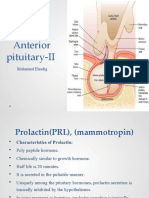
Professional Documents
Culture Documents
Uk Bloomsyndrome
Uk Bloomsyndrome
Uploaded by
MowlidAbdirahman Ali madaaleCopyright:
Available Formats
You might also like
- Mycoses - 2023 - Kato - A Systematic Review and Meta Analysis of Efficacy and Safety of Isavuconazole For The Treatment andDocument10 pagesMycoses - 2023 - Kato - A Systematic Review and Meta Analysis of Efficacy and Safety of Isavuconazole For The Treatment andc gNo ratings yet
- Las Claves Genéticas - Richard RuddDocument29 pagesLas Claves Genéticas - Richard RuddaleNo ratings yet
- Beckwith-Wiedemann SyndromeDocument6 pagesBeckwith-Wiedemann SyndromeAhmad Althof Malihul AdiNo ratings yet
- Molecular Diagnosis of Inherited Disorders: Lessons From HemoglobinopathiesDocument14 pagesMolecular Diagnosis of Inherited Disorders: Lessons From HemoglobinopathiesUmar AliNo ratings yet
- Thein-2004-British Journal of HaematologyDocument11 pagesThein-2004-British Journal of HaematologySri ArianiNo ratings yet
- Journal of Genetic Counseling - 2008 - Pilarski - Cowden Syndrome A Critical ReDocument15 pagesJournal of Genetic Counseling - 2008 - Pilarski - Cowden Syndrome A Critical ReCiudadano “Citizen Dick” VolanteNo ratings yet
- Kes - Martin A. - Mutations in The Adrenoleukodystrophy Gene (1997) (10.1002 - (Sici) 1098-1004 (1997) 9 - 63.0.co - 2-5) - Libgen - LiDocument12 pagesKes - Martin A. - Mutations in The Adrenoleukodystrophy Gene (1997) (10.1002 - (Sici) 1098-1004 (1997) 9 - 63.0.co - 2-5) - Libgen - LiLoplopNo ratings yet
- Fioretos 2001Document9 pagesFioretos 2001sawantleena62No ratings yet
- 2003 - Epigenetic Silencing of Multiple Interferon Pathway Genes After Cellular ImmortalizationDocument10 pages2003 - Epigenetic Silencing of Multiple Interferon Pathway Genes After Cellular ImmortalizationFernando Fernández GarcíaNo ratings yet
- FPDMM2Document9 pagesFPDMM2sccNo ratings yet
- Statistical Applications in Genetics and Molecular BiologyDocument19 pagesStatistical Applications in Genetics and Molecular BiologyJose MoralesNo ratings yet
- 60 453 PDFDocument13 pages60 453 PDFVictor Hugo OrtizNo ratings yet
- Kotlyar 2020Document16 pagesKotlyar 2020Merab KvintradzeNo ratings yet
- Anorectal Malformation: The Etiological FactorsDocument10 pagesAnorectal Malformation: The Etiological FactorsHasri Rina WalastriNo ratings yet
- Abnormal Folate Metabolism and MaternalDocument24 pagesAbnormal Folate Metabolism and MaternalJovicaNo ratings yet
- Zlotogora 2004Document6 pagesZlotogora 2004MARIA JOSE MENDIOLA FIGUEROANo ratings yet
- Predisposing Factors: TH NDDocument3 pagesPredisposing Factors: TH NDArianna Jasmine MabungaNo ratings yet
- Craniofacial MicrosomiaDocument21 pagesCraniofacial MicrosomiaMarcela Cano Ricardo100% (1)
- Shiang 1994Document8 pagesShiang 1994Daniela NarvaezNo ratings yet
- CML Chapter Chereda & Melo Springer 2016Document23 pagesCML Chapter Chereda & Melo Springer 2016JuniaNo ratings yet
- 12.the Impact of Trisomy 21 On Early Human HematopoiesisDocument2 pages12.the Impact of Trisomy 21 On Early Human HematopoiesisCristi Daniel NeagoeNo ratings yet
- Genetica Predisposicion InglesDocument12 pagesGenetica Predisposicion InglesAlejandro PalayNo ratings yet
- Cancer CellDocument140 pagesCancer CellmanbastiNo ratings yet
- 1.0 Genetics and InheritanceDocument13 pages1.0 Genetics and Inheritanceincognitus94100% (4)
- Epidermolysis Bullosa, Junctional, Herlitz Type: (Type The Document Title)Document11 pagesEpidermolysis Bullosa, Junctional, Herlitz Type: (Type The Document Title)kucingkucingNo ratings yet
- 2015 Int J Genet & Mol Biol Article1424860767 - Ortega Et AlDocument7 pages2015 Int J Genet & Mol Biol Article1424860767 - Ortega Et AlAlejandro1-7No ratings yet
- Hum. Reprod. Update 2002 Laml 483 91Document9 pagesHum. Reprod. Update 2002 Laml 483 91Amit GoelNo ratings yet
- Ageing and The Mismatch Repair SystemDocument7 pagesAgeing and The Mismatch Repair Systemeric.cheninNo ratings yet
- Campbell - Walsh-Wein UROLOGY 12th Ed (Dragged) 2Document24 pagesCampbell - Walsh-Wein UROLOGY 12th Ed (Dragged) 2revanth kallaNo ratings yet
- Sickle Cell DiseaseDocument18 pagesSickle Cell DiseasedrheayNo ratings yet
- X-Linked Dominant Chondrodysplasia PunctataDocument5 pagesX-Linked Dominant Chondrodysplasia Punctatalocos3dNo ratings yet
- Analysis of Recombination Along Chromosome 21 During Human Female Pachytene StageDocument11 pagesAnalysis of Recombination Along Chromosome 21 During Human Female Pachytene StageJimmy AvilaNo ratings yet
- Causes of Congenital MalformationsDocument5 pagesCauses of Congenital MalformationsBenyam ZenebeNo ratings yet
- DescargaDocument18 pagesDescargakirbwer macedoNo ratings yet
- Abp-Ivb AnemiaDocument11 pagesAbp-Ivb AnemiaEmerson MadridNo ratings yet
- Historical and Clinical Perspectives On CHR TranslocationsDocument14 pagesHistorical and Clinical Perspectives On CHR TranslocationsAsma HMILANo ratings yet
- 1 s2.0 S0041008X04001334 MainDocument14 pages1 s2.0 S0041008X04001334 MainOscar Alonso LunaNo ratings yet
- Flejter 1996Document6 pagesFlejter 1996Rahul AmbawataNo ratings yet
- Onco Ch23 ESFTDocument31 pagesOnco Ch23 ESFTIlincaNo ratings yet
- Syndromes of The First and Second Pharyngeal Arches: A ReviewDocument7 pagesSyndromes of The First and Second Pharyngeal Arches: A ReviewHARITHA H.PNo ratings yet
- Prion-Like Mechanisms in Alzheimer DiseaseDocument17 pagesPrion-Like Mechanisms in Alzheimer Diseasesabrina whardhaniNo ratings yet
- Genetic Counseling in TranslocationsDocument15 pagesGenetic Counseling in TranslocationsAsma HMILANo ratings yet
- Transient Myeloproliferative Disorder: What's Eosinophilia Got To Do With It?Document1 pageTransient Myeloproliferative Disorder: What's Eosinophilia Got To Do With It?wadeschulzNo ratings yet
- Imprinting DisordersDocument10 pagesImprinting DisordersAhmad KHNo ratings yet
- Craniofacialmicrosomia: Kathleyn A. Brandstetter,, Krishna G. PatelDocument21 pagesCraniofacialmicrosomia: Kathleyn A. Brandstetter,, Krishna G. PatelariskaNo ratings yet
- Imprinting DisordersDocument19 pagesImprinting Disorderssenuberh13No ratings yet
- Current Concepts Review: Ph. Debeer, L. de Smet, W. J. M. Van de Ven, G. Fabry, J.-P. FrynsDocument12 pagesCurrent Concepts Review: Ph. Debeer, L. de Smet, W. J. M. Van de Ven, G. Fabry, J.-P. FrynsDr. Pedro Javier Cadena GonzálezNo ratings yet
- Genetic Background of Cutaneous Forms of Lupus Erythematosus: Update On Current EvidenceDocument10 pagesGenetic Background of Cutaneous Forms of Lupus Erythematosus: Update On Current EvidenceFaradhillah Adi SuryadiNo ratings yet
- Churpek Et Al. - 2015 - Genomic Analysis of Germ Line and Somatic Variants in Familial Myelodysplasiaacute Myeloid Leukemia Key PointsDocument7 pagesChurpek Et Al. - 2015 - Genomic Analysis of Germ Line and Somatic Variants in Familial Myelodysplasiaacute Myeloid Leukemia Key Pointstefod30422No ratings yet
- Genetic Factors of Childhood CancerDocument3 pagesGenetic Factors of Childhood CancerDima HarounNo ratings yet
- FPDMM3Document10 pagesFPDMM3sccNo ratings yet
- Pol 030Document6 pagesPol 030Mirabella AstraNo ratings yet
- Epigenetic and Cancer Therapy, 2019Document12 pagesEpigenetic and Cancer Therapy, 2019AbsjeyNo ratings yet
- Pediatric Genetics Dr. Samed AlsalmiDocument166 pagesPediatric Genetics Dr. Samed AlsalmiabhivnairNo ratings yet
