Professional Documents
Culture Documents
Management of d-PTFE Membrane Exposure For Having Final Clinical Success
Management of d-PTFE Membrane Exposure For Having Final Clinical Success
Uploaded by
Nidia TorreaCopyright:
Available Formats
You might also like
- Population and Community Health Nursing 6th Edition Clark Test BankDocument10 pagesPopulation and Community Health Nursing 6th Edition Clark Test Bankhorsecarbolic1on100% (27)
- ISMP198 Periop Guidelns FINALDocument29 pagesISMP198 Periop Guidelns FINALHIRANGERNo ratings yet
- Retentive Aids in Maxillofacial ProsthesisDocument109 pagesRetentive Aids in Maxillofacial ProsthesisEnu KambojNo ratings yet
- Aussie Emergency CareDocument9 pagesAussie Emergency CarePuspita Sari FiazNo ratings yet
- Pod Uveitis Book RevDocument252 pagesPod Uveitis Book Revelika dwi100% (1)
- Extraction Site Reconstruction For Alveolar Ridge Preservation. Part 2: Membrane-Assisted Surgical TechniqueDocument4 pagesExtraction Site Reconstruction For Alveolar Ridge Preservation. Part 2: Membrane-Assisted Surgical TechniqueLuis MonteroNo ratings yet
- Membranes For Periodontal RegenerationDocument6 pagesMembranes For Periodontal RegenerationinventionjournalsNo ratings yet
- Captura de Tela 2022-09-11 À(s) 19.01.07Document7 pagesCaptura de Tela 2022-09-11 À(s) 19.01.07alefabri-1No ratings yet
- Fugazzotto ImmediateImplantPlacementMolarDocument7 pagesFugazzotto ImmediateImplantPlacementMolarLozovan GregoryNo ratings yet
- Tissue Preservation and Maintenance of Optimum Esthetics: A Clinical ReportDocument7 pagesTissue Preservation and Maintenance of Optimum Esthetics: A Clinical ReportBagis Emre GulNo ratings yet
- Koutouzisetal 2013IJOMIAbutmentdisconnectionClinicalDocument8 pagesKoutouzisetal 2013IJOMIAbutmentdisconnectionClinicalKyoko CPNo ratings yet
- SwithcingDocument6 pagesSwithcingKyoko CPNo ratings yet
- Implant-Retained Auricular Prosthesis: A Case Report: January 2010 - Vol.4Document4 pagesImplant-Retained Auricular Prosthesis: A Case Report: January 2010 - Vol.4noranNo ratings yet
- Removal Of..Document4 pagesRemoval Of..Andrea ValverdeNo ratings yet
- Ejed 15 3 Staehler p288Document18 pagesEjed 15 3 Staehler p288snkidNo ratings yet
- Distalization of Impacted Mandibular SecondDocument6 pagesDistalization of Impacted Mandibular SecondPatricia BurbanoNo ratings yet
- Grizas 2018Document16 pagesGrizas 2018Dome CárdenasNo ratings yet
- Implant-Supported Nasal Prosthesis. Clinical Case Report: Prótesis Nasal Implantosoportada. Reporte de Un Caso ClínicoDocument6 pagesImplant-Supported Nasal Prosthesis. Clinical Case Report: Prótesis Nasal Implantosoportada. Reporte de Un Caso ClínicoMelina Nataly Robles AcuñaNo ratings yet
- Immediate One-Stage Postextraction Implant: A Human Clinical and Histologic Case ReportDocument6 pagesImmediate One-Stage Postextraction Implant: A Human Clinical and Histologic Case ReportBagis Emre GulNo ratings yet
- Post-Prosthetic Surgery: Using Complete Denture Prosthesis For Propriotous Outcome During VestibuloplastyDocument3 pagesPost-Prosthetic Surgery: Using Complete Denture Prosthesis For Propriotous Outcome During VestibuloplastyInternational Journal of Innovative Science and Research TechnologyNo ratings yet
- JOHCD-Maintenance of Implant SupportedDocument3 pagesJOHCD-Maintenance of Implant SupportedManoj HumagainNo ratings yet
- Article 009Document9 pagesArticle 009MedstudNo ratings yet
- Jpis 42 105Document5 pagesJpis 42 105Jean Pierre Alcántara VigoNo ratings yet
- Case SeriesDocument5 pagesCase SeriesPavithra balasubramaniNo ratings yet
- Franceschi2018 PDFDocument12 pagesFranceschi2018 PDFMichal PerkowskiNo ratings yet
- Speroni Desplazamiento Paladar2010Document7 pagesSperoni Desplazamiento Paladar2010SergioNo ratings yet
- ISSN 2347-5579: Use of Porous Polyethylene Implants For Orbital Reconstruction-A ReviewDocument3 pagesISSN 2347-5579: Use of Porous Polyethylene Implants For Orbital Reconstruction-A ReviewVISHAKHANo ratings yet
- Adj 12563Document5 pagesAdj 12563anushiNo ratings yet
- Cyst DecompressionDocument7 pagesCyst DecompressionMugdha FrancisNo ratings yet
- SCD 12407Document5 pagesSCD 12407RochmahAiNurNo ratings yet
- Khoury Bone Augmentation Tunneling-TechniqueDocument4 pagesKhoury Bone Augmentation Tunneling-TechniqueSeptimiu TiplicaNo ratings yet
- Document PDFDocument4 pagesDocument PDFBagis Emre GulNo ratings yet
- Ic Rehabilitation of The Edentulous Patient Maxillectomy UiriDocument5 pagesIc Rehabilitation of The Edentulous Patient Maxillectomy Uiriayush srivastavaNo ratings yet
- Expansion of The Zone of Keratinised Tissue For Healthy Implant Abutment Interface Using Deepithelized Amnion:Chorion AllograftDocument6 pagesExpansion of The Zone of Keratinised Tissue For Healthy Implant Abutment Interface Using Deepithelized Amnion:Chorion AllograftzethyhanumNo ratings yet
- Articol PMFDocument6 pagesArticol PMFclaudia lupuNo ratings yet
- The Use of PRF in Covering Exposed ImplantDocument6 pagesThe Use of PRF in Covering Exposed ImplantMohammad AkheelNo ratings yet
- Immediate, Non-Submerged, Root-Analogue Zirconia Implant in Single Tooth ReplacementDocument3 pagesImmediate, Non-Submerged, Root-Analogue Zirconia Implant in Single Tooth ReplacementNishtha KumarNo ratings yet
- Reusing Dental Implants?: An Experimental Study For Detecting The Success Rates of Re-OsseointegrationDocument7 pagesReusing Dental Implants?: An Experimental Study For Detecting The Success Rates of Re-OsseointegrationSatya AsatyaNo ratings yet
- 10 1016@j Prosdent 2005 10 014Document5 pages10 1016@j Prosdent 2005 10 014everaldocruzNo ratings yet
