Professional Documents
Culture Documents
Mechanism of The Wohl-Ziegler Reaction
Mechanism of The Wohl-Ziegler Reaction
Uploaded by
Abhishek Pandey0 ratings0% found this document useful (0 votes)
15 views1 pageThe Wohl-Ziegler reaction requires keeping the concentration of Br2 and HBr low to prevent unwanted side reactions from ionic addition to the alkene. These reagents are generated in situ from N-bromosuccinimide to control their concentration. Bromine radicals are the active species in this reaction and are formed by homolytic cleavage of Br2, which requires a radical initiator like UV light or AIBN.
Original Description:
Original Title
STF (3).docx
Copyright
© © All Rights Reserved
Available Formats
DOCX, PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentThe Wohl-Ziegler reaction requires keeping the concentration of Br2 and HBr low to prevent unwanted side reactions from ionic addition to the alkene. These reagents are generated in situ from N-bromosuccinimide to control their concentration. Bromine radicals are the active species in this reaction and are formed by homolytic cleavage of Br2, which requires a radical initiator like UV light or AIBN.
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
Download as docx, pdf, or txt
0 ratings0% found this document useful (0 votes)
15 views1 pageMechanism of The Wohl-Ziegler Reaction
Mechanism of The Wohl-Ziegler Reaction
Uploaded by
Abhishek PandeyThe Wohl-Ziegler reaction requires keeping the concentration of Br2 and HBr low to prevent unwanted side reactions from ionic addition to the alkene. These reagents are generated in situ from N-bromosuccinimide to control their concentration. Bromine radicals are the active species in this reaction and are formed by homolytic cleavage of Br2, which requires a radical initiator like UV light or AIBN.
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
Download as docx, pdf, or txt
You are on page 1of 1
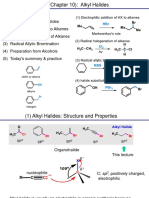
Mechanism of the Wohl-Ziegler Reaction
It is very important to keep the concentration of
Br2 and HBr low to prevent side reactions
derived from simple ionic addition with the
alkene. These reagents are therefore generated
in situ from NBS. The catalytically active
species is Br2, which is almost always present
in NBS samples (red colour).
A radical initiator (UV, AIBN) is needed for the
homolytic bond cleavage of Br2 :
You might also like
- Randox ..... BilirubinDocument4 pagesRandox ..... BilirubinJohn Senufa100% (1)
- CHEM F311 Lecture 4 Diborane Reduction, Birch ReductionDocument20 pagesCHEM F311 Lecture 4 Diborane Reduction, Birch ReductionSAYAN RAYNo ratings yet
- 10-Chapter 9-PreDocument73 pages10-Chapter 9-PreAnh Phương TrầnNo ratings yet
- Chapter 9Document57 pagesChapter 9Quỳnh Anh TrầnNo ratings yet
- Module 5 Reactions With Miscellaneous Reagents: 5.1 N-Bromosuccinimide (NBS)Document47 pagesModule 5 Reactions With Miscellaneous Reagents: 5.1 N-Bromosuccinimide (NBS)Dr Sachin K SinghNo ratings yet
- Class 12 Organic SupplementsDocument14 pagesClass 12 Organic Supplementslovishsindhi185No ratings yet
- Alkyl Halide PDFDocument29 pagesAlkyl Halide PDFSantosh Potdar100% (2)
- JeevanDocument26 pagesJeevanseelampadmaNo ratings yet
- Bilirubin Kit PDFDocument2 pagesBilirubin Kit PDFArasNo ratings yet
- Ankyl Halogenua Phan Ung The Nucleophil Va Tach LoaiDocument103 pagesAnkyl Halogenua Phan Ung The Nucleophil Va Tach LoaiQuang Cường HoàngNo ratings yet
- T&D Bilirubin DMSO MethodDocument1 pageT&D Bilirubin DMSO MethodS MNo ratings yet
- Unit 1. Chemoselectivity and Protecting GroupsDocument84 pagesUnit 1. Chemoselectivity and Protecting GroupsBenjamín BohiguesNo ratings yet
- Plij Gib Me BookDocument16 pagesPlij Gib Me BookDarsheel AmbasthaNo ratings yet
