Professional Documents
Culture Documents
Production of Cyclohexane
Production of Cyclohexane
Uploaded by
Luiz Rodrigo AssisOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Production of Cyclohexane
Production of Cyclohexane
Uploaded by
Luiz Rodrigo AssisCopyright:
Available Formats
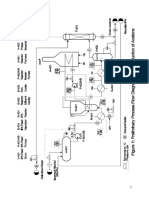
Production of Cyclohexane through Catalytic Hydrogenation of Benzene
Background
Cyclohexane is industrially produced from Benzene as it is not a naturally available
resource. Cyclohexane undergoes oxidation reactions yielding Cyclohexanone and
Cyclohexanol which are precursors for the production of Adipic acid and Caprolactum.
Caprolactum is the raw material used for producing polymer Nylon-6. Benzene reacts with
a mixture of hydrogen and methane in contact with a Nickel based catalyst producing
Cyclohexane. The conversion of this vapour phase reaction is almost 99%.
Reaction involved: Benzene + Hydrogen Cyclohexane (Vapour Phase)
Reactor Used: Catalytic Packed Bed Conversion Reactor
Reactor conditions: Outlet Temperature = 497 K, Pressure Drop = 1.02 atm
Catalyst Used: Nickel Based
Process Description
Fresh benzene (370 kmol/h) and excess hydrogen (1470 kmol/h) is preheated to a
temperature of 422 K and sent to a packed bed reactor. A vapour phase reaction occurs in
the reactor at 497 K which converts benzene to cyclohexane through catalytic
hydrogenation of benzene. The conversion of this reaction is about 99%. The reactor
products are cooled to 370 K and sent through a pressure reduction valve which reduces
the pressure of the stream from 30 atm to 24 atm. A two stage separator separates the
product cyclohexane from unreacted hydrogen and methane- first at a high pressure (24
atm) and then at a lower pressure (3 atm). The unreacted hydrogen-methane mixture is
recovered from the top of the flash column and is sent to a splitter having a splittling ratio
of 9:1. The smaller stream is sent as a recycle stream and mixes with fresh hydrogen, while
the rest is drawn out as fuel gas for incinerators. The bottom stream of the flash column
having 99% (wt/wt) cyclohexane is sent as feed to a distillation column for further
purification. The column consists of 12 stages operating at a reflux ratio of 5:1. The residue
from the column has our desired product with a purity of 99.5% (wt/wt) cyclohexane.
Assumptions
1. Side reactions were ignored.
2. A one pass conversion of 99% was considered.
3. The reactor outlet temperature was considered to be 497 K and pressure drop 1.02
atm.
4. Benzene used is mostly used up and hence, the unreacted benzene is not recovered.
5. The splitting ratio is mainly controlled by the composition of hydrogen in the feed
to the reactor. This was set by trial and error.
6. Custom units were followed for the simulation and not SI unit system
Results
INLET OUTLET
Parameter Unit Benzene Hydrogen Cyclohexane
Temperature K 311 311 427.708
Pressure atm 37.7 37.7 6
Mass Flow kg/h 28936.6 3417.51 23555.6
Mass Fraction
Hydrogen 0 0.816298 1.06E-15
Methane 0 0.183702 1.69E-12
Benzene 1 0 0.00520527
Cyclohexane 0 0 0.994795
Conclusion
Cyclohexane is obtained with a purity of 99.5% on weight basis through hydrogenation of
Benzene.
References
1. Cyclohexane
ARCO Technology Inc.