- 31861.1 20201218131318 CoveredDocument5 pages31861.1 20201218131318 CoveredPatrick StarNo ratings yet
- La Mellarin D2Document2 pagesLa Mellarin D2JESUS DAVID BOLA‹O JIMENEZNo ratings yet
- Imam ShahDocument7 pagesImam Shahdk3873050No ratings yet
- PSU Vol 14, 2000Document2 pagesPSU Vol 14, 2000rajarshikNo ratings yet
- Lecture 2 EnglishDocument24 pagesLecture 2 Englishmondalsawan96No ratings yet
- Genetica Das Doencas Hematologicas As HemoglobinopDocument12 pagesGenetica Das Doencas Hematologicas As HemoglobinopMinésio João DoclasNo ratings yet
- A Model for Gene Therapy: Gene Replacement in the Treatment of Sickle Cell Anemia and ThalassemiaFrom EverandA Model for Gene Therapy: Gene Replacement in the Treatment of Sickle Cell Anemia and ThalassemiaNo ratings yet
- Neuromuscular DisordersDocument1 pageNeuromuscular DisordersMowlidAbdirahman Ali madaaleNo ratings yet
- Epi Lecture 1 Part IIDocument36 pagesEpi Lecture 1 Part IIMowlidAbdirahman Ali madaaleNo ratings yet
- Chain of InfectionDocument35 pagesChain of InfectionMowlidAbdirahman Ali madaale100% (1)
- Microbiology RVDocument91 pagesMicrobiology RVMowlidAbdirahman Ali madaaleNo ratings yet
- Basics of ProbabilityDocument7 pagesBasics of ProbabilityMowlidAbdirahman Ali madaaleNo ratings yet
- CholeraDocument4 pagesCholeraMowlidAbdirahman Ali madaaleNo ratings yet
- Lec-4 The Anterior Pituitary Gland-IIDocument32 pagesLec-4 The Anterior Pituitary Gland-IIMowlidAbdirahman Ali madaaleNo ratings yet
- Biochemistry Answers 161-179Document2 pagesBiochemistry Answers 161-179MowlidAbdirahman Ali madaaleNo ratings yet
- Biochemistry 2Document9 pagesBiochemistry 2MowlidAbdirahman Ali madaaleNo ratings yet
- Toxicology Lecture 10 (Chapter 7 Volatiles)Document22 pagesToxicology Lecture 10 (Chapter 7 Volatiles)MowlidAbdirahman Ali madaaleNo ratings yet
- Lec-5 The Posterior Pituitary GlandDocument39 pagesLec-5 The Posterior Pituitary GlandMowlidAbdirahman Ali madaaleNo ratings yet
- Chapter 6. ....Document11 pagesChapter 6. ....MowlidAbdirahman Ali madaaleNo ratings yet
- General Principles of Prevention and Control of DiseasesDocument17 pagesGeneral Principles of Prevention and Control of DiseasesMowlidAbdirahman Ali madaaleNo ratings yet
- Sexually Transmitted DiseasesDocument54 pagesSexually Transmitted DiseasesMowlidAbdirahman Ali madaaleNo ratings yet
- Floods and DroughtDocument28 pagesFloods and DroughtMowlidAbdirahman Ali madaaleNo ratings yet
- Forensic Med Lecture 11 Chapter 11 Sexual Assaults)Document27 pagesForensic Med Lecture 11 Chapter 11 Sexual Assaults)MowlidAbdirahman Ali madaaleNo ratings yet
- Lec-3 The Pituitary Gland-IDocument37 pagesLec-3 The Pituitary Gland-IMowlidAbdirahman Ali madaaleNo ratings yet
- Transportation: About UsDocument2 pagesTransportation: About UsMowlidAbdirahman Ali madaaleNo ratings yet
- Group 4 DDDocument2 pagesGroup 4 DDMowlidAbdirahman Ali madaaleNo ratings yet
- Genital Infections and SexuallyDocument47 pagesGenital Infections and SexuallyMowlidAbdirahman Ali madaaleNo ratings yet
- Sumayo Xasan Omar 3. Sumayo C/Raxmaan Cali 4. Sabirin Daud Cilmi 5. Sagal Dahir Shire Mrs. Fatima SahraDocument4 pagesSumayo Xasan Omar 3. Sumayo C/Raxmaan Cali 4. Sabirin Daud Cilmi 5. Sagal Dahir Shire Mrs. Fatima SahraMowlidAbdirahman Ali madaaleNo ratings yet
- History of MakaDocument2 pagesHistory of MakaMowlidAbdirahman Ali madaaleNo ratings yet
- Yaya 7Document2 pagesYaya 7MowlidAbdirahman Ali madaaleNo ratings yet
- Yaxye 6Document3 pagesYaxye 6MowlidAbdirahman Ali madaaleNo ratings yet
- White House and Facebook InteractionsDocument31 pagesWhite House and Facebook InteractionstobihiNo ratings yet
- Septic ArthritisDocument12 pagesSeptic ArthritisRashi JainNo ratings yet
- Dr. Ranjita Singh Department of PathologyDocument19 pagesDr. Ranjita Singh Department of PathologyMohan Prasad GuptaNo ratings yet
- Feyer 2020 WvngoshawkDocument13 pagesFeyer 2020 WvngoshawkAgnieszka CzujkowskaNo ratings yet
- Smoking Is DangerousDocument3 pagesSmoking Is DangerousAbdulmalkNo ratings yet
- YökdilDocument195 pagesYökdilglisilsiklin antibiyotikNo ratings yet
- 03 NCP 1 Viral HepatitisDocument17 pages03 NCP 1 Viral Hepatitisamit100% (2)
- Psychophysiological Methods in NeuroscienceDocument17 pagesPsychophysiological Methods in NeuroscienceKariela EstherNo ratings yet
- ABG Ebook PDFDocument13 pagesABG Ebook PDFM DNo ratings yet
- Locomotor Training in People With Parkinson DiseaseDocument10 pagesLocomotor Training in People With Parkinson DiseasemilananandNo ratings yet
- Anees Ahmed Corrected Thesis - RecentDocument135 pagesAnees Ahmed Corrected Thesis - RecentAbdul MunimNo ratings yet
- Brochure Chorustrio Rev.04 enDocument4 pagesBrochure Chorustrio Rev.04 enHaitham Salama Ghareeb100% (1)
- 1.19 Exam PracticeDocument5 pages1.19 Exam Practicesunnr008.211No ratings yet
- Pediatric Assessment TriangleDocument31 pagesPediatric Assessment TriangleblairNo ratings yet
- Psych Narrative Report (1) LoissDocument21 pagesPsych Narrative Report (1) LoissBea Aldueza100% (1)
- Role of The Youth in The Upcoming ElectionDocument15 pagesRole of The Youth in The Upcoming ElectionMitch Escribano MarianoNo ratings yet
- Safety Data Sheet For PolyuDocument4 pagesSafety Data Sheet For PolyuMehedi HasanNo ratings yet
- Reten Modern Dressing 2023 PKDMTDocument51 pagesReten Modern Dressing 2023 PKDMTMasros TukiranNo ratings yet
- Heritability of Behavior in The Abnormally Aggressive Dog, by A. SemyonovaDocument8 pagesHeritability of Behavior in The Abnormally Aggressive Dog, by A. SemyonovaDogsBite.org100% (9)
- Infection Control NY NewDocument6 pagesInfection Control NY NewRC PmedNo ratings yet
- Copy-Of-Chapter 38 Medical Gas TherapyDocument29 pagesCopy-Of-Chapter 38 Medical Gas TherapyMaxinne GorospeNo ratings yet
- Bakteriološki Nalaz U Uretri Muškaraca Sa I Bez Negonoroičnog UretritisaDocument4 pagesBakteriološki Nalaz U Uretri Muškaraca Sa I Bez Negonoroičnog UretritisaNemanjaDragosavacNo ratings yet
- International Journal of Research in AYUSH and Pharmaceutical SciencesDocument6 pagesInternational Journal of Research in AYUSH and Pharmaceutical SciencesDeepak BaghelNo ratings yet
- Chapter 13 - Diseases of White Blood Cells, Lymph Nodes, Spleen, and ThymusDocument10 pagesChapter 13 - Diseases of White Blood Cells, Lymph Nodes, Spleen, and ThymusAgnieszka WisniewskaNo ratings yet
- What Is ECG?Document2 pagesWhat Is ECG?Lê Thanh HàNo ratings yet
- TriageDocument37 pagesTriagekyuleen05No ratings yet
- 10 Secrets in 10 YearsDocument15 pages10 Secrets in 10 YearsasicsNo ratings yet
- Revised Monthly Reporting Format - HWCDocument8 pagesRevised Monthly Reporting Format - HWCChanmari West Sub-Centre50% (2)
- Pola Bleach PDFDocument1 pagePola Bleach PDFSree CumarNo ratings yet
Uk Bloomsyndrome
Uk Bloomsyndrome
Uploaded by
MowlidAbdirahman Ali madaaleOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Uk Bloomsyndrome
Uk Bloomsyndrome
Uploaded by
MowlidAbdirahman Ali madaaleCopyright:
Available Formats
Bloom’s syndrome
Author: Doctor Mounira Amor-Guéret1
Creation date: February 2004
Scientific Editor: Professor Gilbert Tchernia
1
Previous address: Interactions moléculaires et cancer, CNRS UMR 8126, Institut Gustave Roussy, 39
Rue Camille Desmoulins, 94805 VILLEJUIF cedex, France.