- Investigations of Riboflavin Photolysis Via Coloured Light in The Nitro Blue Tetrazolium Assay For Superoxide Dismutase ActivityDocument8 pagesInvestigations of Riboflavin Photolysis Via Coloured Light in The Nitro Blue Tetrazolium Assay For Superoxide Dismutase ActivitySonny RawlsNo ratings yet
- SubstitionDocument36 pagesSubstitionExcellent KhozaNo ratings yet
- Alkenes - Reactions and SynthesisDocument5 pagesAlkenes - Reactions and SynthesisJerica AlondraNo ratings yet
- SN1, SN2, E1 and E2 Reactions in OCDocument60 pagesSN1, SN2, E1 and E2 Reactions in OCHimanshu SharmaNo ratings yet
- LT-17 - Alkyl Halides - 20221108Document35 pagesLT-17 - Alkyl Halides - 20221108Justin HadinataNo ratings yet
- 11 Chemoselectivity (Part 1+2 Redox)Document35 pages11 Chemoselectivity (Part 1+2 Redox)barry allenNo ratings yet
- Mechanism 4Document10 pagesMechanism 4Pushpa ChoudharyNo ratings yet
- Bili R RubinaDocument2 pagesBili R RubinaJorge Tapia MartínezNo ratings yet
- Organic Reaction Mechanism Lecture 7Document21 pagesOrganic Reaction Mechanism Lecture 7Namrah ZakaNo ratings yet
- Reagents NBS SubstituionDocument10 pagesReagents NBS SubstituionPriyanshi JainNo ratings yet
- Chemical Properties 1819 Alkanes Alkyl HalideDocument44 pagesChemical Properties 1819 Alkanes Alkyl HalideAimee MangubatNo ratings yet
- Classification Test For Organic HalidesDocument4 pagesClassification Test For Organic HalidesChynna SyNo ratings yet
- 5) Preparation of Haloalkanes From Hydrocarbons, Alkenes and Halogen Exchange - RemovedDocument2 pages5) Preparation of Haloalkanes From Hydrocarbons, Alkenes and Halogen Exchange - RemovedMansa KaurNo ratings yet
- 03 Chapter 3 PostDocument77 pages03 Chapter 3 PostPhan ThongNo ratings yet
- Chemical Reactions: Lecture by Kalpajyoti Dihingia Department of Chemistry DHSK CollegeDocument21 pagesChemical Reactions: Lecture by Kalpajyoti Dihingia Department of Chemistry DHSK CollegeKalpa DihingiaNo ratings yet
- Usos de NiCl2 PDFDocument4 pagesUsos de NiCl2 PDFAdamarisGonzalezNo ratings yet
- Organic Nucleophilic Substitution Reactions sn1 and sn2 ReactioDocument13 pagesOrganic Nucleophilic Substitution Reactions sn1 and sn2 ReactioDebarjan DattaNo ratings yet
- Immunohistochemistr YDocument28 pagesImmunohistochemistr YNico ScarletNo ratings yet
- WK 2-2 Nucleophilic Substitution and Elimination Jan 18Document54 pagesWK 2-2 Nucleophilic Substitution and Elimination Jan 18cerelia.dkuNo ratings yet
- Substitution ReactionsDocument39 pagesSubstitution ReactionsAnil Kumar KadianNo ratings yet
- Classification of The Nucleophilic Substition Mechanism of Organic HalidesDocument3 pagesClassification of The Nucleophilic Substition Mechanism of Organic Halides3MshopNo ratings yet
- SN Reactions Lecture3Document21 pagesSN Reactions Lecture3Kalpa DihingiaNo ratings yet
- Reviewer Chap 6-14Document4 pagesReviewer Chap 6-14Jana PaduaNo ratings yet
- KOF-mekanisme ReaksiDocument64 pagesKOF-mekanisme ReaksiAudry YuniarNo ratings yet
- Chapter 22. Carbonyl Alpha-Substitution Reactions: Based On Mcmurry'S Organic Chemistry, 9 EditionDocument64 pagesChapter 22. Carbonyl Alpha-Substitution Reactions: Based On Mcmurry'S Organic Chemistry, 9 Edition張湧浩No ratings yet
- fs42-201704 Ultraviolet Reflectance by PaintDocument2 pagesfs42-201704 Ultraviolet Reflectance by PaintCarlos BustamanteNo ratings yet
- Technical Bulletin: Glycoprofile 2-Ab Labeling KitDocument4 pagesTechnical Bulletin: Glycoprofile 2-Ab Labeling KitJime nitaNo ratings yet
- Chapter 4 Substitution ReactionDocument43 pagesChapter 4 Substitution ReactionNahom AmanuelNo ratings yet
- Organic Reactions and Drug MoleculesDocument19 pagesOrganic Reactions and Drug Moleculesvalupalisandeep valupalisandeepNo ratings yet
- Previo 2Document3 pagesPrevio 2david liraNo ratings yet
- Revision - HaloalkanesDocument4 pagesRevision - HaloalkanessafaaNo ratings yet
- Oh HBR ! BRDocument3 pagesOh HBR ! BRAnita KNo ratings yet
