Professional Documents
Culture Documents
B. Tabel: V PH PH/ V
B. Tabel: V PH PH/ V
Uploaded by
Arinda Utami0 ratings0% found this document useful (0 votes)
6 views2 pagesThe document summarizes the results of two titration experiments. The first experiment titrates acetic acid (CH3COOH) with sodium hydroxide (NaOH) and records the changes in volume (v), pH, and pH changes per volume change at each titration point. The second experiment titrates phosphoric acid (H3PO4) with NaOH and similarly records the volume, pH, and pH changes per volume change at each titration point. Both experiments provide data that can be used to analyze the acid-base reactions and determine the end points of the titrations.
Original Description:
titrasi
Original Title
Tabel Perc.1
Copyright
© © All Rights Reserved
Available Formats
DOCX, PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentThe document summarizes the results of two titration experiments. The first experiment titrates acetic acid (CH3COOH) with sodium hydroxide (NaOH) and records the changes in volume (v), pH, and pH changes per volume change at each titration point. The second experiment titrates phosphoric acid (H3PO4) with NaOH and similarly records the volume, pH, and pH changes per volume change at each titration point. Both experiments provide data that can be used to analyze the acid-base reactions and determine the end points of the titrations.
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
Download as docx, pdf, or txt
0 ratings0% found this document useful (0 votes)
6 views2 pagesB. Tabel: V PH PH/ V
B. Tabel: V PH PH/ V
Uploaded by
Arinda UtamiThe document summarizes the results of two titration experiments. The first experiment titrates acetic acid (CH3COOH) with sodium hydroxide (NaOH) and records the changes in volume (v), pH, and pH changes per volume change at each titration point. The second experiment titrates phosphoric acid (H3PO4) with NaOH and similarly records the volume, pH, and pH changes per volume change at each titration point. Both experiments provide data that can be used to analyze the acid-base reactions and determine the end points of the titrations.
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
Download as docx, pdf, or txt
You are on page 1of 2
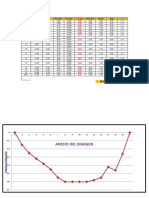
B.
Tabel
1. Titrasi antara CH3COOH dan NaOH
v pH ∆v ∆pH ∆pH/∆v ∆ 2v ∆ 2pH ∆ 2pH/∆ 2
v
0 3,4 0 0 0 0 0 0
1 4,1 1 0,7 0,7 1 0,49 0,49
2 4,3 1 0,2 0,2 1 0,04 0,04
3 4,5 1 0,2 0,2 1 0,04 0,04
4 4,8 1 0,3 0,3 1 0,09 0,09
5 5,0 1 0,2 0,2 1 0,04 0,04
6 5,2 1 0,2 0,2 1 0,04 0,04
7 5,5 1 0,3 0,3 1 0,09 0,09
8 6,0 1 0,5 0,5 1 0,25 0,25
9 11,1 1 5,1 5,1 1 26,01 26,01
10 11,5 1 0,4 0,4 1 0,16 0,16
11 11,6 1 0,1 0,1 1 0,01 0,01
12 11,8 1 0,2 0,2 1 0,04 0,04
13 11,8 1 0 1 1 0 0
14 11,9 1 0,1 0 1 0,01 0,01
15 11,9 1 0 0 1 0 0
2. Titrasi antara H3PO4 dan NaOH
v pH ∆v ∆pH ∆pH/∆v ∆ 2v ∆ 2pH ∆ 2pH/∆ 2
v
0 2,1 0 0 0 0 0 0
2 1,5 2 -0,6 -0,3 4 0,36 0,18
4 1,9 2 0,4 0,2 4 0,16 0,04
6 2,0 2 0,1 0,05 4 0,01 0,0025
8 2,8 2 0,8 0,4 4 0,64 0,16
10 3,5 2 0,7 0,35 4 0,49 0,1225
12 6,5 2 3,0 1,5 4 9 2,25
14 6,9 2 0,4 0,2 4 0,16 0,04
16 6,6 2 -0,3 -0,15 4 0,09 0,0225
18 11 2 4,4 2,2 4 19,36 4,84
20 12,5 2 1,5 0,75 4 2,25 0,5625
22 8,4 2 -4,1 -2,05 4 16,81 4,2025
24 12,7 2 4,3 2,15 4 18,49 4,6225
26 10,8 2 -1,9 -0,95 4 3,61 0,9025
28 12,9 2 2,1 1,05 4 4,41 1,1025
30 11,2 2 -1,7 -0,85 4 2,89 0,7225
You might also like