Present address: Institut Curie - Section de Recherche, CNRS UMR 2027, Bâtiment 110, Centre
Universitaire, F-91405 ORSAY cedex, France. mounira.amor@curie.u-psud.fr
Abstract
Key-words
Disease name/synonyms
Definition
Differential diagnosis
Etiology
Clinical description
Diagnostic methods
Epidemiology
Genetic counseling
Antenatal diagnosis
Management including treatment
Unresolved questions
References
Abstract
Bloom’s syndrome (BS) is a rare human autosomal recessive disorder belonging to a group of
“chromosomal breakage syndromes”. BS is characterized by marked genetic instability, including a high
level of sister chromatid exchanges, associated with a greatly increased predisposition to a wide range of
cancers commonly affecting the general population. The constant clinical features of BS are proportionate
pre- and postnatal growth retardation and cancer predisposition. Additional clinical features include
dolichocephaly, facial sun-sensitive telangiectatic erythema, patchy areas of hyper- and hypopigmentation
of the skin and moderate to severe immunodeficiency manifested by recurrent respiratory tract and
gastrointestinal infections. A 10-fold increase in the rate of sister chromatid exchanges (SCEs) in BS cells
compared to normal cells is the only objective criteria for BS diagnosis. Clinical diagnosis is confirmed
cytogenetically by demonstrating characteristic chromosome instability. BS arises through mutations in
both copies of the BLM gene which encodes a 3’-5’ DNA helicase, a member of the RecQ family. The
function of the BLM protein remains unclear, but several lines of evidence support a major role in
maintaining genomic stability during DNA replication, recombination and repair. BS frequency in the
general population is unknown, probably because this disease is very rare. In Askenazic Jewish
population, the frequency of BS is approximately 1 in 48 000. This is due to a founder effect,
approximately 1% of the Ashkenasi Jewish population being heterozygous carriers for the blmAsh
mutation. There is no curative treatment for BS. However, a physician should carefully follow BS patients
in order to ensure early diagnosis of cancer.
Key-words
Bloom’s syndrome, cancer predisposition, genetic instability, sister chromatid exchanges, RecQ helicase.
Amor-Guéret M. Bloom’s syndrome. Orphanet Encyclopedia. February 2004.
http://www.orpha.net/data/patho/GB/uk-Bloomsyndrome.pdf 1
Disease name/synonyms on chromosome 15 at 15q26.1. Currently, there
Bloom syndrome (Bloom’s syndrome) is no argument for a possible genetic
Synonyms: Bloom-Torre-Mackacek syndrome, heterogeneity in BS. Nonsense or frameshift
Congenital Telangiectatic Erythema. mutations leading to a premature termination
codon as well as missense mutations have been
Definition found in BLM gene from BS patients (Ellis et al.,
Bloom’s syndrome (BS) is a rare human 1995b; Foucault et al., 1997; Barakat et al.,
autosomal recessive disorder characterized by 2000). One particular BLM gene mutation
marked genetic instability associated with a corresponding to a 6-bp deletion and a 7-bp
greatly increased predisposition to a wide range insertion at nucleotide position 2281, referred as
of cancers commonly affecting the general blmAsh mutation, is homozygous in nearly all BS
population. The predominant and constant patients with Ashkenasi Jewish ancestry (Ellis et
clinical feature of BS is proportionate pre- and al., 1995b) due to a founder effect (Ellis et al.,
postnatal growth retardation. Additional clinical 1994).
features are dolichocephaly, narrow facies with BLM gene codes for the 1417 amino acids BLM
nasal prominence and malar and mandibular protein with a predicted molecular mass of 159
hypoplasia, facial sun-sensitive telangiectatic kDa, and which belongs to the DExH box-
erythema in the butterfly area, patchy areas of containing RecQ helicase subfamily (Ellis et al.,
hyper- and hypopigmentation of the skin (café- 1995b). Recombinant (Karow et al., 1997) and
au-lait spots), and moderate to severe endogenous (Dutertre et al., 2002) BLM display
immunodeficiency manifested by recurrent an ATP- and Mg2+ dependent 3’-5’-DNA
respiratory tract and gastrointestinal infections helicase activity that separate the
(German, 1993). BS was first described in 1954 complementary strands of DNA in a 3’-5’
as “congenital telangiectatic erythema direction. But BLM function is still unclear. BLM
resembling lupus erythematosus in dwarfs” protein has been shown to accumulate in S and
(Bloom, 1954). G2 phases of the cell cycle (Dutertre et al., 2000;
Sanz et al., 2000; Bischof et al., 2001a) and to
Differential diagnosis localize in two distinct nuclear structures, PML
Bloom’s syndrome belongs to a group of nuclear bodies (also called ND10) (Ishov et al.,
“chromosomal breakage syndromes” which are 1999) and the nucleolus (Yankiwski et al., 2000).
genetic disorders that are typically transmitted in The preferred substrates for BLM are G-
an autosomal recessive mode. Cells from quadruplex DNA (Sun et al., 1998; Mohaghegh
affected individuals are characterized by an et al., 2001), D-loops structures (van Brabant et
increased frequency of breaks and interchanges al., 2000) and X-junctions (Karow et al., 2000).
occurring either spontaneously or following BLM also promotes branch migration of RecA-
exposure to various DNA-damaging agents. generated Hollyday junctions (Karow et al.,
Patients with these disorders show increased 2000). BLM interacts with several proteins
predisposition to cancer. The commonly involved in the maintenance of genome integrity.
acknowledged chromosomal breakage It participates in a super complex of BRCA1-
syndromes are Fanconi anemia, ataxia associated proteins named BASC (BRCA1-
telangiectasia, Xeroderma Pigmentosum and Associated genome Surveillance Complex) that
Bloom’s syndrome. However, the hallmark of BS includes BRCA1, mutated in some familial breast
cells is an approximately 10-fold increase in the cancers, ATM, NBS1 and MRE11 proteins,
rate of sister chromatid exchanges (SCEs) defective in Ataxia Telangectasia (AT), Nijmegen
compared to normal cells (Chaganti et al., 1974). syndrome and ataxia-telangiectasia-like
The increased level of SCEs is the only objective disorder, respectively, MLH1, MSH2 and MSH6,
criteria for BS diagnosis. It should be noted that involved in Human non-polyposis colorecal
in about 20% of BS patients, normal levels of cancer (HNPCC syndrome), and several other
SCE are observed in a subpopulation of B and T proteins known to be involved in replicational
lymphocytes (Ellis et al., 1995a; Weksberg, and/or post-replicational repair process (Wang et
1995). These low-SCE revertant BS cells result al., 2000). BLM also participates in a complex
from an intragenic crossing over between the containing RPA and topoisomerase IIIa, known
paternal and maternal BLM alleles, generating a to interact independently with BLM (Brosh et al.,
wild type allele in compound heterozygote 2000; Wu et al., 2000; Hu et al., 2001), and five
patients (Ellis et al., 1995a; Foucault et al., of the Fanconi anemia (FA) complementation
1997). group proteins (FANCA, FANCG, FANCC,
FANCE and FANCF): this complex has been
Etiology termed BRAFT (BLM, RPA, FA, Topoisomerase
Bloom’s syndrome arises through mutations in IIIa) (Meetei AR et al., 2003). The other proteins
both copies of the BLM gene, which is located
Amor-Guéret M. Bloom’s syndrome. Orphanet Encyclopedia. February 2004.
