016@j Cogsc 2017 03 010rahmanNo ratings yet
- ISOLATION AND EXTRACTION OF nATURAL PRODCUTSDocument4 pagesISOLATION AND EXTRACTION OF nATURAL PRODCUTSAlvina RiazNo ratings yet
- Extraction: Miranda, Marilyn, de Guzman, Anne KDocument4 pagesExtraction: Miranda, Marilyn, de Guzman, Anne KAegeeNo ratings yet
- Biosep Short OverviewDocument9 pagesBiosep Short OverviewMeyakorberNo ratings yet
- Extraction and Purification of CaffeineDocument3 pagesExtraction and Purification of CaffeineAnonymous GO6JVW9Wud100% (2)
- Solvent Extraction Lab ReportDocument11 pagesSolvent Extraction Lab ReportWan Nurshahira100% (2)
- Extraction of Caffeine From TeaDocument7 pagesExtraction of Caffeine From Teaامنة محمد حسن احمدNo ratings yet
- 06 Exp 04 Extraction 2Document8 pages06 Exp 04 Extraction 2lklepoNo ratings yet
- Full Exp 4Document8 pagesFull Exp 4Rashdan CskNo ratings yet
- G02 CM133L Experiment6Document6 pagesG02 CM133L Experiment6Alexandria Nicole CaalimNo ratings yet
- CHM457 Exp1Document5 pagesCHM457 Exp1AMIRAH ISHAMI ISHAKNo ratings yet
- BiotechnologyDocument6 pagesBiotechnologysben03675No ratings yet
- Department of Pharmacy: The Extraction MR - Ahmed AlmohhmmedDocument10 pagesDepartment of Pharmacy: The Extraction MR - Ahmed AlmohhmmedcrtgyhujikNo ratings yet
- Food Chemistry: Yongqiang Wang, Yujie Gao, Hui Ding, Shejiang Liu, Xu Han, Jianzhou Gui, Dan LiuDocument7 pagesFood Chemistry: Yongqiang Wang, Yujie Gao, Hui Ding, Shejiang Liu, Xu Han, Jianzhou Gui, Dan LiuDessy Erlyani Mugita SariNo ratings yet
- Expt. 3 - Separation and Purification of Org. Compounds (Part 2) (20230214121826)Document5 pagesExpt. 3 - Separation and Purification of Org. Compounds (Part 2) (20230214121826)Kaye SaavedraNo ratings yet
- Ahmad Zakwan Bin Kassim - Lab 1 - CHM557Document13 pagesAhmad Zakwan Bin Kassim - Lab 1 - CHM557Ahmad ZakwanNo ratings yet
- Isolation and Sublimation of Caffeine From Tea LeavesDocument8 pagesIsolation and Sublimation of Caffeine From Tea Leavesmaryamarfa762No ratings yet
- Preview PDFDocument103 pagesPreview PDFPhú NguyễnNo ratings yet
- Organic Chemistry Laboratory: Report 8: Fischer Ester SynthesisDocument7 pagesOrganic Chemistry Laboratory: Report 8: Fischer Ester SynthesisPhú NguyễnNo ratings yet
- Ethics of Research Using Hybrids, Chimeras and Cytoplasmic Hybrids?Document7 pagesEthics of Research Using Hybrids, Chimeras and Cytoplasmic Hybrids?Phú NguyễnNo ratings yet
- Organic Chemistry Laboratory: Report 5: Simple DistillationDocument8 pagesOrganic Chemistry Laboratory: Report 5: Simple DistillationPhú NguyễnNo ratings yet
- Melting Point: Point at Which Solid and Liquid Phases Are in EquilibriumDocument6 pagesMelting Point: Point at Which Solid and Liquid Phases Are in EquilibriumPhú NguyễnNo ratings yet
- Lab 2 CosDocument4 pagesLab 2 CosPhú NguyễnNo ratings yet
- ORGANIC CHEMISTRY LABORATORY - Lab 2Document10 pagesORGANIC CHEMISTRY LABORATORY - Lab 2Phú NguyễnNo ratings yet