- Total Maxillectomy With Prosthetic Reconstruction Technique: Case ReportDocument3 pagesTotal Maxillectomy With Prosthetic Reconstruction Technique: Case ReportArun PrasadNo ratings yet
- Multidisciplinary Treatment Approach To A Complicated Crown Fracture: A Case ReportDocument5 pagesMultidisciplinary Treatment Approach To A Complicated Crown Fracture: A Case Reportramadan ariesthaNo ratings yet
- 2022 AiutoDocument7 pages2022 AiutovivianaNo ratings yet
- Aumento de Reborde Con Tejido BlandoDocument8 pagesAumento de Reborde Con Tejido BlandoSilvia SolisNo ratings yet
- An Innovative Technique To Restore The Non Vital Teeth With Wide Open Canals - A Clinical StudyDocument4 pagesAn Innovative Technique To Restore The Non Vital Teeth With Wide Open Canals - A Clinical StudyIOSRjournalNo ratings yet
- Surgical and Endodontic Management of Large Cystic Lesion: AbstractDocument5 pagesSurgical and Endodontic Management of Large Cystic Lesion: AbstractanamaghfirohNo ratings yet
- Implant ResearchDocument8 pagesImplant ResearchFBI LUC1FeRNo ratings yet
- Amato 1 (2018), Immediate Implant PlacementDocument8 pagesAmato 1 (2018), Immediate Implant PlacementPhương Thảo TrầnNo ratings yet
- Immediate Dentoalveolar Restoration in Compromised Sockets: Technique and Bone BiologyDocument14 pagesImmediate Dentoalveolar Restoration in Compromised Sockets: Technique and Bone BiologyMichal PerkowskiNo ratings yet
- Madohc MS Id 000103Document6 pagesMadohc MS Id 000103Manjulika TysgiNo ratings yet
- Therapeutic Management For Immediate Implant Placement in Sites With Periapical Deficiencies Where Coronal Bone Is Present: Technique and Case ReportDocument6 pagesTherapeutic Management For Immediate Implant Placement in Sites With Periapical Deficiencies Where Coronal Bone Is Present: Technique and Case ReportRaul Alfonsin SanchezNo ratings yet
- Case Report: The Prosthetic Management of A Case of Epulis Fissuratum by A Novel TechniqueDocument6 pagesCase Report: The Prosthetic Management of A Case of Epulis Fissuratum by A Novel TechniqueMichelle VelezNo ratings yet
- Implementingsocketsealsurgery JP2008Document11 pagesImplementingsocketsealsurgery JP2008Gabriela ArgeseanuNo ratings yet
- Article 1434132803Document5 pagesArticle 1434132803antariksha DodNo ratings yet
- Yu 그리고 Qiu - 2018 - Efficacy of the split-thickness labial flap methodDocument6 pagesYu 그리고 Qiu - 2018 - Efficacy of the split-thickness labial flap methodsupercool0120No ratings yet
- Tissue ManagementDocument16 pagesTissue ManagementSaleh AlsadiNo ratings yet
- Simion 2012)Document13 pagesSimion 2012)SergioNo ratings yet
- Sticky Bone For Space Maintenance in The Treatment of Miller's Class I Gingival Recession Defects With VISTA Technique A Case SeriesDocument4 pagesSticky Bone For Space Maintenance in The Treatment of Miller's Class I Gingival Recession Defects With VISTA Technique A Case SeriesInternational Journal of Innovative Science and Research TechnologyNo ratings yet
- Oh 2010Document5 pagesOh 2010gbaez.88No ratings yet
- Protesis Fija Con Ataches. Rep Caso. 2020. Cureus. SivakumarDocument13 pagesProtesis Fija Con Ataches. Rep Caso. 2020. Cureus. SivakumarAngela AcostaNo ratings yet
- EDI Journal - A Graftless Solution For Adjacent Implants in The Aesthetic Zone - Dr. Tunkiwala Et AlDocument6 pagesEDI Journal - A Graftless Solution For Adjacent Implants in The Aesthetic Zone - Dr. Tunkiwala Et AlNaina TalwarNo ratings yet
- Complications of Using Retraction CordDocument4 pagesComplications of Using Retraction CordAya Ibrahim YassinNo ratings yet
- A New Porcine-Derived Collagen Matrix To Thicken Peri-Implant Soft Tissues in High Aestethic Value AreasDocument1 pageA New Porcine-Derived Collagen Matrix To Thicken Peri-Implant Soft Tissues in High Aestethic Value AreasEdoardo BiancoNo ratings yet
- 2015 - A New Technique For Increasing Keratinized Tisue Around Dental Implants. The Partialy Epithelialized Free Conective Tisue Graft. Retrospective Analysis of A Case Series - Eberhard FrischDocument7 pages2015 - A New Technique For Increasing Keratinized Tisue Around Dental Implants. The Partialy Epithelialized Free Conective Tisue Graft. Retrospective Analysis of A Case Series - Eberhard FrischJose LuisNo ratings yet
- Short ImplantsFrom EverandShort ImplantsBoyd J. TomasettiNo ratings yet
- Biology TMJ 2013Document8 pagesBiology TMJ 2013Nidia TorreaNo ratings yet
- The Speaking Method in Measuring Vertical DimensionDocument5 pagesThe Speaking Method in Measuring Vertical DimensionNidia TorreaNo ratings yet
- Antibodies To Watch in 2019: Hélène Kaplon & Janice M. ReichertDocument21 pagesAntibodies To Watch in 2019: Hélène Kaplon & Janice M. ReichertNidia TorreaNo ratings yet
- Journal of Dentistry: Review ArticleDocument13 pagesJournal of Dentistry: Review ArticleNidia TorreaNo ratings yet
- Intraoperative and Postoperative Complications, Accidents and Failures Related With Dental Implant ApplicationsDocument6 pagesIntraoperative and Postoperative Complications, Accidents and Failures Related With Dental Implant ApplicationsNidia TorreaNo ratings yet
- Research FinalDocument67 pagesResearch FinalhelshynieNo ratings yet
- Research 11Document22 pagesResearch 11Jenn TajanlangitNo ratings yet
- Pcos 170123144542Document86 pagesPcos 170123144542Dewi Puspita Apsari100% (2)
- Evaluation of Er:YAG and Er, Cr:YSGG Laser Irradiation For The Debonding of Prefabricated Zirconia CrownsDocument9 pagesEvaluation of Er:YAG and Er, Cr:YSGG Laser Irradiation For The Debonding of Prefabricated Zirconia Crownsdr.m3.abazaNo ratings yet