http://www.orpha.net/data/patho/GB/uk-Bloomsyndrome.pdf 2
known to interact physically and/or functionally Clinical description
with BLM are: A program of surveillance referred to as Bloom’s
• the tumor suppressor protein p53 Syndrome Registry has been established in
(Garkavtsev et al., 2001; Wang et al., 1960 in which the follow-up of 168 BS patients
2001; Yang et al., 2002; Sengupta et al., (93 males, 75 females) has been reported until
2003) 1991 (German et al., 1977; 1979; 1984; German
• the WRN protein, a RecQ helicase and Passarge, 1989). As reported in details by
defective in the Werner syndrome, a German (1995) on the basis of the data collected
premature aging disease (von Kobbe et in the Bloom’s Syndrome Registry, the two
al., 2002), constant clinical features associated with BS are
• MLH1, which participates in the growth retardation starting in utero and
mismatch repair pathway (Langland et persisting throughout life with normal
al., 2001; Pedrazzi et al., 2001), proportioning and accompanied by
• RAD51, the key protein of the dolichocephaly, and predisposition to all types of
homologous recombination (Wu et al., cancers. The mean adult height for men is 147,5
2001), cm (range 130 to 162), and for women is 138,6
• TRF2, a double-stranded telomeric DNA cm (range 122 to 151). German (1995) has also
binding protein likely involved in the noted eleven additional clinical features that are
regulation of telomeric length (Opresko not constant and that vary in severity among BS
et al., 2002; Stavropoulos et al., 2002). patients: (1) a “bird-like” facies with a narrow
The BLM protein has also been shown to be: face and prominent nose, and malar and
• cleaved into 40-47 kDa N-terminal and mandibular hypoplasia; (2) sun-sensitive
110-120 kDa C-terminal major erythema affecting the butterfly area of the face
fragments by caspase-3 in response to (similar to that caused by lupus erythematosis),
several apoptosis-inducing agents such and sometimes the dorsa of the hands and
as anti-Fas antibody, etoposide, forearms; (3) spots of hyper- and
hydroxyurea and UV-C irradiation hypopigmentation of the skin (“café au lait”
(Bischof et al., 2001b; Freire et al., 2001; spots); (4) a high-pitched voice (Mickey mouse
Ababou et al., 2002a), voice); (5) a variable degree of “vomiting and
• phosphorylated in mitotic cells (Dutertre diarrhea” during infancy; (6) diabetes mellitus
et al., 2000; Beamish et al., 2002), and (diagnosed at a mean age of 24,9 years in 20 of
in response to DNA damaging agents the 168 BS patients in the Registry); (7) small
(Ababou et al., 2000; Bishof et al., 2001; testes accompanied by a total failure of
Ababou et al., 2002b; Franchitto and spermatogenesis in men and early cessation of
Pichierri, 2002), menstruation accompanied by reduced fertility in
women; (8) immunodeficiency manifested by
• involved in DNA double-strand breaks
recurrent respiratory tract infections complicated
repair (Rünger and Kraemer, 1989;
by otitis media and pneumonia (life-threatening
Gaymes et al., 2002; Langland et al.,
ear and lung infections are common) and by the
2002; Onclercq-Delic et al., 2003),
gastrointestinal problems mentioned in 5; (9)
• associated with telomeres and ribosomal
some minor anatomic abnormalities such as
DNA repeats (Schawalder et al., 2003).
obstructing anomalies of the urethra which have
Concerning mice models of BS, among the five
been of major clinical importance in several
BS knocktout alleles that have been generated,
cases; (10) average intelligence (sometimes
four led to embryonic lethality when the targeted
mental deficiency); (11) clinical features which
allele was homozygous (Chester et al., 1998;
occurred in only one or few BS patients and that
Luo et al., 2000; Goss et al., 2002), and only one
are not to be considered part of BS itself, such
resulted in viable “BS” mice through a complex
as congenital thrombocytopenia, mild anemia,
rearrangement of the targeted region (Luo et al.,
asthma, psoriatic arthritis. These clinical
2000). Luo et al. (2000) proposed that an
information have also been reported in German
increased rate of loss of heterozygosity resulting
and Ellis (1997).
from mitotic recombination constitutes the
The 100 cancers that had arisen in 71 of the 168
underlying mechanism causing tumour
BS patients recorded in the Bloom’s Syndrome
susceptibility in these mice.
Registry have been reported (German and Ellis,
Altogether these data support a major role for
1997; German, 1997), and the distribution of
BLM in maintaining genomic stability during DNA
sites and types of these cancers is similar to that
replication, recombination and DNA repair
found in the general population. The main
(reviewed in Nakayama, 2002; Bachrati and
conclusions of this report are that nearly half of
Hickson, 2003).
the registered BS (71/168) patients have had at
least one cancer at a mean age of 24,7 years,
Amor-Guéret M. Bloom’s syndrome. Orphanet Encyclopedia. February 2004.
http://www.orpha.net/data/patho/GB/uk-Bloomsyndrome.pdf 3
40% of whom having had more than one primary
cancer (29/71). Acute leukemias, lymphomas
and rare tumors (medulloblastoma, Wilm’s
tumor, osteogenic sarcoma) represent 21%,
23% and 5% of the cancers, respectively, and
predominate in the first two decades of life,
whereas carcinomas represent 51% of the
cancers and occur later in the second decade
(German and Ellis; German, 1997).
Diagnostic methods
The hallmark of BS cells is the elevated level of
SCEs that represent the only objective criteria
for the diagnosis of the disease. SCE detection
is based on differential labeling of sister
chromatids, which is classically done by the
method developed by Wolff and Perry (1974)
and modified by Morgan et al. (1983). In brief,
blood lymphocytes stimulated by Figure 1: Normal cell metaphase
phytohemagglutinin or skin fibroblasts are
cultured in a 5-bromodeoxyuridine (BrdU)-
containing medium during two rounds of cell
replication (BrdU is an analogue of thymidine
that is incorporated specifically into DNA of
replicating cells), resulting in one of the sister
chromatids unifilarly substituted with BrdU and
the other one bifilarly substituted. Then,
colcemid is added for the last hours before
fixation to arrest the cells in metaphase.
Differential staining of BrdU-substituted
chromatids is obtained by fluorescence-plus-
Giemsa coloration of spread mitotic
chromosomes. The unifilarly substituted
chromatids are darkly stained (black) and the
bifilarly substituted ones are stained lightly
(white) (see figures 1 and 2).
SCEs frequency averages 0.24 per chromosome
in normal cells and 2.12 per chromosome in BS
cells (McDaniel and Schultz, 1992). Figure 2: BS cell metaphase
The screening for BLM gene mutations could
also be performed by the analysis of the 21 Epidemiology
coding exons (4437 bp in length), and it is Bloom’s syndrome is a very rare disease and its
possible to look for specific changes in the BLM frequency in the general population is unknown.
gene, in particular for the blmAsh mutation. However, BS is more common in Askenazic
Jewish population, reaching a frequency of
approximately 1 in 48 000 (Shahrabani-Gargir et
al., 1998).
Genetic counseling
Due to the autosomal recessive transmission
mode of BS, sibs of two heterozygous carriers
are at 25% risk of having BS and 50% risk of
being a carrier. It should be noted that among
the Ashkenazi Jewish population, the
heterozygous carrier frequency of this mutation
is approximately 1% (Ellis et al., 1998; Li et al.,
1998; Shahrabani-Gargir et al., 1998). Two
studies conducted to assess the cancer risk
among blmAsh heterozygotes gave contradictory
results (Gruber et al., 2002; Cleary et al., 2003).
Amor-Guéret M. Bloom’s syndrome. Orphanet Encyclopedia. February 2004.
http://www.orpha.net/data/patho/GB/uk-Bloomsyndrome.pdf 4
Antenatal diagnosis References
When the risk of BS transmission has been well Ababou M, Dutertre S, Lécluse Y, Onclercq R,
evaluated, prenatal diagnosis can be proposed Chatton B, Amor-Guéret M. ATM-dependent
(SCEs analysis of fetal cells or looking for a phosphorylation and accumulation of
specific BLM gene mutation when causative endogenous BLM protein in response to ionizing
mutations are identified). radiation, Oncogene, 19, 5955-5963, 2000.