- Relevamiento Slew Drive B05-S01Document12 pagesRelevamiento Slew Drive B05-S01yamileNo ratings yet
- Manual Técnico: Tabela de Perda de Carga em TubulaçõesDocument1 pageManual Técnico: Tabela de Perda de Carga em TubulaçõesLafaete JuniorNo ratings yet
- Tabela Perda de Carga Tubos FoFo e Aço GalvanizadoDocument6 pagesTabela Perda de Carga Tubos FoFo e Aço GalvanizadoWilliamNo ratings yet
- Relatório Sev Guara 2Document1,646 pagesRelatório Sev Guara 2Lucas Vieira LimaNo ratings yet
- Liebherr LTM1130 CW9.8tDocument57 pagesLiebherr LTM1130 CW9.8tguruhNo ratings yet
- Answer For GOM Assignment IndividualDocument3 pagesAnswer For GOM Assignment IndividualPerta MinaNo ratings yet
- Tabel D.1 Data Seluruh Arus Jenuh Untuk 56 Siklus Hijau Lajur GabunganDocument14 pagesTabel D.1 Data Seluruh Arus Jenuh Untuk 56 Siklus Hijau Lajur GabunganKrisna DeknaNo ratings yet
- Analitica P2Document6 pagesAnalitica P2juan arellano olivaresNo ratings yet
- Triangulos Cuenca 1 PDFDocument1 pageTriangulos Cuenca 1 PDFandrEEs moREENONo ratings yet
- Curva de Titulación: V Naoh PH Primera Derivada DPH/DV Segunda DerivadaDocument3 pagesCurva de Titulación: V Naoh PH Primera Derivada DPH/DV Segunda DerivadaJorgeFlolopNo ratings yet
- Relatório SEV BarretosDocument1,647 pagesRelatório SEV BarretosLucas Vieira LimaNo ratings yet
- Industrial Static Mixers Static Mixers: Static Mixers From Stock Static Mixers From StockDocument8 pagesIndustrial Static Mixers Static Mixers: Static Mixers From Stock Static Mixers From StockElleuch HaithemNo ratings yet
- Calculo IO MAT01212023201 P1Document1 pageCalculo IO MAT01212023201 P1Priscila FerminoNo ratings yet
- Ejercicio 3 de La Clase 3Document2 pagesEjercicio 3 de La Clase 3Andrés RestrepoNo ratings yet
- 23-24 Phys-S1001 Cfs RésultatsDocument9 pages23-24 Phys-S1001 Cfs Résultatscotam58578No ratings yet
- ANESTESIADocument2 pagesANESTESIAMarco MurilloNo ratings yet
- Horario Doméstica Hospital Colegios Jardines Deportivo Hostelería-Bares - Restaurante S Industrial - Comercial - OficinasDocument2 pagesHorario Doméstica Hospital Colegios Jardines Deportivo Hostelería-Bares - Restaurante S Industrial - Comercial - OficinasLaura CarreñoNo ratings yet
- PatronesDocument2 pagesPatronesLaura CarreñoNo ratings yet
- Lampiran Hambatan AlatDocument8 pagesLampiran Hambatan AlatSyahrial ApriandyNo ratings yet
- Chi CuadradoDocument3 pagesChi CuadradoNailetiNo ratings yet
- MST3Document1 pageMST3Aimi KagawaNo ratings yet
- NOTAS GENERAL ECOAGROPECUARIA 2022 B Ene 2023Document1 pageNOTAS GENERAL ECOAGROPECUARIA 2022 B Ene 2023ISABELLA ACOSTA RUIZNo ratings yet
- K20 KR21 HesaplamaDocument2 pagesK20 KR21 Hesaplamabuse1111uNo ratings yet
- 0° 5° K KP V: ProgramsDocument1 page0° 5° K KP V: ProgramsFernando FanteNo ratings yet
- Tabela Pressão X Temperatura - CompletaDocument4 pagesTabela Pressão X Temperatura - CompletaCarlos TripodiNo ratings yet
- Common Refrigerants Poster - tcm310-131260 PDFDocument4 pagesCommon Refrigerants Poster - tcm310-131260 PDFr.miguelNo ratings yet
- Common Refrigerants Poster - tcm316-131260Document4 pagesCommon Refrigerants Poster - tcm316-131260Marcelo GereniereNo ratings yet
- UGM UI USM Undip IPB UB Unair UNYDocument11 pagesUGM UI USM Undip IPB UB Unair UNYriza kurniawanNo ratings yet
- Common Refrigerants Poster - tcm17-131260Document4 pagesCommon Refrigerants Poster - tcm17-131260aliakbar tavafiNo ratings yet
- NOTAS Opcional 1 PDFDocument1 pageNOTAS Opcional 1 PDFKaren Gisela RealpeNo ratings yet