- Pma Memo No 2022 10 24 020Document9 pagesPma Memo No 2022 10 24 020Emma AustriaNo ratings yet
- Funda NotesDocument9 pagesFunda Noteszyrenejoy.motita.mnlNo ratings yet
- France and Saudi ArabiaDocument17 pagesFrance and Saudi ArabiaHajar HossamNo ratings yet
- Practical Research 1 Worksheet 3-1Document2 pagesPractical Research 1 Worksheet 3-1Brylle Christian C. LopeNo ratings yet
- Chicago London: Your ItineraryDocument3 pagesChicago London: Your ItineraryZubaidah ZamNo ratings yet
- Reproductive Health AssignmentDocument3 pagesReproductive Health AssignmentRajat Kumar SabharwalNo ratings yet
- Blood and Marrow Transplant Quality Management PlanDocument47 pagesBlood and Marrow Transplant Quality Management PlansumathiNo ratings yet
- Carbohydrate-Related Diseases Term PaperDocument5 pagesCarbohydrate-Related Diseases Term Paperheiress comiaNo ratings yet
- 10 COMPLETE PRACTICE TESTS (1) 30 CopiesDocument129 pages10 COMPLETE PRACTICE TESTS (1) 30 Copiesghulam abbasNo ratings yet
- Bachelor of Dental Surgery (BDS)Document2 pagesBachelor of Dental Surgery (BDS)Rohit GhuseNo ratings yet
- FARID SURA WIJAYA, Dr. H. M. Untung Tranggono, MS, PA (K), SP.B, Sp.UDocument2 pagesFARID SURA WIJAYA, Dr. H. M. Untung Tranggono, MS, PA (K), SP.B, Sp.UM. Sabirin AsikinNo ratings yet
- Quality Nutrition and Dietetics PracticeDocument3 pagesQuality Nutrition and Dietetics PracticeNurlienda HasanahNo ratings yet
- Existential Psychology - Review QuestionnaireDocument7 pagesExistential Psychology - Review Questionnairerop serrNo ratings yet
- Ps1 - CFD Analysis Cold ChainDocument9 pagesPs1 - CFD Analysis Cold ChainMark SantosNo ratings yet
- Read Online Textbook Behavior Modification What It Is and How To Do It 10Th Edition Garry Martin Ebook All Chapter PDFDocument22 pagesRead Online Textbook Behavior Modification What It Is and How To Do It 10Th Edition Garry Martin Ebook All Chapter PDFhilda.rufe918100% (2)
- PDF]Jan-Febc 2010 - Jaypee Brothers Medical Publishers jaypeebrothers.com/pdf/cataloginmonths/jan-feb2010.pdf by S Bhat - 2010 A unique book on clinical methods in surgery along with interactive DVD-ROM ... and clinical approach to each and individual surgical cases .... including a brief outline on prognosis. • Provides in-depth ..... Susmita Bhattacharya. Epidemiology. mbbs books - Scribd https://www.scribd.com/doc/239765747/mbbs-books Sep 15, 2014 - mbbs books - Download as Word Doc (.doc / .docx), PDF File (.pdf), Text ... BHATTACHARYA SHORT CASES IN SURGERY 5TH 2002 200.00 BOOKS1 - Scribd https://www.scribd.com/doc/99385577/BOOKS1 Jul 7, 2012 - BOOKS1 - Ebook download as PDF File (.pdf), Text file (.txt) or read book ... LAWRENCE W. SHORT CASES IN SURGERY / BHATTACHARYA, ... Medical Books - mbbs exam questions for second third and ... hafeesh.blogspot.com/2009/12/medical-books.html Dec 25, 2009 - GUPTE SHORT TEXTBOOK OF MEDICAL MICROBIOLOGY 8TH 2002 .... BHATTACHARYA SHORT CASESDocument3 pagesPDF]Jan-Febc 2010 - Jaypee Brothers Medical Publishers jaypeebrothers.com/pdf/cataloginmonths/jan-feb2010.pdf by S Bhat - 2010 A unique book on clinical methods in surgery along with interactive DVD-ROM ... and clinical approach to each and individual surgical cases .... including a brief outline on prognosis. • Provides in-depth ..... Susmita Bhattacharya. Epidemiology. mbbs books - Scribd https://www.scribd.com/doc/239765747/mbbs-books Sep 15, 2014 - mbbs books - Download as Word Doc (.doc / .docx), PDF File (.pdf), Text ... BHATTACHARYA SHORT CASES IN SURGERY 5TH 2002 200.00 BOOKS1 - Scribd https://www.scribd.com/doc/99385577/BOOKS1 Jul 7, 2012 - BOOKS1 - Ebook download as PDF File (.pdf), Text file (.txt) or read book ... LAWRENCE W. SHORT CASES IN SURGERY / BHATTACHARYA, ... Medical Books - mbbs exam questions for second third and ... hafeesh.blogspot.com/2009/12/medical-books.html Dec 25, 2009 - GUPTE SHORT TEXTBOOK OF MEDICAL MICROBIOLOGY 8TH 2002 .... BHATTACHARYA SHORT CASESSaiSuryaTeja0% (1)
- A Reading of Contemporary Hysteria From LacanDocument9 pagesA Reading of Contemporary Hysteria From LacanMarcelo AraújoNo ratings yet
- Our Materials: Ethical Shoe PromiseDocument5 pagesOur Materials: Ethical Shoe PromiseSharoon S RaoNo ratings yet
- Sample Family Case Study 1 1 - CompressDocument55 pagesSample Family Case Study 1 1 - CompressGladys JhayeNo ratings yet
- Saqa - 14909 - Nhlapo Bellina JV17099Document16 pagesSaqa - 14909 - Nhlapo Bellina JV17099sibonisoNo ratings yet
- 2018-089. NASKAH PUBLIKASI Elsi Nursyahdin MayaranaDocument12 pages2018-089. NASKAH PUBLIKASI Elsi Nursyahdin MayaranaElsi NursyahdinNo ratings yet
- Chain Exercise Terhadap Penurunan Intensitas Nyeri Pada LansiaDocument9 pagesChain Exercise Terhadap Penurunan Intensitas Nyeri Pada LansiaFitria NHNo ratings yet
Management of d-PTFE Membrane Exposure For Having Final Clinical Success
Management of d-PTFE Membrane Exposure For Having Final Clinical Success
Uploaded by
Nidia TorreaOriginal Description:
Original Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Management of d-PTFE Membrane Exposure For Having Final Clinical Success
Management of d-PTFE Membrane Exposure For Having Final Clinical Success
Uploaded by
Nidia TorreaCopyright:
Available Formats
CASE LETTER
Management of d-PTFE Membrane Exposure for Having
Final Clinical Success
Paolo C. Maridati, DDS, PhD, MSc1
Sergio Cremonesi, DDS, MSc2
Filippo Fontana, DDS, MSc1
Marco Cicciù, DDS, PhD, MSc3*
Carlo Maiorana, MD, DDS, MSc4
INTRODUCTION a nonresorbable membrane application for bone regeneration.