Ababou M, Dumaire V, Lécluse Y Amor-Guéret
Management including treatment M. Bloom’s syndrome protein response to
There is no curative treatment for BS. However, ultraviolet C radiation and hydroxyurea mediated
a physician should carefully follow BS patients in DNA synthesis inhibition. Oncogene, 21, 2079-
order to ensure early diagnosis of cancer. 2088, 2002a.
Ababou M, Dumaire V, Lécluse Y, M Amor-
Unresolved questions Guéret. Cleavage of BLM protein and sensitivity
The underlying mechanisms causing BS are not of Bloom’s syndrome cells to hydroxyurea and
well defined since BLM protein’ s function(s) is UVC radiation. Cell Cycle, 4, 262-266, 2002b.
still unclear. Bachrati CZ, Hickson ID. RecQ helicases:
suppressors of tumorigenesis and premature
aging. Biochem J, 374, 577-606, 2003.
Barakat A, Ababou M, Onclercq R, Dutertre S,
Chadli E, Had N, Benslimane A, Amor-Guéret M.
Identification of a novel BLM missense mutation
(T2706C) in a moroccan patient with Bloom’s
syndrome. Hum Mut, 15, 584-585, 2000.
Beamish H, Kedar P, Kaneko H, Chen P, Fukao
T, Peng C, Beresten S, Gueven N, Purdie D,
Lees-Miller S, Ellis N, Kondo N, Lavin MF.
Functional link between BLM defective in
Bloom's syndrome and the ataxia-telangiectasia-
mutated protein, ATM. J Biol Chem, 277, 30515-
30523, 2002.
Bischof O, Kim SH, Irving J Beresten S, Ellis
NA, Campisi J. Regulation and localization of the
Bloom syndrome protein in response to DNA
damage. J Cell Biol,153, 367-380, 2001a.
Bischof O, Galande S, Farzaneh F, Kohwi-
Shigematsu T, Campisi J. Selective cleavage of
BLM, the Bloom syndrome protein, during
apoptotic cell death. J Biol Chem, 276, 12068-
12075, 2001b.
Bloom D. Congenital telangiectatic erythema
resembling lupus erythematosus in dwarfs. Am.
J Dis Child, 88, 754-758, 1954.
Brosh RM Jr, Li JL, Kenny MK, Karow JK,
Cooper MP, Kureekattil RP, Hickson ID, Bohr
VA. Replication protein A physically interacts
with the Bloom's syndrome protein and
stimulates its helicase activity. J Biol Chem, 275,
23500-23508, 2000.
Chaganti RSK, Schonberg S, German J. A
manyfold increase in sister chromatide
exchanges in Bloom's syndrome lymphocytes.
Proc Natl Acad Sci USA, 71, 4508-4512, 1974.
Cleary SP, Zhang W, Di Nicola N, Aronson M,
Aube J, Steinman A, Haddad R, Redston M,
Gallinger S, Narod SA, Gryfe R. Heterozygosity
for the BLM(Ash) mutation and cancer risk.
Cancer Res, 63, 1769-1771, 2003.
Chester N, Kuo F, Kozak C, O'Hara CD, Leder
P. Stage-specific apoptosis, developmental
delay, and embryonic lethality in mice
Amor-Guéret M. Bloom’s syndrome. Orphanet Encyclopedia. February 2004.
http://www.orpha.net/data/patho/GB/uk-Bloomsyndrome.pdf 5
homozygous for a targeted disruption in the Bloom's syndrome. Oncogene, 21, 2525-2533,
murine Bloom's syndrome gene. Genes Dev, 12, 2002.
3382-3393, 1998. German J, Bloom D, Passarge E. Bloom's
Dutertre S, Ababou M, Onclercq R, Delic J, syndrome. V. Surveillance for cancer in affected
Chatton B, Jaulin C, Amor-Gueret M. families. Clin Genet, 12, 162-168, 1977.
Endogenous Bloom’s syndrome helicase German J, Bloom D, Passarge E. Bloom's
expression during the cell cycle. Oncogene, 19, syndrome. VII. Progress report for 1978. Clin
2731-2738, 2000. Genet, 15, 361-367, 1979.
Dutertre S, Sekhri R, Tintgnac L.A, Onclercq- German J, Bloom D, Passarge E. Bloom's
Delic R, Chatton B, Jaulin C Amor-Guéret M. syndrome. XI. Progress report for 1983. Clin
Dephosphorylation and subcellular compartment Genet, 25, 166-174, 1984.
change of the mitotic Bloom’s syndrome DNA German J, Passarge E. Bloom's syndrome. XII.
helicase in response to ionizing radiation. J Biol Report from the Registry for 1987. Clin Genet,
Chem, 277, 6280-6286, 2002. 35, 57-69, 1989.
Ellis NA, Roe AM, Kozloski J, Proytcheva M, German J. Bloom syndrome : a Mendelian
Falk C, German J. Linkage disequilibrium prototype of somatic mutational disease.
between the FES, D15S127, and BLM loci in Medicine. 72, 393-406, 1993.
Ashkenazi Jews with Bloom syndrome. Am J German J. Bloom's syndrome. Dermatol Clin,
Hum Genet, 55, 453-460, 1994. 13, 7-18, 1995.
Ellis NA, Lennon DJ, Proytcheva M, Alhadeff B, German J. Bloom's syndrome. XX. The first 100
Henderson EE, German J. Somatic intragenic cancers. Cancer Genet Cytogenet, 93, 100-106,
recombination within the mutated locus BLM can 1997.
correct the high sister-chromatid exchange German J, Ellis NA. Bloom syndrome. The
phenotype of Bloom syndrome cells. Am J Hum genetic basis of human cancer. Editors :
Genet, 57, 1019-1027, 1995a. Vogelstein B, Kinzler KW. Chapter 30, 733-751,
Ellis NA, Groden J, Ye TZ, Straughen J, Lennon 1997.
DJ, Ciocci S, Proytcheva M, German J. The Goss KH, Risinger MA, Kordich JJ, Sanz MM,
Bloom's syndrome gene product is homologous Straughen JE, Slovek LE, Capobianco AJ,
to RecQ helicases. Cell. 83, 655-666, 1995b. German J, Boivin GP, Groden J. Enhanced
Ellis NA, Ciocci S, Proytcheva M, Lennon D, tumor formation in mice heterozygous for Blm
Groden J, German J. The Ashkenazic Jewish mutation. Science, 297, 2051-2053, 2002.
Bloom syndrome mutation blmAsh is present in Gruber SB, Ellis NA, Scott KK, Almog R,
non-Jewish Americans of Spanish ancestry. Am. Kolachana P, Bonner JD, Kirchhoff T, Tomsho
J Hum Genet, 63, 1685-1693, 1998. LP, Nafa K, Pierce H, Low M, Satagopan J,
Foucault F, Vaury, C, Barakat A., Thibout D., Rennert H, Huang H, Greenson JK, Groden J,
Planchon P., Jaulin C., Praz F, M Amor-Guéret. Rapaport B, Shia J, Johnson S, Gregersen PK,
Characterization of a new BLM mutation Harris CC, Boyd J, Offit K. BLM heterozygosity
associated with a topoisomerase II defect in a and the risk of colorectal cancer. Science, 297,
patient with Bloom's syndrome. Hum Mol Genet, 2013, 2002.
6, 1427-1434, 1997. Hu P, Beresten SF, van Brabant AJ, Ye TZ,
Franchitto A, Pichierri P. Bloom's syndrome Pandolfi PP, Johnson FB, Guarente L, Ellis NA.
protein is required for correct relocalization of Evidence for BLM and Topoisomerase IIIalpha
RAD50/MRE11/NBS1 complex after replication interaction in genomic stability. Hum Mol Genet,
fork arrest. J Cell Biol., 157, 19-30, 2002. 10, 1287-1298, 2001.
Freire R, d'Adda Di Fagagna F, Wu L, Pedrazzi Ishov AM, Sotnikov AG, Negorev D, Vladimirova
G, Stagljar I, Hickson ID, Jackson SP. Cleavage OV, Neff N, Kamitani T, Yeh ET, Strauss JF 3rd,
of the Bloom's syndrome gene product during Maul GG. PML is critical for ND10 formation and
apoptosis by caspase-3 results in an impaired recruits the PML-interacting protein daxx to this
interaction with topoisomerase III alpha. Nucleic nuclear structure when modified by SUMO-1. J
Acids Res, 29, 3172-3180, 2001. Cell Biol, 147, 221-234, 1999.