Hydrocarbon Processing, November 1977, p 143
2. Soave G.
"Equilibrium constants from a modified Redlich-Kwong equation of state" C.E.S.,
27, 6,1197-1203 (1972)
You might also like
- Production of N Octane From Ethylene and I ButaneDocument2 pagesProduction of N Octane From Ethylene and I ButaneRamyaNo ratings yet
- Project Group 9 CC01 PDFDocument24 pagesProject Group 9 CC01 PDFHuy NinhNo ratings yet
- Nitrobenzene Plant ReportDocument74 pagesNitrobenzene Plant ReportPankaj Borana100% (1)
- Although This Process Is No Longer in Common UseDocument15 pagesAlthough This Process Is No Longer in Common Usedia_aldy100% (1)
- AIChEJournalVolume57issue32011doi10 1002 - Aic 12289WilliamL Luyben - Designandcontroloftheethylbenzeneprocess1 PDFDocument16 pagesAIChEJournalVolume57issue32011doi10 1002 - Aic 12289WilliamL Luyben - Designandcontroloftheethylbenzeneprocess1 PDFTobias De SomerNo ratings yet
- Engineers Guide - Cumene Peroxidation Process For Phenol ProductionDocument2 pagesEngineers Guide - Cumene Peroxidation Process For Phenol ProductionEdrian A. Mañalong100% (1)
- Hydrodealkylation SimulationDocument8 pagesHydrodealkylation SimulationSchaieraNo ratings yet
- EthylbenzeneDocument4 pagesEthylbenzeneMouaath Al-Kalbani75% (4)
- AnilineDocument2 pagesAnilineKPAC333100% (2)
- Chemical Design EthylbenzeneDocument32 pagesChemical Design Ethylbenzeneafnan_lion94100% (1)
- Fundamentals of MasstransferandkineticshydrogenationDocument14 pagesFundamentals of MasstransferandkineticshydrogenationRamandhaPrasetyaAdibrataNo ratings yet
- AnilineDocument1 pageAnilineCherry Pearl MiparanumNo ratings yet
- Kinetics of Catalytic Dehydrogenation of Ethylbenzene To StyreneDocument5 pagesKinetics of Catalytic Dehydrogenation of Ethylbenzene To Styreneibrahim3318No ratings yet
- AcetoneDocument7 pagesAcetoneGeorgiana AndreeaNo ratings yet
- Project 6 - Ethylene Oxide PDFDocument13 pagesProject 6 - Ethylene Oxide PDFStephanie Hawkins100% (1)
- Overall Flowsheet Simulation Benzene Cyclohexane TW6Document7 pagesOverall Flowsheet Simulation Benzene Cyclohexane TW6Mitesh ParmarNo ratings yet
- Cumene Methods 2520of ProductionDocument4 pagesCumene Methods 2520of ProductionYunardi YusufNo ratings yet
- Presentation CumeneDocument39 pagesPresentation Cumeneممدوح الرويليNo ratings yet
- Cumene A PDFDocument4 pagesCumene A PDFdanena88No ratings yet
- Presentation CumeneDocument39 pagesPresentation CumeneBis ChemNo ratings yet
- MEK in School SecondDocument13 pagesMEK in School Secondifiok100% (1)
- A Project Report Submitted By: in Partial Fulfilment For The Award of The DegreeDocument91 pagesA Project Report Submitted By: in Partial Fulfilment For The Award of The DegreeHari BharathiNo ratings yet
- ETHYLBENZENEDocument19 pagesETHYLBENZENEolaNo ratings yet
- Physical Properties of Cumene Property ValueDocument4 pagesPhysical Properties of Cumene Property ValueC.Çağrı Yekeler50% (2)
- CHE655 - Plant Design Project #5 Summer 2010 Design of An Ehtyl Benzene Production ProcessDocument13 pagesCHE655 - Plant Design Project #5 Summer 2010 Design of An Ehtyl Benzene Production ProcessAyşe ÖztürkNo ratings yet
- Project: Design of A Reactor For The Aniline ProductionDocument19 pagesProject: Design of A Reactor For The Aniline ProductionLUIS ESTEBAN VÁSQUEZ CASTANEDANo ratings yet
- Ethylbenzene ProductionDocument30 pagesEthylbenzene ProductionUum LukmanNo ratings yet
- Ethyl Benzene Production ReactionsDocument2 pagesEthyl Benzene Production ReactionsMohd Hakimie0% (1)
- Benzene: Chemical Economics HandbookDocument3 pagesBenzene: Chemical Economics HandbookMaría VásquezNo ratings yet
- CumeneDocument21 pagesCumeneDiv SavaliyaNo ratings yet
- Production of Acrylonitrile by Ammoxidation of PropyleneDocument33 pagesProduction of Acrylonitrile by Ammoxidation of PropyleneJ José B VelasquezNo ratings yet