Thus, a dense polytetrafluoroethylene (d-PTFE) device was
R
econstruction of alveolar bone atrophy by means of
selected to handle this bone defect.
nonresorbable membrane is a well-known technique.
Downloaded from http://meridian.allenpress.com/doi/pdf/10.1563/aaid-joi-D-15-00074 by guest on 13 April 2020
Amoxicillin and clavulanic acid (1000 mg bid), ibuprofen
Expanded polytetrafluoroethylene membrane (e-PTFE)
(600 mg bid) and chlorhexidine digluconate (0.2 % mouth rinse)
classically required perfect soft tissue closure to
were prescribed.8–10
prevent wound dehiscence. The consequence of membrane
After injection of local anesthesia, a mid-crestal incision in
exposure ranges from a minor problem necessitating mem-
the keratinized mucosa was extended to the gingival sulcus of
brane removal to a major problem including treatment failure
2 adjacent teeth both mesially and distally. A full thickness
and implant loss.1–4 In the last few years, e-PTFE membrane has
flap was elevated to reach the bone defect. The implant site
been discontinued from the dental market. An alternative to
was prepared with calibrated surgical burs and a 3.4 mm
this barrier is the high-density polytetrafluoroethylene (d-PTFE)
diameter x 11 mm length implant (Xive S, Dentsply,
membrane. It is a nonresorbable device made of a high-density
Mannheim, Germany) was positioned with a recorded stability
PTFE with submicron (,0.3 lm) porosity size that has been
over 30Ncm. The bone augmentation procedure was per-
originally tested in postextraction sockets without primary soft
formed by combining a titanium-reinforced d-PTFE (Cytoplast,
tissue closure.5–7 Thanks to its structure, the d-PTFE barrier
Osteogenics Biomedical, Lubbock, Texas) with deproteinized
seems to have more resistance to bacterial penetration,
bovine bone graft (Bio-Oss, Geistlich, Wolhusen, Switzerland).
protecting the regenerating bone or implant. Some authors5,6
Then, four pins were placed to fix the membrane over the
have claimed the possibility that this membrane may remain
graft. Periosteal releasing incisions were done at the level of
exposed to the oral cavity with reduced risk of possible
the buccal flap to ensure proper passivation prior to suture.
complications, such as bacterial contamination, infection, and The suturing technique consisted of horizontal internal
loss of the graft. mattress and single suture.
This article describes the case of a d-PTFE membrane Postoperative instructions were given: cold pack, soft food
exposure and its management. diet, no hot drinks or food, no demanding physical work or
exercise, and no prosthesis on treated area. Sutures were
removed 14 days after surgery (Figures 1 through 8).
DESCRIPTION OF THE CASE At the time of suture removal, wound dehiscence was
A 63-year-old female patient presented for treatment of a left noticed. A membrane exposure larger than 3 mm without any
maxillary premolar. After clinical and radiographic examination, sign of infection was recorded. The membrane was left in place
a longitudinal fracture of tooth 2.4 was detected. The initial for additional 2 weeks to ensure bone regeneration.
treatment option consisted in tooth removal and delayed Four weeks after the regenerative procedure, a full
implant insertion with a ridge augmentation procedure. thickness flap was elevated to remove the d-PTFE membrane
After the tooth extraction and an uneventful healing of 2 and the fixation pins. The underlying bone graft was clinically
months, the site was re-evaluated. Radiographic analysis healthy without any sign of infection and thus was left in place
showed a 3-dimensional alveolar bone atrophy that required so as not to alter the regeneration process. A connective tissue
graft was harvested from the palate and placed on the top of
bone graft area to obtain wound closure.
1
Department of Dental Implants, U. O. C. Maxillofacial Surgery & Abutment connection was carried out 7 months after the
Odontostomatology, Fondazione IRCCS Cà Granda, University of Milan,
Milan, Italy. first surgery.
2
Private practice, Milan, Italy. After 3 weeks, a polyether impression was made to
3
Human Pathology Department, University of Messina, Messina, Italy. manufacture a screw-retained provisional restoration. After an
4
Oral Surgery and Department of Dental Implants, U. O. C. Maxillofacial additional 2 months, a definitive full ceramic restoration (IPS
Surgery & Odontostomatology, Fondazione IRCCS Cà Granda. University
of Milan, Milan, Italy. e.max CAD LT, Ivoclar, Schaan, Principality of Liechtenstein)
* Corresponding author, e-mail: acromarco@yahoo.it was cemented on a cad/cam zirconia abutment (Atlantis,
DOI: 10.1563/aaid-joi-D-15-00074 Dentsply).
Journal of Oral Implantology 289
Management of d-PTFE Membrane Exposure
Downloaded from http://meridian.allenpress.com/doi/pdf/10.1563/aaid-joi-D-15-00074 by guest on 13 April 2020
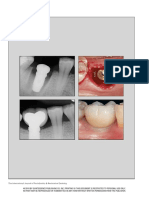
FIGURES 1–9. FIGURE 1. Clinical lateral view of the bone defect 2 months after tooth extraction. FIGURE 2. X ray of the bone defect. FIGURE 3.
Buccal view of the implant position. FIGURE 4. Occlusal view of the implant position. FIGURE 5. A deproteinized bovine bone graft combined
with a titanium reinforced high-density polytretrafluoroethylene was used to handle the bone defect. FIGURE 6. X ray after the regenerative
procedure. FIGURE 7. Membrane exposure at 3 weeks follow-up postop. FIGURE 8. Preoperative picture before membrane removal. FIGURE 9.
The bone graft under the membrane appeared clinically healthy; occlusal view.
A clinical and radiographic follow up was made 12 months on the suture or to the flap during periosteal incision with
after the placement of the definitive crown (Figures 9 through subsequent soft tissue necrosis.