- Organic Chemistry Laboratory: Report 6: Reflux ReactionDocument7 pagesOrganic Chemistry Laboratory: Report 6: Reflux ReactionPhú NguyễnNo ratings yet
- Organic Chemistry Laboratory: Report 4: Column ChromatographyDocument5 pagesOrganic Chemistry Laboratory: Report 4: Column ChromatographyPhú NguyễnNo ratings yet
- Subject: Organic chemistry laboratory. Lab partner: Lê Hồ Thi Đỗ Trương Anh Thư Bùi Hữu Đức Trần Hoàng Thanh Tuyền Instructor's name: Dr. Hoang Le SonDocument46 pagesSubject: Organic chemistry laboratory. Lab partner: Lê Hồ Thi Đỗ Trương Anh Thư Bùi Hữu Đức Trần Hoàng Thanh Tuyền Instructor's name: Dr. Hoang Le SonPhú NguyễnNo ratings yet
- Assignment 2 Fermentation PDFDocument2 pagesAssignment 2 Fermentation PDFPhú NguyễnNo ratings yet
- NUTRACEUTICALS LABORATORY Lab 2Document10 pagesNUTRACEUTICALS LABORATORY Lab 2Phú NguyễnNo ratings yet
- Organic Chemistry Laboratory: Report 3: Thin Layer ChromatographyDocument7 pagesOrganic Chemistry Laboratory: Report 3: Thin Layer ChromatographyPhú NguyễnNo ratings yet
- Assignment 1 - NguyenTanPhuBTBCIU16057Document4 pagesAssignment 1 - NguyenTanPhuBTBCIU16057Phú NguyễnNo ratings yet
- Nutraceuticals Laboratory Report 3: Determination of Total Flavonoid ContentDocument7 pagesNutraceuticals Laboratory Report 3: Determination of Total Flavonoid ContentPhú NguyễnNo ratings yet
- Organic Chemistry Semester 2 Homework 1 1Document2 pagesOrganic Chemistry Semester 2 Homework 1 1Phú NguyễnNo ratings yet
- ActivitiesDocument1 pageActivitiesPhú NguyễnNo ratings yet
- Solution HW1Document4 pagesSolution HW1Phú NguyễnNo ratings yet
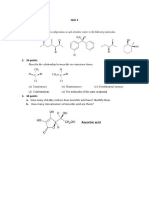
- Quiz 2Document1 pageQuiz 2Phú NguyễnNo ratings yet
- Organic Chemistry Semester 2 Homework 1 1Document3 pagesOrganic Chemistry Semester 2 Homework 1 1Phú NguyễnNo ratings yet
- Part A: Preparation of Kmno Solution. Experiment I:: N 0.0025 (Mol) M 0.395 (G)Document5 pagesPart A: Preparation of Kmno Solution. Experiment I:: N 0.0025 (Mol) M 0.395 (G)Phú NguyễnNo ratings yet
- Organic Chemistry 7 by John McmurryDocument10 pagesOrganic Chemistry 7 by John McmurryPhú NguyễnNo ratings yet
- CHEM 2425. Chapter 28. Biomolecules - Nucleic Acids (Homework) WDocument15 pagesCHEM 2425. Chapter 28. Biomolecules - Nucleic Acids (Homework) WPhú NguyễnNo ratings yet
- Homework 2: StrereoisomersDocument3 pagesHomework 2: StrereoisomersPhú NguyễnNo ratings yet
- Homework 3: Organic Chemistry 7 by John McmurryDocument1 pageHomework 3: Organic Chemistry 7 by John McmurryPhú NguyễnNo ratings yet
- Homework 2: StrereoisomersDocument3 pagesHomework 2: StrereoisomersPhú NguyễnNo ratings yet
- Quiz 1Document2 pagesQuiz 1Phú NguyễnNo ratings yet
- MULTIPLE CHOICE. Choose The One Alternative That Best Completes The Statement or Answers The QuestionDocument24 pagesMULTIPLE CHOICE. Choose The One Alternative That Best Completes The Statement or Answers The QuestionPhú NguyễnNo ratings yet
- Report 2: Genetics LaboratoryDocument5 pagesReport 2: Genetics LaboratoryPhú NguyễnNo ratings yet
- FT Course Syllabus Sep 2019Document4 pagesFT Course Syllabus Sep 2019Phú NguyễnNo ratings yet