- EnnnDocument9 pagesEnnnSajid AliNo ratings yet
- Cumene Production Robert SchmidtDocument14 pagesCumene Production Robert SchmidtVatsalNo ratings yet
- Ethyl Benzene Plant DesignDocument45 pagesEthyl Benzene Plant DesignfaridzawiNo ratings yet
- Hydrogenation of Nitrobenzene To AnilineDocument8 pagesHydrogenation of Nitrobenzene To AnilineYu HuiNo ratings yet
- CPE639 Mini Project - Production of Acetonitrile Using Fluidized Bed Reactor PDFDocument41 pagesCPE639 Mini Project - Production of Acetonitrile Using Fluidized Bed Reactor PDFnoorNo ratings yet
- Styrene From Ethane and BenzeneDocument6 pagesStyrene From Ethane and BenzeneAmy Puah100% (2)
- Ethyl BenzeneDocument11 pagesEthyl BenzeneIan Jasper SabordoNo ratings yet
- Production of Aniline by Direct AminationDocument29 pagesProduction of Aniline by Direct AminationSatyshikh SrivNo ratings yet
- Cumene Production Process DescriptionDocument1 pageCumene Production Process DescriptionAudrey Patrick KallaNo ratings yet
- Cumene212 PDFDocument10 pagesCumene212 PDFtiaradyepiNo ratings yet
- Design of EthylbenzeneDocument5 pagesDesign of Ethylbenzenesahar vahdatifarNo ratings yet
- Liquidphasealkylationofbenzene With Ethylene 160713071057 PDFDocument111 pagesLiquidphasealkylationofbenzene With Ethylene 160713071057 PDFFrancesca GarciaNo ratings yet
- LECTURE - 6: Ethylene Derivatives: Ethylene Oxide and Ethanol Amines 6.1 Ethylene OxideDocument7 pagesLECTURE - 6: Ethylene Derivatives: Ethylene Oxide and Ethanol Amines 6.1 Ethylene Oxideمحمود محمدNo ratings yet
- Types of Phenol Manufacturing ProcessDocument4 pagesTypes of Phenol Manufacturing ProcessIsma AzraNo ratings yet
- FYP ProposalDocument11 pagesFYP ProposalArslan SamNo ratings yet
- CumeneDocument5 pagesCumeneNasmiyeth Rodriguez VittaNo ratings yet
- ReportDocument20 pagesReportCrazy HelloNo ratings yet
- Viewcontent11 PDFDocument54 pagesViewcontent11 PDFEr Mayur PatilNo ratings yet
- Reactor ModelDocument12 pagesReactor ModelTanuja ThanuNo ratings yet
- Ethylbenzene MSDS PDFDocument6 pagesEthylbenzene MSDS PDFyuanitaNo ratings yet
- Report BTPDocument47 pagesReport BTPvpsrpuchNo ratings yet
- Mek From N Butene PDFDocument111 pagesMek From N Butene PDFAlexis PulhinNo ratings yet
- CiclohexanoDocument6 pagesCiclohexanoSebastian BelloNo ratings yet
- AADocument30 pagesAAAhmed MajidNo ratings yet
- Hysys SimulationDocument24 pagesHysys SimulationNeybil100% (1)
- Process Control CompleteDocument71 pagesProcess Control CompleteJanusNo ratings yet
- Abstract:: Pinacol Pinacolone Rearrangement Reaction (Preparation of Benzopinacolone)Document6 pagesAbstract:: Pinacol Pinacolone Rearrangement Reaction (Preparation of Benzopinacolone)Bryan Gerard GuillermoNo ratings yet
- First Review Report On Production of Phenol: Done by GuideDocument27 pagesFirst Review Report On Production of Phenol: Done by GuideRuban RkNo ratings yet
- Exp 2Document7 pagesExp 2zanjinyadzaNo ratings yet
- Jaro Education JDDocument4 pagesJaro Education JDsushant kadamNo ratings yet
- Adv For The Post of Apprentices 2021Document6 pagesAdv For The Post of Apprentices 2021sushant kadamNo ratings yet
- CR RemovalDocument4 pagesCR Removalsushant kadamNo ratings yet
- Recruitment of Specialist OfficersDocument3 pagesRecruitment of Specialist Officerssushant kadamNo ratings yet
- Sem 8 SyallbusDocument16 pagesSem 8 Syallbussushant kadamNo ratings yet
- Cyclohexane PDFDocument14 pagesCyclohexane PDFsushant kadamNo ratings yet
- A Review On Phenolic Resin and Its Composites: Current Analytical Chemistry October 2017Document14 pagesA Review On Phenolic Resin and Its Composites: Current Analytical Chemistry October 2017sushant kadamNo ratings yet
- Brief Summary Subject: Proposed Coal Tar Distillation Project To Manufacture 84,000 MT/Annum Coal TarDocument2 pagesBrief Summary Subject: Proposed Coal Tar Distillation Project To Manufacture 84,000 MT/Annum Coal Tarsushant kadamNo ratings yet