- Potenciometria HalurosDocument7 pagesPotenciometria HalurosDayan OcampoNo ratings yet
- Vertical Progresiva Pro. Obs. Vel. Pun. - Gas. Unit. Ancho ÁreaDocument4 pagesVertical Progresiva Pro. Obs. Vel. Pun. - Gas. Unit. Ancho ÁreaKatherina CuelloNo ratings yet
- Blind Rivets: d1 d2 Clamp. Range L e Grade SUDocument1 pageBlind Rivets: d1 d2 Clamp. Range L e Grade SUVishnuNo ratings yet
- Juan Diego RoaDocument4 pagesJuan Diego RoaMiguel AngelNo ratings yet
- Exercício Avaliativo - Ponto Final de Titulação - João Pedro Gass Mendes PasqualettoDocument5 pagesExercício Avaliativo - Ponto Final de Titulação - João Pedro Gass Mendes Pasqualettojoão pedro gassNo ratings yet
- Observatorio Del Delito 2003-2015Document252 pagesObservatorio Del Delito 2003-2015Jorge SalazarNo ratings yet
- Ib GR44Document1 pageIb GR44abranchelo08No ratings yet
- Gebbrilla Anjeni Sadewa 002Document5 pagesGebbrilla Anjeni Sadewa 002GabbyNo ratings yet
- Tabelul 7.2-Tabel Pentru Determinarea Suprafetei de Baza Multipla-14-20Document1 pageTabelul 7.2-Tabel Pentru Determinarea Suprafetei de Baza Multipla-14-20Adrian NegoitaNo ratings yet
- Armonico 1Document6 pagesArmonico 1Oscar MartínezNo ratings yet
- 3 PDFsam PDFsam Duct FittingsDocument1 page3 PDFsam PDFsam Duct FittingsJosephRusselVizmanosNo ratings yet
- Anillo Seeger CompletoDocument25 pagesAnillo Seeger CompletoAngelNo ratings yet
- BTH Ferm AlcoolicaDocument2 pagesBTH Ferm AlcoolicaAlexandra Ramona MareşNo ratings yet
- Diagrama de Cortante (Q-KN) Diagrama de Momento (M-KNM) Diagrama de Normal (N-KN)Document1 pageDiagrama de Cortante (Q-KN) Diagrama de Momento (M-KNM) Diagrama de Normal (N-KN)lucasNo ratings yet
- Tablas y GraficasDocument8 pagesTablas y GraficasAngel Jair Pulido SantiagoNo ratings yet
- Bab IvDocument6 pagesBab IvAnonymous AdZe7vqQNo ratings yet
- Notas PDFDocument2 pagesNotas PDFWilmer CórdobaNo ratings yet
- Circlips ExternDocument5 pagesCirclips ExternserkanNo ratings yet
- Tabelul 7.2-Tabel Pentru Determinarea Suprafetei de Baza Multipla-6-12Document1 pageTabelul 7.2-Tabel Pentru Determinarea Suprafetei de Baza Multipla-6-12Adrian NegoitaNo ratings yet
- Angle PropertiesDocument9 pagesAngle PropertiesMdShahbazAhmedNo ratings yet
- Nominal Dimensions Section Properties, Static DataDocument9 pagesNominal Dimensions Section Properties, Static DataMdShahbazAhmedNo ratings yet
- Khách hàng Số tiền chi Đăng ký thành viênDocument6 pagesKhách hàng Số tiền chi Đăng ký thành viênAnh NguyenNo ratings yet
- Dose Chart For Xyl Bupr Ket Premed and Diaz Propofol InductionDocument2 pagesDose Chart For Xyl Bupr Ket Premed and Diaz Propofol InductionUNICORN TIMENo ratings yet
- Abad - Trisha PS5Document12 pagesAbad - Trisha PS5Claire TaborNo ratings yet
- Radial Clearance of BearingDocument1 pageRadial Clearance of Bearingstenly massieNo ratings yet
- Lich Thuy Trieu Cua Ong FinalDocument12 pagesLich Thuy Trieu Cua Ong FinalminhnghienNo ratings yet
- GRAFICOSDocument4 pagesGRAFICOSANDERSON STEVEN PINEDA MORALESNo ratings yet
- Tutorial Data Mining Menggunakan Metode Fuzzy C-MeansDocument3 pagesTutorial Data Mining Menggunakan Metode Fuzzy C-Meansarie_sppNo ratings yet
- Input OutputDocument1,209 pagesInput Outputgamidi67No ratings yet
- Math Practice Simplified: Decimals & Percents (Book H): Practicing the Concepts of Decimals and PercentagesFrom EverandMath Practice Simplified: Decimals & Percents (Book H): Practicing the Concepts of Decimals and PercentagesRating: 5 out of 5 stars5/5 (3)