Garkavtsev IV, Kley N, Grigorian IA, Gudkov A. Karow JK, Chakraverty RK, Hickson ID. The
The Bloom syndrome protein interacts and Bloom's syndrome gene product is a 3'-5' DNA
cooperates with p53 in regulation of transcription helicase. J Biol Chem, 49, 464-469, 1997.
and cell growth control. Oncogene, 20, 8276- Karow JK, Constantinou A, Li,JL, West SC,
8280, 2001. Hickson ID. The Bloom's syndrome gene
Gaymes TJ, North PS, Brady N, Hickson ID, product promotes branch migration of holliday
Mufti GJ, Rassool FV. Increased error-prone non junctions. Proc Natl Acad Sci U S A, 97, 6504-
homologous DNA end-joining--a proposed 6508, 2000.
mechanism of chromosomal instability in Langland G, Kordich J, Creaney J, Goss KH,
Lillard-Wetherell K, Bebenek K, Kunkel TA,
Amor-Guéret M. Bloom’s syndrome. Orphanet Encyclopedia. February 2004.
http://www.orpha.net/data/patho/GB/uk-Bloomsyndrome.pdf 6
Groden J. The Bloom's syndrome protein (BLM) Sengupta S, Linke SP, Pedeux R, Yang Q,
interacts with MLH1 but is not required for DNA Farnsworth J, Garfield SH, Valerie K, Shay JW,
mismatch repair. J Biol Chem., 276, 30031- Ellis NA, Wasylyk B, Harris,CC. BLM helicase-
30035, 2001. dependent transport of p53 to sites of stalled
Langland G, Elliott J, Li Y, Creaney J, Dixon K, DNA replication forks modulates homologous
Groden J. The BLM helicase is necessary for recombination. EMBO J, 22, 1-13, 2003.
normal DNA double-strand break repair. Cancer Shahrabani-Gargir L, Shomrat R, Yaron Y, Orr-
Res, 62, 2766-2770, 2002. Urtreger A, Groden J, Legum C. High frequency
Li L, Eng C, Desnick RJ, German J, Ellis NA. of a common Bloom syndrome Ashkenazi
Carrier frequency of the Bloom syndrome mutation among Jews of Polish origin. Genet
blmAsh mutation in the Ashkenazi Jewish Test, 2, 293-296, 1998.
population. Mol Genet Metab, 64, 286-290, Schawalder J, Paric E, Neff NF. Telomere and
1998. ribosomal DNA repeats are chromosomal targets
Luo G, Santoro IM, McDaniel LD, Nishijima I, of the bloom syndrome DNA helicase. BMC Cell
Mills M, Youssoufian H, Vogel H, Schultz RA, Biol, 4, 15, 2003.
Bradley A. Cancer predisposition caused by Stavropoulos DJ, Bradshaw PS, Li X, Pasic I,
elevated mitotic recombination in Bloom mice. Truong K, Ikura M, Ungrin M, Meyn MS. The
Nat Genet, 26, 424-429, 2000. Bloom syndrome helicase BLM interacts with
McDaniel LD, Schultz RA. Elevated sister TRF2 in ALT cells and promotes telomeric DNA
chromatid exchange phenotype of Bloom synthesis. Hum Mol Genet, 11, 3135-3144,
syndrome cells is complemented by human 2002.
chromosome 15. Proc Natl Acad Sci U S A, 89, Sun H, Karow JK, Hickson ID, Maizels N. The
7968-7972, 1992. Bloom's syndrome helicase unwinds G4 DNA. J
Meetei AR, Sechi S, Wallisch M, Yang D, Young Biol Chem, 273, 27587-27592, 1998.
MK, Joenje H, Hoatlin ME, Wang W. A van Brabant AJ, Ye T, Sanz M, German III JL,
multiprotein nuclear complex connects Fanconi Ellis NA, Holloman WK. Binding and melting of
anemia and Bloom syndrome. Mol Cell Biol,23, D-loops by the Bloom syndrome helicase.
3417-3426, 2003. Biochemistry, 39, 14617-14625, 2000.
Mohaghegh P, Karow JK, Brosh Jr RM Jr, Bohr von Kobbe C, Karmakar P, Dawut L, Opresko
VA, Hickson ID. The Bloom's and Werner's P, Zeng X, Brosh RM Jr, Hickson ID, Bohr VA.
syndrome proteins are DNA structure-specific Colocalization, physical, and functional
helicases. Nucleic Acids Res, 29, 2843-2849, interaction between Werner and Bloom
2001. syndrome proteins. J Biol Chem,277, 22035-
Morgan WF, Schwartz JL, Murnane JP, Wolff S. 22044, 2002.
Effect of 3-aminobenzamide on sister chromatid Wang Y, Cortez D, Yazdi P, Neff N, Elledge SJ,
exchange frequency in X-irradiated cells. Radiat Qin J. BASC, a super complex of BRCA1-
Res, 93, 567-571, 1983. associated proteins involved in the recognition
Nakayama H. RecQ family helicases: roles as and repair of aberrant DNA structures. Genes
tumor suppressor proteins. Oncogene, 21, 9008- and Dev, 14, 927-939, 2000.
9021, 2002. Wang X.W, Tseng A, Ellis N.A, Spillare EA,
Onclercq-Delic R, Calsou P, Delteil C, Salles B, Linke SP, Robles AI, Seker H, Yang Q, Hu P,
Papadopoulo D, Amor-Guéret M. Possible anti- Beresten S, Bemmels NA, Garfield S, Harris CC.
recombinogenic role of Bloom’s syndrome Functional interaction of p53 and BLM DNA
helicase in double-strand break processing. helicase in apoptosis. J Biol Chem, 276, 32948-
Nucleic Acids Res, 31, 6272-6282, 2003. 32955, 2001.
Opresko PL, von Kobbe C, Laine JP, Harrigan Weksberg R. Low-sister-chromatid-exchange
J, Hickson ID, Bohr VA. Telomere-binding Bloom syndrome cell lines: an important new
protein TRF2 binds to and stimulates the Werner tool for mapping the basic genetic defect in
and Bloom syndrome helicases. J Biol Chem, Bloom syndrome and for unraveling the biology
277, 41110-41119, 2002. of human tumor development. Am J Hum Genet,
Pedrazzi G, Perrera C, Blaser H, Kuster P, 57, 994-997,1995.
Marra G, Davies SL, Ryu GH, Freire R, Hickson Wolff S, Perry P. Differential Giemsa staining of
ID, Jiricny J, Stagljar I. Direct association of sister chromatids and the study of chromatid
Bloom's syndrome gene product with the human exchanges without autoradiography.
mismatch repair protein MLH1. Nucleic Acids Chromosoma, 48, 341-353, 1974.
Res, 29, 4378-86, 2001. Wu L, Davies SL, North PS, Goulaouic H, Riou
Rünger TM, Kraemer KH. Joining of linear JF, Turley H, Gatter KC, Hickson ID. The
plasmid DNA is reduced and error prone in Bloom's syndrome gene product interacts with
Bloom's syndrome cells. EMBO J, 8, 1419-1425, topoisomerase III. J Biol Chem, 275, 9636-9644,
1989. 2000.
Amor-Guéret M. Bloom’s syndrome. Orphanet Encyclopedia. February 2004.
http://www.orpha.net/data/patho/GB/uk-Bloomsyndrome.pdf 7
Wu L, Davies SL, Levitt NC, Hickson ID. Brosh Jr RM, Karmakar P, Bohr VA, Harris CC.
Potential role for the BLM helicase in The processing of Holliday junctions by BLM and
recombinational repair via a conserved WRN helicases is regulated by p53. J Biol
interaction with RAD51. J Biol Chem, 276, Chem., 277, 31980-31987, 2002.
19375-19381, 2001. Yankiwski V, Marciniak RA, Guarente L, Neff
Yang Q, Zhang R, Wang XW, Spillare EA, Linke NF. Nuclear structure in normal and Bloom
SP, Subramanian D, Griffith JD, Li JL, Hickson syndrome cells. Proc Natl Acad Sci U S A, 97,
ID, Shen JC, Loeb LA, Mazur SJ, Appella E, 5214-5219, 2000.