15). The management of a membrane exposure is still a
controversial issue because most of the reported information
is not evidence-based but rather derived from the clinical
DISCUSSION experience of the surgeon. Fontana et al12 recently proposed a
clinical classification of complications with e-PTFE membrane
Today, guided bone regeneration by using of nonresorbable for an easier identification of the treatment procedure. In case
PTFE membrane is a predictable procedure for the reconstruc- of large membrane exposures (.3 mm) without purulent
tion of the atrophic jaw ridges. However, it is not lacking exudate, the authors suggest immediate membrane removal so
difficulties. Complications may vary from dehiscence with as not to jeopardize the underlying bone graft. This clinical
limited consequence to an abscess with the consequent approach was appropriate with e-PTFE membrane since this
treatment failure. Membrane exposure is considered the most barrier had a ‘‘labyrinth-like’’ structure with medium–high
common drawback. In the early 1990s, Buser2 experienced 41% porosity. Once exposed to the oral environment, microorgan-
of wound dehiscence in horizontal-guided bone regeneration. isms could quickly invade the surface and pass through the
From this pioneering study, a better knowledge of soft tissue membrane in 3 to 4 weeks, as reported by Simion.13
and membrane handling has drastically reduced the possibility On the contrary, the d-PTFE barrier is made of two layers of
of exposure.8,11 high-density polytetrafluoroethylene with less than 0.2–0.3 lm
Incomplete wound closure and consequent barrier expo- porosity size. For this reason, d-PTFE has been tested in
sure is usually the consequence of a clinical mistake at one step postextraction socket without primary soft tissue closure since
of the surgical procedure, as reported in previous study.12 In the 1990s.5–7 Barber et al5 described the possibility to achieve
this case report, the leading factor may be related to bone regeneration around implants with d-PTFE membrane
insufficient flap release with consequent tension and damage without primary soft tissue closure. At the time of tooth
290 Vol. XLII / No. Three / 2016
Maridati et al
Downloaded from http://meridian.allenpress.com/doi/pdf/10.1563/aaid-joi-D-15-00074 by guest on 13 April 2020
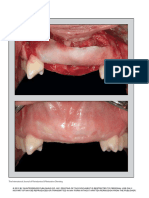
FIGURES 10–15. FIGURE 10. A connective tissue graft was placed to cover the regenerating bone graft. FIGURE 11. Soft tissue healing after 2
weeks. FIGURE 12. X ray of the submerged healing. FIGURE 13. Healing abutment connection. FIGURE 14. Buccal view of the final restoration
after 1 year. FIGURE 15. X ray after 1 year.
extraction, the implant was placed in association to a bone 2. Buser D, Dula K, Belser U, Hirt HP, Berthold H. Localized ridge
graft. A membrane was used to cover the bone graft and the augmentation using guided bone regeneration. 1. Surgical procedure in the
maxilla. Int J Periodontics Restorative Dent. 1993;13:29–45.
surgical defect with minimal flap reflection. The barrier was 3. Jovanovic SA, Spiekermann H, Richter EJ. Bone regeneration
removed 4 to 6 weeks after, and a well-regenerating bone was around titanium dental implants in dehisced defect sites: a clinical study.
observed. The area was left to heal for additional 3 months Int J Oral Maxillofac Implants. 1992;7:233–245.
before abutment connection. 4. Simion M, Trisi P, Piattelli A. Vertical ridge augmentation using a
membrane technique associated with osseointegrated implants. Int J
It may be hypothesized that this device may offer more Periodontics Restorative Dent. 1994:14:496–511.
resistance to bacterial contamination and penetration. For this 5. Barber HD, Lignelli J, Smith BM, Bartee BK. Using a dense PTFE
reason, in this clinical report, the device was left in place for 4 membrane without primary closure to achieve bone and tissue regenera-
weeks to ensure proper space-making effect. Barber5 suggest- tion. J Oral Maxillofac Surg. 2007;65:748–752.
6. Bartee BK. A simplified technique for ridge preservation after tooth
ed removing the membrane within 6 weeks to avoid major risk extraction. Dent Today. 1995:14:62–67.
of complications; however, in this case, the soft tissues around 7. Bartee BK. The use of high-density polytetrafluoroethylene
the exposed device were stable without any sign of infection membrane to treat osseous defects: clinical reports. Implant Dent. Spring.
and, thus, device removal was postponed to enhance bone 1995;4:21–26.
8. Fontana F, Santoro F, Maiorana C, Iezzi G, Piattelli A, Simion M.
quality. As of now, we do not have sufficient experience to Clinical and histological evaluation of allogenous bone matrix versus
define which is the ‘‘point of no return’’ in d-PTFE membrane autogenous bone chips associated with titanium reinforced e-PTFE
removal. membrane for vertical ridge augmentation: a prospective pilot study in
The use of d-PTFE membrane has been recently claimed by the human. Int J Oral Maxillofac Implants. 2008;23:1003–1012.
9. Ronda M, Rebaudi A, Torelli L, Stacchi C. Expanded vs. dense
several authors9,11 as a valid alternative to e-PTFE to rebuild polytetrafluoroethylene membranes in vertical ridge augmentation around
large bony defect and atrophic maxillary and mandibular dental implants: a prospective randomized controlled clinical trial. Clin Oral
arches. Nevertheless, long-term clinical studies are needed to Implants Res. 2014;25:859–866.
confirm this hypothesis. 10. Simion M, Fontana F, Rasperini G, Maiorana C. Vertical ridge
augmentation by expanded-polytetrafluoroethylene membrane and a
combination of intraoral autogenous bone graft and deproteinized
anorganic bovine bone (Bio-Oss). Clin Oral Implants Res. 2007;18:620–629.
ABBREVIATIONS 11. Urban IA, Lozada JL, Jovanovic SA, Nagursky H, Nagy K. Vertical
ridge augmentation with titanium-reinforced, dense-PTFE membranes and a
d-PTFE: high-density polytetrafluoroethylene combination of particulated autogenous bone and anorganic bovine bone-
e-PTFE: expanded polytetrafluoroethylene derived mineral: a prospective case series in 19 patients. Int J Oral Maxillofac
Implants. 2014;29:185–193.
12. Fontana F, Grossi GB, Fimanò M, Maiorana C. Osseointegrated
implants in vertical ridge augmentation with a nonresorbable membrane: a
REFERENCES retrospective study of 75 implants with 1 to 6 years of follow-up. Int J
Periodontics Restorative Dent. 2015;35:29–39.
1. Buser D, Bragger U, Lang NP, Nyman S. Regeneration and 13. Simion M, Baldoni M, Rossi P, Zaffe D. A comparative study of the
enlargement of jaw bone using guided tissue regeneration. Clin Oral effectiveness of e-PTFE membranes with and without early exposure during
Implants Res. 1990;1:22–32. the healing period. Int J Periodontics Restorative Dent. 1994;14:166–180.