- 12 - Chapter 3 PDFDocument122 pages12 - Chapter 3 PDFsushant kadamNo ratings yet
- IJCT 10 (1) CR RemovalDocument7 pagesIJCT 10 (1) CR Removalsushant kadamNo ratings yet
- Name: - Subject: Science Grade: 5 Sec: PRWS: 4 (L-13) Roll No.: Date: - / - / - (2016-17)Document3 pagesName: - Subject: Science Grade: 5 Sec: PRWS: 4 (L-13) Roll No.: Date: - / - / - (2016-17)Priyanka GandhiNo ratings yet
- Gas TestDocument1 pageGas TestAbdelaaliNo ratings yet
- Katalog Almega SejahteraDocument35 pagesKatalog Almega SejahteraRuri YuliantiNo ratings yet
- Modeling Suspended Growth Systems: - See Grady, Daigger & LimDocument43 pagesModeling Suspended Growth Systems: - See Grady, Daigger & LimOlaleye Akeem KehindeNo ratings yet
- JEE Advanced 2023 Surface Chemistry Revision Notes - Free PDF DownloadDocument7 pagesJEE Advanced 2023 Surface Chemistry Revision Notes - Free PDF Downloadhishamkalliyath19No ratings yet
- Ep 3920 Machinable Grade: Product InformationDocument3 pagesEp 3920 Machinable Grade: Product InformationDimset DieptabNo ratings yet
- Industrial Crops & Products: Didem Sutay Kocabas, Merve Erkoç Akçelik, Erinç Bahçegül, Hatice Neval OzbekDocument13 pagesIndustrial Crops & Products: Didem Sutay Kocabas, Merve Erkoç Akçelik, Erinç Bahçegül, Hatice Neval Ozbekjans carlosNo ratings yet
- Antimalarial DrugsDocument36 pagesAntimalarial DrugsKasim UmarNo ratings yet
- Microbiology 214 Skills Test Notes PDFDocument21 pagesMicrobiology 214 Skills Test Notes PDFTayla Nel100% (1)
- Protein Isolates From Bambara Groundnut Voandz.Document19 pagesProtein Isolates From Bambara Groundnut Voandz.ilyas.tassineNo ratings yet
- XII Organic Reasoning QuestionsDocument7 pagesXII Organic Reasoning QuestionslakshvanthbalaNo ratings yet
- MSDS - Irgazin Yellow 3 RLTNDocument6 pagesMSDS - Irgazin Yellow 3 RLTNfernando.rsrNo ratings yet
- Dowclene 1601Document4 pagesDowclene 1601Castle Jing100% (1)
- Eco-Friendly Dyes and Dyeing: Asim Kumar Roy CHOUDHURYDocument33 pagesEco-Friendly Dyes and Dyeing: Asim Kumar Roy CHOUDHURYUtsho ParvezNo ratings yet
- PH8HF Holder PH Meter, YokogawaDocument39 pagesPH8HF Holder PH Meter, YokogawaRangga TaufiqurahmanNo ratings yet
- The Chequered History of The Development and Use of Simultaneous Equations For The Accurate Determination of Chlorophylls A and BDocument8 pagesThe Chequered History of The Development and Use of Simultaneous Equations For The Accurate Determination of Chlorophylls A and BFitri SukmawatiNo ratings yet
- EEMPA Process 1Document9 pagesEEMPA Process 1Maythee SaisriyootNo ratings yet
- Organic-IB-MC-Exam Qu-AnsDocument3 pagesOrganic-IB-MC-Exam Qu-Ansbernardowusubempah123No ratings yet
- Preliminary ExaminationDocument3 pagesPreliminary ExaminationJulie Jr GulleNo ratings yet
- Full Download Book Titanium Dioxide Tio2 and Its Applications PDFDocument41 pagesFull Download Book Titanium Dioxide Tio2 and Its Applications PDFkatie.gonzales634100% (25)
- Rekap Pasien Arry JohanDocument18 pagesRekap Pasien Arry JohanArry Johan DANo ratings yet
- Astm F 1545Document8 pagesAstm F 1545Ivan Alaniz100% (2)
- Chemistry CH No 10Document5 pagesChemistry CH No 10Syed Salman SaeedNo ratings yet
- ThreeBond 1539B Elastic AdhesiveDocument5 pagesThreeBond 1539B Elastic AdhesiveGobinath BalasubramaniamNo ratings yet
- B 4 Revision PackDocument64 pagesB 4 Revision PackKrish SharmaNo ratings yet
- Ut 1 2Document10 pagesUt 1 2MoNo ratings yet
- Module 1: FireDocument13 pagesModule 1: FireMarielle CaralipioNo ratings yet
- Astm D 6886Document6 pagesAstm D 6886Navneet YadavNo ratings yet
- Science-IA Guide LanternaDocument11 pagesScience-IA Guide LanternaIffat KamaalNo ratings yet
- Industrial Crops & Products: A A B B B ADocument8 pagesIndustrial Crops & Products: A A B B B ASiddharthBhasneyNo ratings yet