- US2662052Document4 pagesUS2662052DŨNG VŨ NGUYỄN TUẤNNo ratings yet
- 0126 Cat-C Chemistry (Xi, Xii) Paper BDocument2 pages0126 Cat-C Chemistry (Xi, Xii) Paper Bsrikanthdip007No ratings yet
- Introduction To Biotechnology 3rd Edition Thieman Test BankDocument26 pagesIntroduction To Biotechnology 3rd Edition Thieman Test BankDanielleNelsonxfiq100% (64)
- Field Guide TemplateDocument2 pagesField Guide TemplateAlberto RicardoNo ratings yet
- EAPCETDocument2 pagesEAPCETP. Phani prasadNo ratings yet
- Amines QuestionsDocument12 pagesAmines QuestionsAnuradha DamaleNo ratings yet
- 823 1731 1 PBDocument9 pages823 1731 1 PBRegita ReynataNo ratings yet
- Standard For Food Additive Use - Beijing, ChinaDocument188 pagesStandard For Food Additive Use - Beijing, ChinaCM MacaliaNo ratings yet
- Effects of Organic and Mineral Fertilizer Nitrogen On Greenhouse Gas Emissions and Plant-Captured Carbon Under Maize Cropping in ZimbabweDocument15 pagesEffects of Organic and Mineral Fertilizer Nitrogen On Greenhouse Gas Emissions and Plant-Captured Carbon Under Maize Cropping in ZimbabweFitaTriWantikaNo ratings yet
- Catalogo Perlastan PDFDocument2 pagesCatalogo Perlastan PDFYuri Katerine Vargas ReyesNo ratings yet
- Hamza ProjectDocument40 pagesHamza ProjectDedan GideonNo ratings yet
- Aldol ReactionDocument4 pagesAldol ReactionhafizNo ratings yet
- Class 11 Chemistry Support MaterialDocument292 pagesClass 11 Chemistry Support MaterialMehar Fathima100% (2)
- Soap Based Chain Conveyor Lubricant - Basf Wyandotte CorporationDocument7 pagesSoap Based Chain Conveyor Lubricant - Basf Wyandotte CorporationShaara NeyNo ratings yet
- ControlDocument10 pagesControlAhmed FathiNo ratings yet
- Assay of Ascorbic Acid in Vitamin C SyrupDocument6 pagesAssay of Ascorbic Acid in Vitamin C SyrupOyemomi Samuel AdeyemiNo ratings yet
- Topic 7.3 Review Practice QuestionsDocument5 pagesTopic 7.3 Review Practice QuestionsGlen JojiNo ratings yet
- Sapporo JapanDocument2 pagesSapporo JapanBaitulNo ratings yet
- Pva CoatingDocument2 pagesPva Coatingmuhammad shoaibNo ratings yet
- Experiment 1 Solubility of Organic CompoundsDocument5 pagesExperiment 1 Solubility of Organic CompoundsKirstin Blaire MagdadaroNo ratings yet
- Introduction To Nylon FibreDocument3 pagesIntroduction To Nylon FibreDebasish GhoshNo ratings yet
- Organosilanes Brochure EEMDocument16 pagesOrganosilanes Brochure EEMSandipan SahaNo ratings yet
- Aspek Genetik Dalam ReproduksiDocument40 pagesAspek Genetik Dalam ReproduksiNaflah NadilaNo ratings yet
- Hydrochlorothiazide Monograph USPDocument2 pagesHydrochlorothiazide Monograph USPBhaven PatelNo ratings yet
- PCH Assignment 1 EditedDocument3 pagesPCH Assignment 1 EditedTadiwanashe ManyandeNo ratings yet
- Electrophilic SubstitutionDocument4 pagesElectrophilic SubstitutionPrasad BidweNo ratings yet
- Baha Vol67Document26 pagesBaha Vol67Agnes SulistyaNo ratings yet
- Generic NamesDocument3 pagesGeneric NameszyfranciscoNo ratings yet