- Lecture 8 Halide RXDocument84 pagesLecture 8 Halide RXquang trầnNo ratings yet
- Understanding The Elimination ReactionDocument33 pagesUnderstanding The Elimination ReactionnirmlaNo ratings yet
- 4.true False-Haloalknaes & HaloarenesDocument7 pages4.true False-Haloalknaes & HaloarenesrajeshwariNo ratings yet
- Alkenes GRDocument8 pagesAlkenes GRcrownhimanshu445No ratings yet
- Chapter 7Document37 pagesChapter 7민규강No ratings yet
- CHEM1920 Lecture 17v2Document29 pagesCHEM1920 Lecture 17v2Mr SpaciousNo ratings yet
- Clayden AryneDocument3 pagesClayden AryneDániel NemeskériNo ratings yet
- Org Chem Lec m8 AlkylhalidesDocument5 pagesOrg Chem Lec m8 AlkylhalidesAbigail P. ARANGGANo ratings yet
- Amide BondFormation Via One-Pot Nitrone in SituFormationDocument8 pagesAmide BondFormation Via One-Pot Nitrone in SituFormationvarnish-gearbox-0fNo ratings yet
- 2020 Haloalkanes and Haloarenes Krishna SirDocument27 pages2020 Haloalkanes and Haloarenes Krishna SirShreyaNo ratings yet
- Chapter 5 Elimination ReactionDocument16 pagesChapter 5 Elimination Reactionahmedmustefa773No ratings yet
- Baek 1985Document4 pagesBaek 1985FELIPE DANIEL MONTERO BRUNINo ratings yet
- Oxidative Photochlorination of Electron Rich Arenes Via in Situ BrominationDocument5 pagesOxidative Photochlorination of Electron Rich Arenes Via in Situ BrominationABCNo ratings yet
- Chapter 4 Major Organic ReactionDocument55 pagesChapter 4 Major Organic ReactionTolera TadesseNo ratings yet
- Controlled Reduction of Tertiary Amides To The Corresponding Alcohols, Aldehydes, or Amines Using Dialkylboranes and Aminoborohydride ReagentsDocument10 pagesControlled Reduction of Tertiary Amides To The Corresponding Alcohols, Aldehydes, or Amines Using Dialkylboranes and Aminoborohydride ReagentsSaurav PaulNo ratings yet
- Computer Fundamentals: Q.1 Write A Short Note On ROMDocument3 pagesComputer Fundamentals: Q.1 Write A Short Note On ROMAbhishek PandeyNo ratings yet
- Hysteroscope Quotation 2Document2 pagesHysteroscope Quotation 2Abhishek PandeyNo ratings yet
- Computer Fundamentals: Q.1 Write A Short Note On ROMDocument3 pagesComputer Fundamentals: Q.1 Write A Short Note On ROMAbhishek PandeyNo ratings yet
- Mechanism of The Wohl-Ziegler ReactionDocument1 pageMechanism of The Wohl-Ziegler ReactionAbhishek PandeyNo ratings yet
- Product Digital Electronic Colposcope Make Kernel Model KN2200 Technical FeaturesDocument1 pageProduct Digital Electronic Colposcope Make Kernel Model KN2200 Technical FeaturesAbhishek PandeyNo ratings yet
- Business CommunicationDocument3 pagesBusiness CommunicationAbhishek PandeyNo ratings yet
- Lap Set'Document1 pageLap Set'Abhishek PandeyNo ratings yet
- Ational Bureau of StandardsDocument1 pageAtional Bureau of StandardsAbhishek PandeyNo ratings yet
- N Led Light Source - 120 Watt: 3) Insuffalator Vega 30 Liter/MinuteDocument1 pageN Led Light Source - 120 Watt: 3) Insuffalator Vega 30 Liter/MinuteAbhishek PandeyNo ratings yet
- Bromosuccinimide (NBS)Document1 pageBromosuccinimide (NBS)Abhishek PandeyNo ratings yet
- Video Bronchoscope BFP-240: RGB OutpuDocument1 pageVideo Bronchoscope BFP-240: RGB OutpuAbhishek PandeyNo ratings yet
- Lap Set'Document1 pageLap Set'Abhishek PandeyNo ratings yet
- Arthroscopic Shaver System Standard Configuration: Control Unit: Hand Piece With Connecting Cable 1unit of Dyonics/Smith and Nephew BladesDocument1 pageArthroscopic Shaver System Standard Configuration: Control Unit: Hand Piece With Connecting Cable 1unit of Dyonics/Smith and Nephew BladesAbhishek PandeyNo ratings yet
- Tantra SiddhiDocument1 pageTantra SiddhiAbhishek PandeyNo ratings yet
- Tantra SarDocument1 pageTantra SarAbhishek PandeyNo ratings yet
- Tantra SiddhiDocument1 pageTantra SiddhiAbhishek PandeyNo ratings yet
- Tantra SarDocument1 pageTantra SarAbhishek PandeyNo ratings yet