Amor-Guéret M. Bloom’s syndrome. Orphanet Encyclopedia. February 2004.
http://www.orpha.net/data/patho/GB/uk-Bloomsyndrome.pdf 8
You might also like
- Mycoses - 2023 - Kato - A Systematic Review and Meta Analysis of Efficacy and Safety of Isavuconazole For The Treatment andDocument10 pagesMycoses - 2023 - Kato - A Systematic Review and Meta Analysis of Efficacy and Safety of Isavuconazole For The Treatment andc gNo ratings yet
- Las Claves Genéticas - Richard RuddDocument29 pagesLas Claves Genéticas - Richard RuddaleNo ratings yet
- Beckwith-Wiedemann SyndromeDocument6 pagesBeckwith-Wiedemann SyndromeAhmad Althof Malihul AdiNo ratings yet
- Molecular Diagnosis of Inherited Disorders: Lessons From HemoglobinopathiesDocument14 pagesMolecular Diagnosis of Inherited Disorders: Lessons From HemoglobinopathiesUmar AliNo ratings yet
- Thein-2004-British Journal of HaematologyDocument11 pagesThein-2004-British Journal of HaematologySri ArianiNo ratings yet
- Journal of Genetic Counseling - 2008 - Pilarski - Cowden Syndrome A Critical ReDocument15 pagesJournal of Genetic Counseling - 2008 - Pilarski - Cowden Syndrome A Critical ReCiudadano “Citizen Dick” VolanteNo ratings yet
- Kes - Martin A. - Mutations in The Adrenoleukodystrophy Gene (1997) (10.1002 - (Sici) 1098-1004 (1997) 9 - 63.0.co - 2-5) - Libgen - LiDocument12 pagesKes - Martin A. - Mutations in The Adrenoleukodystrophy Gene (1997) (10.1002 - (Sici) 1098-1004 (1997) 9 - 63.0.co - 2-5) - Libgen - LiLoplopNo ratings yet
- Fioretos 2001Document9 pagesFioretos 2001sawantleena62No ratings yet
- 2003 - Epigenetic Silencing of Multiple Interferon Pathway Genes After Cellular ImmortalizationDocument10 pages2003 - Epigenetic Silencing of Multiple Interferon Pathway Genes After Cellular ImmortalizationFernando Fernández GarcíaNo ratings yet
- FPDMM2Document9 pagesFPDMM2sccNo ratings yet
- Statistical Applications in Genetics and Molecular BiologyDocument19 pagesStatistical Applications in Genetics and Molecular BiologyJose MoralesNo ratings yet
- 60 453 PDFDocument13 pages60 453 PDFVictor Hugo OrtizNo ratings yet
- Kotlyar 2020Document16 pagesKotlyar 2020Merab KvintradzeNo ratings yet
- Anorectal Malformation: The Etiological FactorsDocument10 pagesAnorectal Malformation: The Etiological FactorsHasri Rina WalastriNo ratings yet
- Abnormal Folate Metabolism and MaternalDocument24 pagesAbnormal Folate Metabolism and MaternalJovicaNo ratings yet
- Zlotogora 2004Document6 pagesZlotogora 2004MARIA JOSE MENDIOLA FIGUEROANo ratings yet
- Predisposing Factors: TH NDDocument3 pagesPredisposing Factors: TH NDArianna Jasmine MabungaNo ratings yet
- Craniofacial MicrosomiaDocument21 pagesCraniofacial MicrosomiaMarcela Cano Ricardo100% (1)
- Shiang 1994Document8 pagesShiang 1994Daniela NarvaezNo ratings yet
- CML Chapter Chereda & Melo Springer 2016Document23 pagesCML Chapter Chereda & Melo Springer 2016JuniaNo ratings yet
- 12.the Impact of Trisomy 21 On Early Human HematopoiesisDocument2 pages12.the Impact of Trisomy 21 On Early Human HematopoiesisCristi Daniel NeagoeNo ratings yet
- Genetica Predisposicion InglesDocument12 pagesGenetica Predisposicion InglesAlejandro PalayNo ratings yet
- Cancer CellDocument140 pagesCancer CellmanbastiNo ratings yet
- 1.0 Genetics and InheritanceDocument13 pages1.0 Genetics and Inheritanceincognitus94100% (4)
- Epidermolysis Bullosa, Junctional, Herlitz Type: (Type The Document Title)Document11 pagesEpidermolysis Bullosa, Junctional, Herlitz Type: (Type The Document Title)kucingkucingNo ratings yet
- 2015 Int J Genet & Mol Biol Article1424860767 - Ortega Et AlDocument7 pages2015 Int J Genet & Mol Biol Article1424860767 - Ortega Et AlAlejandro1-7No ratings yet
- Hum. Reprod. Update 2002 Laml 483 91Document9 pagesHum. Reprod. Update 2002 Laml 483 91Amit GoelNo ratings yet
- Ageing and The Mismatch Repair SystemDocument7 pagesAgeing and The Mismatch Repair Systemeric.cheninNo ratings yet
- Campbell - Walsh-Wein UROLOGY 12th Ed (Dragged) 2Document24 pagesCampbell - Walsh-Wein UROLOGY 12th Ed (Dragged) 2revanth kallaNo ratings yet
- Sickle Cell DiseaseDocument18 pagesSickle Cell DiseasedrheayNo ratings yet
- X-Linked Dominant Chondrodysplasia PunctataDocument5 pagesX-Linked Dominant Chondrodysplasia Punctatalocos3dNo ratings yet
- Analysis of Recombination Along Chromosome 21 During Human Female Pachytene StageDocument11 pagesAnalysis of Recombination Along Chromosome 21 During Human Female Pachytene StageJimmy AvilaNo ratings yet
- Causes of Congenital MalformationsDocument5 pagesCauses of Congenital MalformationsBenyam ZenebeNo ratings yet
- DescargaDocument18 pagesDescargakirbwer macedoNo ratings yet
- Abp-Ivb AnemiaDocument11 pagesAbp-Ivb AnemiaEmerson MadridNo ratings yet
- Historical and Clinical Perspectives On CHR TranslocationsDocument14 pagesHistorical and Clinical Perspectives On CHR TranslocationsAsma HMILANo ratings yet
- 1 s2.0 S0041008X04001334 MainDocument14 pages1 s2.0 S0041008X04001334 MainOscar Alonso LunaNo ratings yet
- Flejter 1996Document6 pagesFlejter 1996Rahul AmbawataNo ratings yet
- Onco Ch23 ESFTDocument31 pagesOnco Ch23 ESFTIlincaNo ratings yet
- Syndromes of The First and Second Pharyngeal Arches: A ReviewDocument7 pagesSyndromes of The First and Second Pharyngeal Arches: A ReviewHARITHA H.PNo ratings yet
- Prion-Like Mechanisms in Alzheimer DiseaseDocument17 pagesPrion-Like Mechanisms in Alzheimer Diseasesabrina whardhaniNo ratings yet
- Genetic Counseling in TranslocationsDocument15 pagesGenetic Counseling in TranslocationsAsma HMILANo ratings yet
- Transient Myeloproliferative Disorder: What's Eosinophilia Got To Do With It?Document1 pageTransient Myeloproliferative Disorder: What's Eosinophilia Got To Do With It?wadeschulzNo ratings yet
- Imprinting DisordersDocument10 pagesImprinting DisordersAhmad KHNo ratings yet
- Craniofacialmicrosomia: Kathleyn A. Brandstetter,, Krishna G. PatelDocument21 pagesCraniofacialmicrosomia: Kathleyn A. Brandstetter,, Krishna G. PatelariskaNo ratings yet
- Imprinting DisordersDocument19 pagesImprinting Disorderssenuberh13No ratings yet
- Current Concepts Review: Ph. Debeer, L. de Smet, W. J. M. Van de Ven, G. Fabry, J.-P. FrynsDocument12 pagesCurrent Concepts Review: Ph. Debeer, L. de Smet, W. J. M. Van de Ven, G. Fabry, J.-P. FrynsDr. Pedro Javier Cadena GonzálezNo ratings yet
- Genetic Background of Cutaneous Forms of Lupus Erythematosus: Update On Current EvidenceDocument10 pagesGenetic Background of Cutaneous Forms of Lupus Erythematosus: Update On Current EvidenceFaradhillah Adi SuryadiNo ratings yet
- Churpek Et Al. - 2015 - Genomic Analysis of Germ Line and Somatic Variants in Familial Myelodysplasiaacute Myeloid Leukemia Key PointsDocument7 pagesChurpek Et Al. - 2015 - Genomic Analysis of Germ Line and Somatic Variants in Familial Myelodysplasiaacute Myeloid Leukemia Key Pointstefod30422No ratings yet
- Genetic Factors of Childhood CancerDocument3 pagesGenetic Factors of Childhood CancerDima HarounNo ratings yet
- FPDMM3Document10 pagesFPDMM3sccNo ratings yet