Journal of Oral Implantology 291
You might also like
- Population and Community Health Nursing 6th Edition Clark Test BankDocument10 pagesPopulation and Community Health Nursing 6th Edition Clark Test Bankhorsecarbolic1on100% (27)
- ISMP198 Periop Guidelns FINALDocument29 pagesISMP198 Periop Guidelns FINALHIRANGERNo ratings yet
- Retentive Aids in Maxillofacial ProsthesisDocument109 pagesRetentive Aids in Maxillofacial ProsthesisEnu KambojNo ratings yet
- Aussie Emergency CareDocument9 pagesAussie Emergency CarePuspita Sari FiazNo ratings yet
- Pod Uveitis Book RevDocument252 pagesPod Uveitis Book Revelika dwi100% (1)
- Extraction Site Reconstruction For Alveolar Ridge Preservation. Part 2: Membrane-Assisted Surgical TechniqueDocument4 pagesExtraction Site Reconstruction For Alveolar Ridge Preservation. Part 2: Membrane-Assisted Surgical TechniqueLuis MonteroNo ratings yet
- Membranes For Periodontal RegenerationDocument6 pagesMembranes For Periodontal RegenerationinventionjournalsNo ratings yet
- Captura de Tela 2022-09-11 À(s) 19.01.07Document7 pagesCaptura de Tela 2022-09-11 À(s) 19.01.07alefabri-1No ratings yet
- Fugazzotto ImmediateImplantPlacementMolarDocument7 pagesFugazzotto ImmediateImplantPlacementMolarLozovan GregoryNo ratings yet
- Tissue Preservation and Maintenance of Optimum Esthetics: A Clinical ReportDocument7 pagesTissue Preservation and Maintenance of Optimum Esthetics: A Clinical ReportBagis Emre GulNo ratings yet
- Koutouzisetal 2013IJOMIAbutmentdisconnectionClinicalDocument8 pagesKoutouzisetal 2013IJOMIAbutmentdisconnectionClinicalKyoko CPNo ratings yet
- SwithcingDocument6 pagesSwithcingKyoko CPNo ratings yet
- Implant-Retained Auricular Prosthesis: A Case Report: January 2010 - Vol.4Document4 pagesImplant-Retained Auricular Prosthesis: A Case Report: January 2010 - Vol.4noranNo ratings yet
- Removal Of..Document4 pagesRemoval Of..Andrea ValverdeNo ratings yet
- Ejed 15 3 Staehler p288Document18 pagesEjed 15 3 Staehler p288snkidNo ratings yet
- Distalization of Impacted Mandibular SecondDocument6 pagesDistalization of Impacted Mandibular SecondPatricia BurbanoNo ratings yet
- Grizas 2018Document16 pagesGrizas 2018Dome CárdenasNo ratings yet
- Implant-Supported Nasal Prosthesis. Clinical Case Report: Prótesis Nasal Implantosoportada. Reporte de Un Caso ClínicoDocument6 pagesImplant-Supported Nasal Prosthesis. Clinical Case Report: Prótesis Nasal Implantosoportada. Reporte de Un Caso ClínicoMelina Nataly Robles AcuñaNo ratings yet
- Immediate One-Stage Postextraction Implant: A Human Clinical and Histologic Case ReportDocument6 pagesImmediate One-Stage Postextraction Implant: A Human Clinical and Histologic Case ReportBagis Emre GulNo ratings yet
- Post-Prosthetic Surgery: Using Complete Denture Prosthesis For Propriotous Outcome During VestibuloplastyDocument3 pagesPost-Prosthetic Surgery: Using Complete Denture Prosthesis For Propriotous Outcome During VestibuloplastyInternational Journal of Innovative Science and Research TechnologyNo ratings yet
- JOHCD-Maintenance of Implant SupportedDocument3 pagesJOHCD-Maintenance of Implant SupportedManoj HumagainNo ratings yet
- Article 009Document9 pagesArticle 009MedstudNo ratings yet
- Jpis 42 105Document5 pagesJpis 42 105Jean Pierre Alcántara VigoNo ratings yet
- Case SeriesDocument5 pagesCase SeriesPavithra balasubramaniNo ratings yet
- Franceschi2018 PDFDocument12 pagesFranceschi2018 PDFMichal PerkowskiNo ratings yet
- Speroni Desplazamiento Paladar2010Document7 pagesSperoni Desplazamiento Paladar2010SergioNo ratings yet
- ISSN 2347-5579: Use of Porous Polyethylene Implants For Orbital Reconstruction-A ReviewDocument3 pagesISSN 2347-5579: Use of Porous Polyethylene Implants For Orbital Reconstruction-A ReviewVISHAKHANo ratings yet
- Adj 12563Document5 pagesAdj 12563anushiNo ratings yet
- Cyst DecompressionDocument7 pagesCyst DecompressionMugdha FrancisNo ratings yet
- SCD 12407Document5 pagesSCD 12407RochmahAiNurNo ratings yet
- Khoury Bone Augmentation Tunneling-TechniqueDocument4 pagesKhoury Bone Augmentation Tunneling-TechniqueSeptimiu TiplicaNo ratings yet
- Document PDFDocument4 pagesDocument PDFBagis Emre GulNo ratings yet
- Ic Rehabilitation of The Edentulous Patient Maxillectomy UiriDocument5 pagesIc Rehabilitation of The Edentulous Patient Maxillectomy Uiriayush srivastavaNo ratings yet
- Expansion of The Zone of Keratinised Tissue For Healthy Implant Abutment Interface Using Deepithelized Amnion:Chorion AllograftDocument6 pagesExpansion of The Zone of Keratinised Tissue For Healthy Implant Abutment Interface Using Deepithelized Amnion:Chorion AllograftzethyhanumNo ratings yet
- Articol PMFDocument6 pagesArticol PMFclaudia lupuNo ratings yet
- The Use of PRF in Covering Exposed ImplantDocument6 pagesThe Use of PRF in Covering Exposed ImplantMohammad AkheelNo ratings yet
- Immediate, Non-Submerged, Root-Analogue Zirconia Implant in Single Tooth ReplacementDocument3 pagesImmediate, Non-Submerged, Root-Analogue Zirconia Implant in Single Tooth ReplacementNishtha KumarNo ratings yet
- Reusing Dental Implants?: An Experimental Study For Detecting The Success Rates of Re-OsseointegrationDocument7 pagesReusing Dental Implants?: An Experimental Study For Detecting The Success Rates of Re-OsseointegrationSatya AsatyaNo ratings yet
- 10 1016@j Prosdent 2005 10 014Document5 pages10 1016@j Prosdent 2005 10 014everaldocruzNo ratings yet
- Total Maxillectomy With Prosthetic Reconstruction Technique: Case ReportDocument3 pagesTotal Maxillectomy With Prosthetic Reconstruction Technique: Case ReportArun PrasadNo ratings yet
- Multidisciplinary Treatment Approach To A Complicated Crown Fracture: A Case ReportDocument5 pagesMultidisciplinary Treatment Approach To A Complicated Crown Fracture: A Case Reportramadan ariesthaNo ratings yet
- 2022 AiutoDocument7 pages2022 AiutovivianaNo ratings yet
- Aumento de Reborde Con Tejido BlandoDocument8 pagesAumento de Reborde Con Tejido BlandoSilvia SolisNo ratings yet
- An Innovative Technique To Restore The Non Vital Teeth With Wide Open Canals - A Clinical StudyDocument4 pagesAn Innovative Technique To Restore The Non Vital Teeth With Wide Open Canals - A Clinical StudyIOSRjournalNo ratings yet
- Surgical and Endodontic Management of Large Cystic Lesion: AbstractDocument5 pagesSurgical and Endodontic Management of Large Cystic Lesion: AbstractanamaghfirohNo ratings yet
- Implant ResearchDocument8 pagesImplant ResearchFBI LUC1FeRNo ratings yet
- Amato 1 (2018), Immediate Implant PlacementDocument8 pagesAmato 1 (2018), Immediate Implant PlacementPhương Thảo TrầnNo ratings yet
- Immediate Dentoalveolar Restoration in Compromised Sockets: Technique and Bone BiologyDocument14 pagesImmediate Dentoalveolar Restoration in Compromised Sockets: Technique and Bone BiologyMichal PerkowskiNo ratings yet
- Madohc MS Id 000103Document6 pagesMadohc MS Id 000103Manjulika TysgiNo ratings yet
- Therapeutic Management For Immediate Implant Placement in Sites With Periapical Deficiencies Where Coronal Bone Is Present: Technique and Case ReportDocument6 pagesTherapeutic Management For Immediate Implant Placement in Sites With Periapical Deficiencies Where Coronal Bone Is Present: Technique and Case ReportRaul Alfonsin SanchezNo ratings yet
- Case Report: The Prosthetic Management of A Case of Epulis Fissuratum by A Novel TechniqueDocument6 pagesCase Report: The Prosthetic Management of A Case of Epulis Fissuratum by A Novel TechniqueMichelle VelezNo ratings yet
- Implementingsocketsealsurgery JP2008Document11 pagesImplementingsocketsealsurgery JP2008Gabriela ArgeseanuNo ratings yet
- Article 1434132803Document5 pagesArticle 1434132803antariksha DodNo ratings yet
- Yu 그리고 Qiu - 2018 - Efficacy of the split-thickness labial flap methodDocument6 pagesYu 그리고 Qiu - 2018 - Efficacy of the split-thickness labial flap methodsupercool0120No ratings yet
- Tissue ManagementDocument16 pagesTissue ManagementSaleh AlsadiNo ratings yet
- Simion 2012)Document13 pagesSimion 2012)SergioNo ratings yet
- Sticky Bone For Space Maintenance in The Treatment of Miller's Class I Gingival Recession Defects With VISTA Technique A Case SeriesDocument4 pagesSticky Bone For Space Maintenance in The Treatment of Miller's Class I Gingival Recession Defects With VISTA Technique A Case SeriesInternational Journal of Innovative Science and Research TechnologyNo ratings yet
- Oh 2010Document5 pagesOh 2010gbaez.88No ratings yet
- Protesis Fija Con Ataches. Rep Caso. 2020. Cureus. SivakumarDocument13 pagesProtesis Fija Con Ataches. Rep Caso. 2020. Cureus. SivakumarAngela AcostaNo ratings yet
- EDI Journal - A Graftless Solution For Adjacent Implants in The Aesthetic Zone - Dr. Tunkiwala Et AlDocument6 pagesEDI Journal - A Graftless Solution For Adjacent Implants in The Aesthetic Zone - Dr. Tunkiwala Et AlNaina TalwarNo ratings yet
- Complications of Using Retraction CordDocument4 pagesComplications of Using Retraction CordAya Ibrahim YassinNo ratings yet
- A New Porcine-Derived Collagen Matrix To Thicken Peri-Implant Soft Tissues in High Aestethic Value AreasDocument1 pageA New Porcine-Derived Collagen Matrix To Thicken Peri-Implant Soft Tissues in High Aestethic Value AreasEdoardo BiancoNo ratings yet
- 2015 - A New Technique For Increasing Keratinized Tisue Around Dental Implants. The Partialy Epithelialized Free Conective Tisue Graft. Retrospective Analysis of A Case Series - Eberhard FrischDocument7 pages2015 - A New Technique For Increasing Keratinized Tisue Around Dental Implants. The Partialy Epithelialized Free Conective Tisue Graft. Retrospective Analysis of A Case Series - Eberhard FrischJose LuisNo ratings yet
- Short ImplantsFrom EverandShort ImplantsBoyd J. TomasettiNo ratings yet
- Biology TMJ 2013Document8 pagesBiology TMJ 2013Nidia TorreaNo ratings yet
- The Speaking Method in Measuring Vertical DimensionDocument5 pagesThe Speaking Method in Measuring Vertical DimensionNidia TorreaNo ratings yet
- Antibodies To Watch in 2019: Hélène Kaplon & Janice M. ReichertDocument21 pagesAntibodies To Watch in 2019: Hélène Kaplon & Janice M. ReichertNidia TorreaNo ratings yet
- Journal of Dentistry: Review ArticleDocument13 pagesJournal of Dentistry: Review ArticleNidia TorreaNo ratings yet
- Intraoperative and Postoperative Complications, Accidents and Failures Related With Dental Implant ApplicationsDocument6 pagesIntraoperative and Postoperative Complications, Accidents and Failures Related With Dental Implant ApplicationsNidia TorreaNo ratings yet
- Research FinalDocument67 pagesResearch FinalhelshynieNo ratings yet
- Research 11Document22 pagesResearch 11Jenn TajanlangitNo ratings yet
- Pcos 170123144542Document86 pagesPcos 170123144542Dewi Puspita Apsari100% (2)
- Evaluation of Er:YAG and Er, Cr:YSGG Laser Irradiation For The Debonding of Prefabricated Zirconia CrownsDocument9 pagesEvaluation of Er:YAG and Er, Cr:YSGG Laser Irradiation For The Debonding of Prefabricated Zirconia Crownsdr.m3.abazaNo ratings yet
- Pma Memo No 2022 10 24 020Document9 pagesPma Memo No 2022 10 24 020Emma AustriaNo ratings yet
- Funda NotesDocument9 pagesFunda Noteszyrenejoy.motita.mnlNo ratings yet
- France and Saudi ArabiaDocument17 pagesFrance and Saudi ArabiaHajar HossamNo ratings yet