- Pol 030Document6 pagesPol 030Mirabella AstraNo ratings yet
- Epigenetic and Cancer Therapy, 2019Document12 pagesEpigenetic and Cancer Therapy, 2019AbsjeyNo ratings yet
- Pediatric Genetics Dr. Samed AlsalmiDocument166 pagesPediatric Genetics Dr. Samed AlsalmiabhivnairNo ratings yet
- 31861.1 20201218131318 CoveredDocument5 pages31861.1 20201218131318 CoveredPatrick StarNo ratings yet
- La Mellarin D2Document2 pagesLa Mellarin D2JESUS DAVID BOLA‹O JIMENEZNo ratings yet
- Imam ShahDocument7 pagesImam Shahdk3873050No ratings yet
- PSU Vol 14, 2000Document2 pagesPSU Vol 14, 2000rajarshikNo ratings yet
- Lecture 2 EnglishDocument24 pagesLecture 2 Englishmondalsawan96No ratings yet
- Genetica Das Doencas Hematologicas As HemoglobinopDocument12 pagesGenetica Das Doencas Hematologicas As HemoglobinopMinésio João DoclasNo ratings yet
- A Model for Gene Therapy: Gene Replacement in the Treatment of Sickle Cell Anemia and ThalassemiaFrom EverandA Model for Gene Therapy: Gene Replacement in the Treatment of Sickle Cell Anemia and ThalassemiaNo ratings yet
- Neuromuscular DisordersDocument1 pageNeuromuscular DisordersMowlidAbdirahman Ali madaaleNo ratings yet
- Epi Lecture 1 Part IIDocument36 pagesEpi Lecture 1 Part IIMowlidAbdirahman Ali madaaleNo ratings yet
- Chain of InfectionDocument35 pagesChain of InfectionMowlidAbdirahman Ali madaale100% (1)
- Microbiology RVDocument91 pagesMicrobiology RVMowlidAbdirahman Ali madaaleNo ratings yet
- Basics of ProbabilityDocument7 pagesBasics of ProbabilityMowlidAbdirahman Ali madaaleNo ratings yet
- CholeraDocument4 pagesCholeraMowlidAbdirahman Ali madaaleNo ratings yet
- Lec-4 The Anterior Pituitary Gland-IIDocument32 pagesLec-4 The Anterior Pituitary Gland-IIMowlidAbdirahman Ali madaaleNo ratings yet
- Biochemistry Answers 161-179Document2 pagesBiochemistry Answers 161-179MowlidAbdirahman Ali madaaleNo ratings yet
- Biochemistry 2Document9 pagesBiochemistry 2MowlidAbdirahman Ali madaaleNo ratings yet
- Toxicology Lecture 10 (Chapter 7 Volatiles)Document22 pagesToxicology Lecture 10 (Chapter 7 Volatiles)MowlidAbdirahman Ali madaaleNo ratings yet
- Lec-5 The Posterior Pituitary GlandDocument39 pagesLec-5 The Posterior Pituitary GlandMowlidAbdirahman Ali madaaleNo ratings yet
- Chapter 6. ....Document11 pagesChapter 6. ....MowlidAbdirahman Ali madaaleNo ratings yet
- General Principles of Prevention and Control of DiseasesDocument17 pagesGeneral Principles of Prevention and Control of DiseasesMowlidAbdirahman Ali madaaleNo ratings yet
- Sexually Transmitted DiseasesDocument54 pagesSexually Transmitted DiseasesMowlidAbdirahman Ali madaaleNo ratings yet
- Floods and DroughtDocument28 pagesFloods and DroughtMowlidAbdirahman Ali madaaleNo ratings yet
- Forensic Med Lecture 11 Chapter 11 Sexual Assaults)Document27 pagesForensic Med Lecture 11 Chapter 11 Sexual Assaults)MowlidAbdirahman Ali madaaleNo ratings yet
- Lec-3 The Pituitary Gland-IDocument37 pagesLec-3 The Pituitary Gland-IMowlidAbdirahman Ali madaaleNo ratings yet
- Transportation: About UsDocument2 pagesTransportation: About UsMowlidAbdirahman Ali madaaleNo ratings yet
- Group 4 DDDocument2 pagesGroup 4 DDMowlidAbdirahman Ali madaaleNo ratings yet
- Genital Infections and SexuallyDocument47 pagesGenital Infections and SexuallyMowlidAbdirahman Ali madaaleNo ratings yet
- Sumayo Xasan Omar 3. Sumayo C/Raxmaan Cali 4. Sabirin Daud Cilmi 5. Sagal Dahir Shire Mrs. Fatima SahraDocument4 pagesSumayo Xasan Omar 3. Sumayo C/Raxmaan Cali 4. Sabirin Daud Cilmi 5. Sagal Dahir Shire Mrs. Fatima SahraMowlidAbdirahman Ali madaaleNo ratings yet
- History of MakaDocument2 pagesHistory of MakaMowlidAbdirahman Ali madaaleNo ratings yet
- Yaya 7Document2 pagesYaya 7MowlidAbdirahman Ali madaaleNo ratings yet
- Yaxye 6Document3 pagesYaxye 6MowlidAbdirahman Ali madaaleNo ratings yet
- White House and Facebook InteractionsDocument31 pagesWhite House and Facebook InteractionstobihiNo ratings yet
- Septic ArthritisDocument12 pagesSeptic ArthritisRashi JainNo ratings yet
- Dr. Ranjita Singh Department of PathologyDocument19 pagesDr. Ranjita Singh Department of PathologyMohan Prasad GuptaNo ratings yet
- Feyer 2020 WvngoshawkDocument13 pagesFeyer 2020 WvngoshawkAgnieszka CzujkowskaNo ratings yet
- Smoking Is DangerousDocument3 pagesSmoking Is DangerousAbdulmalkNo ratings yet
- YökdilDocument195 pagesYökdilglisilsiklin antibiyotikNo ratings yet
- 03 NCP 1 Viral HepatitisDocument17 pages03 NCP 1 Viral Hepatitisamit100% (2)
- Psychophysiological Methods in NeuroscienceDocument17 pagesPsychophysiological Methods in NeuroscienceKariela EstherNo ratings yet
- ABG Ebook PDFDocument13 pagesABG Ebook PDFM DNo ratings yet
- Locomotor Training in People With Parkinson DiseaseDocument10 pagesLocomotor Training in People With Parkinson DiseasemilananandNo ratings yet
- Anees Ahmed Corrected Thesis - RecentDocument135 pagesAnees Ahmed Corrected Thesis - RecentAbdul MunimNo ratings yet
- Brochure Chorustrio Rev.04 enDocument4 pagesBrochure Chorustrio Rev.04 enHaitham Salama Ghareeb100% (1)
- 1.19 Exam PracticeDocument5 pages1.19 Exam Practicesunnr008.211No ratings yet
- Pediatric Assessment TriangleDocument31 pagesPediatric Assessment TriangleblairNo ratings yet
- Psych Narrative Report (1) LoissDocument21 pagesPsych Narrative Report (1) LoissBea Aldueza100% (1)
- Role of The Youth in The Upcoming ElectionDocument15 pagesRole of The Youth in The Upcoming ElectionMitch Escribano MarianoNo ratings yet
- Safety Data Sheet For PolyuDocument4 pagesSafety Data Sheet For PolyuMehedi HasanNo ratings yet
- Reten Modern Dressing 2023 PKDMTDocument51 pagesReten Modern Dressing 2023 PKDMTMasros TukiranNo ratings yet
- Heritability of Behavior in The Abnormally Aggressive Dog, by A. SemyonovaDocument8 pagesHeritability of Behavior in The Abnormally Aggressive Dog, by A. SemyonovaDogsBite.org100% (9)
- Infection Control NY NewDocument6 pagesInfection Control NY NewRC PmedNo ratings yet
- Copy-Of-Chapter 38 Medical Gas TherapyDocument29 pagesCopy-Of-Chapter 38 Medical Gas TherapyMaxinne GorospeNo ratings yet
- Bakteriološki Nalaz U Uretri Muškaraca Sa I Bez Negonoroičnog UretritisaDocument4 pagesBakteriološki Nalaz U Uretri Muškaraca Sa I Bez Negonoroičnog UretritisaNemanjaDragosavacNo ratings yet
- International Journal of Research in AYUSH and Pharmaceutical SciencesDocument6 pagesInternational Journal of Research in AYUSH and Pharmaceutical SciencesDeepak BaghelNo ratings yet
- Chapter 13 - Diseases of White Blood Cells, Lymph Nodes, Spleen, and ThymusDocument10 pagesChapter 13 - Diseases of White Blood Cells, Lymph Nodes, Spleen, and ThymusAgnieszka WisniewskaNo ratings yet
- What Is ECG?Document2 pagesWhat Is ECG?Lê Thanh HàNo ratings yet
- TriageDocument37 pagesTriagekyuleen05No ratings yet
- 10 Secrets in 10 YearsDocument15 pages10 Secrets in 10 YearsasicsNo ratings yet
- Revised Monthly Reporting Format - HWCDocument8 pagesRevised Monthly Reporting Format - HWCChanmari West Sub-Centre50% (2)
- Pola Bleach PDFDocument1 pagePola Bleach PDFSree CumarNo ratings yet