- Practical Research 1 Worksheet 3-1Document2 pagesPractical Research 1 Worksheet 3-1Brylle Christian C. LopeNo ratings yet
- Chicago London: Your ItineraryDocument3 pagesChicago London: Your ItineraryZubaidah ZamNo ratings yet
- Reproductive Health AssignmentDocument3 pagesReproductive Health AssignmentRajat Kumar SabharwalNo ratings yet
- Blood and Marrow Transplant Quality Management PlanDocument47 pagesBlood and Marrow Transplant Quality Management PlansumathiNo ratings yet
- Carbohydrate-Related Diseases Term PaperDocument5 pagesCarbohydrate-Related Diseases Term Paperheiress comiaNo ratings yet
- 10 COMPLETE PRACTICE TESTS (1) 30 CopiesDocument129 pages10 COMPLETE PRACTICE TESTS (1) 30 Copiesghulam abbasNo ratings yet
- Bachelor of Dental Surgery (BDS)Document2 pagesBachelor of Dental Surgery (BDS)Rohit GhuseNo ratings yet
- FARID SURA WIJAYA, Dr. H. M. Untung Tranggono, MS, PA (K), SP.B, Sp.UDocument2 pagesFARID SURA WIJAYA, Dr. H. M. Untung Tranggono, MS, PA (K), SP.B, Sp.UM. Sabirin AsikinNo ratings yet
- Quality Nutrition and Dietetics PracticeDocument3 pagesQuality Nutrition and Dietetics PracticeNurlienda HasanahNo ratings yet
- Existential Psychology - Review QuestionnaireDocument7 pagesExistential Psychology - Review Questionnairerop serrNo ratings yet
- Ps1 - CFD Analysis Cold ChainDocument9 pagesPs1 - CFD Analysis Cold ChainMark SantosNo ratings yet
- Read Online Textbook Behavior Modification What It Is and How To Do It 10Th Edition Garry Martin Ebook All Chapter PDFDocument22 pagesRead Online Textbook Behavior Modification What It Is and How To Do It 10Th Edition Garry Martin Ebook All Chapter PDFhilda.rufe918100% (2)
- PDF]Jan-Febc 2010 - Jaypee Brothers Medical Publishers jaypeebrothers.com/pdf/cataloginmonths/jan-feb2010.pdf by S Bhat - 2010 A unique book on clinical methods in surgery along with interactive DVD-ROM ... and clinical approach to each and individual surgical cases .... including a brief outline on prognosis. • Provides in-depth ..... Susmita Bhattacharya. Epidemiology. mbbs books - Scribd https://www.scribd.com/doc/239765747/mbbs-books Sep 15, 2014 - mbbs books - Download as Word Doc (.doc / .docx), PDF File (.pdf), Text ... BHATTACHARYA SHORT CASES IN SURGERY 5TH 2002 200.00 BOOKS1 - Scribd https://www.scribd.com/doc/99385577/BOOKS1 Jul 7, 2012 - BOOKS1 - Ebook download as PDF File (.pdf), Text file (.txt) or read book ... LAWRENCE W. SHORT CASES IN SURGERY / BHATTACHARYA, ... Medical Books - mbbs exam questions for second third and ... hafeesh.blogspot.com/2009/12/medical-books.html Dec 25, 2009 - GUPTE SHORT TEXTBOOK OF MEDICAL MICROBIOLOGY 8TH 2002 .... BHATTACHARYA SHORT CASESDocument3 pagesPDF]Jan-Febc 2010 - Jaypee Brothers Medical Publishers jaypeebrothers.com/pdf/cataloginmonths/jan-feb2010.pdf by S Bhat - 2010 A unique book on clinical methods in surgery along with interactive DVD-ROM ... and clinical approach to each and individual surgical cases .... including a brief outline on prognosis. • Provides in-depth ..... Susmita Bhattacharya. Epidemiology. mbbs books - Scribd https://www.scribd.com/doc/239765747/mbbs-books Sep 15, 2014 - mbbs books - Download as Word Doc (.doc / .docx), PDF File (.pdf), Text ... BHATTACHARYA SHORT CASES IN SURGERY 5TH 2002 200.00 BOOKS1 - Scribd https://www.scribd.com/doc/99385577/BOOKS1 Jul 7, 2012 - BOOKS1 - Ebook download as PDF File (.pdf), Text file (.txt) or read book ... LAWRENCE W. SHORT CASES IN SURGERY / BHATTACHARYA, ... Medical Books - mbbs exam questions for second third and ... hafeesh.blogspot.com/2009/12/medical-books.html Dec 25, 2009 - GUPTE SHORT TEXTBOOK OF MEDICAL MICROBIOLOGY 8TH 2002 .... BHATTACHARYA SHORT CASESSaiSuryaTeja0% (1)
- A Reading of Contemporary Hysteria From LacanDocument9 pagesA Reading of Contemporary Hysteria From LacanMarcelo AraújoNo ratings yet
- Our Materials: Ethical Shoe PromiseDocument5 pagesOur Materials: Ethical Shoe PromiseSharoon S RaoNo ratings yet
- Sample Family Case Study 1 1 - CompressDocument55 pagesSample Family Case Study 1 1 - CompressGladys JhayeNo ratings yet
- Saqa - 14909 - Nhlapo Bellina JV17099Document16 pagesSaqa - 14909 - Nhlapo Bellina JV17099sibonisoNo ratings yet
- 2018-089. NASKAH PUBLIKASI Elsi Nursyahdin MayaranaDocument12 pages2018-089. NASKAH PUBLIKASI Elsi Nursyahdin MayaranaElsi NursyahdinNo ratings yet
- Chain Exercise Terhadap Penurunan Intensitas Nyeri Pada LansiaDocument9 pagesChain Exercise Terhadap Penurunan Intensitas Nyeri Pada LansiaFitria NHNo ratings yet





















































































![PDF]Jan-Febc 2010 - Jaypee Brothers Medical Publishers jaypeebrothers.com/pdf/cataloginmonths/jan-feb2010.pdf by S Bhat - 2010 A unique book on clinical methods in surgery along with interactive DVD-ROM ... and clinical approach to each and individual surgical cases .... including a brief outline on prognosis. • Provides in-depth ..... Susmita Bhattacharya. Epidemiology. mbbs books - Scribd https://www.scribd.com/doc/239765747/mbbs-books Sep 15, 2014 - mbbs books - Download as Word Doc (.doc / .docx), PDF File (.pdf), Text ... BHATTACHARYA SHORT CASES IN SURGERY 5TH 2002 200.00 BOOKS1 - Scribd https://www.scribd.com/doc/99385577/BOOKS1 Jul 7, 2012 - BOOKS1 - Ebook download as PDF File (.pdf), Text file (.txt) or read book ... LAWRENCE W. SHORT CASES IN SURGERY / BHATTACHARYA, ... Medical Books - mbbs exam questions for second third and ... hafeesh.blogspot.com/2009/12/medical-books.html Dec 25, 2009 - GUPTE SHORT TEXTBOOK OF MEDICAL MICROBIOLOGY 8TH 2002 .... BHATTACHARYA SHORT CASES](https://imgv2-1-f.scribdassets.com/img/document/270707806/149x198/154b6610c1/1605034206?v=1)





